Abstract
Background and objective:
Surgical drains are widely utilized in hepatopancreaticobiliary surgery to prevent intra-abdominal collections and identify postoperative complications. Surgical drain monitoring ranges from simple-output measurements to specific analysis for constituents such as amylase. This systematic review aimed to determine whether surgical drain monitoring can detect postoperative complications and impact on patient outcomes.
Methods:
A systematic review was performed, and the following databases searched between 02/03/20 and 26/04/20: MEDLINE, EMBASE, The Cochrane Library, and Clinicaltrials.gov. All studies describing surgical drain monitoring of output and content in adult patients undergoing hepatopancreaticobiliary surgery were considered. Other invasive methods of intra-abdominal sampling were excluded.
Results:
The search returned 403 articles. Following abstract review, 390 were excluded and 13 articles were included for full review. The studies were classified according to speciality and featured 11 pancreatic surgery and 2 hepatobiliary surgery studies with a total sample of 3262 patients. Postoperative monitoring of drain amylase detected pancreatic fistula formation and drain bilirubin testing facilitated bile leak detection. Both methods enabled early drain removal. Improved patient outcomes were observed through decreased incidence of postoperative complications (pancreatic fistulas, intra-abdominal infections, and surgical-site infections), length of stay, and mortality rate. Isolated monitoring of drain output did not confer any clinical benefits.
Conclusions:
Surgical drain monitoring has advantages in the postoperative care for selected patients undergoing hepatopancreaticobiliary surgery. Enhanced surgical drain monitoring involving the testing of drain amylase and bilirubin improves the detection of complications in the immediate postoperative period.
Introduction
Rationale
The use of surgical drains is commonplace across surgical practice, with records describing their use dating back to the Hippocratic era. 1 In certain gastrointestinal surgical procedures, abdominal drains are inserted intra-operatively to remove anticipated accumulation of abdominal fluid such as peritoneal fluid, blood, bile, and pancreatic secretions, with the aim of reducing the risk of postoperative collections. 2 Another function of surgical drains is to detect the development of postoperative complications such as anastomotic leak, ureteric injury, pancreatic fistulas (PFs), intra-abdominal infection, or hemorrhage. 3 This is frequently the reasoning for their use in hepatopancreaticobiliary (HPB) surgery, in procedures such as pancreaticoduodenectomy (PD) where the risk of PF formation is high. 4
Current clinical management of drains is largely orientated around daily drain output monitoring, measuring crude volume output and describing content following visual inspection. 5 If specific clinical concerns are raised, fluid samples can be sent for formal laboratory analysis of constituents such as electrolytes, amylase, lactate, or cytokines. However, the use of these is center specific, limited by resource and access to surgical services. 6 While surgical drains can aid in the identification of complications, prolonged surgical drain use can increase the risk of surgical site infections (SSIs), cause patient discomfort, and contribute to delays in patient discharge. 7 Accurate monitoring of surgical drain output is also dependent on rigorous checks and documentation which is subjective and often open to human error. 5 Furthermore, current surgical practice involves the enhanced recovery after surgery (ERAS) approach which advocates for restrictive use of surgical drains to support early mobilization and improve patient comfort. 8 It is therefore paramount to establish the benefits of surgical drain use.
The literature is replete with studies that have focused on prophylactic abdominal drainage inserted at the time of gastrointestinal surgery. 9 Comparisons have focused on the benefits of drainage versus no drainage with little focus on the analysis of surgical drain output monitoring. Testing drain constituents has the potential to facilitate early detection and treatment of postoperative complications. For example, gastrointestinal anastomotic leaks alone cause significant morbidity and mortality. 10 Prompt identification might be facilitated by monitoring surgical drain output resulting in expedient intervention and improve patient outcomes. The aim of this systematic review was to determine if monitoring drain volume and content confers any benefit on the detection of postoperative complications and improves patient outcomes in HPB surgery.
Objectives
To identify what current surgical drain output monitoring methods exist following HPB surgery.
To identify postoperative complications that can be detected through the use of surgical drain output monitoring and to assess the impact on patient outcomes.
Methods
Protocol and registration
The protocol for this review was guided by the PRISMA and AMSTAR 2 guidelines and was registered with PROSPERO (registration number CRD42020170845).11,12
Eligibility criteria
Study type
Case series, cohort, and case–control studies, randomized control trials, and systematic reviews were included in this study.
Participants
The population included adult patients over the age of 18 who had an abdominal surgical drain inserted following HPB surgery.
Intervention
Studies that described monitoring of abdominal surgical drain output and constituents in patients undergoing HPB surgery were included.
Exclusion criteria
All news, editorials, comments, letters, study protocols, non-English language articles, and technical notes were excluded. Studies describing pediatric cohorts (patients under the age of 18) or involving other invasive methods of intra-abdominal sampling, such as ascitic taps or radiologically guided drains, were excluded.
Outcomes measured
The variables recorded were categorized by the reviewers into either patient related or surgical drain related.
Patient outcomes:
Postoperative complication.
Mortality.
Time to diagnosis (of complication).
SSI Rates.
Length of stay (LOS).
Surgical drain monitoring outcomes:
Drain output volume measured postoperatively
Drain output content analyzed for constituents such as amylase.
The secondary outcomes included in this study focused on the postoperative complications identified using surgical drain monitoring.
Information sources
The studies published prior to April 2020 were considered for inclusion after searching the following sources: MEDLINE, EMBASE, The Cochrane Library, and Clinicaltrials.gov. “My NCBI” (National Center for Biotechnology Information) email alert service provided by PubMed was utilized to identify new published studies during the review period. Completed but unpublished trials with results published online were searched for using the Clinicaltrials.gov website.
Referenced studies within identified literature were also considered for inclusion in the review. All unpublished and incomplete data from the gray literature such as news, editorials, comments, letters, study protocols, or technical notes were excluded from the review.
Search
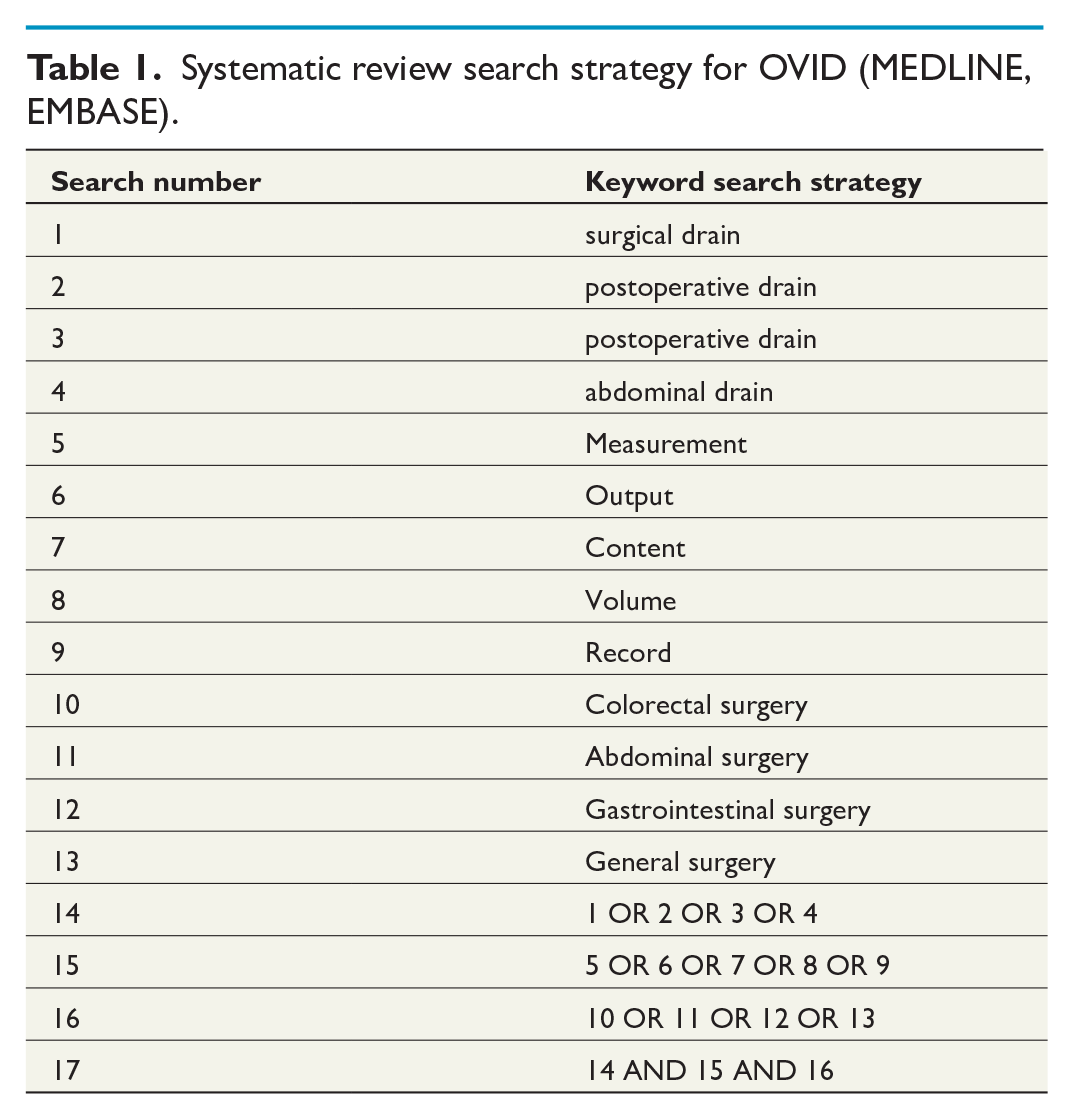
The systematic search was performed by two independent investigators (M.K. and W.B.). The databases were searched using the strategy outlined in Table 1. All studies published between 30 March 2000 and 30 March 2020 were considered for eligibility. The last search was run on the 26th April 2020.
Systematic review search strategy for OVID (MEDLINE, EMBASE).
Study selection
Studies were selected using a staged review of titles and abstracts, followed by full-text review. M.K. independently retrieved article abstracts with W.B. cross-checking. The search-identified abstracts and those from additional sources were screened independently by M.K. and W.B. to identify studies that potentially met the inclusion criteria outlined earlier. Discordance was settled by arbitration by the senior author J.B.
Data collection process
Two independent investigators (M.K. and W.B.) extracted data using a standardized data collection form. The form was first tested using sample articles and adjusted to improve efficiency and accuracy of data collection. As with study assessment for inclusion, the third investigator (J.B.) was consulted regarding any discrepancies in data extraction.
Data items
The two investigators carried out independent data collection of study demographics, interventions, characteristics, and outcomes using the following data fields:
Demographics: population studied and type of surgery involved (grouped by surgical specialty).
Interventions: type of surgical drain output monitoring implemented.
Study characteristics: study design, data collection period, number of subjects, randomization, blinding, allocation concealment, funding, and country of origin.
Outcomes: patient and surgical drain related.
Risk of bias in individual studies
Due to the heterogeneity of studies identified, it was deemed inappropriate to utilize a standard scoring tool for risk of bias and we attempted to pragmatically include all eligible studies to enrich the narrative synthesis.
Summary measures
The treatment effect was displayed in odds ratios (ORs) or risk ratios (RRs) with 95% confidence intervals (CIs) for dichotomous data. Continuous data treatment effects were expressed as mean differences with 95% CIs.
Synthesis of results
A narrative synthesis approach was chosen as this allowed the analysis of a broad range of studies in a meaningful manner. We did not perform a meta-analysis of effect estimates. The narrative synthesis of quantitative data was conducted in line with the Guidance on the Conduct of Narrative Synthesis in Systematic Reviews from the Economic and Social Research Council. 13 Included studies were tabulated and grouped according to the general surgical subspecialties managing the conditions described in each study population. The evidence provided by the included literature was then synthesized to provide a structured narrative that was relevant to the research question.
Results
Study selection
The search strategy returned 403 articles following duplication removal. Following abstract screening, 390 articles were excluded. The main reasons for exclusion were study interventions not including surgical drains, studies concerning surgical specialties other than HPB surgery and study focus on comparison of drain versus no drain with no monitoring of drain output or constituents. The remaining 13 articles were included for a full review using the prepared data collection spreadsheet. Two systematic reviews were identified which were used to identify further studies through reference searching.14,15 This selection process is outlined in the PRISMA flowchart in Fig. 1.
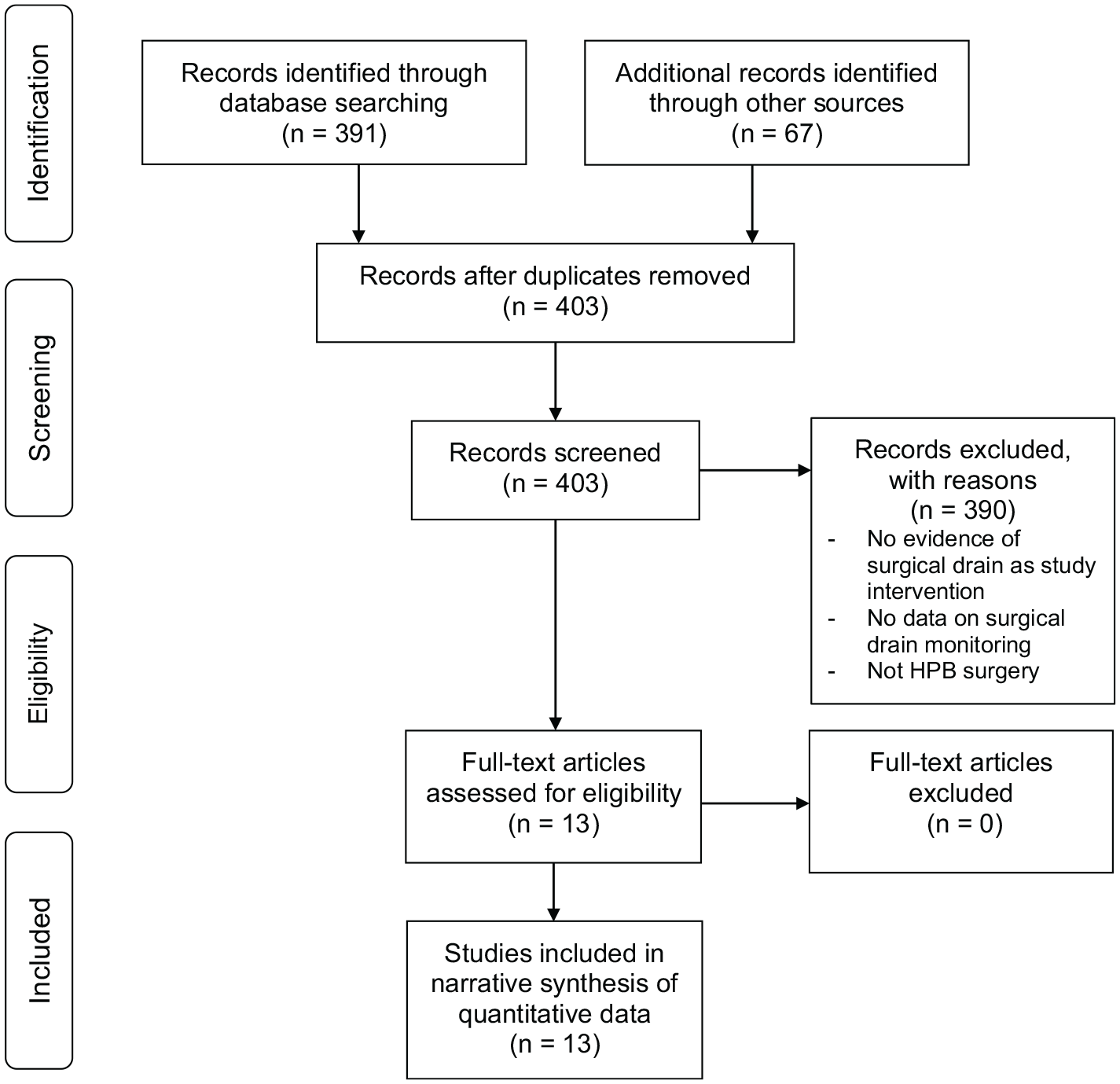
PRIMSA flow diagram for study selection. Adapted from Liberati et al. 16
Individual study characteristics
Studies were grouped according to surgical subspecialty: pancreatic surgery and hepatobiliary surgery. These results are summarized in Table 2.
Individual study results grouped to pancreatic surgery and hepatobiliary surgery.
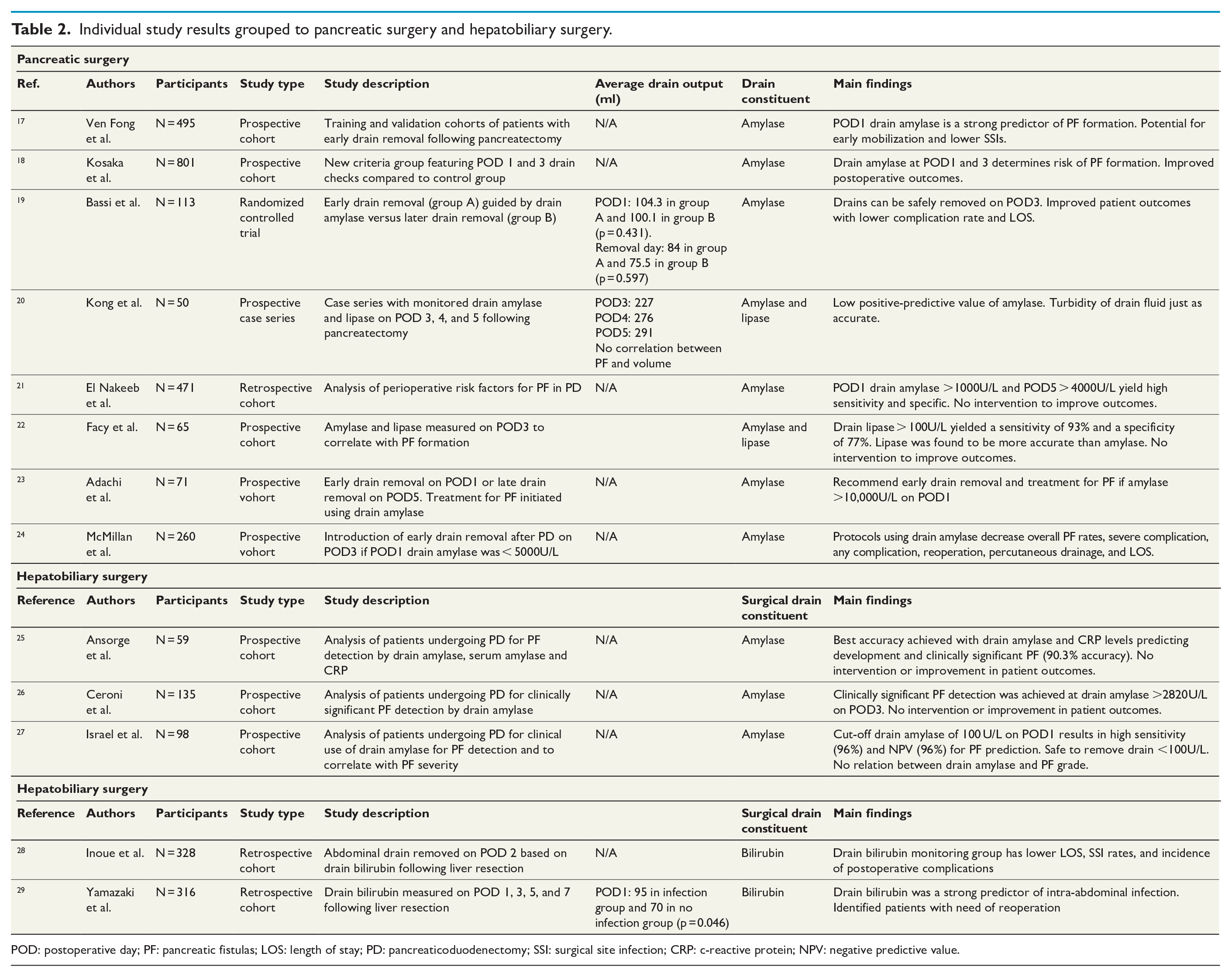
POD: postoperative day; PF: pancreatic fistulas; LOS: length of stay; PD: pancreaticoduodenectomy; SSI: surgical site infection; CRP: c-reactive protein; NPV: negative predictive value.
Pancreatic surgery
Eleven studies featured surgical drain monitoring in pancreatic surgery. The majority of articles aimed at detecting PFs and establishing a safe time for drain removal. Ven Fong et al. 17 carried out a prospective cohort study of 495 patients undergoing PD and found that a postoperative day (POD) 1 drain amylase <600 U/L was a strong predictor for the absence of PF formation. Although no improved patient outcomes were shown, the study suggested that early drain removal in aforementioned conditions would benefit patients through early mobilization and lower risk of SSIs. The authors are currently validating the early drain removal protocol with the intention to implement it into their clinical practice. Further cohort studies on patient undergoing PD have validated the use of amylase as a predictor of PF formation. El Nakeeb et al. 21 identified POD 1 drain amylase >1000 U/L and POD 5 > 4000 U/L as a highly sensitive and specific marker for PF formation (ROC 0.797, p < 0.0001, ROC 0.96, p < 0.0001). This was also confirmed by Ceroni et al. 26 who found that clinically significant PF detection was achieved at drain amylase >2820 U/L on POD 3. Israel et al. 27 investigated the use of drain amylase as a negative predictor for PF formation and found that it was safe to remove drains when drain amylase was below 100 U/L on POD 1. The summarized studies did not investigate the impact of applying cut-off levels and interventions on patient outcomes.
Two other cohort studies carried out similar analysis with the use of additional markers. Ansorge et al. 25 validated the use of drain amylase in a cohort of patients undergoing PD with the addition of C-reactive protein (CRP). The authors discovered that the best accuracy was achieved with POD 1 drain amylase > 1322 U/L, POD 2 drain amylase > 314 U/L, and POD 3 CRP > 202 mg/L in predicting development and clinically significant PF (90.3% accuracy). Facy et al. 22 utilized drain lipase and found that it was more accurate than drain amylase on POD3 with levels less than 100 U/L yielding a sensitivity of 93% and a specificity of 77%. However, these studies also did not evaluate the use of this as interventions to improve patient outcomes.
McMillan et al. investigated the use of drain amylase in relation to patient outcomes. In a cohort of patients undergoing PD, they removed drains on POD 3 if POD 1 drain amylase was <5000 U/L. They concluded that this cut-off level identified patients who benefit from early drain removal. Patients who were treated using drain amylase monitoring had decreased overall rates of PF formation, severe complication, any complication, reoperation, percutaneous drainage and length of stay (LOS). 24 Kosaka et al. conducted a similar cohort study involving 801 patients. The patient cohort was split into a control group with drain amylase checked on POD 1 and a sequentially checked group which featured drain amylase checks on POD 1 and 3. 18 Drains were removed in the sequentially checked group on POD 3 if drain amylase was <5000 U/L on POD 1 and <3000 U/L on POD 3. The sequentially checked group exhibited lower incidence of intra-abdominal abscesses (p < 0.001) and lower risk of PF formation (OR = 0.601, p < 0.05) when compared to the control group. In addition, a decreased morality rate was observed, and the authors concluded that amylase measurement on POD 1 and 3 determines the risk of PF formation and influences patient outcomes. Interventions based on drain amylase were also researched by Adachi et al., who carried out a cohort study of patients undergoing distal pancreatectomy. Their protocol involved early drain removal with drain amylase levels >10,000 U/L prompting treatment with gabexate mesilate, octreotide, and carbapenem antibiotics to prevent PF formation. Patients who were treated with the outlined triple therapy did not require further management and the authors concluded that the early drain removal protocol was safe to use. 23 The timing of drain removal was also studied by Bassi et al. 19 in a randomized controlled trial (RCT) on 113 patients undergoing PD or distal pancreatectomy. Patients were randomized to early or late drain removal groups, with the former being guided by drain amylase levels. A higher rate of all postoperative complications was observed in the late drain removal group and timing of drain removal was an independent risk factor for PF formation. The process linking the timing of drain removal and PF formation was explained by pressure erosion from intraluminal migration of surgical drains that may occur at the site of a pre-existing anastomotic leak. The study also found that drain output volume did not differ between the two groups (p = 0.431 on POD1 and p = 0.597 on day of removal). The early drain removal group showed that drains can be safely removed on POD 3 using amylase drain levels which avoid major abdominal complications.
The remaining study related to pancreatic surgery did not show such clear evidence for surgical drain monitoring. A cohort study in Australia measured amylase drain levels on POD 3, 4, and 5 with the aim of identifying PF formation. 20 The positive predictive value of amylase levels for PF formation was 50%, and the results showed that observing the turbidity of drain fluid was an equal to drain amylase monitoring in identifying postoperative complications. There was no correlation between drain output volume and risk of PF formation.
Hepatobiliary surgery
The literature related to hepatobiliary surgery is directed at drain bilirubin measurements. A retrospective cohort study analyzed patients undergoing liver resections for hepatic tumors. 28 The study implemented drain removal on POD 2 if drain bilirubin was one-third the serum levels. The group with drains removed on POD 2 exhibited significantly lower rates of SSIs, postoperative complications, and LOS. The monitoring of drain bilirubin and its correlations with drain removal therefore improved patient outcomes. However, it is unclear from this study if this was due to surgical drain monitoring or as a result of uncomplicated surgery with lower drain bilirubin levels. Yamazaki et al. 29 also studied drain bilirubin monitoring following liver resection. Their study compared patients who developed postoperative infections defined with positive bacteriological cultures and also included analysis of drain volumes in addition to bilirubin monitoring. Drain bilirubin over 3 mg/dL was a strong predictor of infection (OR = 15.11, p < 0.001) and successfully identified patients who required reoperation. Drain volumes were less useful and only found to be significantly increased in the infection group on POD 1 (p = 0.046), with the median drainage volumes being less than 100 mL throughout the study. The authors concluded a “3 × 3 rule” involving drain removal on POD 3 if drain bilirubin was <3 mg/dL and bacteriological cultures remained negative. This enables early identification of bile leak (which in turn is associated with a high risk of infection) and facilitates early drain removal, preventing the development of retrograde infection. The study therefore describes a safe rule that identifies patients at risk of postoperative infection; however, the direct effect on patient outcomes was not reported.
Discussion
Summary of evidence
Improved patient outcomes using surgical drain monitoring
In specific cases, surgical drain monitoring may improve patient outcomes. This is suggested in the use of drain amylase testing in pancreatic surgery. The ability to predict PF formation is crucial in the postoperative period as mortality secondary to PF has been reported as high as 25%. 30 The existing evidence described in this review focuses on varying drain amylase measurements utilizing different protocols. Future research should involve increasing the frequency or performing continuous amylase testing. Promising results have also been observed in hepatobiliary surgery with drain bilirubin monitoring. Intra-abdominal infections following liver resections have a negative impact on patient survival. 31 The potential for timely bilirubin measurement to safely identify bile leak without the introduction of retrograde infection is a novel approach and this practise should be validated through further research.
Feasibility of enhanced surgical drain monitoring
Surgical drain amylase and bilirubin have been identified as useful markers to guide postoperative care.17–19,28,29 The findings summarized in this study suggest that their use in enhanced surgical drain monitoring compared to standard procedures on the ward may be useful, although the process is potentially compromised by the accuracy of daily output recording. Additional limitations to this may include the extra cost and processing time for non-standard or established tests. Jiang et al. 32 showed that amylase screening–based methods can facilitate early discharge and subsequently decrease overall healthcare costs through a decrease in LOS and complication rates. Currently, there are no studies on the cost-effectiveness of drain bilirubin and frequent testing as part of postoperative surgical monitoring is likely to prove expensive. The earlier detection of potentially expensive complications must be cost-saving and improve outcomes enough to offset the cost of extra diagnostic tests. Enhanced surgical drain monitoring will need to be achieved through a low-cost, accurate, and continuous monitoring system. Implementation of low-cost biosensors to surgical drain systems that detect amylase or bilirubin may enable wider use of surgical drain monitoring in the future of HPB surgery.
Maximizing the advantages of surgical drains
Multiple studies report increased rates of wound infections in patients who had an abdominal drain inserted following gastrointestinal surgery.33–36 It is therefore paramount to appropriately select the patients most likely to benefit from the potential advantages of surgical drain use to balance the risks associated with wound infections. Enhancing surgical drain monitoring could further increase the benefit and utility of surgical drains. Novel electronic surgical drain devices have the ability to record surgical drain outputs in real-time. 37 These devices could be coupled with biosensors that can detect drain amylase or bilirubin at increased intervals enabling further guidance on surgical drain care and detection of postoperative complications. This could have implications on patient outcomes in HPB surgery and strengthen the argument for appropriate surgical drain use.
Strengths and limitations
Strengths of this study are recognized. A narrative synthesis of the literature allowed the inclusion of a broad range of study types. This was important, given the high degree of study heterogeneity and meant that a broad research question could be investigated. Via this synthesis, we aimed to generate hypotheses and stimulate areas of research to improve the functionality and clinical utility of surgical drains. This study provides a foundation on which to build this clinically significant area of research. Limitations are also recognized. The heterogeneity of the included literature prevented the conduct of a meta-analysis. Even in specific topics such as PF detection, a large variation on drain amylase monitoring and measurements was observed. Our study is therefore unable to give clear evidence-based guidance. Other surgical specialties that manage gastrointestinal disease with surgical drains were not included in this study and may have provided additional insights. Finally, we are unable to formally assess the risk of bias in the included studies and opted for a pragmatic inclusion of eligible papers to enrich the narrative.
Conclusion
Surgical drain monitoring has established advantages in the postoperative care of selected HPB patients. Improved patient outcomes have been demonstrated in pancreatic surgery through the use drain amylase to detect PF formation and facilitate early drain removal. In hepatobiliary surgery, drain bilirubin can be used to identify bile leak and also enable early drain removal. Isolated monitoring of drain output was found to be of minimal clinical benefit. Further recommendations for research include increased frequency of drain amylase testing, cost-effectiveness analysis of drain bilirubin testing, and innovation in surgical drain sensors. Research into electronic surgical drains with integrated biosensors is recommended as a method of achieving real-time and continuous monitoring for postoperative surgical drain care.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
