Abstract
Background and objective:
The aim of this study was to describe short-term changes in morbidity and mortality associated with the implementation of screening for colorectal cancer in Denmark.
Methods:
Prospective cohort study with inclusion of all patients aged 50–75 years treated for colorectal cancer between 1 March 2014 and 31 December 2015 in Denmark. Adjusted hazard ratios were calculated for 30 and 90 days mortality using Cox Regression. We made two adjusted models—a “basic” adjusted for screening status, sex, age, smoking, alcohol consumption, and cancer type and an “advanced” that also included body mass index and American society of Anesthesiologists score in analyses. Relative risks were calculated for postoperative surgical and medical complications.
Results:
In total, 5348 patients were included. In the “basic model,” adjusted risk of 30 and 90 days total mortality was reduced in the screen-detected group (p < 0.01, HR = 0.43, CI = 0.24–0.76) and (p < 0.01, HR = 0.45, CI = 0.30–0.69). In the “advanced model,” only 90 days total mortality was significantly reduced in the screen-detected group (p = 0.01, HR 0.59, CI = 0.39–0.90). No significant changes were found with regard to surgical and medical complications, respectively, (p = 0.05 (CI = 0.76–1.00) and p = 0.47(CI = 0.74–1.15)).
Conclusions:
This nationwide study showed that screening for colorectal cancer was associated with a lower 90 days total mortality although no significant improvements were seen with regard to morbidity.
Introduction
Colorectal cancer (CRC) is a common cancer with more than 1.4 million new cases worldwide each year and nearly 700,000 dying of the disease every year. 1 CRC develops through the adenoma-carcinoma sequence and fulfills the World Health Organization (WHO) screening criteria.2,3 In CRC, screening is known to reduce the disease-specific mortality with 16% using the guaiac fecal occult blood test (gFOBT) 4 possibly due to early detection of cases, removal of benign adenomas, and a reduced need for emergency surgery.5,6 Little is known regarding and mortality following the implementation of CRC screening. However, screening could lead to improved outcomes on both.7,8 In Denmark, screening for CRC was implemented nationwide on 1 March 2014 and all Danes aged 50–75 years are invited. The cut-off value for the fecal immune test (FIT) is 100 µg hemoglobin/L. Following a positive FIT test, patients are offered fast-track diagnostics and treatment in according to the National Cancer Plans to reduce diagnostic and therapeutic delays.9,10
Surgery is the only curative option in CRC. This includes an inborn risk of morbidity and mortality with several well-identified risk factors, that is, American Society of Anesthesiologists physical state score (ASA) and location of CRC.11,12 The optimal strategy is elective resection where the mortality rate is below 2% 10 , although endoscopic approaches can be used in selected cases. 13 Endoscopy may be advantageous due to lower complication rates. 13 In general, these studies indicate advantages associated with screening, that is, less emergency surgery and fewer complications but has methodological shortcomings, such as exclusion of emergency cases, no control group, no endoscopic therapy, and no reporting on 90 days mortality.6,8,14 –16 As previously stated, screening for CRC leads to the finding of lower disease stages 4 which could make endoscopic treatment a viable option 13 and reduce the need for resections with high morbidity rates following surgery.17 –19 It could therefore be argued that screening may also improve short-term outcomes following treatment for CRC. We decided to conduct this study to describe short-term changes in morbidity and mortality associated with the implementation of screening for CRC in Denmark.
Material and Methods
Study population
In Denmark, treatment for CRC is provided solely by the public healthcare system. All patients diagnosed with CRC between 1 March 2014 and 31 December 2015 were primarily included in this prospective cohort study. In Denmark, it is mandatory to report cases of CRC to the Danish Colorectal Cancer Database (DCCG). We included patients between 50 and 75 years of age at the time of detection as this is the age group invited to screening for CRC in Denmark. Further exclusion criteria were no CRC treatment that is due to refusal of treatment, disseminated disease leading to unresectable disease, no surgical procedure specified, unknown screening status, and other unspecified reasons. We also excluded patients who died before treatment could be initiated. Due to few cases, no analyses were performed with regard to complications after endoscopic treatment as final procedure.
Patients were extracted from the DCCG and Danish bowel screening Database (DTS). This study was approved by the Danish data protection agency (no. 2012-58-0004). No approval from ethic committees was necessary.
Patients were tracked using the Danish computerized, unique central personal registration number (CPR). Status (dead/alive) and exact date of death were ascertained through linkage of the patients’s CPR with the Danish Civil registration system. 20 All cancers were histologically confirmed and staged according to the Union for International Cancer Control (UICC) stages.
Data, extraction, variable, and management
The following data were extracted from DCCG: sex; status of screening (yes/no); UICC stages (I–IV); alcohol intake divided into 0, 1–14, above 15 units per week and missing values, respectively (1 unit = 12 g alcohol); smoking (non-smokers, active smokers, previous smokers, missing values); complications (Clavien-Dindo grades 1–3a, 3b–5, and unknown) 21 ; ASA score (I–V); tumor site (colon/rectum); goal of treatment (curative, compromised resection, or palliative); treatment approach grouped according to surgical magnitude (laparotomy, minimally invasive (i.e. laparoscopy or robotic), and endoscopic (i.e. stent, EMR, or TEM)); emergency surgery; postoperative complications; and 30-day and 90-day mortality. Body mass index (BMI) was grouped as follows: below 19, 20–24.99, 25–29.99, 30–34.99, and 35 and above. Age was grouped into the following groups: 50–59, 60–69, and 70–75 years, respectively. Data from DTS were compared to DCCG to ensure validity of the screening variable in DCCG.
Outcome measures
Main outcomes were 30/90 days total mortality and postoperative complications dichotomized according to the Clavien-Dindo classification where a stratification was done at grade 3b as this is a complication that requires intervention under general anesthesia. 21 Besides this, the severity of complications was classified as the most severe medical and/or surgical complication a patient suffered postoperatively. The exposure was whether the CRC was detected through screening.
Statistics
Comparison of the 3535 non-screened-detected patients to the 1813 screen-detected patients was done using Pearson’s chi-square and Fisher’s exact tests. Adjusted hazard ratios (HR) were calculated for 30 and 90 days mortality using Cox Regression. As screening works by finding the cancer at an earlier stage, we would take away the screening effect if adjusting for stage. Similarly detecting the cancer at an earlier stage by screening will result in better BMI and ASA. This would take away some of the screening effect if adjusting for BMI and ASA. We therefore, made one model that did not include these variables (the basic model). The variables in the “basic model” can though not adjust for the healthy user bias. To get this in to the model, we included BMI and ASA in an “advanced model” to adjust for some of the healthy user bias, knowing that this will underestimate the screening effect.8,14 It should though be noticed that no adjustment was made for alcohol in medical complications due to few complications which made adjustment impossible. Using SAS version 9.4 (SAS Institute, NC, USA), logistic regression with the above-mentioned variables, we used to calculate adjusted relative risks (RR) for postoperative surgical complications and postoperative medical complications for each variable. To achieve convergence for the analysis for postoperative medical complications, we had to use the method proposed by Deddens et al. 22 A p-value of 0.05 or lower was considered statistically significant.
Results
A total of 5348 patients were included in the study. There were 1813 (33.8%) in the screened-detected group and 3535 (66.2%) in the non-screened group. For analysis regarding complications, a total of 4896 patients undergoing surgery were included excluding the 452 treated endoscopically of which 257 were in the screened-detected group (Fig. 1 and Table 2).
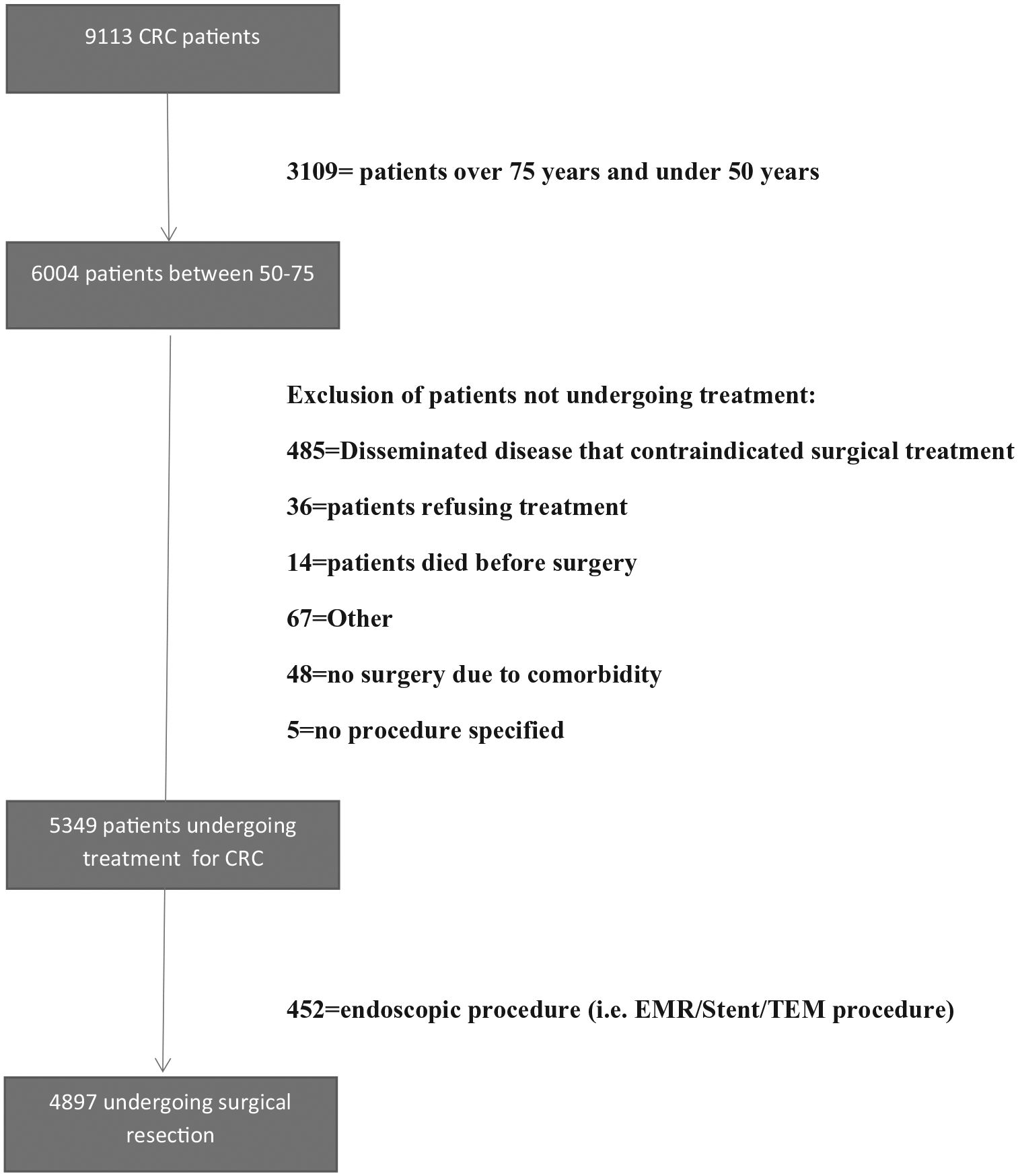
Flowchart of study.
There were several demographic differences between the groups in unadjusted analysis. However, 30 and 90 days mortality was significantly lower in the screen-detected group (p = 0.01 and p < 0.001) (Table 1).
Demographic variables.
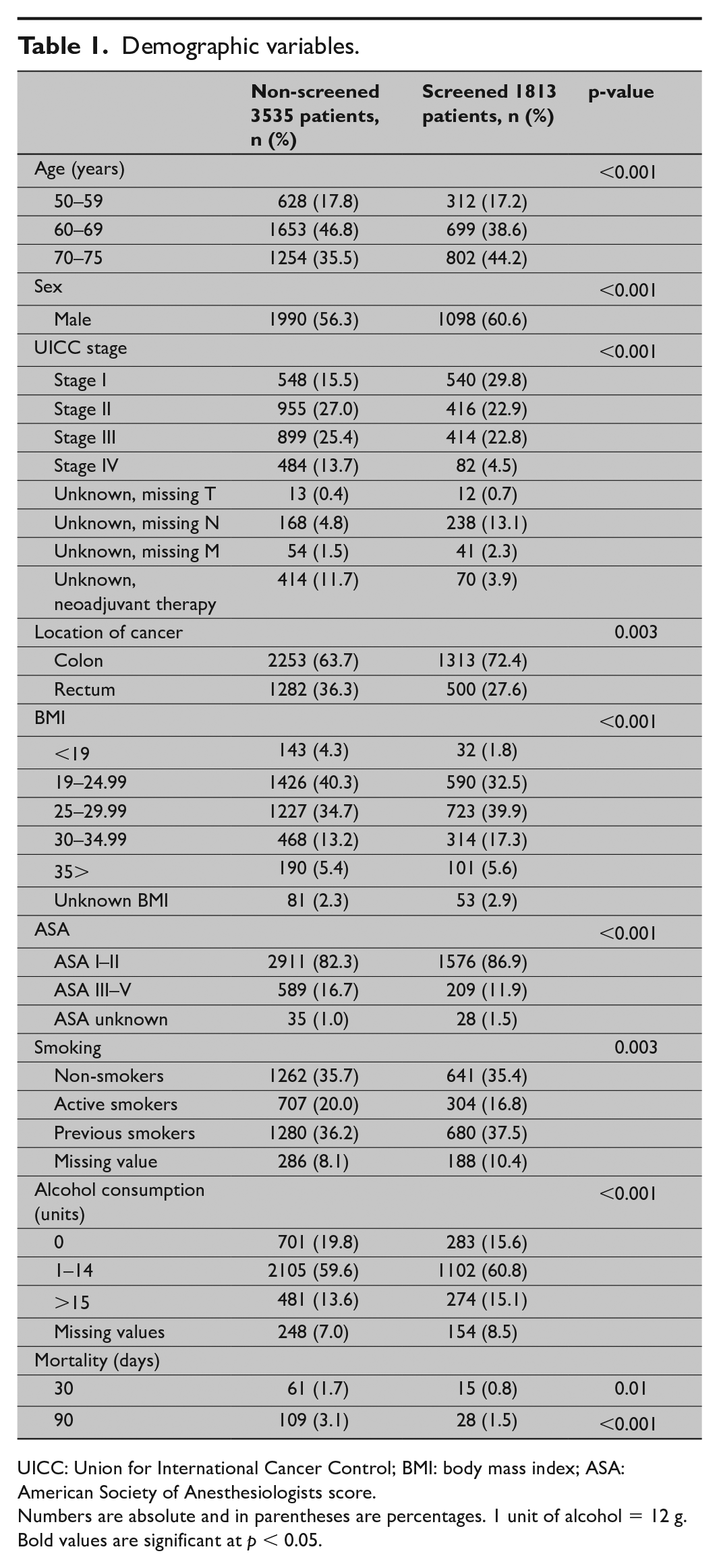
UICC: Union for International Cancer Control; BMI: body mass index; ASA: American Society of Anesthesiologists score.
Numbers are absolute and in parentheses are percentages. 1 unit of alcohol = 12 g.
Bold values are significant at p < 0.05.
The therapeutic procedures also differed with more endoscopic and minimally invasive surgery, fewer emergency resections, less supplemental resections of adjacent organs, and more treatment with curative intent in the screen-detected group compared to the non-screened group (Table 2).
Overview of procedures.
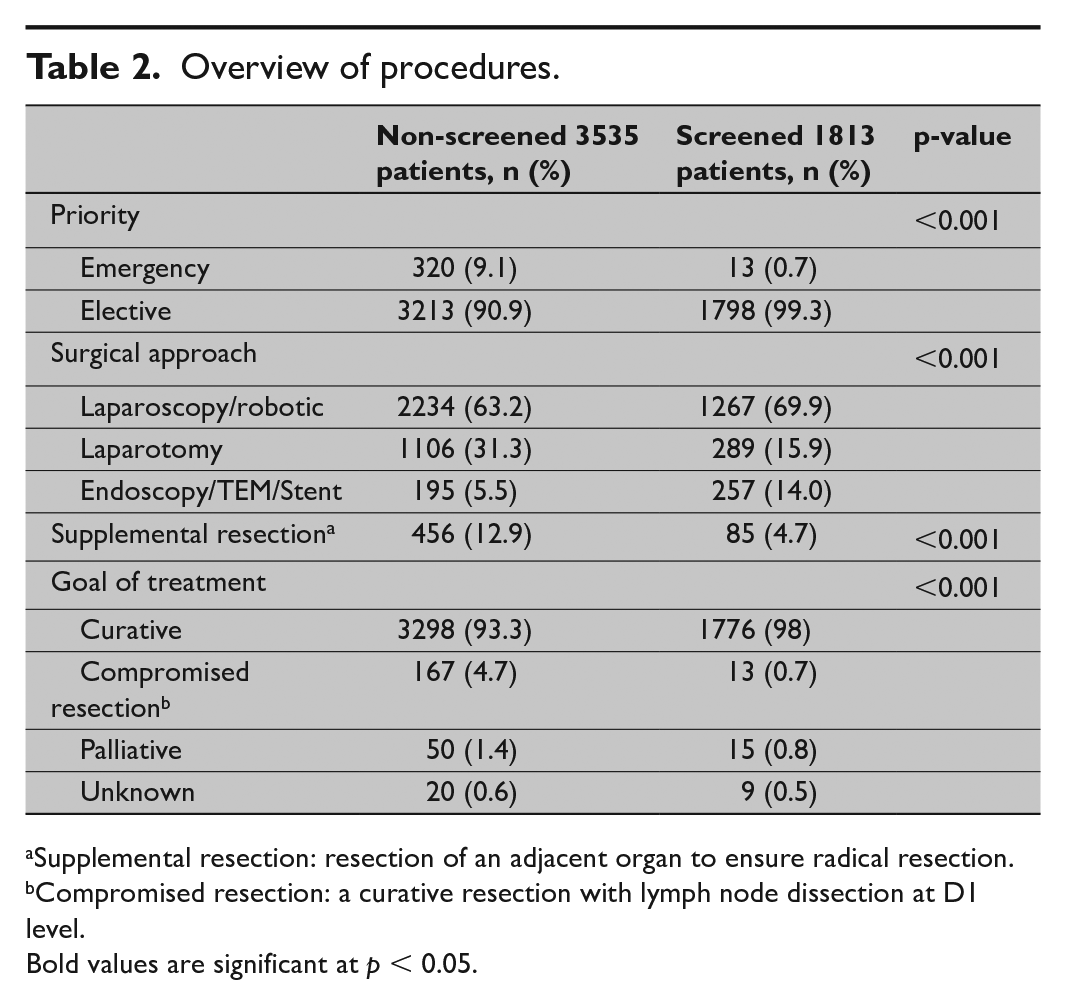
Supplemental resection: resection of an adjacent organ to ensure radical resection.
Compromised resection: a curative resection with lymph node dissection at D1 level.
Bold values are significant at p < 0.05.
The incidence of surgical complications was significantly lower in the screen-detected group compared to the non-screened group (p = 0.02), while no significant difference in the severity of surgical complications was found (p = 0.06). With regard to medical complications, no significant differences were found in neither incidence (p = 0.14) nor severity (p = 0.58) (Table 3). For severity of individual complications and detailed overview of individual procedures, please refer to the Supplemental Appendices 1 and 2, respectively.
Overview of complications.
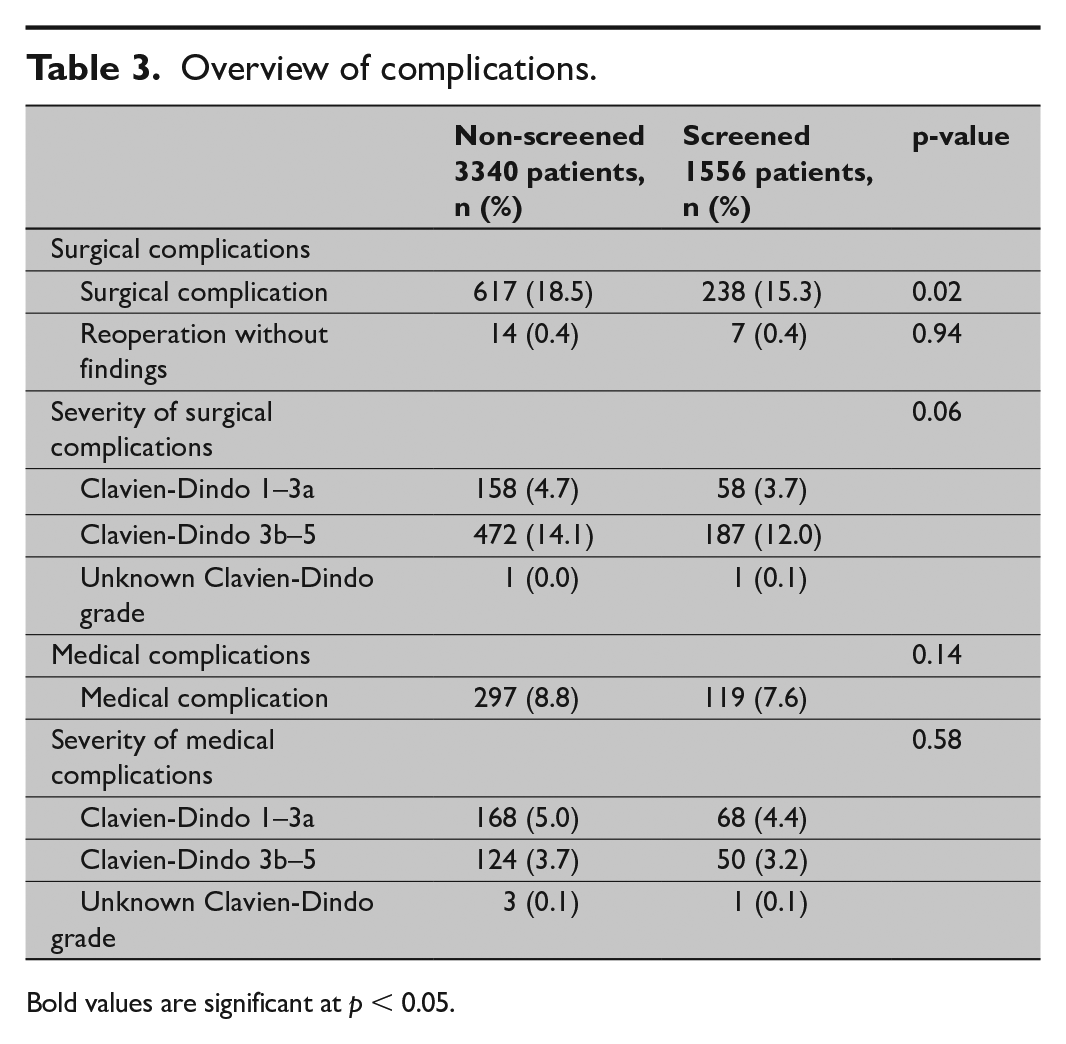
Bold values are significant at p < 0.05.
In the “basic model,” adjusted risk of 30- and 90-day total mortality was reduced in the screen-detected group (p < 0.01, HR = 0.43, CI = 0.24–0.76) and (p < 0.01, HR = 0.45, CI = 0.30–0.69) (Table 4). In the “advanced model,” including BMI and ASA score, 30 days total mortality was not significantly reduced in the screened group compared to the non-screened group (p = 0.07) but 90 days total mortality was significantly reduced in the screened group (p = 0.01, HR = 0.59, CI = 0.39–0.90) (Table 4).
HR for 30 and 90 days total mortality.
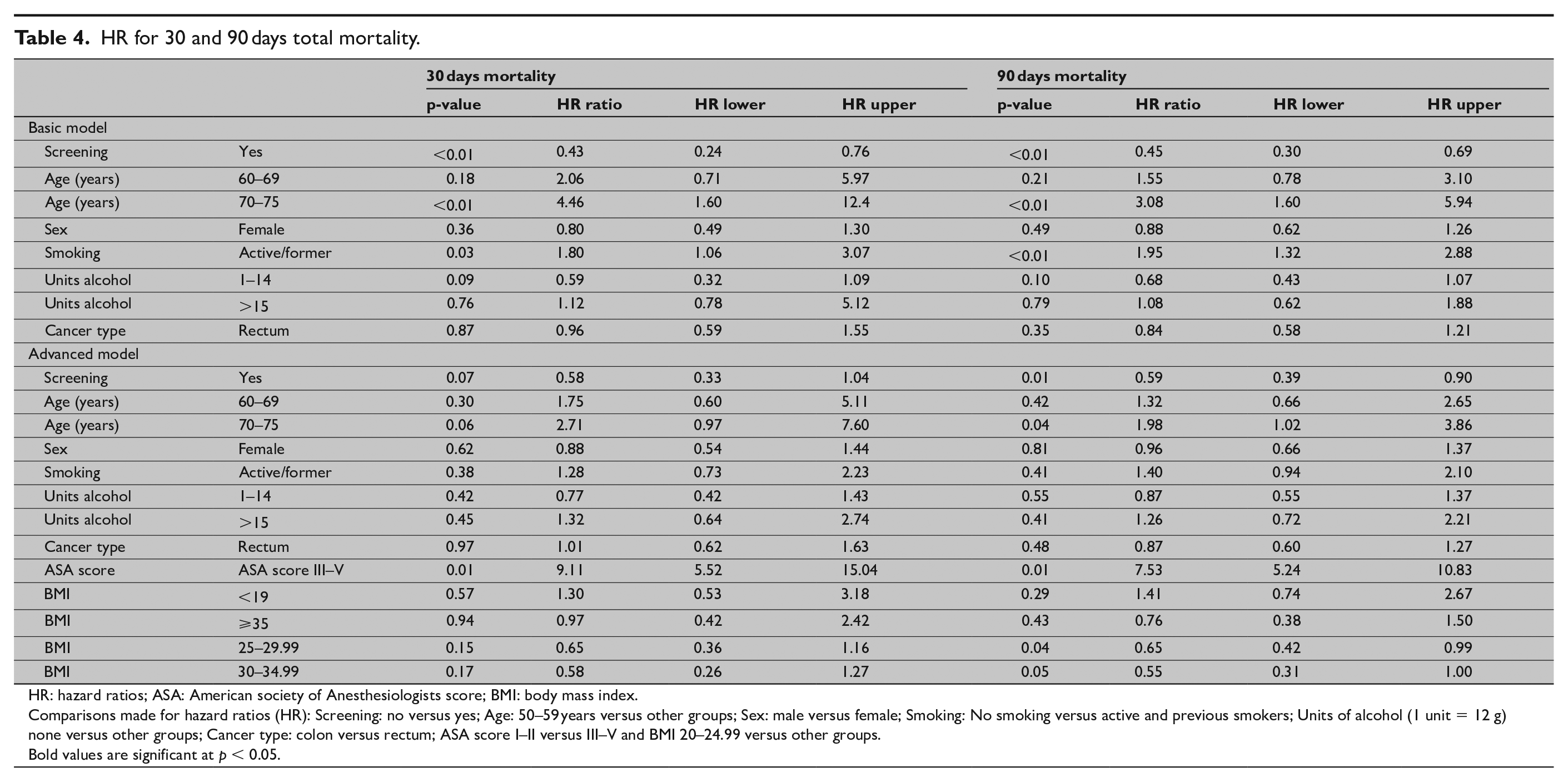
HR: hazard ratios; ASA: American society of Anesthesiologists score; BMI: body mass index.
Comparisons made for hazard ratios (HR): Screening: no versus yes; Age: 50–59 years versus other groups; Sex: male versus female; Smoking: No smoking versus active and previous smokers; Units of alcohol (1 unit = 12 g) none versus other groups; Cancer type: colon versus rectum; ASA score I–II versus III–V and BMI 20–24.99 versus other groups.
Bold values are significant at p < 0.05.
In adjusted analysis using the “advanced model,” risk of surgical and medical complications was not reduced in the screened group compared to the non-screened group, respectively, (p = 0.05, RR = 0.87 (CI = 0.76–1.00) and p = 0.47, RR = 0.93 (CI = 0.74–1.15)) (Table 5).
Association between demographic variables and postoperative complications.
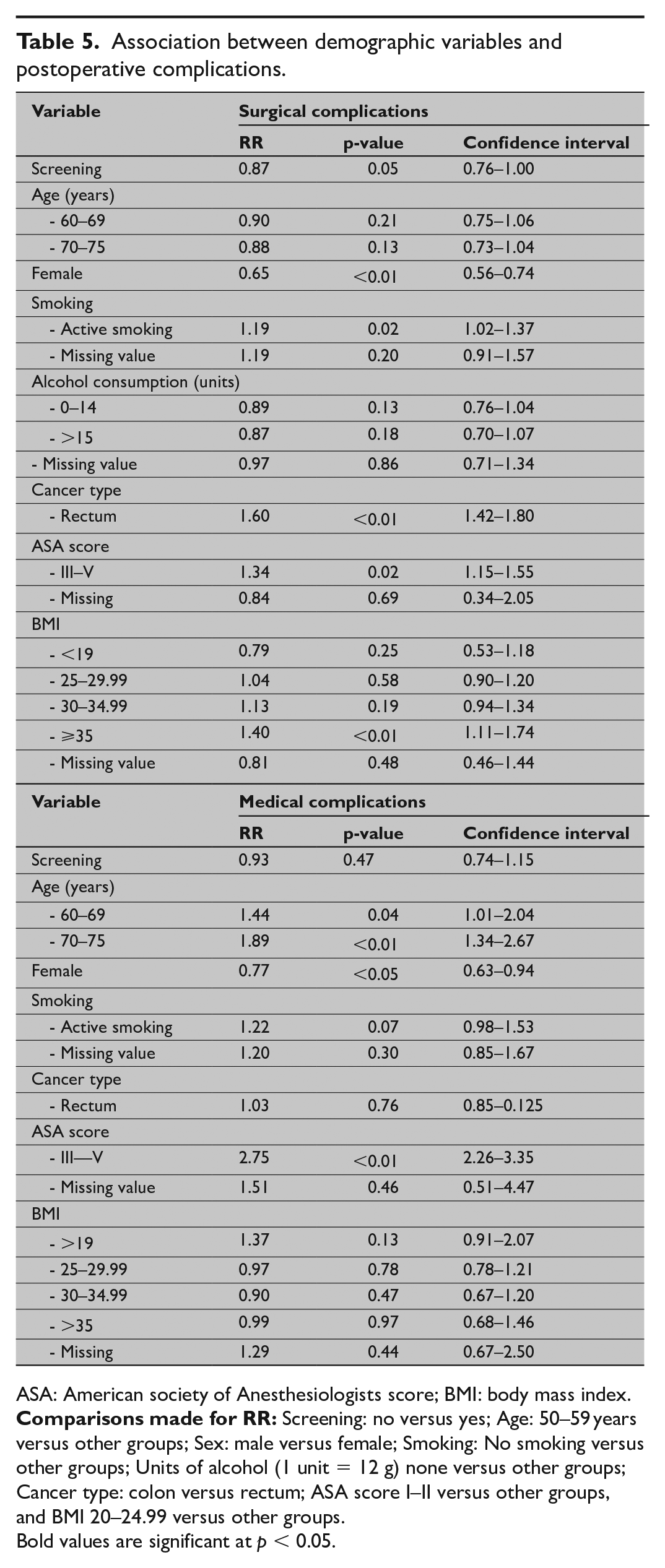
ASA: American society of Anesthesiologists score; BMI: body mass index.
Bold values are significant at p < 0.05.
Discussion
The main finding of this study is a lower 90 days total mortality in the screen-detected group. No improvements with regard to complications were found in adjusted analysis. Besides this, several changes in the therapeutic approach were found, that is, more curative treatment, more elective treatment, and more endoscopic treatment.
In this nationwide population-based study, there were more patients in the screen-detected group above 70 years of age compared to the non-screened group due to the way invitations were issued in the CRC screening program. This resulted in many patients above 70 years of age in order not to miss cases of CRCs among patients approaching the end of the screening program (9). In general, increasing age is a strong risk factor for complications and mortality following surgery for CRC. 19
We have tried to adjust for the “healthy user effect” which is a possible bias in screening studies by including BMI and ASA score in the “advanced model” as screening works through the detection of earlier cancers and thereby patients are in a better general condition before undergoing treatment. Doing this, we only saw an effect of screening on 90 days mortality perhaps due to the elimination of some of the screening effect.
However, 90 days mortality, which is a better marker for postoperative mortality, has not been used previously in screening literature.6,8,14,15,23 We also found that more patients could be treated with curative intent in the screened group, more could be treated with endoscopic resections, and that emergency surgery was almost eliminated in the screening group which is also new compared to other studies.8,24 –26 This is important as emergency treatment is associated with high mortality rates and morbidity rates17,18,27, whereas endoscopic treatment is associated with lower mortality and morbidity compared with surgery.15,28
The methodological limitations of this study include a setup that leaves room for selection bias and healthy user bias. 29 These biases were minimized by including all patients in the screening population to limit selection bias. Besides this, we also used two different statistical models to minimize healthy user bias. To our knowledge, healthy user bias has not been addressed by any previous studies on short-term outcomes after screening for CRC. It can also be argued that adjustments for TNM stage should be made. However, other studies on short-term outcomes following the implementation of screening do not adjust for TNM stage either.8,14,26 Strengths of this study include its nationwide population-based design with very few missing variables and inclusion of all cases and 90 days mortality, which has been lacking in previous studies.8,15,24,30 It can be argued that complications for endoscopically treated patients should be included but this would not be meaningful as we have only 452 of these procedures and very few complications arise following endoscopic procedures.24,28 This is underlined by another study with a larger sample size of 2984 screening colonoscopies with 1 patient dying and 44 patients with complications 15 Therefore, the results in our study represent everyday practice in Denmark where endoscopically treated patients may be treated incidentally, but surgically treated patients are generally treated by specialists. 10
In conclusion, we found that the treatment of CRC following screening is associated with more curative treatment, elective treatment, and improvements with regard to number of endoscopic resections and lower 90 days total mortality, despite inclusion of many patients above 70 years in the screened group and inclusion of emergency cases.
Supplemental Material
sj-pdf-1-sjs-10.1177_14574969211019824 – Supplemental material for Impact of screening on short-term mortality and morbidity following treatment for colorectal cancer
Supplemental material, sj-pdf-1-sjs-10.1177_14574969211019824 for Impact of screening on short-term mortality and morbidity following treatment for colorectal cancer by Michael Wilhelmsen, Sisse Helle Njor, Ole Roikjær, Morten Rasmussen and Ismail Gögenur in Scandinavian Journal of Surgery
Supplemental Material
sj-pdf-2-sjs-10.1177_14574969211019824 – Supplemental material for Impact of screening on short-term mortality and morbidity following treatment for colorectal cancer
Supplemental material, sj-pdf-2-sjs-10.1177_14574969211019824 for Impact of screening on short-term mortality and morbidity following treatment for colorectal cancer by Michael Wilhelmsen, Sisse Helle Njor, Ole Roikjær, Morten Rasmussen and Ismail Gögenur in Scandinavian Journal of Surgery
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
