Abstract
Introduction:
Minimally invasive liver surgery for hepatocellular carcinoma has gained widespread interest as an alternative to conventional open liver surgery. However, long-term survival benefits of this approach seem unclear. This meta-analysis was conducted to investigate long-term survival following minimally invasive liver surgery.
Method:
A systematic review was performed to identify studies comparing long-term survival after minimally invasive liver surgery and open liver surgery until January 2020. The I2 test was used to test for statistical heterogeneity and publication bias was assessed using Egger test. Random-effects meta-analysis was performed for all-cause 5-year (main outcome) and 3-year mortality, and disease-specific 5-year and 3-year mortality. Meta-regression was performed for the 5-year and 3-year survival outcomes with adjustment for study factors (region, design), annual center volume, patient factors (American Society of Anesthesiologists (ASA) grade, gender, age, body mass index, cirrhosis, tumor size, and number), and resection extent. Sensitivity analyses were performed on studies by study year, region, annual center volume, and resection type.
Result:
The review identified 50 relevant studies including 13,731 patients undergoing liver resection for hepatocellular carcinoma of which 4071 (25.8%) underwent minimally invasive liver surgery. Pooled analysis revealed similar all-cause (odds ratio: 0.83, 95% confidence interval: 0.70–1.11, p = 0.3) and disease-specific (odds ratio: 0.93, 95% confidence interval: 0.80–1.09, p = 0.4) 5-year mortality after minimally invasive liver surgery compared with open liver surgery. Sensitivity analysis of published studies from 2010 to 2019 demonstrated a significantly lower disease-specific 3-year mortality (odds ratio: 0.75, 95% confidence interval: 0.59–0.96, p = 0.022) and all-cause 5-year mortality (odds ratio: 0.63, 95% confidence interval: 0.50–0.81, p = 0.002). Meta-regression identified no confounding factors in all analyses.
Conclusions:
Improvement in minimally invasive liver surgery techniques over the past decade appears to demonstrate superior disease-specific mortality with minimally invasive liver surgery compared to open liver surgery. Therefore, minimally invasive liver surgery can be recommended as an alternative surgical approach for hepatocellular carcinoma.
Keywords
Introduction
Advancements in minimally invasive techniques over the last decade have increased the importance of these methods in curative treatment of hepatocellular carcinoma (HCC). However, it has taken longer for the field of hepatic surgery to adopt this technology when compared to other specialties. 1 The majority of patients with HCC suffer from chronic liver disease and cirrhosis, which often limits the choice of treatment. In addition, these patients are often associated with high morbidity which further compounds the problem. 2 These surgical complexities have limited the use of minimally invasive methods, but recent technical refinements and improvements in instruments have strengthened the case for mainstream use.1,2
Despite this, evidence surrounding the long-term survival following minimally invasive liver surgery (MILS) remains conflicting. Several studies have reported that MILS is associated with improved long-term survival,3–6 while some studies have demonstrated no statistically significant impact.1,2,7–28 For example, a single-center, high-volume study by Yoon et al. 1 and multi-center study by Yamamoto et al. 2 reported no difference in survival following MILS when compared to open liver surgery (OLS). However, a recent single-center study by Xiang et al. 21 found improved long-term survival following MILS.
As there is a lack of clarity in the long-term survival benefits following minimally invasive methods, this systematic review and meta-analysis aims to summarize the evidence concerning the impact of MILS on long-term survival. This study also performed a meta-regression to identify study-, hospital-, and patient-level factors confounding study findings.
Methods
Search strategy
This study was conducted according to the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) group guidelines. 29 A systematic and comprehensive search was undertaken of MEDLINE, EMBASE, and Cochrane Library databases, searching for studies published until 20 December 2019. Search terms included the following, individually or in combination: “robotics” or “robotic surgery” or “robot-assisted” or “laparoscopic” or “laparoscopic-assisted” or “open” and “hepatectomy” or “segmentectomy” or “liver resection” and “complications” or “mortality” or “survival.” The full search strategy with all the included search terms is presented in Supplementary Table 1.
Inclusion and exclusion
The inclusion criteria were (1) studies exploring survival outcomes following MILS (laparoscopic and/or robotic) and OLS and (2) studies published in the English language. Exclusion criteria were (1) case reports (<5 patients), review articles, conference abstracts; (2) papers with metastatic liver disease; (3) studies not reporting survival outcomes in patients with HCC; and (4) all studies describing hand-assisted or hybrid resections (without subgroup data on pure laparoscopic resections).
After performing the literature search and removing all duplicates, two researchers (R.R.G., S.K.K.) independently screened the remaining titles and abstracts. Where a paper was considered for inclusion, the full text was examined for closer review. In doing this, any potential discrepancies between the judgment of the two primary researchers were resolved. Where multiple studies analyzed the same dataset or population, the most recent paper was selected unless different outcomes were reported.
Study outcomes
Primary outcome measure was all-cause 5-year mortality defined as time from surgery to last known follow-up or death. Secondary outcome measures were all-cause and disease-specific 3-year mortality and disease-specific 5-year mortality. Both all-cause and disease-specific mortality at 3 years and 5 years were derived from survival curves of studies, where available, as previously described.30,31
Data extraction
One researcher (R.R.G.) extracted data on study characteristics (author, year of publication, country, study interval, number of participants), patient characteristics (age, sex, body mass index (BMI), American Society of Anesthesiologists (ASA) grade, cirrhosis), details of surgical approach, resection type, and reported clinical outcomes.
Definitions
Hepatic resection was considered to be major (resection of three or more segments) and minor (resection of less than three segments) in accordance with the Brisbane nomenclature of hepatic anatomy and resection. 32
Assessment of study quality
Methodological quality and standard of the outcome reporting study was assessed in each study by two independent researchers (R.R.G., S.K.K.). Any disagreements were settled through discussion between these researchers. The Risk Of Bias In Non-randomized Studies of Interventions (ROBINS-I) was used to formally assess the quality in cohort studies, 33 and the Revised Cochrane risk-of-bias tool for randomized trials (RoB 2) was used to assess the methodological quality where appropriate. 34
Statistical analysis
This systematic review and network meta-analysis was conducted in accordance with the recommendations of the Cochrane Library and PRISMA guidelines.29,35 Data analysis was undertaken using R Foundation Statistical software (R 3.2.1) as previously described, 36 to produce a random-effects meta-analysis for each outcome, providing pooled odds ratio (OR) with 95% confidence intervals (95% CIs). The choice of a random-effects analysis was based on the variability of minimally invasive techniques within each comparison group (e.g. totally robotic, totally laparoscopic) between the studies, which might influence the magnitude of the treatment effect. 37 The I2 test was used to evaluate statistical heterogeneity of the included studies, with levels of heterogeneity defined as not important (I2: 0%–40%), moderate (I2: 30%–60%), substantial (I2: 50%–90%), or considerable (I2: 75%–100%). 31 The chi-square test was used for the same purpose, with a statistical significance level of p < 0.05, indicating presence of statistical heterogeneity. Publication bias and small study effects were assessed using Egger test for statistical asymmetry in the funnel plot, with a statistical significance level of p < 0.1 indicating publication bias or small-study effects. Random-effects meta-regression was used to adjust for potential confounding by the following variables: year of publication, continent of origin, study design (retrospective cohort study, prospective cohort study, randomized controlled trials), case matching (yes, no), annual center volume (total, Minimally invasive pancreatiocoduodenectomy (MIPD), open), patient age (median), gender (male, female), and ASA physical status (I, II, III, and IV) upon the results of the all-cause 3-year and 5-year mortality outcomes.
Results
Study characteristics
Of the 7453 unique studies, 50 studies met the inclusion criteria and were included into the paper (Fig. 1). Of the 50 studies, these included a total of 13,731 patients undergoing liver resection for HCC, of which 23.6% (n = 3234) underwent MILS. Of the 50 papers, only 2 papers reported use of robotic liver resections and the remaining 48 papers were laparoscopic liver resections. Majority of these studies were from Asia (n = 36), followed by Europe (n = 10), North America (n = 3), and Africa (n = 1). Prospective cohort studies accounted for 18 of the included papers with one randomized control trial; the remaining studies were all retrospective cohort studies. A summary of the quality assessment of the included studies using the ROBINS-I and RoB 2 tools is presented in Supplementary Tables 3 and 4.33,34
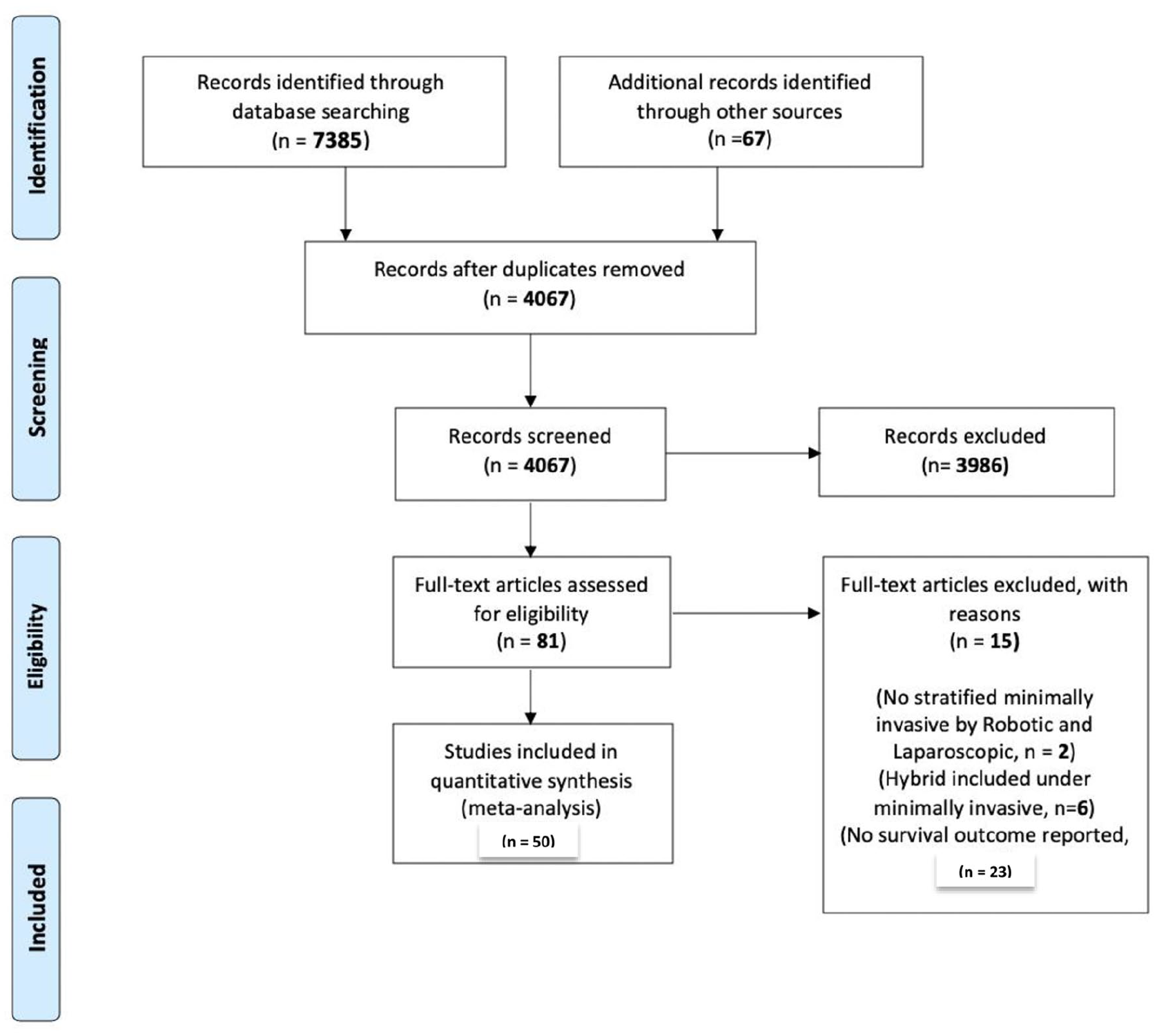
PRISMA Diagram of included studies.
Patient and tumor characteristics
Baseline characteristics of patient demographics are presented in Supplementary Table 2. Age was reported in 46 studies and median age was similar between patients receiving MILS and OLS (median: 59 vs 60 years). Gender was reported in 48 studies and rates of male gender were comparable between both MILS and OLS (71% vs 75%). Furthermore, cirrhosis rates were reported in 44 studies with comparable rates of cirrhosis in MILS and OLS (74% vs 70%). Tumor number was reported in 25 studies and rates of solitary tumor were comparable between MILS and OLS (78% vs 80%). Barcelona Clinic Liver Cancer (BCLC) staging system was only reported in six studies. Again, the rates of BCLC stage 0 (27% vs 21%), A (37% vs 37%), and B (30% vs 36%) cancers were comparable between MILS and OLS. Adjuvant therapy following resection was reported in seven studies. The rates of radiofrequency ablation (3% vs 1%), transarterial chemoembolization (12% vs 9%), chemotherapy (0% vs 1%), re-resection (3% vs 1%), and liver transplantation (1% vs 1%) were similar between MILS and OLS.
All-cause mortality
3-year mortality
A total of 47 studies reported all-cause 3-year mortality (Fig. 2). The pooled analysis revealed 3% decreased 3-year mortality after MILS compared with OLS (OR: 0.97, 95% CI: 0.78–1.20, p = 0.8). The statistical heterogeneity of the studies was “substantial” (I2: 65%). The funnel plot was symmetrical both according to visual and statistical testing (Egger test = 0.27), arguing against small-study effects or publication bias. The sensitivity analysis of contemporary studies from 2010 to 2019 included 12 studies and revealed a similar OR as in the main analysis (OR: 1.28, 95% CI: 0.79–2.06, p = 0.3).
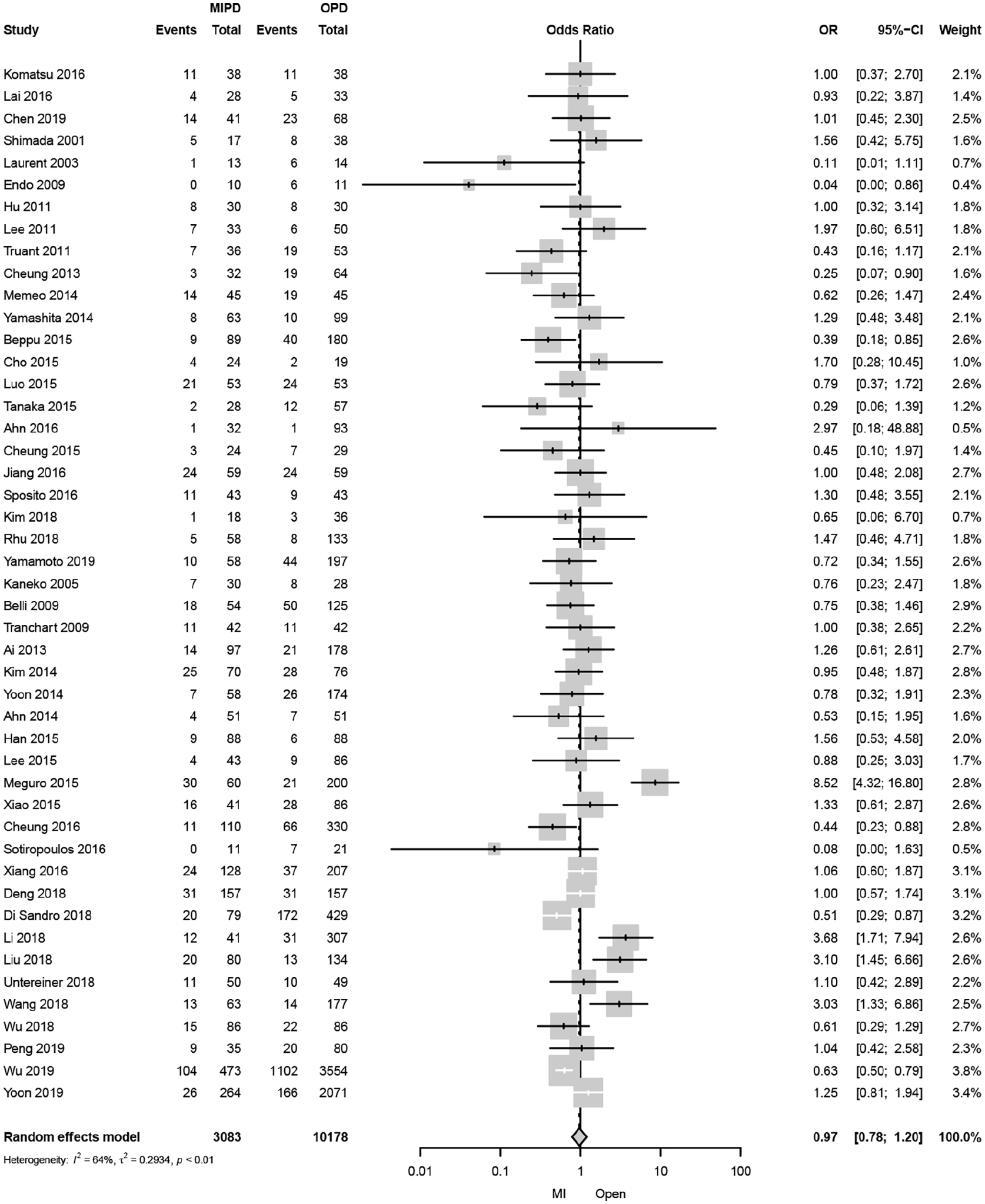
Forest plot of all-cause 3-year mortality comparing minimally invasive and open liver resection for hepatocellular carcinoma.
5-year mortality
A total of 33 studies reported all-cause 5-year mortality (Fig. 3). The pooled analysis revealed 12% decreased 5-year mortality after MILS compared with OLS (OR: 0.88, 95% CI: 0.70–1.11, p = 0.3). The statistical heterogeneity of the studies was “substantial” (I2: 68%). The funnel plot was symmetrical both according to visual and statistical testing (Egger test = 0.16), arguing against small-study effects or publication bias. The sensitivity analysis of contemporary studies from 2010 to 2019 included four studies and revealed a significantly lower risk of death with MILS than OLS (OR: 0.63, 95% CI: 0.50–0.81, p = 0.002). Meta-regression indicated no confounding by the covariates of region, study design, annual center volume, ASA grade, gender, age, BMI, cirrhosis, and resection type (Supplementary Tables 5–7).
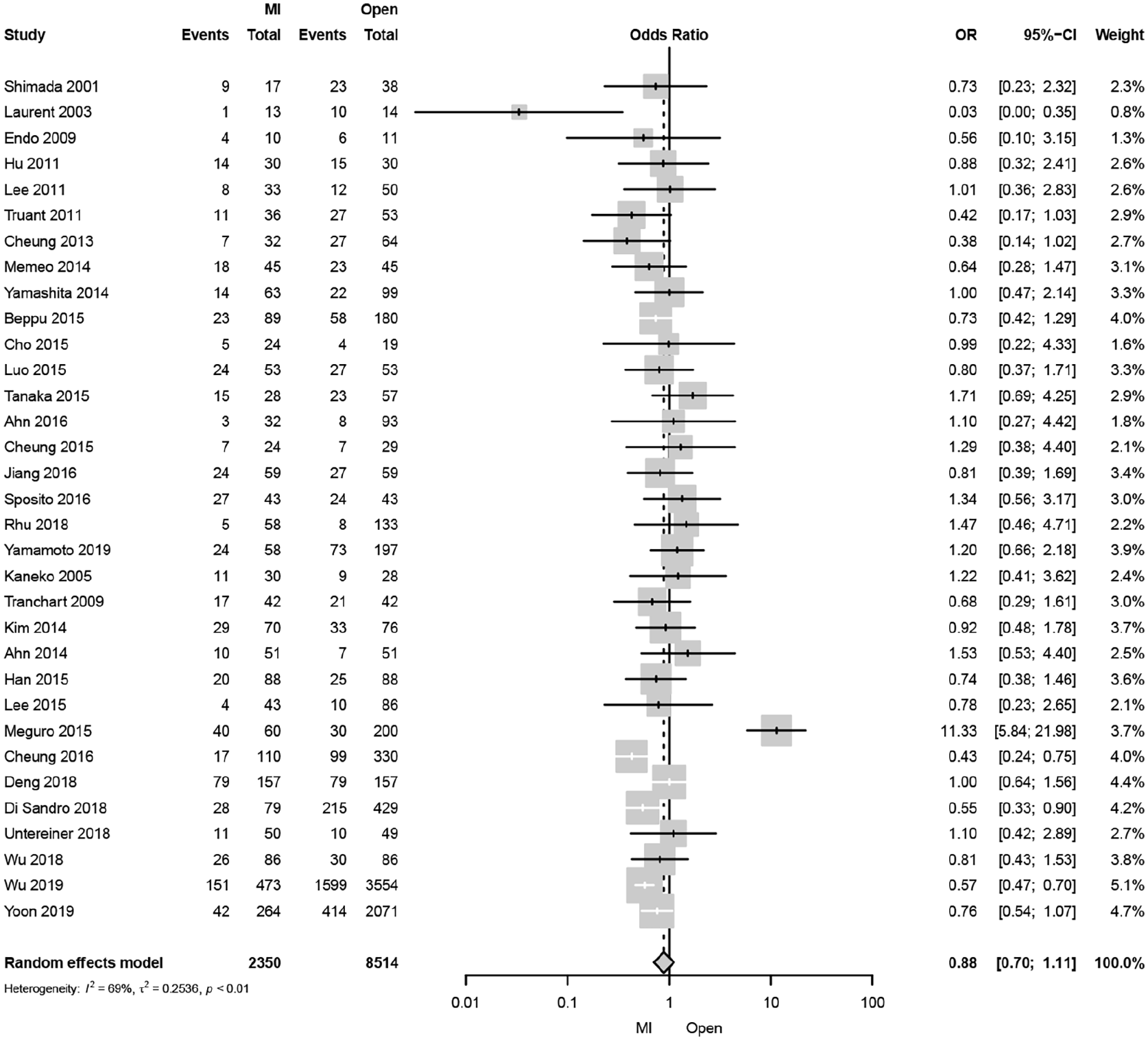
Forest plot of all-cause 5-year mortality comparing minimally invasive and open liver resection for hepatocellular carcinoma.
Disease-specific mortality
3-year mortality
A total of 39 studies reported disease-specific 3-year mortality (Fig. 4). The pooled analysis revealed a 18% decrease in disease-specific 3-year mortality after MILS compared with OLS (OR: 0.86, 95% CI: 0.77–0.97, p = 0.013). The statistical heterogeneity of the studies was “not important” (I2: 1%). The funnel plot was symmetrical both according to visual and statistical testing (Egger test = 0.98), arguing against small-study effects or publication bias. The sensitivity analysis of contemporary studies from 2010 to 2019 including eight studies demonstrated a significantly lower disease-specific 3-year mortality (OR: 0.75, 95% CI: 0.59–0.96, p = 0.022). The statistical heterogeneity of the studies was “not important” (I2: 0%).
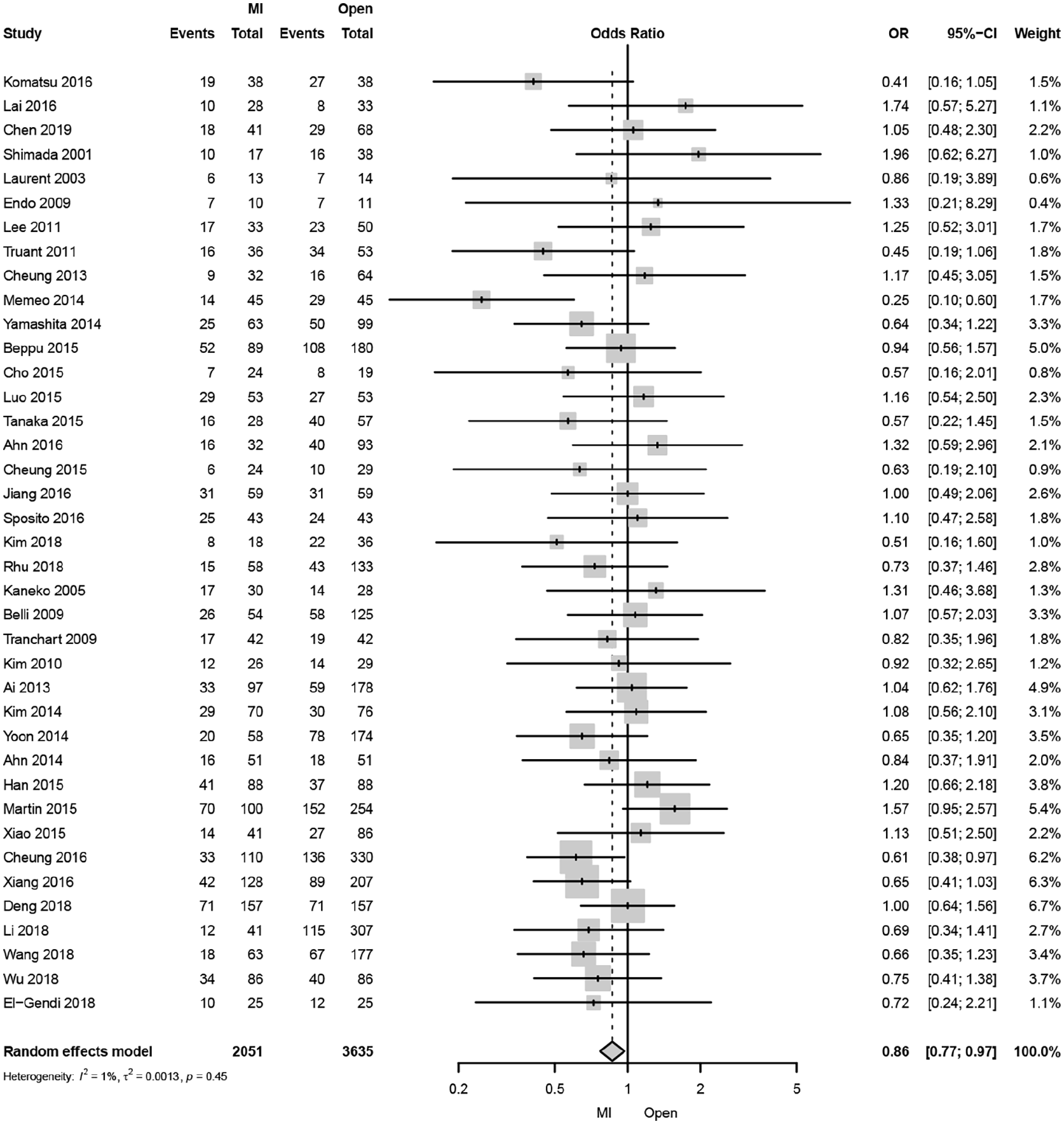
Forest plot of disease-specific 3-year mortality comparing minimally invasive and open liver resection for hepatocellular carcinoma.
5-year mortality
A total of 26 studies reported disease-specific 5-year mortality (Fig. 5). The pooled analysis revealed a 7% decrease in disease-specific 5-year mortality after MILS compared with OLS (OR: 0.93, 95% CI: 0.80–1.09, p = 0.4). The statistical heterogeneity of the studies was “not important” (I2: 6%). The funnel plot was symmetrical both according to visual and statistical testing (Egger test = 0.57), arguing against small-study effects or publication bias. The sensitivity analysis of contemporary studies from 2010 to 2019 including two studies demonstrated a similar disease-specific 5-year mortality with MILS compared to OLS (OR: 0.95, 95% CI: 0.58–1.53, p = 0.8). The statistical heterogeneity of the studies was “not important” (I2: 0%). Meta-regression indicated no confounding by the covariates of region, study design, annual center volume, ASA grade, gender, age, BMI, cirrhosis, and resection type (Supplementary Table 8).
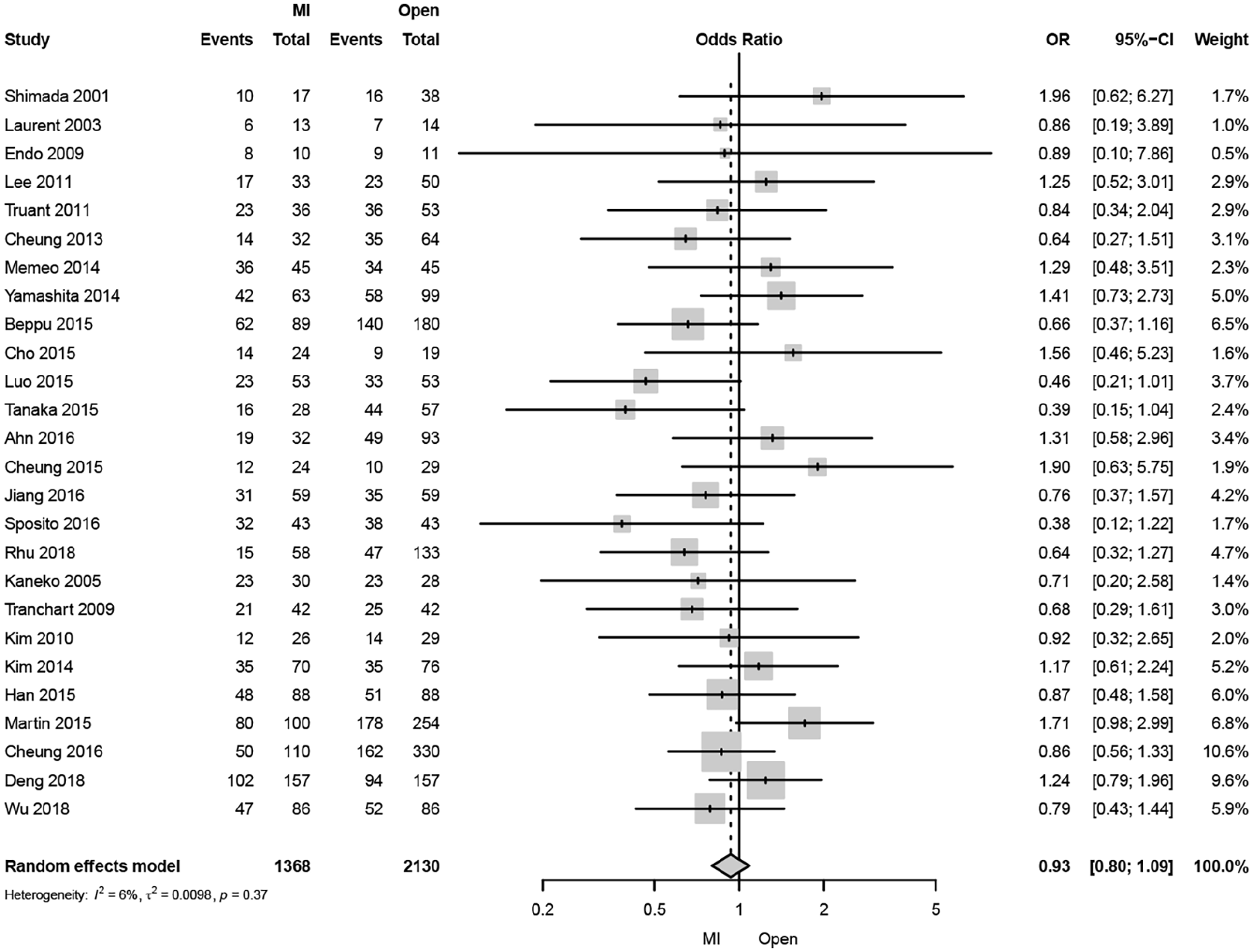
Forest plot of disease-specific 5-year mortality comparing minimally invasive and open liver resection for hepatocellular carcinoma.
Discussion
Since the Southampton Consensus Guidelines for Laparoscopic Liver Surgery (LLS) conference in 2017, 38 the optimism regarding the wider implementation of a minimally invasive approach seems justified on the basis of accruing evidence within laparoscopic treatment of colorectal liver metastases.39–41 However, the greatest controversy that remains to be addressed for laparoscopic resection of HCC is its long-term impact on survival. This is systematic review and meta-analysis including 50 studies and almost 14,000 patients demonstrated MILS is not associated with improvement in 5-year all-cause and disease-specific mortality. Sensitivity analyses of contemporary published data, ranging from 2010 to 2019, demonstrated MILS had a significantly lower 5-year all-cause mortality and 3-year disease-specific mortality compared to OLS. From a conservative viewpoint, these results can therefore be interpreted to indicate that laparoscopy is at least not inferior to the standard open approach.
This study has both strengths and weaknesses. An advantage is the extensive literature search, covering cohort studies, randomized clinical trials, unpublished data, and conference abstracts, which provided a large number of included studies and patients. In general, the included studies were of relatively high quality according to the quality measurement tools. The low presence of heterogeneity, small-study effects, and publication bias are other advantages. Studies that used historical cohorts of OLS were excluded in sensitivity analyses with subsequent results differing from the main analysis. This indicates that bias due to improved overall treatment over time may perhaps explain the results. The meta-regression was adjusted for important prognostic factors, including extent of resection, and showed no confounding by these factors. Yet, residual confounding cannot be excluded, especially for age and ASA grade, which was only reported in relatively few of the included papers. There is a possibility that patients with beneficial prognostic variables, for example, younger age and less comorbidity, were more readily selected for MILS rather than open surgery. The funnel plots were symmetrical for studies with 3 years and 5 years of follow-up, including the main outcome of all-cause mortality, strengthening the validity of the results for these outcomes. Furthermore, the pooled ORs were similar (0.82–0.91) for all four outcomes, implying consistency of the study findings. Different surgical methods were used in the included studies, which makes it difficult to more specifically point out if there was any singular MILS technique that was the most beneficial for long-term survival, that is, laparoscopy or robotic or the combination of these. In fact, the present study compromises several surgical procedures in both the MILS and OLS groups. However, as hybrid procedures and hand-assisted techniques were not included in the MILS group, the variance was reduced.42–48 The surgical approach is often determined by the location of the tumor to allow adequate lymphadenectomy. 49
Previous meta-analyses comparing laparoscopic and open liver resection have been performed. However, the variation in minimally invasive techniques such as pure laparoscopic and hand-assisted has not been accounted for in some studies, introducing potential bias when considering conclusions.50–54 A recent meta-analysis that did consider these factors lacks many large-scale studies published in the years following and they only evaluated the effect of purely laparoscopic resection compared with OLS for HCC. 55 This prompted us to re-examine the topic with a strict methodology and criteria to update findings with the best available evidence. We included robotic liver resections to truly examine the effect of MILS when compared to OLS. This, in combination with the increased total study size of 15,784 compared to 5100, found a significant reduction in 3-year disease-specific mortality unlike the previous meta-analysis. This association remained significant under sensitivity analysis and 5-year disease-specific mortality also a showed significant decrease with MILS under the similar conditions.
An increasing evidence base within MILS now includes the recent Oslo-CoMet39,56 and LapOpHuva 40 randomized trials, affirming that it does not compromise intraoperative oncologic adequacy, but offers other benefits in terms of reducing complications, length of hospital stay, and improving health-related quality of life and cost-effectiveness. Nonetheless, patients in the LapOpHuva 40 and Oslo-CoMet39,56 trials have only been followed up for a minimum of 1 and 3 years, respectively, whereas long-term outcomes from the Orange-II study have not been reported. 57 Overall survival has been advocated as the primary measure of efficacy in oncologic trials58,59 and is recognized to be influenced by a multitude of postoperative factors beyond immediate surrogate endpoints such as resection margins. In fact, recent studies of hepatectomy for colorectal liver metastases have questioned the utility of resection margins as a surrogate endpoint for overall survival.60,61
This study cannot assess the mechanisms behind the suggested reduced long-term mortality after MILS compared with OLS. Although OLS may be associated with improved radical surgical clearance of the tumor (R0 resection rates) and/or lymph nodes yield, MILS may be associated with a lower rate of severe complications compared with OLS, 27 which is likely due to there being less surgical trauma. Major postoperative complications and reoperations after liver resection have been found to be associated with reduced long-term survival. 62 Pulmonary infections have been shown to be less common after MILS, probably explained by the avoidance of laparotomy for certain operations. 63 Consequently, it is possible that MILS leads to fewer, and perhaps, less severe complications, which may, in turn, permit a higher proportion of patients to retain enough immuno-competence to delay or avoid tumor recurrence and ensuing death.27,64,65
Conclusion
In conclusion, contemporary data appears to suggest superior disease-specific mortality with MILS compared to OLS with no change in all-cause mortality. Thus, MIPD can be recommended as an alternative surgical approach for HCC. Further studies in the topic are still needed, as an increase in high-quality cohort studies and randomized clinical trials would improve the state of current evidence.
Supplemental Material
sj-pdf-1-sjs-10.1177_14574969211042455 – Supplemental material for Does minimally invasive liver resection improve long-term survival compared to open resection for hepatocellular carcinoma? A systematic review and meta-analysis
Supplemental material, sj-pdf-1-sjs-10.1177_14574969211042455 for Does minimally invasive liver resection improve long-term survival compared to open resection for hepatocellular carcinoma? A systematic review and meta-analysis by Sivesh K. Kamarajah, Rohan R. Gujjuri, Moh’d A. Hilal, Derek M. Manas and Steven A. White in Scandinavian Journal of Surgery
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
