Abstract
Background and objective:
Intra-abdominal hypertension is frequently present in critically ill patients and is an independent predictor for mortality. In this narrative review, we aim to provide a comprehensive overview of current insights into intra-abdominal pressure monitoring, intra-abdominal hypertension, and abdominal compartment syndrome. The focus of this review is on the pathophysiology, risk factors and outcome of intra-abdominal hypertension and abdominal compartment syndrome, and on therapeutic strategies, such as non-operative management, surgical decompression, and management of the open abdomen. Finally, future steps are discussed, including propositions of what a future guideline should focus on.
Conclusions:
Pathological intra-abdominal pressure is a continuum ranging from mild intra-abdominal pressure elevation without clinically significant adverse effects to substantial increase in intra-abdominal pressure with serious consequences to all organ systems. Intra-abdominal pressure monitoring should be performed in all patients at risk of intra-abdominal hypertension. Although continuous intra-abdominal pressure monitoring is feasible, this is currently not standard practice. There are a number of effective non-operative medical interventions that may be performed early in the patient’s course to reduce intra-abdominal pressure and decrease the need for surgical decompression. Abdominal decompression can be life-saving when abdominal compartment syndrome is refractory to non-operative treatment and should be performed expeditiously. The objectives of open abdomen management are to prevent fistula and to achieve delayed fascial closure at the earliest possible time. There is still a lot to learn and change. The 2013 World Society of Abdominal Compartment Syndrome guidelines should be updated and multicentre studies should evaluate the effect of intra-abdominal hypertension treatment on patient outcome.
Keywords
Background and aims
Intra-abdominal hypertension (IAH) and abdominal compartment syndrome (ACS) are conditions that result from increased intra-abdominal pressure (IAP). IAH is frequently present in critically ill patients and is an independent predictor for mortality.1,2 The World Society of the Abdominal Compartment Syndrome (WSACS) was founded in 2004 (www.wsacs.org) and its early accomplishments were the development of uniform definitions for IAH and ACS and standardized techniques for IAP monitoring to facilitate research and improve patient care. 3 The definitions of IAP and ACS are based on consensus, and the absolute IAP levels and thresholds used facilitate comparisons. However, in clinical practice, pathological IAP is a continuum ranging from mild IAP elevations without clinically significant adverse effects to substantial increases in IAP with multi-organ failure. 3
In this review, we aim to provide a comprehensive overview of current insights into IAP measurement, IAH, and ACS. Its focus is on pathophysiology, risk factors, and outcomes of IAH and ACS and on therapeutic strategies, such as non-operative management, surgical decompression, and management of the open abdomen. Finally, future steps are discussed, including propositions of what a future guideline should focus on.
How to measure intra-abdominal pressure
IAP is the steady-state pressure within the abdominal cavity. 4 Since the abdominal compartment generally transduces pressure evenly throughout the cavity, IAP can be measured in nearly every part of the abdomen.5,6 Clinical assessment of IAP based on palpation of the abdomen is unreliable, and clinically significant IAH may be present in the absence of abdominal distension. 7 IAP can be measured directly from catheters placed in the peritoneal cavity; for example, during laparoscopy when the abdomen is insufflated with CO2 gas. Indirect IAP may be measured in hollow organs in the abdominal cavity with easy contact with the atmosphere: the bladder, stomach, rectum, or vagina. 6 In critical care, IAP measurements through either bladder or stomach are most practical and intravesicular IAP measurement is considered the gold standard. 5 However, measurements may be inaccurate; for example, when artifacts in the IAP waveform lead to signal over- or underdamping or when the pressure transducer is malpositioned relative to the reference point. 6
Furthermore, regional inter-compartmental pressure differences between the upper and lower abdomen have been described in patients following liver transplantation. 8 A standardized approach to IAP measurement is important to ensure reproducibility, 5 and the WSACS consensus definitions state that IAP should be expressed in mmHg and measured at end-expiration in the complete supine position after ensuring that abdominal muscle contractions are absent and with the transducer zeroed at the level of the midaxillary line (Table 1). The reference standard for intermittent IAP measurement is through the bladder with a maximal instillation volume of 25 mL sterile saline. 3
World Society of Abdominal Compartment Syndrome (WSACS) consensus definitions.

From a theoretical viewpoint, the only correct reference level is the mid-bladder level at the tip of the Foley catheter, but this level cannot be identified at the bedside. The WSACS-recommended zero reference level is the midaxillary line at the level of the iliac crest as this bony structure is easy to palpate, even in obese patients.
Clinically relevant changes in IAP occur at the head-of-bed increases above 20° 9 and when measured at 30° and 45° head-of-bed elevation, the IAP is, respectively, 4 and 9 mmHg higher on average than in the supine position. 10
Normal IAP is approximately 5–7 mmHg in critically ill adults, 4 but there is a positive correlation between IAP and body mass index (BMI). 11 Baseline IAP levels are between 9 and 14 mmHg in morbidly obese patients. 10
IAP monitoring
The WSACS-recommended IAP is measured every 4–6 h in critically ill patients with one or more risk factors for the development of IAH or ACS, and protocols for IAP measurement should be developed for each intensive care unit (ICU). 5 Continuous IAP monitoring is feasible, but is currently not standard practice. 12 The development of new technologies in IAP monitoring is promising. For example, the Accuryn urinary catheter (Potrero Medical, San Francisco, CA) measures IAP from a small balloon seamlessly integrated directly below the catheter tip. IAP will be measured and displayed at the pressing of a button. It is technically possible to continuously monitor IAP using this system. Furthermore, the Accuryn urinary catheter continuously measures urine output. 13
In 2004, the cost of intermittent IAP measurements was estimated to be between 20 and 80 euros per patient. Set-up costs for continuous IAP monitoring are higher by at least a factor 3. Set-up cost of IAP monitoring using a microchip was estimated to be a factor 40 higher than intermittent intravesicular measurement. 6 In the Netherlands, in 2020, the cost of the Accuryn urinary catheter is approximately 200 euro per patient once the set-up cost of the reusable monitor (approximately 9800 euro) has been covered.
Abdominal compliance
Abdominal compliance (Cab) is defined as a measure of the ease of abdominal expansion. This is determined by the elasticity of the abdominal wall and diaphragm. It should be expressed as the change in intra-abdominal volume (IAV) per change in IAP (in ml/mmHg). Since the abdominal pressure–volume relationship is curvilinear, a small decrease in IAV, for example, by drainage of abdominal free fluid, may result in a substantial decrease in IAP in patients with ACS. Measurement of Cab at the bedside is difficult, which may explain why it has been called a neglected parameter in critically ill patients. Measurement can only be done in case of a change in IAV; for example, when drainage of intra-abdominal free fluid is performed. Normal Cab is around 250–450 mL/mmHg. Decreased Cab means that the same increase in IAV will result in a greater increase in IAP compared to normal. Factors associated with decreased Cab include male gender, android body composition, fluid overload, and prone positioning. Treatment of patients with decreased Cab and patients with IAH are largely based on the same principles.14,15
Pathophysiology
Increased abdominal pressure has adverse effects on many different organ systems. Most often this involves the heart, lungs, and kidneys, but the intestines, liver, and brain may also be affected.
The ACS is comparable to a compartment syndrome in general, where increased pressure within an anatomic compartment leads to decreased blood flow and ultimately to tissue hypoxia. 16
The pathophysiology of IAH and ACS is not completely understood, but a cascade is set in motion leading to a vicious cycle of deterioration and multi-organ failure. 17 Elevated IAP leads to compression of the vena cava causing reduced venous return to the heart. This decrease in preload leads to reduced cardiac output resulting in reduced blood flow to the organs (including kidneys and intestines). Furthermore, high intrathoracic pressures may result from elevation of the diaphragm due to high IAP. Thoracic compliance is reduced leading to increased airway pressures or increased work of breathing. Fluid resuscitation will further increase edema of the abdominal and thoracic wall and decrease abdominal and thoracic compliance. Edema of the bowel will increase bowel volume adding to this vicious cycle of increasing IAP. 18 Decreased splanchnic perfusion leads to decreased mucosal blood flow and bacterial translocation, as Diebel described in a rodent model of ACS. 19 Bowel ischemia and multi-organ failure may follow. 20 Hepatic venous, arterial, and microcirculatory blood flow decreases significantly with even slight increases in IAP. Hepatic dysfunction may lead to decreased clearance of plasma lactate, further adding to metabolic acidosis. 21
The increase in intra-thoracic pressure impairs the venous return of the brain, thereby increasing intra-cranial pressure (ICP) and consequently, decreasing cerebral blood flow. 22
Pathophysiology of acute kidney injury
The pathophysiology of acute kidney injury (AKI) is complex and has not been completely elucidated. Decreased cardiac output leading to decreased renal perfusion contributes to AKI, but multiple factors play a role. 17 Oliguria may be one of the earliest signs of ACS. 23 IAH reduces renal blood flow and glomerular filtration rate through compression of the renal veins and arterial vasoconstriction. This leads to activation of the renin–angiotensin–aldosterone hormone system (RAAS) signaling cascade. RAAS regulates blood pressure, fluid, and electrolyte balance, and systemic vascular resistance. When renal blood flow is reduced, renin is secreted into the circulation by juxtaglomerular cells. Angiotensinogen is converted to angiotensin I by renin, and angiotensin I is subsequently converted to angiotensin II by the angiotensin-converting enzyme (ACE). Angiotensin II is a potent vasoconstrictor leading to an increase in systemic vascular resistance. Angiotensin II also stimulates the secretion of aldosterone from the adrenal cortex. Aldosterone causes the renal tubules to increase the reabsorption of sodium and water in the blood, which increases the volume of extracellular fluid in the body. The intended effect is to increase renal perfusion. 24 Vasoconstriction may also be mediated by the sympathetic nervous system. Although mean arterial blood pressure (MAP) may increase at first, it soon stabilizes or decreases. 18 The inflammatory response triggered by IAH may also add to AKI.16,17 In IAP > 20 mmHg, an increase in circulating levels of a variety of inflammatory mediators has been described in rats. 25 Renal tissue and plasma tumor necrosis factor-alpha (TNF-alpha) and interleukin-6 (IL-6) levels were significantly higher in rats undergoing increases in IAP compared to sham rats. There was an association with histopathological changes in renal tissue, and renal tubular changes were most prominent. 26 In a study of living kidney donors, an increase in inflammatory mediators was found in patients whose IAP was only 12 mmHg. 27 In this study, living kidney donors were randomly assigned to hand-assisted laparoscopy (with induction of a pneumoperitoneum of 12 mmHg) and open nephrectomy. Postoperative levels of C-reactive protein (CRP) and IL-6 were higher in the laparoscopy group even though the procedure time was shorter.
Epidemiology
Prevalence of IAH and ACS
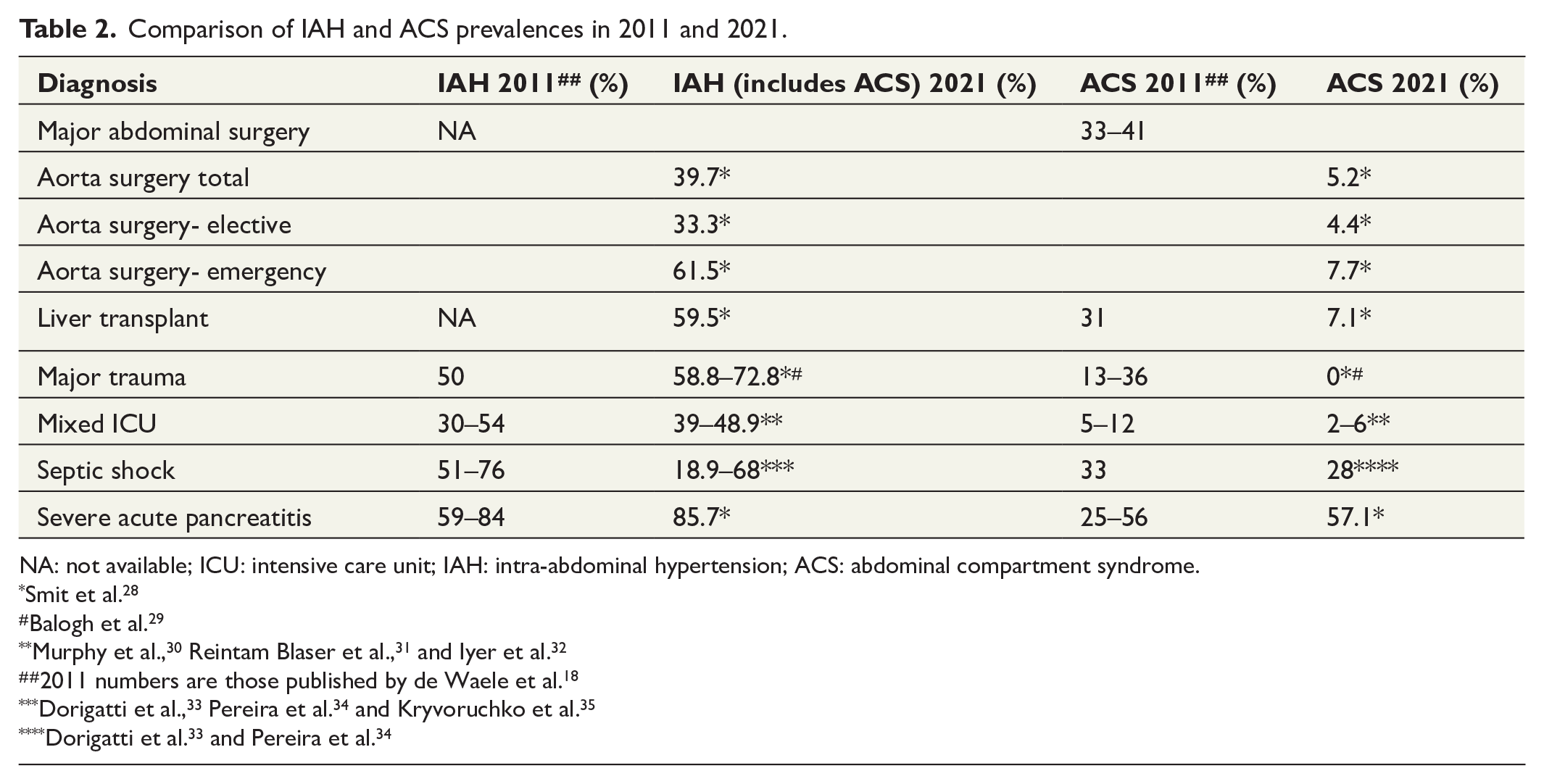
There are substantial differences in incidence and prevalence within subgroups of patients, for example, medical, surgical, and trauma patients. Reported incidences of IAH and ACS by case mix were summarized by De Waele in 2011 (Table 2). 18 These data were based on the limited number of publications available at the time, and varying definitions of IAH and ACS were used. In more recent studies, IAH and ACS have been defined according to the WSACS. These studies are compared to the incidences reported by De Waele in Table 2.
Comparison of IAH and ACS prevalences in 2011 and 2021.
NA: not available; ICU: intensive care unit; IAH: intra-abdominal hypertension; ACS: abdominal compartment syndrome.
Smit et al. 28
Balogh et al. 29
2011 numbers are those published by de Waele et al. 18
Data from a mixed medical–surgical ICU where all consecutive ICU patients were included showed that ACS occurred in 3% of patients. 30 In a mixed multi-center ICU population of 491 consecutive ICU patients, ACS occurred in 6% of patients. The difference between the studies was attributed to the difference in case mix. 31
A study in 81 trauma patients showed the incidence of ACS was 0% and IAH was 75%. 29 In a study of 503 high risk ICU patients, prevalences of IAH and ACS were 33% and 3.6%, respectively. 28 Highest prevalence of ACS occurred in subgroups of patients with pancreatitis (57%), after orthotopic liver transplant (OLT) (7%), and after abdominal aorta surgery (5%).
IAH persists and may increase as critically ill patients increasingly survive initial insults. 36 Overall, ACS is decreasing (Table 2), possibly through early recognition and targeted management of IAH. 36 Other factors probably include improved peri-operative and intensive care management, including restrictive peri-operative fluid management and meticulous hemostasis. However, ACS continues to be a major problem in specific subgroups, for example, in the obese and in pancreatitis. 28
Outcome
IAH is an independent predictor for mortality.1,2,30 ICU mortality is significantly higher compared to those who never developed IAH (6%–30% versus 1%–11%), 30 and the grade of IAH is inversely related to outcome. 31 The 90-day mortality of ACS remains high at 39%–76% but shows a large variation.28,31 Besides patient selection, variation may well reflect differences in patient management. In addition to increased mortality, IAH and ACS are associated with increased length of ICU stay and increased severity of organ failure, particularly renal and respiratory failure.28,37 This is illustrated by an increased requirement for renal replacement therapy and an increased duration of mechanical ventilation. 28 Timing of treatment is important in preventing progression of AKI and development of chronic kidney disease. 18
Risk factors
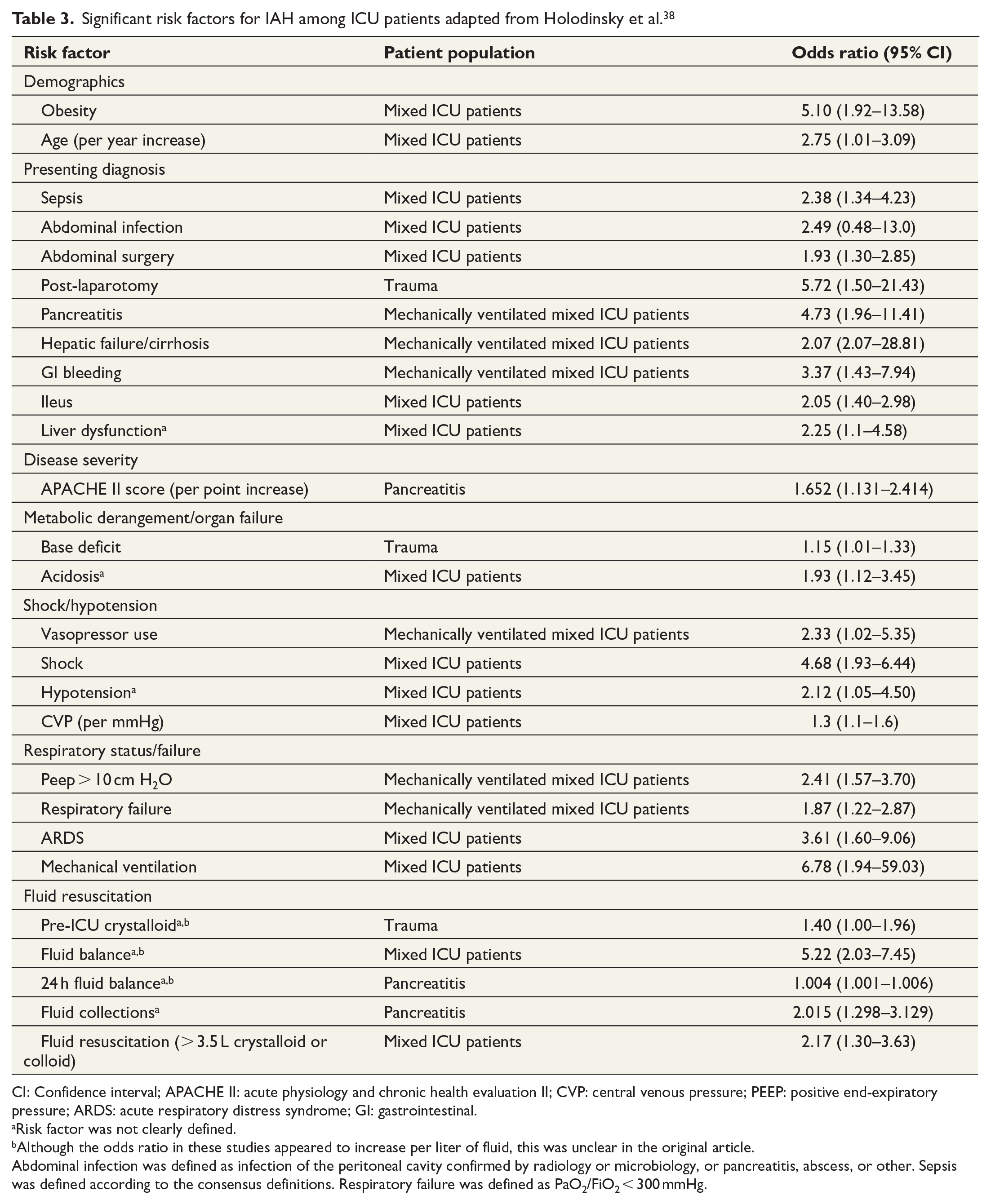
A number of studies of IAH and ACS risk factors have been published, but interpretation is difficult due to significant heterogeneity between the studies. A systematic review and meta-analysis, including 14 studies and 2500 critically ill adults, were conducted in 2013 (Tables 3 and 4). 38 A presenting diagnosis of sepsis, abdominal infection, abdominal surgery, hepatic failure/cirrhosis, gastrointestinal (GI) bleeding, and ileus were the risk factors for the development of IAH among ICU patients (Table 3). Risk factors for ACS were studied in trauma and severe acute pancreatitis (SAP) patients (Table 4). Although several risk factors transcended across presenting patient diagnoses (e.g. large-volume crystalloid resuscitation and the presence of shock/hypotension), many were specific to the type of patient population under study. The authors stated that their findings were partially limited by clinical heterogeneity and the quality of statistical analyses conducted in the included studies and therefore these risk factors should be considered candidate evidence-based risk factors until formally evaluated in a prospective multi-center observational study. However, thus far this is the best available evidence.
Significant risk factors for IAH among ICU patients adapted from Holodinsky et al. 38
CI: Confidence interval; APACHE II: acute physiology and chronic health evaluation II; CVP: central venous pressure; PEEP: positive end-expiratory pressure; ARDS: acute respiratory distress syndrome; GI: gastrointestinal.
Risk factor was not clearly defined.
Although the odds ratio in these studies appeared to increase per liter of fluid, this was unclear in the original article.
Abdominal infection was defined as infection of the peritoneal cavity confirmed by radiology or microbiology, or pancreatitis, abscess, or other. Sepsis was defined according to the consensus definitions. Respiratory failure was defined as PaO2/FiO2 < 300 mmHg.
Significant risk factors for ACS among ICU patients adapted from Holodinsky et al. 38
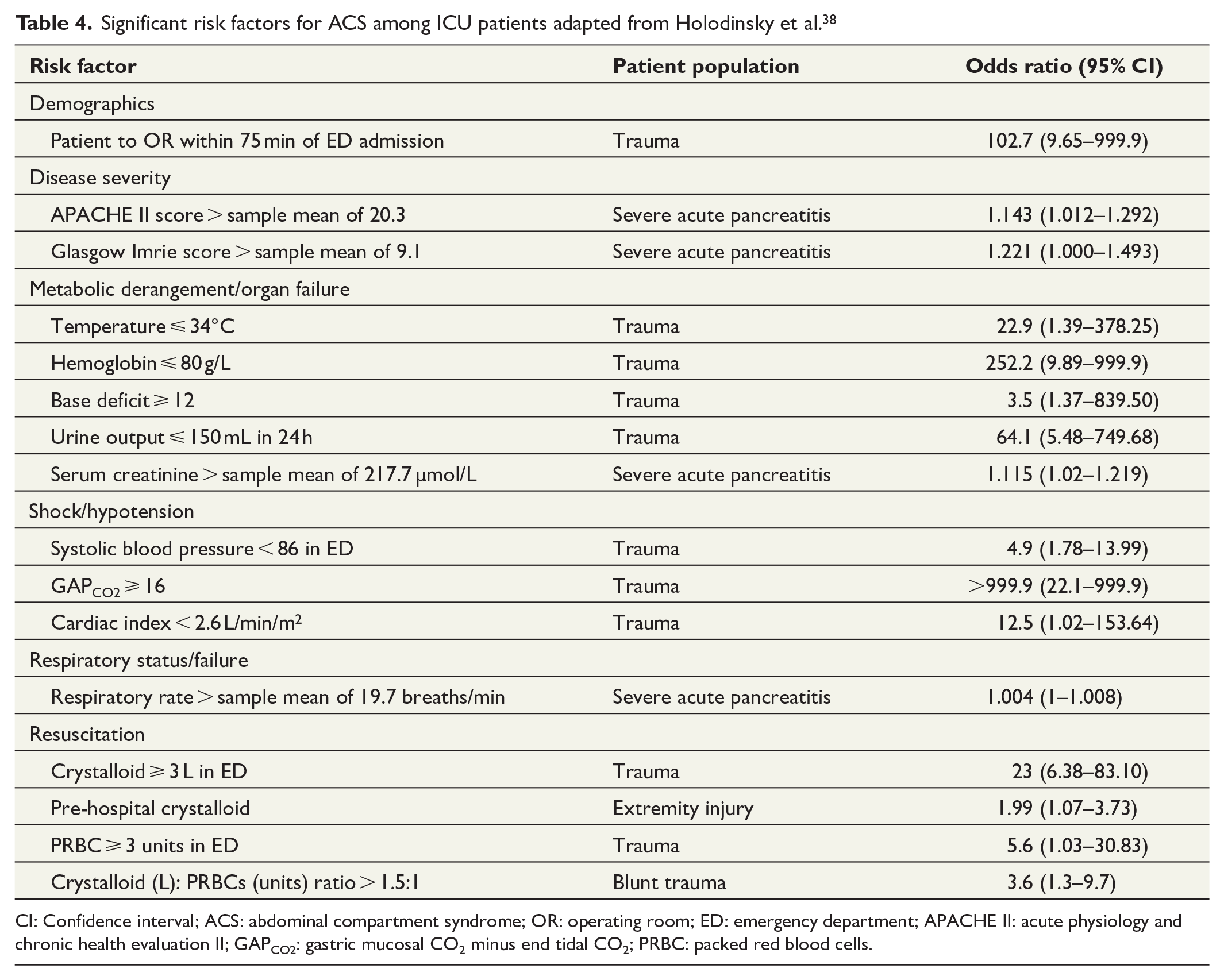
CI: Confidence interval; ACS: abdominal compartment syndrome; OR: operating room; ED: emergency department; APACHE II: acute physiology and chronic health evaluation II; GAPCO2: gastric mucosal CO2 minus end tidal CO2; PRBC: packed red blood cells.
Management
IAH has consistently been associated with morbidity and mortality in observational studies. However, it remains uncertain whether treating or preventing this condition improves patient outcomes. 4
To date, there have been no intervention studies to answer the important question which patient needs non-operative management or surgical management of ACS. Therefore, the position and timing of decompression laparotomy in ACS are still unknown.39,40
Non-operative management of ACS
In a 2008 study of 83 patients admitted to the ICU, 10 patients developed ACS (12%). These patients all received nasogastric suctioning, rectal decompression, diuretics, deep sedation, and neuromuscular blockade. None underwent surgical decompression. Two patients (20%) survived with non-operative ACS management. 37
Non-operative management has advanced since 2008, and there are a number of effective non-operative interventions that may be performed to reduce IAP and thereby decrease the need for surgical decompression.4,41
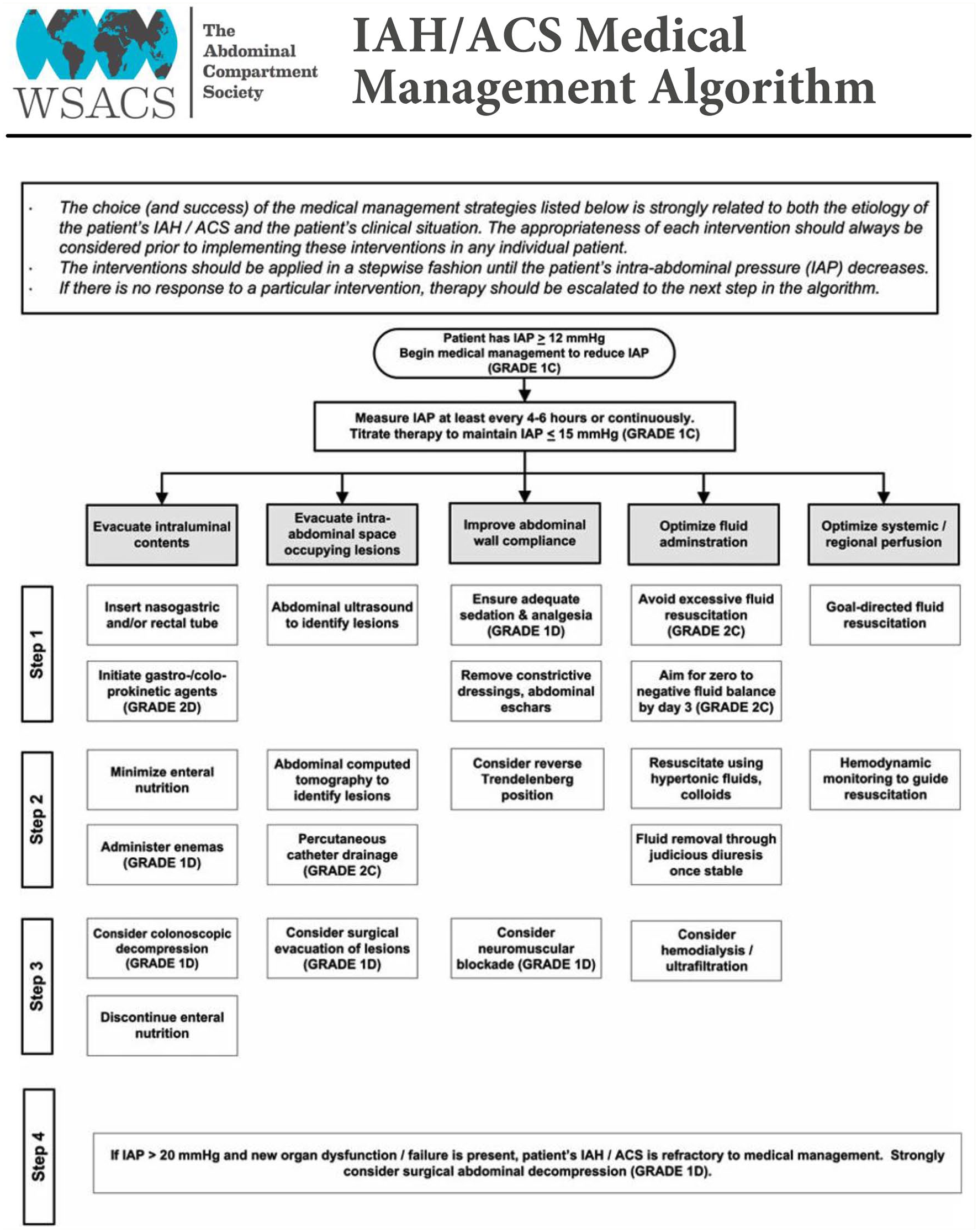
In 2013, the WSACS published an IAH/ACS medical management algorithm consisting of five therapeutic interventions 4 (Table 5). However, overall quality of evidence available to guide the development of recommendations was low. 4 Small trials have investigated single measures and its effect on IAP, not on outcome.
IAH/ACS medical management algorithm. 4

Adapted with permission according to open access CC BY License 4.0 from Kirkpatrick et al. (https://link.springer.com/article/10.1007/s00134-013-2906-z).
In 2007, the first prospective trial of neuromuscular blockade in IAH management, reporting temporary reductions in IAP in 9 out of 10 patients was published. 42 In a prospective, blinded trial of epidural versus intravenous postoperative pain therapy, the two therapies were demonstrated to have equivalent efficacy for reducing IAP. 43 Drainage of any intra-abdominal fluid collections identified is a safe and effective technique for reducing IAP. 44 Removal of even a few hundred milliliters of fluid can result in a marked decrease in IAP in the presence of significant IAH and loss of Cab. 41
Surgical decompression
“Open the abdomen and keep it open” was the adage used in the early 2000s when surgical management was considered the only therapeutic option available for the patient with ACS. 45 Currently there is still no consensus on indications for decompression, both in terms of IAP values and of timing, 46 but abdominal decompression can be life-saving when ACS is refractory to non-operative treatment and should then be performed expeditiously. 41
However, the evidence supporting decompressive laparotomy is limited, as only small non-randomized trials have been performed. In 2010, a study protocol of a multicentre, randomized, controlled study investigating decompressive laparotomy versus percutaneous puncture with the placement of abdominal catheter in patients with ACS during acute pancreatitis was published. 47 However, the results of this study are still pending. In a recent systematic review and meta-analysis, 15 articles and 286 patients (including children) were included. 48 Decompressive laparotomy resulted in a significantly lower IAP and improvement in hemodynamic, respiratory, and renal parameters. Mortality after decompressive laparotomy remained high at 49.7% in adult patients. There was a correlation between the timing of decompressive laparotomy and mortality. The results of this meta-analysis confirm the recommendation that decompressive laparotomy should be considered when medical options fail, even though it remains unclear which patients would benefit most from decompressive laparotomy.
Among the surgical techniques used to decompress the abdomen are median laparotomy and bilateral transverse subcostal laparotomy. 49 A third, less invasive option is the subcutaneous linea alba fasciotomy (SLAF), where three short, horizontal skin incisions are made. 50 This technique is effective in only 50%–70% of patients. 49 Few data support the use of other techniques than median laparotomy. 39
Open abdomen
After surgical decompression, the fascial edges of the abdomen are not approximated (laparostomy). The abdominal contents are exposed and protected with a temporary cover. 39 Since the open abdomen may require multiple reoperations and there is a risk of significant complications, such as entero-atmospheric fistulas, loss of abdominal wall domain, and large hernias, 51 open abdomen management should immediately follow surgical decompression. The objectives of open abdomen management are to prevent complications and to achieve the earliest possible delayed fascial closure. 52
Negative pressure wound therapy (NPWT) or vacuum therapy is the procedure currently recommended in the treatment of the open abdomen. 53 Roberts et al. performed a systematic review to determine the comparative efficacy and safety of NPWT versus alternate temporary abdominal closure techniques in critically ill adults with open abdominal wounds. 54 In this study of 1018 adults, two randomized controlled trials and nine cohort studies (three prospective and six retrospective) were included. These were all controlled cohort studies. Methodological quality of the prospective studies was moderate and low for most of the retrospective studies. The limited data suggested that NPWT may be linked with improved outcomes versus alternate temporary closing techniques. Cirocchi et al. performed a review and meta-analysis on the effectiveness of NPWT in patients treated with open abdomen. 55 In this study, data of controlled and uncontrolled studies were pooled, 1225 patients were analyzed, and 723 (59%) underwent NPWT. Comparing the NPWT group and the group without NPWT, there was no statistically significant difference in fascial closure, postoperative 30 day overall morbidity, postoperative entero-atmospheric fistulas rate (2.1% versus 5.8%, p = 0.57), postoperative bleeding rate, and postoperative abdominal abscess rate. There were significant differences in the postoperative mortality rate (28.5% versus 41.4%, p = 0.03) and in the length of stay in the ICU. The data should be interpreted with substantial caution given the non-randomized nature of many of the included studies, the small sample sizes, and high variability between studies. Furthermore, the pooling of controlled and uncontrolled data is a confounding factor and limits the validity of the conclusions further. Both studies highlight the need for randomized controlled trials with homogeneous inclusion criteria to assess the use of NPWT for the management of open abdomen.
Kirkpatrick et al. performed a single-center randomized controlled trial where 45 adults with abdominal injury or intra-abdominal sepsis were randomly allocated to ABThera (n = 23) or Barker Vacuum pack (n = 22). 56 The cumulative incidence of primary fascial closure at 90 days was similar between groups. However, 90-day mortality was improved in the ABThera group (p = 0.04). This survival difference did not seem to be mediated by an improvement in peritoneal fluid drainage, fascial closure rates, or markers of systemic inflammation.
A laparostomy registry entitled Open Abdomen Route by European Registry of Abdominal Wall Hernias has been implemented in 2015 by CAMIN (surgical working group for military and emergency surgery) of DGAV (German Society for General and Visceral Surgery) 52 ACS was the reason for open abdomen treatment in 18% of the 82 patients. Average duration of open abdomen treatment was 19 days (range 2–120 days). Small bowel fistula occurred in 8 of the 82 patients (9.8%); in 2 of these patients occurred after the treatment for ACS.
In the International Register of Open Abdomen (IROA), 649 adult patients with open abdomen were registered, 2.9% after ACS. Entero-atmospheric fistulas developed in 8.9% of patients. There was a linear correlation between the days of open abdomen and time to nutrition with development of entero-atmospheric fistulas. 57
Treatment of entero-atmospheric fistulas is very complex; therefore, this complication should be prevented whenever possible after decompressive laparotomy: by performing fascial closure at the earliest possible opportunity. 51
Future steps
First, recent reports indicate that a new disease, such as COVID-19, and the increasing application of extra-corporeal life support in the ICU, may impact the prevalence of IAH and its complications.58,59 Further study will follow, but in the meantime IAP monitoring may be considered in these new clinical contexts.
Second, IAP monitoring should be easier, accessible, and preferably continuous. Ball et al. noted in 2006 that IAP measurement was routine in the critically ill in their hospital and that IAP was considered the “fifth vital sign”. 60 However, this is not standard care in every ICU. When IAP measurements become easier and less time-consuming, IAP trends may be easily followed and IAP may indeed become a vital sign. This will be an opportunity to collect an abundance of data, allowing us to learn more about the dynamics of IAP in different clinical settings or during interventions and individual differences. The development and distribution of a cost-effective and continuous IAP monitoring device to be used in every ICU should be followed closely.
Third, an update in the 2013 WSACS guidelines should be considered. A future guideline should focus more on the clinical context accompanying IAP increase than on specific numbers, bearing in mind that pathological IAP is a continuum and the clinical context ultimately determines the need for therapy. Furthermore, the guideline should take into account that it is not always possible or practical to measure IAP in the supine position and allow for IAP measurement in different positions. It is standard of care in the ICU to elevate the head-of-bed to 30° to prevent ventilator-associated pneumonia. 61 Since IAP increases when the head-of-bed is elevated, inaccuracies may occur.9,10 Therefore, we propose a simple reference table correcting IAP for head-of-bed position (Table 6). This reference table should be validated in a prospective study where IAP measurements are repeated in different positions per patient.
Proposed reference table for correction of intra-abdominal pressure in different head-of-bed positions.
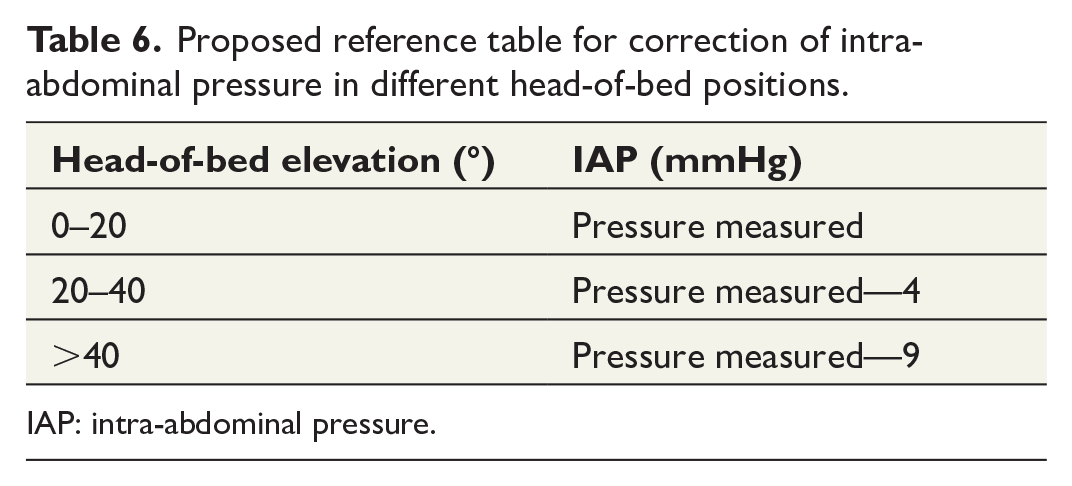
IAP: intra-abdominal pressure.
Finally, the consequence of the heterogeneous patient population at risk for ACS in combination with the low incidence of ACS is that intervention studies can only be performed in large multi-center studies. A multi-center study which randomizes between early decompression surgery with open abdomen management and bundles of non-operative treatment in ACS patients might answer some of the many remaining important clinical questions. Furthermore, the effect of treatment of IAH on outcome needs further study. IAH has consistently been associated with morbidity and mortality in observational studies, but it is uncertain whether treating or preventing this condition improves patient outcomes. A large multi-center trial randomizing patients at risk of IAH between bundles of non-operative management in IAH (but not ACS) versus no treatment of IAH should be performed as soon as possible. We recommend that this study is performed in a homogeneous patient population with a relatively high prevalence of IAH, for example, in patients with SAP.
Conclusion
Pathological IAP is a continuum ranging from mild IAP elevations without clinically significant adverse effects to substantial increases in IAP with serious consequences to all organ systems in the body. In ACS, increased pressure within the abdominal compartment leads to decreased blood flow and ultimately to multi-organ failure. IAP monitoring should be performed in all patients at risk of IAH. Although continuous IAP monitoring is feasible, this is currently not standard practice. There are a number of effective non-operative medical interventions that may be performed early in the patient’s course to reduce IAP and decrease the need for surgical decompression. Abdominal decompression can be life-saving when ACS is refractory to non-operative treatment and should be performed expeditiously. The objectives of open abdomen management are to prevent fistula and to achieve delayed fascial closure at the earliest possible time. There is still a lot to learn and change. First and foremost, the 2013 WSACS guidelines should be updated and multicentre studies should evaluate the effect of IAH treatment on patient outcome.
Footnotes
Author contributions
M.S. reviewed the article; M.S., M.v.M., and J.G.Z. interpreted the article; M.S. wrote several drafts of the article, including the final version; M.v.M. and J.G.Z. revised the article; J.G.Z. participated in conceptualization of the article; all authors read and approved the final article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Institutional funding only.