Abstract
Background:
Theoretical advantages of robotic surgery compared to conventional laparoscopic surgery include improved instrument dexterity, 3D visualization, and better ergonomics. This systematic review and meta-analysis aimed to determine advantages of robotic surgery over laparoscopic surgery in patients undergoing liver resections.
Method:
A systematic literature search was conducted for studies comparing robotic assisted or totally laparoscopic liver resection. Meta-analysis of intraoperative (operative time, blood loss, transfusion rate, conversion rate), oncological (R0 resection rates), and postoperative (bile leak, surgical site infection, pulmonary complications, 30-day and 90-day mortality, length of stay, 90-day readmission and reoperation rates) outcomes was performed using a random effects model.
Result:
Twenty-six non-randomized studies including 2630 patients (950 robotic and 1680 laparoscopic) were included, of which 20% had major robotic liver resection and 14% had major laparoscopic liver resection. Intraoperatively, robotic liver resection was associated with significantly less blood loss (mean: 286 vs 301 mL, p < 0.001) but longer operating time (mean: 281 vs 221 min, p < 0.001). There were no significant differences in conversion rates or transfusion rates between robotic liver resection and laparoscopic liver resection. Postoperatively, there were no significant differences in overall complications, bile leaks, and length of hospital stay between robotic liver resection and laparoscopic liver resection. However, robotic liver resection was associated with significantly lower readmission rates than laparoscopic liver resection (odds ratio: 0.43, p = 0.005).
Conclusions:
Robotic liver resection appears to offer some advantages compared to conventional laparoscopic surgery, although both techniques appear equivalent. Importantly, the quality of evidence is generally limited to cohort studies and a high-quality randomized trial comparing both techniques is needed.
Introduction
Over the last decade, robotic surgery has emerged as a potentially valid alternative to conventional laparoscopic surgery. Advantages of current robotic surgical platforms include 3D visualization of the surgical field and improved instrument dexterity which may facilitate complex dissection and surgical reconstructions. In addition, the use of an ergonomic surgical console may reduce surgeon fatigue for long and complex procedures as well as easier control of bleeding which are two of the reasons why the laparoscopic approach remains unpopular for major resections (1). In addition, robotic liver resections (RLR) may therefore encourage more surgeons to take up complex minimally invasive techniques which they previously would not have considered doing. Robotic surgery has been shown to be superior to conventional laparoscopic surgery in a variety of different complex surgical procedures. For instance, robotic surgery for distal pancreatectomy is associated with significantly lower conversion rates and shorter hospital stay compared to laparoscopic surgery (2).
Minimally invasive liver resections (MILR) has been gaining popularity in recent years, with systematic reviews suggesting minimally invasive pancreaticoduodenectomy (MIPD) (combining both laparoscopic (LLR) and RLR) were associated with significantly less blood loss, and lower rates of margin-positive resections compared to conventional (open) liver resections (3–5). A recent meta-analysis comparing LLR and open liver resection for the management of hepatocellular carcinoma has concluded that the use of the laparoscopic approach is associated with less intraoperative blood loss, the need for a blood transfusion, increased R0 resection rates and shorter length of hospital stay. However, LLR and open liver resections have similar overall survival, disease free survival and rates of recurrence (3). The laparoscopic approach was, however, associated with a shorter duration of hospital stay and lower overall complications (6). The ongoing ORANGE SEGMENTS and ORANGE-II PLUS randomized controlled trials will provide high-quality evidence on the benefit of LLR over open liver resection (7). Whether the use of a robotic approach may prove to be superior to both the conventional laparoscopic and open techniques for liver resections still remains to be determined (8).
To date, evidence comparing the benefits of RLR and LLR is limited (9), and more data are still needed to offer clearer guidance on the future development of minimally invasive liver surgery. The aim of this systematic review and meta-analysis was to evaluate the current evidence regarding RLR and to compare its advantages with the conventional laparoscopic approaches.
Methods
Search Strategy
A systematic search of PubMed, EMBASE, and the Cochrane Library databases was conducted on the 21 March 2019 by two independent investigators (S.K.K., J.B.). The search terms used were “robotic surgery,” or “laparoscopic surgery,” or “open surgery,” and “hepatectomy,” or “liver resection” either individually or in combination. The “related articles” function was used to broaden the search, and all citations were considered for relevance. A manual search of reference lists in recent reviews and eligible studies was also undertaken. A summary of search terms are presented in Supplementary Table 1. This paper is reported according to the PRISMA guidelines and flow diagram presented in Fig. 1 (10). This study was prospectively registered with the PROSPERO database (Registration CRD42019131493).
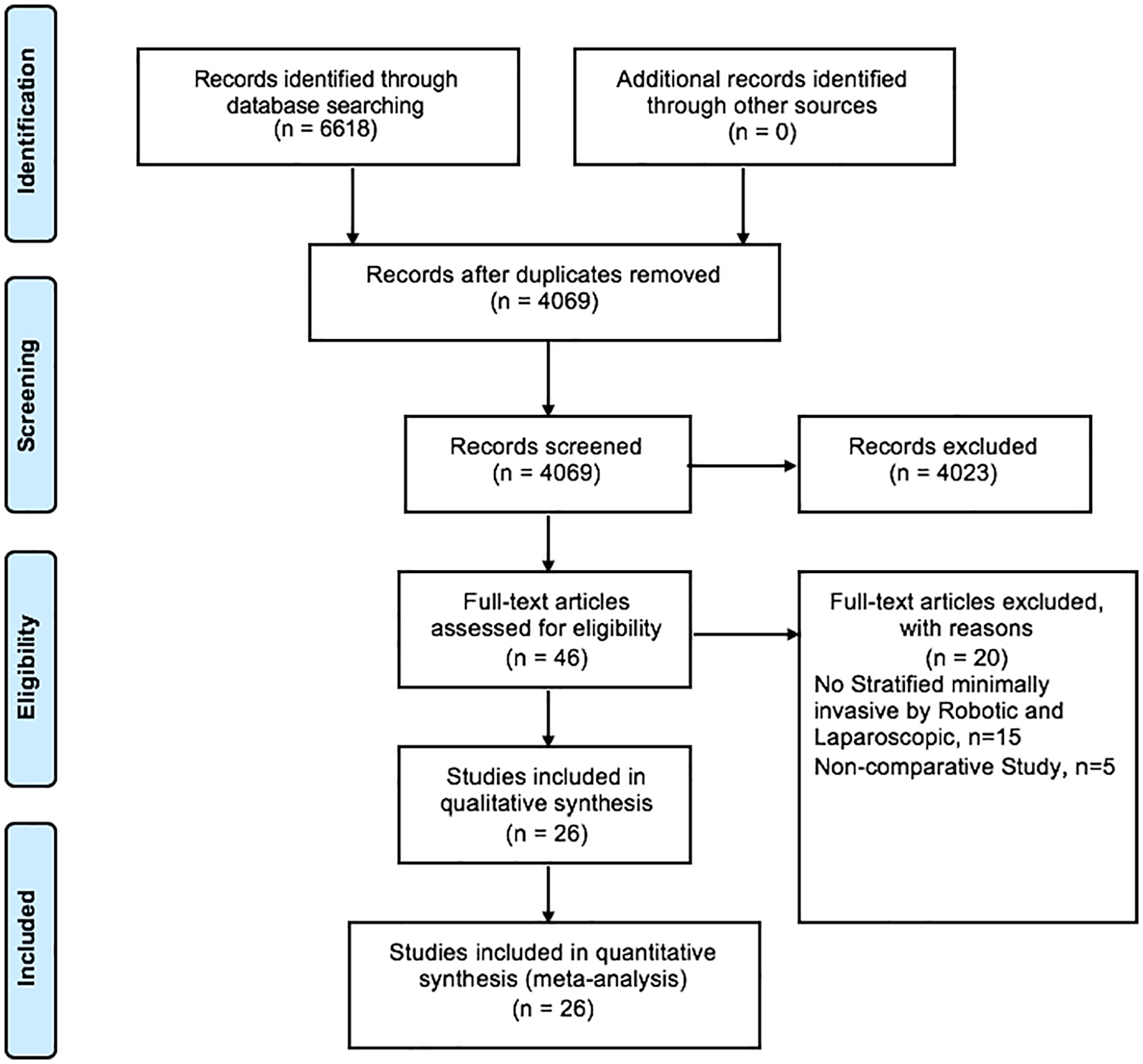
Flowchart of PRISMA diagram.
Inclusion and Exclusion Criteria
Inclusion criteria were as follows: (1) studies comparing the use of robotic and laparoscopic for liver resections for benign and malignant indications and (2) published in the English language. Exclusion criteria were as follows: (1) conference abstracts, review articles, and case reports (<5 patients), (2) non-comparative analysis between minimally invasive surgery, and (3) studies where outcomes were not reported separately for robotic and laparoscopic surgery. After excluding duplicates, two researchers (S.K.K., J.B.) independently reviewed the titles and abstracts of studies identified by the literature search. Where a study was considered to be potentially relevant to the research question a full copy of the publication was obtained for further review. The reference lists of all included studies were hand-searched in order to identify other potentially relevant studies. Any areas of disagreement between the two primary researchers were resolved through discussion.
Study Outcomes
The primary outcome measure was post-operative complications such as overall and major complications (⩾Grade III reported according to Clavien-Dindo Classification) (11). Secondary outcome measures were intraoperative (operative time, blood loss, transfusion rate, conversion rate), oncological (R0 resection rates), and surgery-specific complications such as bile leak, surgical site infection, pulmonary complications, 30-day and 90-day mortality, length of stay, 90-day readmission and reoperation rates. Intraoperative techniques such as Pringle maneuver and vascular clamping were also evaluated.
Definitions
TR was defined as complete use of the robotic technique for liver resection without laparoscopic or hand-assisted techniques during the procedure, this also included the use of laparoscopic ports by a surgical assistant as part of the robotic procedure. TL was defined as complete use of a laparoscopic technique for liver resection without robotic or hand-assisted techniques. Overall and major complications were defined according to the Clavien-Dindo classification, whereby major complications were defined as ⩾Grade III Clavien-Dindo complications (11–14).
Data Extraction
The following data were extracted from the papers: name of first author, year of publication, country of study conducted, study design, number of patients in robotic and laparoscopic group, patients’ characteristics (age, gender, ASA score, body mass index (BMI), pathology type, size of lesion), surgical techniques (use of Pringle’s manouvre, parenchymal transection), intra-operative variables (operative time, total blood loss, transfusion rate, conversion rate, R0 margin status), and post-operative variables (length of stay, 90 days readmission rate, 90 days reoperation rate, complication rate, major complication rate, bile leaks, pulmonary complications, and surgical site infections).
Quality Assessment
Methodological quality and standard of outcome reporting within included studies were assessed by two independent researchers (S.K.K., J.B.). Methodological quality was formally assessed using the Newcastle-Ottawa score (NOS) for cohort studies (S.K.K., J.B.) and the Cochrane Risk of Bias Tool for randomized controlled trial, where applicable (15,16).
Statistical Analysis
This systematic review and meta-analysis were conducted in accordance with the recommendations of the Cochrane Library and PRISMA guidelines (17,18). For categorical variables, analysis was performed by calculating the odds ratio (OR). The random effects called the DerSimonian-Laird method was used for the meta-analysis of outcomes. Funnel plots were used to visually assess publication bias of included studies. Heterogeneity between studies was assessed using the I2 value in order to determine the degree of variation not attributable to chance alone. I2 values were considered to represent low, moderate, and high degrees of heterogeneity where values were <25%, 25–75%, and >75%, respectively. Funnel plot asymmetry was assessed using the Egger test. Statistical significance was considered when p < 0.05. Statistical analyses were performed using the R Foundation Statistical software (R 3.2.1) and Stata 15 (Version 15.1, StataCorp, College Station, Texas) as previously described (2,18).
Results
Patients’ Characteristics and Pre-Operative Variables
This review included 26 studies involving 2630 patients (950 robotic and 1680 laparoscopic) undergoing liver resections. Study selection as a PRISMA flowchart is summarized in Fig. 1. Study-level and patient-level characteristics of RLR and LLR groups are listed in Table 1. Median NOS study quality assessment score was 7, ranging from 6 to 9 across the studies (Table 2), reflecting high study qualities. None of the studies had a blinded assessment of the outcomes or performed a prospective calculation of sample size.
Details of included studies reporting minimally invasive surgery for hepatectomy.
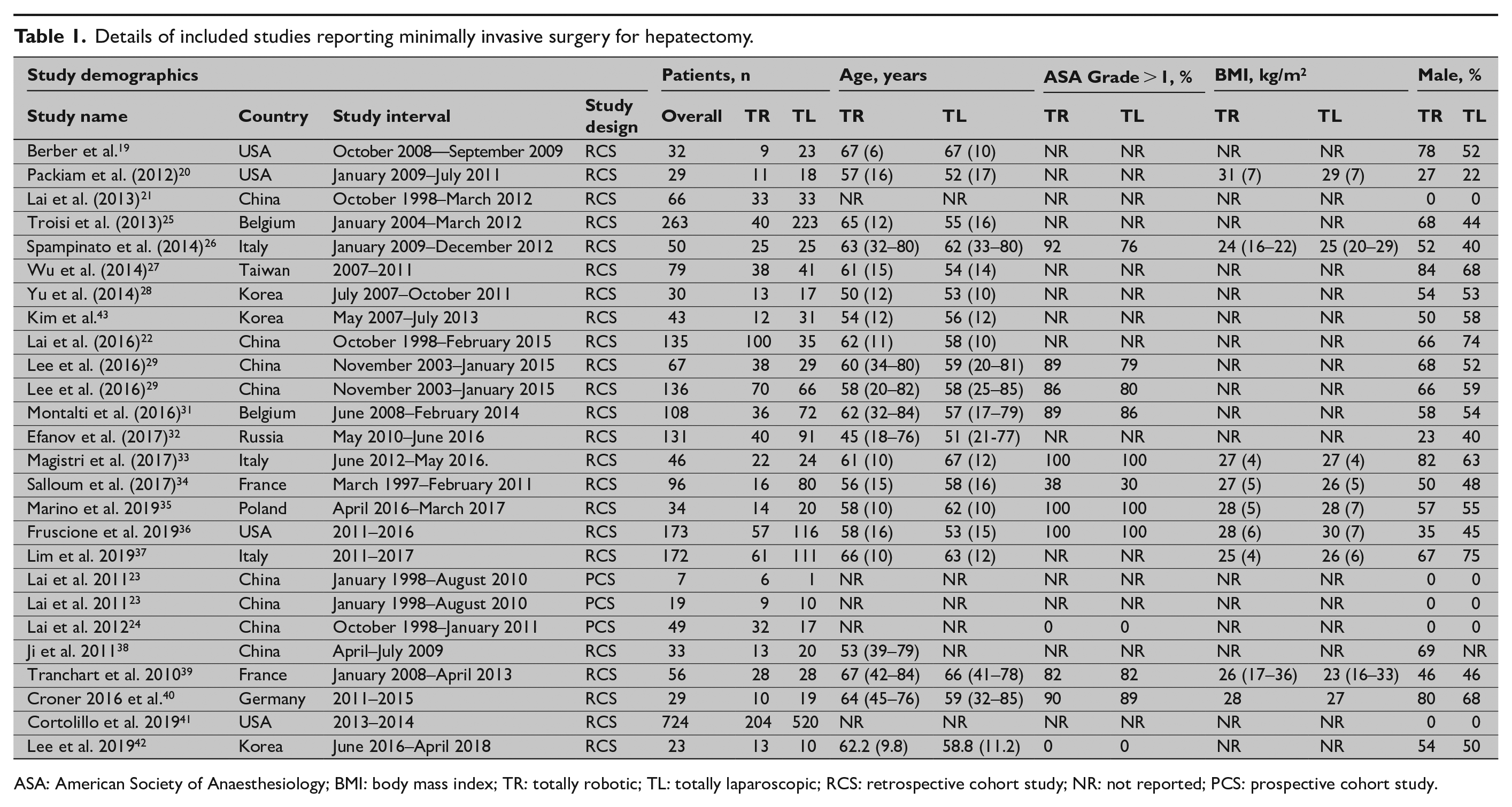
ASA: American Society of Anaesthesiology; BMI: body mass index; TR: totally robotic; TL: totally laparoscopic; RCS: retrospective cohort study; NR: not reported; PCS: prospective cohort study.
Study quality according to Newcastle Ottawa Scale.
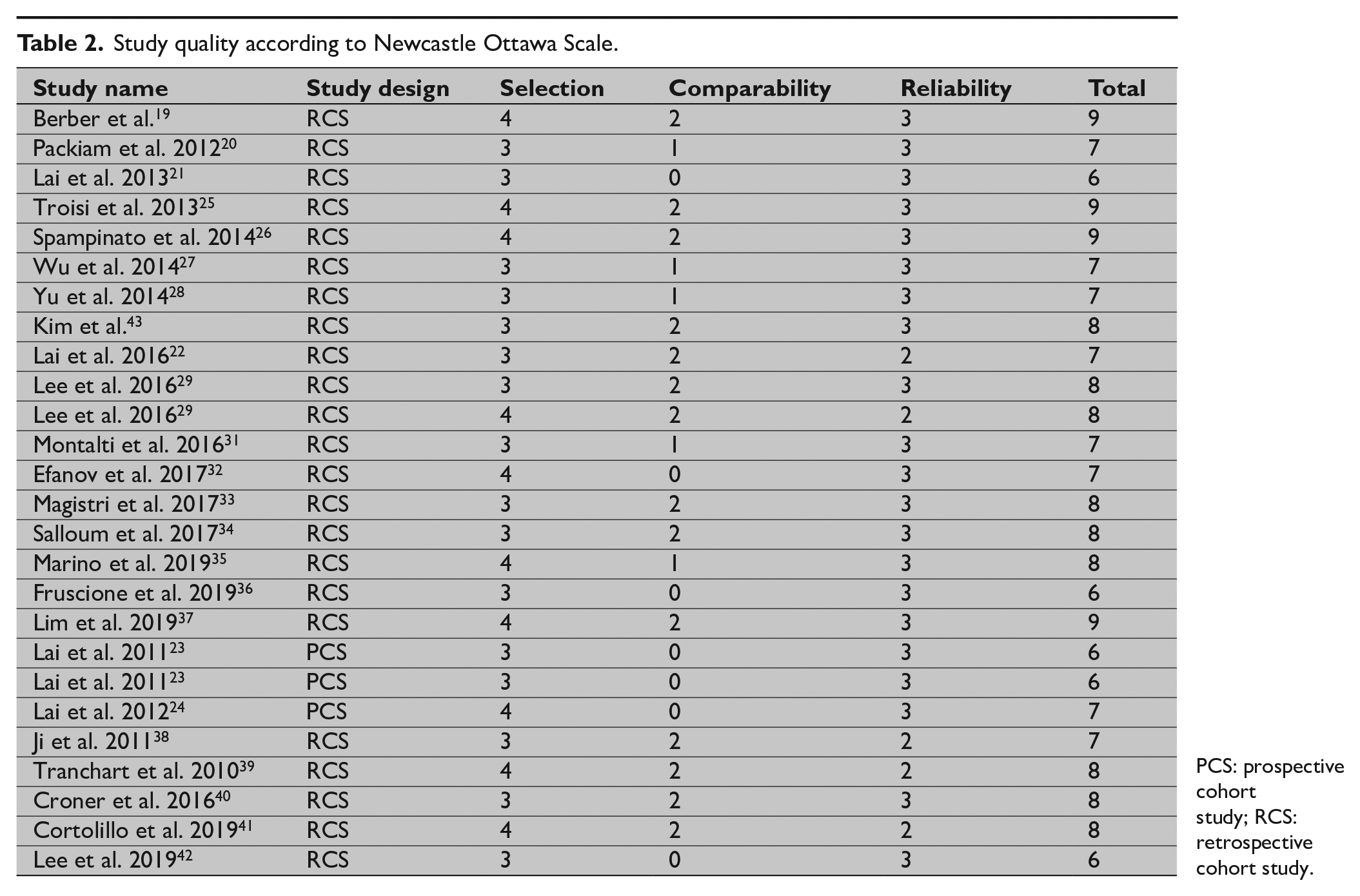
PCS: prospective cohort study; RCS: retrospective cohort study.
Patient-level characteristics were analyzed through meta-analysis. There were no significant differences in age, gender, American Society of Anaesthesiologist (ASA) grade, BMI, previous abdominal surgery, and malignant tumor pathology between both groups (Table 1, Supplementary Fig. 1A to F). There were also no significant differences in tumor number, tumor size, and tumor type (both hepatocellular carcinoma (HCC) and colorectal liver metastases (CRLM)). Patients undergoing RLR had significantly lower rates of cirrhosis than LLR (OR: 0.67, p = 0.017, I2 = 0%, Supplementary Fig. 1G). Types of liver resections for each study are presented in Supplementary Table 2.
Extent of Surgical Resection
Of the 26 studies included in the review, 14 studies included a mix of major and minor resections, 10 studies included minor resections only, and 2 studies included major resections. Of the 10 studies reporting minor resections, only 3 studies reported outcomes for left lateral section resections. A summary of the extent of resection are presented in Supplementary Table 2.
Parenchymal Transection
Of the 26 studies, 21 studies reported the type of parenchymal transection for both RLR and LLR. Harmonic scalpel (20 vs 15 studies) and Cavitron Ultrasonic Surgical Aspirator (5 vs 12 studies) were the most commonly used transection techniques for RLR and LLR, respectively. Parenchymal transection techniques are presented in Supplementary Table 3.
Intra-Operative Outcomes
Twenty-five studies reported operative times, all of which included docking times. RLR had significantly longer operating time than LLR (mean: 281 vs 221 min, p < 0.001; I2 = 90%) (Fig. 2A). Twenty-four studies reported blood loss showing significantly less after RLR compared to LLR (mean: 286 vs 301 mL, p < 0.001; I2 = 99%) (Fig. 2B). Pringle’s maneuver and vascular clamping were reported in eight and four studies, respectively. There were no significant differences between use of Pringle maneuver and vascular clamping between RLR and LLR (Table 3). Transfusion rates, conversion rates, and R0 resection rates were reported in 11, 17, and 13 studies, respectively. There were no significant differences in transfusion rate (10% vs 8%, OR: 1.13, CI95%: 0.49–2.58, p = 0.8; I2 = 50%; Fig. 2C), conversion rate (6% vs 8%, OR: 0.86, CI95%: 0.49–1.51, p = 0.6; I2 = 32%; Fig. 2D) and R0 resection rate (89% vs 86%, OR: 1.24, CI95%: 0.85–1.79, p = 0.3; I2 = 0%; Fig. 2E) between RLR and LLR.

Meta-analysis of intraoperative and oncological outcomes (A) operating time (B) blood loss (C) pringle maneuver (D) use of vascular clamping (E) duration of vascular clamping (F) blood transfusion (G) conversion (H) R0 resection.
Summary of the meta-analysis regarding baseline demographics, intraoperative and postoperative outcomes.
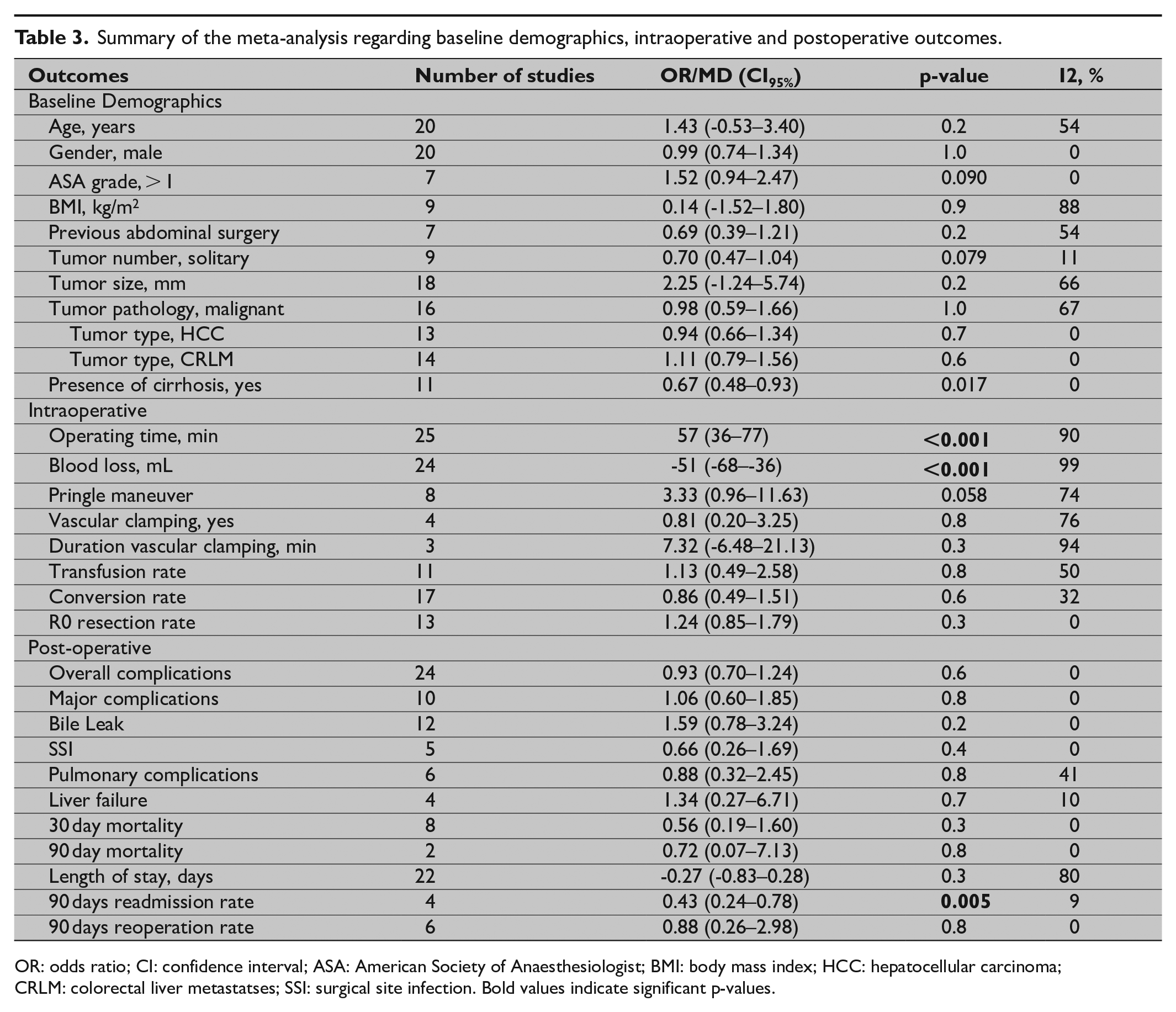
OR: odds ratio; CI: confidence interval; ASA: American Society of Anaesthesiologist; BMI: body mass index; HCC: hepatocellular carcinoma; CRLM: colorectal liver metastatses; SSI: surgical site infection.
Bold values indicate significant p-values.
Post-Operative Outcomes
Overall and major complications were reported in 24 and 10 studies, respectively. There were no significant differences in complication rates between RLR and LLR for overall (OR: 0.93, CI95%: 0.70–1.24, p = 0.6; I2 = 0%; Fig. 3A) and major complications (OR: 1.06, CI95%: 0.60–1.85, p = 0.8; I2 = 0%; Fig. 3B). Bile leaks, surgical site infections, pulmonary complications and liver failure were reported in 12, 5, 6, and 4 studies, respectively. There were no significant differences in bile leak (3% vs 2%, OR: 1.59, CI95%: 0.78–3.24, p = 0.2; I2 = 0%; Fig. 3C), surgical site infections (SSI) (OR: 0.66, CI95%: 0.26–1.69, p = 0.4; I2 = 0%; Fig. 3D), pulmonary complications (OR: 0.88, CI95%: 0.32–2.45, p = 0.8; I2 = 0%; Fig. 3E) and liver failure (OR: 1.34, CI95%: 0.27–6.71, p = 0.7; I2 = 10%; Fig. 3F) between RLR and LLR.

Meta-analysis of postoperative outcomes (A) overall complications (B) major complications (C) bile leak (D) surgical site infection (E) pulmonary complications (F) liver failure (G) 30-day mortality (H) 90-day mortality (I) length of stay (J) 90-day readmission (K) 90-day reoperation.
Peri-operative 30-day and 90-day mortality were reported in eight and two studies, respectively. There was no significant difference in 30 day (0% vs 1%, OR: 0.56, CI95%: 0.19–1.60, p = 0.3; I2 = 0%) and 90 day (0% vs 1%, OR: 0.72, CI95%: 0.07–7.13, p = 0.8; I2 = 0%) mortality rates between RLR and LLR.
Length of hospital stay was reported in all 22 studies. There were no significant differences in length of hospital stay between RLR and LLR (mean: 7 vs 8 days, p = 0.3; I2 = 80%). Readmission rates at 90 days were reported in all four studies. Patients undergoing RLR had significantly lower readmission rates than LLR (4% vs 11%, OR: 0.43, CI95%: 0.24–0.78, p = 0.005; I2 = 9%) (Fig. 3G).
Publication Biases
Funnel plot analysis for the intra-operative and post-operative variables was done to assess the possibility of publication bias. All of the studies were within 95% CI, and there was no evidence of publication bias in this study for all outcomes studied.
Discussion
Over the last decade, both laparoscopic and robotic approaches have been applied to a variety of HPB resections. However, minimally invasive liver resection remains limited to specialist centers with no general consensus regarding the translatability of minimally invasive liver surgery for improving clinical outcomes, although recent guidelines have been developed to guide surgeons as to its appropriate application (44). Several meta-analyses have published outcomes comparing robotic and open liver resections, which have demonstrated that RLR were associated with significantly lower overall complications, blood loss and blood transfusion along with a shorter hospital stay by 3–5 days (3,5). Oncologically, RLR achieves slightly better but still significant R0 resection rates (93% vs 94%).
Theoretical advantages of robotic technology include improved dexterity, visual magnification and precision by eliminating surgical tremor (45). For experienced liver surgeons who are only familiar with an open approach, the learning curve for robotic-assisted surgery may be less steep compared with conventional laparoscopic surgery (46–48). This is evident from lower rates of conversion in a recent meta-analysis in distal pancreatic resections and also to be a robotic surgeon the skills are not necessarily transferable from laparoscopic (2). Since the first reported robotic-assisted liver resection performed in 2003 in Italy (49), case reports and single institution series of RLR have been published, highlighting the feasibility of the robotic approach for major and minor hepatectomies (50). Despite the encouraging early outcomes demonstrated by these studies, no single study has provided conclusive results in favor of either approach Furthermore, robotic liver resection may offer expansion to more complex cases such as major hepatectomies, extended hepatectomies with biliary reconstruction and difficult segmentectomies of the posterior-superior segments. For instance, LLR of posterosuperior (PS) segments (Segments 1, 4a, 7, and 8) are considered to be challenging due to liver mobilization and access along with control of any bleeding (51–53). Many authors do not consider lesions in PS segments to be amenable to a pure laparoscopic approach, while others advocate ‘‘hand assistance” (54).
Compared with previous published meta-analysis (55–57), more high-quality studies and patients undergoing minimally invasive laparoscopic surgery (MILS) have been included, providing more reliable evidence. This systematic review and meta-analysis highlights (1) the current evidence supporting both RLR and LLR to be similar but limited to retrospective cohort studies (2) although the robotic approach was associated with a longer operating time, RLR was associated with less blood loss and readmission rates (3) comparable outcomes in terms of complications, blood transfusion and conversion rates reaffirm some of the difficulties faced in performing minimally invasive liver resections. Although we accept that RLR took longer there was still no statistical difference in terms of hospital stay or R1 resection rate, which confirms the two approaches are similar in terms of safety and effectiveness. This highlights the importance of continuing to develop RLR so as to develop further marginal gains.
Similar trends were seen between robotic and laparoscopic approaches in other surgical procedures such as nephrectomy (58), hysterectomy (59), anterior resections for rectal cancers (60) and distal pancreatectomy (2), where similar morbidity and mortality of the two minimally invasive approaches have been reported, but the majority all show lower conversion rates during robotic surgery for hysterectomy (59) (OR: 0.29, CI95%: 0.18–0.46) and anterior resections (60) (OR: 0.58, CI95%: 0.35–0.97, p = 0.04), although non-significant in radical nephrectomy (58) (OR: 0.44, CI95%: 0.18–1.09, p = 0.08) were noted.
This review also found significantly less blood loss during RLR compared to LLR. To our knowledge, bleeding can be controlled more easily and freely in robotic surgery due to the seven degrees of freedom afforded by the EndoWrist and the three-dimensional optics. However, these advantages do not embody in the present study. On the one hand, more major hepatectomies have prolonged operative time and are associated with more blood loss. On the other hand, the robotic system does not provide an ultrasonic dissector, which is the most popular device for parenchymal transection in the United Kingdom (61). During robotic surgery, parenchymal transection is usually conducted using either the harmonic scalpel, Endowrist sealer, scissors or bipolar forceps (43). This is because the Cavitron Ultrasonic Surgical Aspirator (CUSA) is not available in a robotic format. However, it can be used through an assistant port but in general has been obsolete during our robotic liver resections. This area needs further development and engagement with companies who manufacture parenchymal transection devices.
Previously, it was thought that intermittent inflow occlusion (Pringles maneuver) was more commonly used after RLR and seldomly performed in LLR. This maneuver creates an ischemia/reperfusion injury particularly in patients with cirrhotic livers. However, this review found no significant differences in rates of Pringle’s maneuver or vascular clamping between RLR and LLR. Also, robotic enthusiasts would also argue that the superior vision quality allows a more bloodless field as smaller vessels can be seen more readily and dealt with before they start bleeding.
Since limited studies in this review report total costs of RLR, we did not evaluate the cost associated with RLR, although it has been well described elsewhere that this procedure does typically incur an increased cost for the institution (19,62). Notwithstanding this disadvantage having a robotic program may actually have the opposite effect by increasing the number of referrals (patient choice) thus actually increasing income for the institution but again this is yet to be proven. It is perceived that over coming years the development of new robotic systems will increase competition in the marketplace and lower the overall cost of robotic surgery making it more accessible for institutions and thus making RLR more likely to achieve at least cost neutrality when compared to LLR. Also, lower conversion rates as shown previous review may translate to lower overall hospital costs (2). Nevertheless, it could also be argued that robotic systems will become technologically more advanced thus actually increasing cost.
Our review has strengths important to note such as high-quality rating of all included studies and detailed data extraction encompassing several intra-operative and postoperative outcomes. This review has limitations which are important to address. First, all studies included in this review were mainly retrospective, lacking any randomized controlled trials. This may reflect that RLR is still in the early phase of the learning curve. Second, none of the studies have evaluated the long-term outcomes such as survival, which limits our ability to draw useful prognostic conclusions and improvements in quality of life. Finally, all studies in this review do not stratify outcomes between indication (benign and malignant) and neither extent of surgery (minor or major) hepatectomy which are also important to know. However, only two studies included major resections and similar results were observed for patients undergoing minor resections only.
Conclusion
In summary, this systematic review and meta-analysis comparing RLR and LLR suggest that both techniques can be used safely. RLR appears to offer some advantages compared to LLR, although both techniques appear equivalent. Further studies and in particular a randomized controlled trial (RCT) is warranted to enable us to draw meaningful conclusions regarding patients’ survival, quality of life, cost and long-term oncological outcome.
Supplemental Material
sj-pdf-1-sjs-10.1177_1457496920925637 – Supplemental material for Robotic versus conventional laparoscopic liver resections: A systematic review and meta-analysis
Supplemental material, sj-pdf-1-sjs-10.1177_1457496920925637 for Robotic versus conventional laparoscopic liver resections: A systematic review and meta-analysis by Sivesh Kathir Kamarajah, James Bundred, Derek Manas, Long Jiao, Mohammad Abu Hilal and S. A. White in Scandinavian Journal of Surgery
Footnotes
Author contributions
All authors listed in this manuscript meet the authorship requirements set out by the International Committee of Medical Journal Editors (ICMJE). Conception and design was done by S.K. and S.A.W. Analysis and interpretation was done S.K., J.B., and S.A.W. Data collection was by SK and JB. The article was written by S.K., D.M.M., L.R.J., M.A.B., and S.A.W. Critical revision of the article was done by S.K., D.M.M., L.R.J., M.A.B., and S.A.W.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
