Abstract
Introduction:
Endovascular aneurysm sealing represents an alternative to advanced technology devices for compromised patients with abdominal aortic aneurysms. We report our results of 15 fragile patients with very low-quality infrarenal necks treated with endovascular aneurysm sealing.
Material and methods:
All patients treated with Nellix device in our hospital between June 2015 and October 2016 were retrospectively reviewed. The primary endpoints are the following: overall survival and freedom from reintervention rates. The secondary endpoints are the following: technical success; 30-day mortality; abdominal aortic aneurysm–related mortality; and freedom from endoleak rate, complications, and surgical conversion rate.
Results:
Nellix was used in 15 patients, median age 75.5 years, of which 67% were unfit for open surgery. Mean aneurysm diameter was 60 mm. One-third (5/15) of the patients were inside the Nellix instructions for use. Technical success rate was 93.3%. No perioperative complications existed, and 30-day mortality was 0%. Median follow-up was 35 (interquartile range: 11–37) months. Survival rates at 1 and 3 years were 80% and 59.3%. Abdominal aortic aneurysm–related mortality occurred in 3 of 15 cases. Freedom from rupture rates at 1 and 3 years were 92.9% and 66%. Freedom from endoleak rates at 1 and 3 years were 92.9% and 74.5%. Freedom from reintervention rates at 1 and 3 years were 86.7% and 70.6%, with a dramatic drop to 37.1% at 4 years of follow-up. Three open surgery conversions were needed. There were no statistically significant differences in results between patients treated inside and outside instructions for use.
Conclusion:
The endovascular aneurysm sealing has shown encouraging short-term results, but its safety and effectiveness during time is questionable, because this system still carries high rates of reintervention, conversions for type IA endoleaks, and secondary aneurysm ruptures.
Keywords
Introduction
Endovascular aneurysm repair (EVAR) changed the treatment of abdominal aortic aneurysm (AAA) dramatically as being significantly less invasive with lower mortality compared with open surgery (1 –4). However, the initial enthusiasm got its drawback in the beginning of millennium as the rate of late complications of the first-generation stent graft started to show their nature. Several complications, such as migration, endoleak, infection, and rupture, are feared from the surgeons during the follow-up because they often require a reintervention, and in some cases, the explantation of the graft.
During the last two decades, in light of these results, new devices have come to market and new technologies have been realized to assure best results in the future of EVAR era. Infrarenal neck is often the main issue for suitability in first instance for endovascular repair and in second instance for a basic procedure of EVAR and for the choice of the proper graft. The importance of the proximal sealing zone is well-known (5, 6), and during the years, several strategies have been proposed to avoid graft migration (suprarenal fixation, endoanchors). Also, good iliac sealing has showed to be important in preventing graft migration (4 –9).
The new concept of endovascular aneurysm sealing is born out from all these considerations (9): it is made to oppose to the perpendicular force in the aneurysm sac, obliterating the sac lumen, and to manage the cases in which proximal sealing is not properly fit for a traditional endovascular repair, creating sealing into the aneurysm sac. Endovascular aneurysm sealing (EVAS) was first approved in 2013 as a good alternative to open repair. Since then, this device showed a high aneurysm exclusion rate and low frequency of complications, especially endoleaks (5, 7, 8 –13). The EVAS seemed to represent a valid choice also in cases with poor anatomy with short infrarenal neck that may need advanced technologies such as fenestrated devices. The use of EVAS increased in many centers. However, as every kind of new device, also this new option needs to be verified as a safe and durable product in the follow-up.
In this article, we describe the experience of our center with the Nellix Endovascular Aneurysm Sealing System (Endologix, Inc., Irvine, CA, USA) that obliterates the aneurysm sac and creates sealing in this way, based on the 2013 and 2016 versions of the instructions for use (IFU).
Material and Methods
Study Design
A retrospective cohort study was conducted of all patients treated with the first-generation Nellix from June 2015 to October 2016 in our center (Helsinki University Hospital). All patients treated had a large asymptomatic AAA.
Patient Selection and Ifu Assignment
Patients were collected in a prospectively maintained retrospective database. We collected information on the baseline demographics and aorto-iliac dimensions. In general, patients had a high cardiologic risk, high risk for open surgery, and in some cases poor life expectancy. The reason to use EVAS technique was the fact that patients had poor anatomical characteristics unfit for other available standard devices and that would have needed an advanced technology treatment (e.g. with fenestrated custom-made endoprosthesis).
The first guidelines for Nellix were published in 2013. Since then, some complications occurred as showed in unpublished 2-year results of the EVAS Investigational Device Exemption (IDE) trial (especially migration, endoleak, and AAA sac growth). To address these complications and improve outcomes, IFU were redefined in 2016. We followed until the beginning of 2016 the first IFU (11 patients), and then for the last four patients, we adopted the 2016 IFU. The changes include the following: a reduced maximum infrarenal neck diameter and reduced degree of neck diameter change from ⩽20% to ⩽10%; the evaluation of the aortic lumen ratio was added, comparing the maximum diameter of the aortic aneurysm to the maximum aortic blood lumen diameter in order to limit the proportion of thrombus allowed in the aneurysm and thus provide better seal and prevent migration; and the distal seal criteria were made more stringent, the maximum iliac artery diameter was limited to 20 mm, and the distal seal zone length was added.
In our study, pre- and postoperative computed tomography angiography (CTA) scans were assessed by an expert vascular surgeon. Diameter measurements were determined by a three-dimensional (3D) dedicated software (3Mensio Vascular; Pie Medical Imaging, Bilthoven, The Netherlands). The maximum aneurysm and maximum aortic flow lumen diameters were used to calculate the diameter ratio for the refined IFU of 2016, also for patients who responded to 2013 IFU. Suprarenal and infrarenal angulations were measured according to a previously reported standard (14). Preoperative and postoperative seal lengths were assessed in 3D CTA analysis to determine the placement of the device.
After a meticulous study of the IFU, we determined whether patients had anatomical characteristics inside or outside the IFU of 2013 and 2016, respectively. After the measurements, we collected all anatomical data on each patient.
Device Description
The Nellix sac-anchoring endoprosthesis consists of two identical catheter-based flow channels, one for each side (5). Each system has the following components: (a) two endoframes: a stainless-steel balloon-expandable endoskeleton designed to support the flow channel mounted on a balloon which is positioned in a kissing configuration at the aortic neck proximally, and each extends into the common iliac artery distally; (b) the endobag: made from a nonporous expanded polytetrafluoroethylene-based material that surrounds the endoframe and filled by polymer that conforms to the aneurysm sac providing proximal and distal sealing; and (c) polymer: a biocompatible, non-biodegradable polyethylene glycol–based solution mixed with radiopaque contrast agent, visible under fluoroscopy. It takes from 3 to 5 min to solidify at 37°C, and filling is achieved by an injection system furnished with a handle with a controlled mechanism to monitoring fill volume and pressure.
Evas Procedure
The EVAS device and procedure have been well described previously (15). All patients were treated with antibiotic prophylaxis and systemic heparinization with 100 UI/kg of sodic heparin. Bilateral femoral accesses were obtained always percutaneously. The Nellix stent graft was advanced bilaterally over stiff wires and deployed in a juxta-renal position and the sheaths were retracted. Then a vacuum was applied to test the integrity of the endobags, and the balloon in the stents was inflated simultaneously to 7 atm. The endobags were prefilled with saline solution to verify the calculated polymer quantity and to assess the absence of endoleaks with the intended endobag pressure of 180 mmHg. Then the endobags were deflated and a polyethylene glycol–based polymer was instilled into the endobags with a filling pressure aimed at 180 mmHg while the position was maintained. If required, a secondary fill was performed to correct insufficient sac filling (15).
Follow-Up
Follow-up involved outpatient clinic visits and CTA at 3 months, Duplex Ultrasound (DUS) at 6 months, and thereafter yearly DUS and/or CTA unless events required closer examination.
Endpoints and Definitions
The primary endpoints of the study were overall survival and freedom from reintervention rates. The secondary endpoints were technical success (TS); 30-day mortality; AAA-related mortality; and freedom from endoleak rate, complication rate, and surgical conversion rate.
Technical success was defined as successful deployment of the device to exclude the aneurysmal flow and absence of endoleak or stent thrombosis on completion angiography. Endoleaks were classified according to the reporting standards (16). All endoleaks diagnosed on DUS were confirmed on CTA.
Statistical Analysis
Continuous variables were presented as the ±median and interquartile range (IQR: Q1–Q3) depending on the results of normalcy testing. Categorical variables were presented as frequencies and percentages. Proportions and nominal variables were compared using the chi-square test or Fisher exact test for small sample size. The Kaplan–Meier analysis was employed to estimate rates of survival, freedom from reintervention, and freedom from endoleak. The threshold of statistical significance was p < .05. All statistical analyses were performed using IBM SPSS Statistics (version 25.0; IBM Corporation, Armonk, NY, USA).
Results
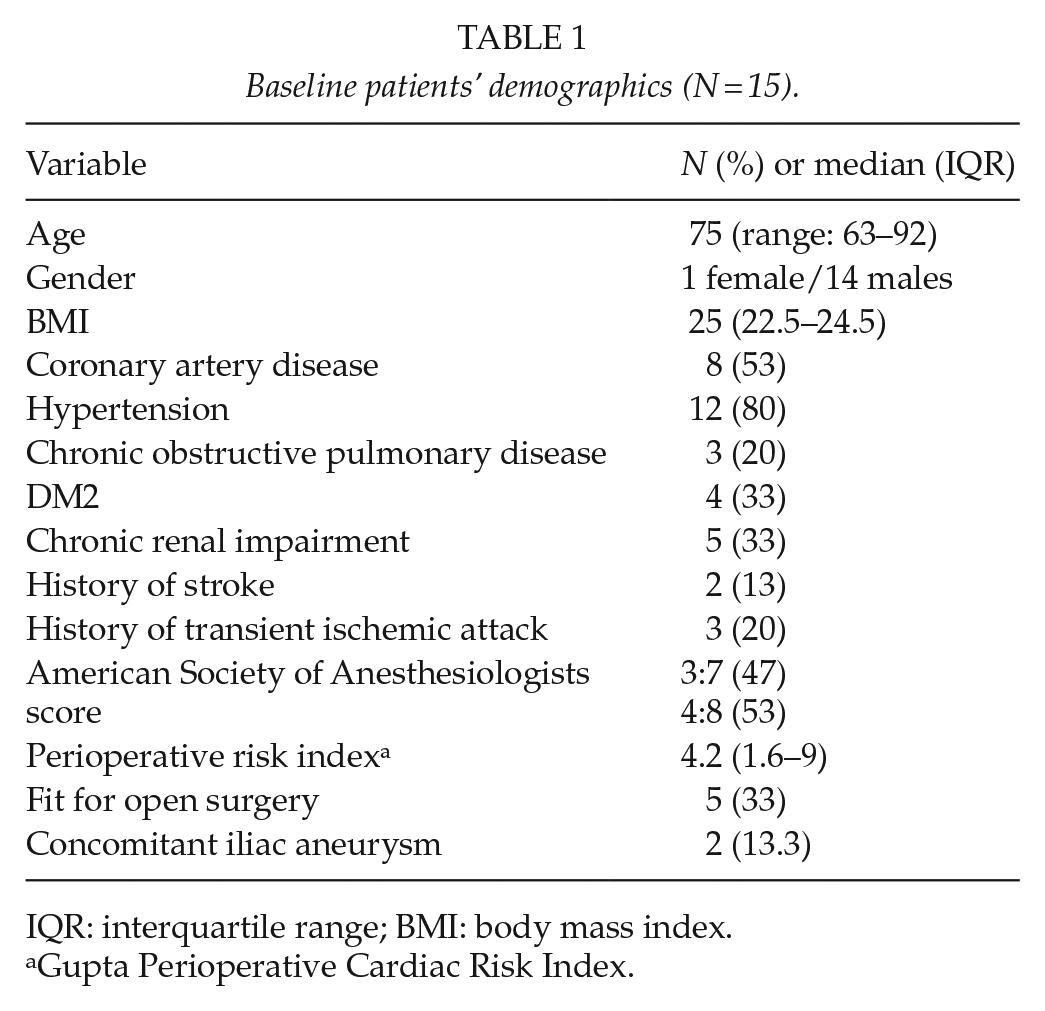
During the study period, we performed 180 endovascular aortic procedures. Nellix was used in 15 (8.3%) cases. Baseline demographics of patients are reported in Table 1. As a remarkable data, the majority of patients (n = 10; 67%) were unfit for open repair.
Baseline patients’ demographics (N = 15).
IQR: interquartile range; BMI: body mass index.
Gupta Perioperative Cardiac Risk Index.
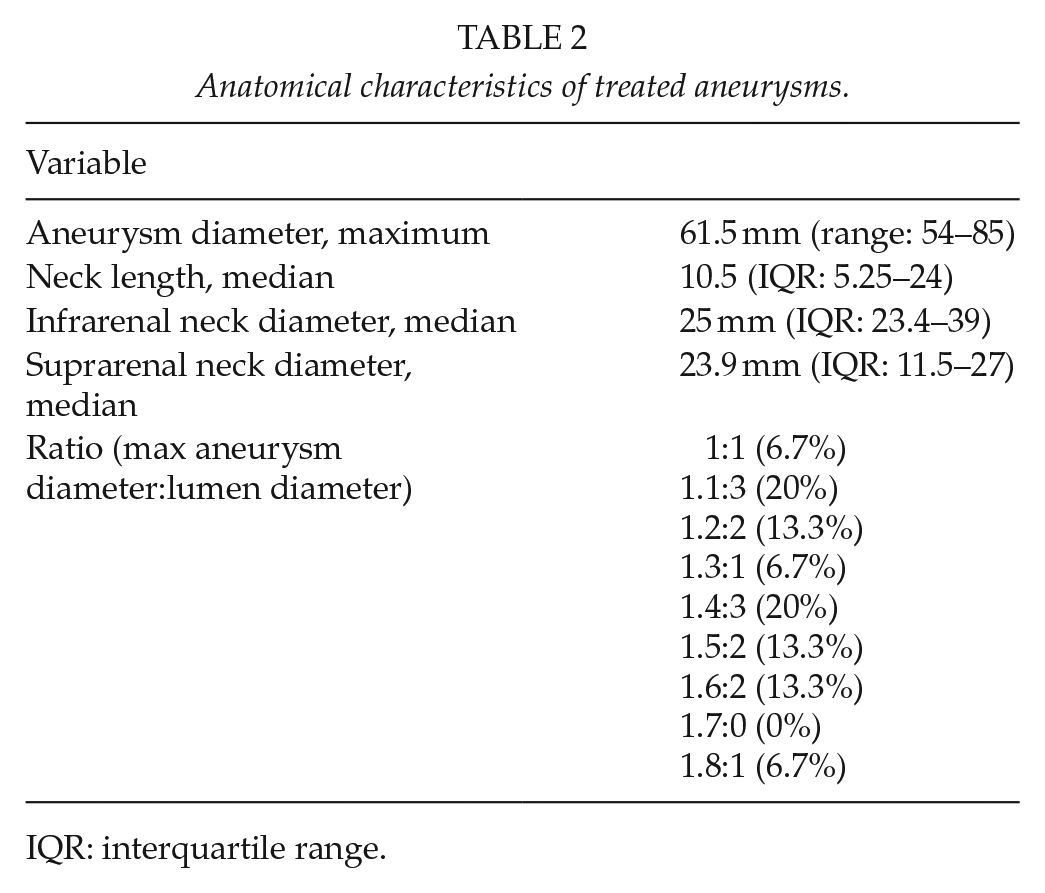
One-third (5/15) of the patients were inside the Nellix IFU, according to the period in which they received treatment (one for the 2016 IFU, four for the 2013 IFU), while 66.7% were outside the IFUs. The reasons were the following: large iliac arteries (diameter of distal landing zone >25 mm for 2013 IFU and >20 mm for 2016 IFU) in eight cases; short neck (<10 mm) in five patients; wide neck (>32 mm for 2013 IFU and >28 mm for 2016 IFU) in three cases; lumen diameter ratio higher than the consented value (>1.4) in five cases; and neck angulation (beta angle >60°) in four cases. The baseline anatomical features of the patients are showed in Table 2.
Anatomical characteristics of treated aneurysms.
IQR: interquartile range.
Intra E Periprocedural Results
Technical success was 93.3%. One (6.7%) type IA endoleak was detected at the end of the initial procedure. In this case, the graft was deployed too distally in the neck and the endobags were filled with an insufficient amount of polymer because the amount of polymer exceeded significantly the pre-calculated volume. The endoleak was successfully treated after 3 months with selective embolization with coils and glue (Fig. 1).
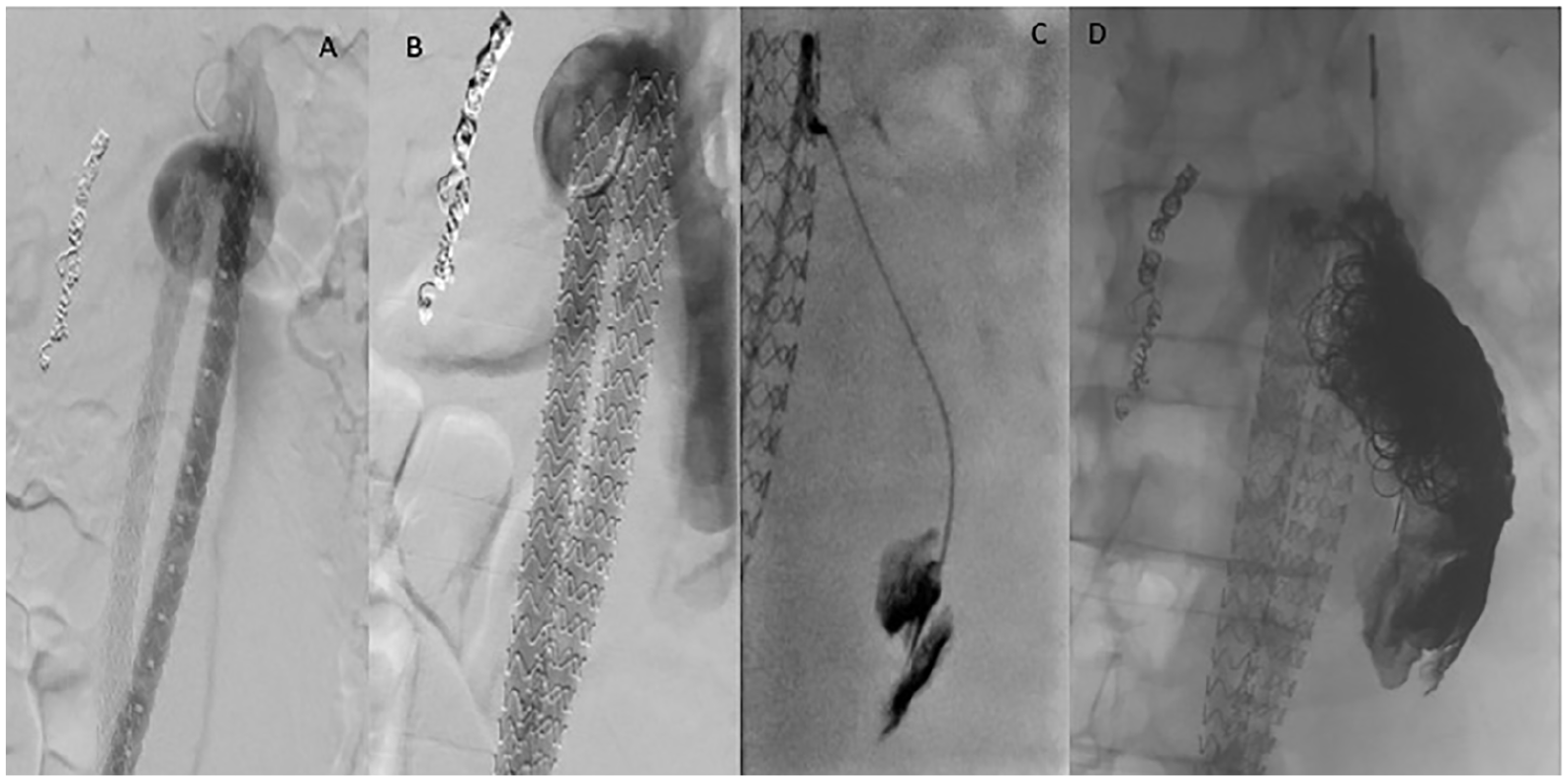
A case of primary type IA endoleak (A and B), treated with embolization with glue (C) and coiling (D).
In four cases (26.7%), it was necessary, for the really short neck, to adopt the chimney technique for one renal artery in order not to cover the lowest renal artery. Only one of these patients (25%) developed a type IA endoleak during follow-up. Mean length of stay in hospital was of 3.5 days after the procedure (range: 1–8 days). Mortality at 30 days was 0%. No perioperative complications occurred.
Follow-up Results
Mean follow-up was 35 (IQR: 11–37) months. Seven (47%) patients died during follow-up. Survival rate at 1, 2, 3, and 4 years was 80% ± 10.3%, 66.7% ± 12.2%, 59.3% ± 12.9%, and 47.4% ± 14.8%, respectively (Fig. 2).
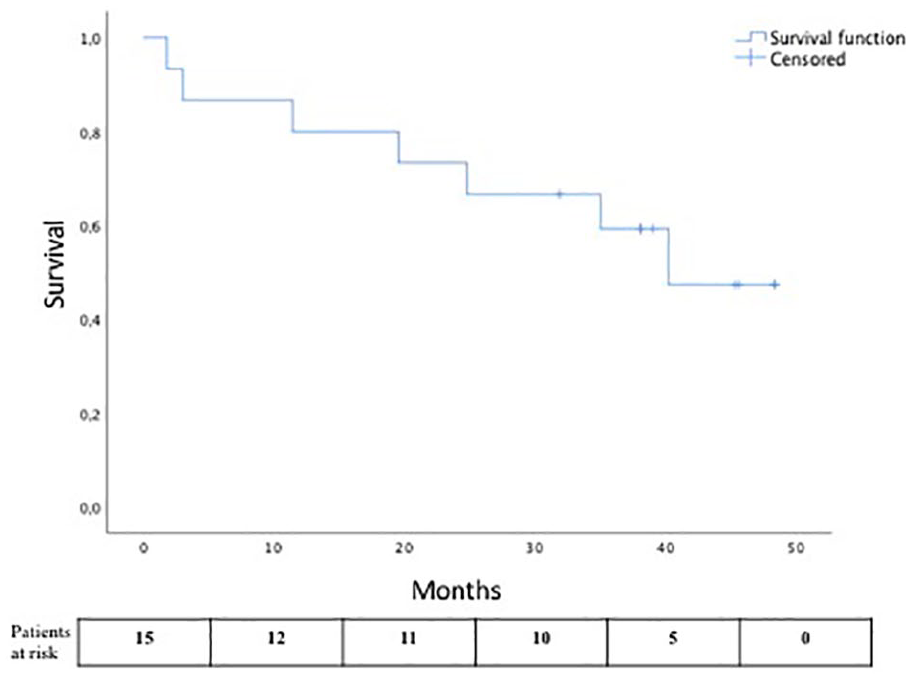
The Kaplan–Meier curve showing survival rate during follow-up.
None of the baseline risk factors were significantly related to mortality. Three patients died due to cardiac reasons after implantation. One case was converted to open surgery 1.5 months after EVAS due to graft infection. This patient died due to coronary disease after open surgery. Aneurysm-related mortality occurred in 3 (20%) of 15 cases during the follow-up (three deaths for ruptured aneurysms).
Freedom from rupture rate at 1, 2, and 3 years was 92.9% ± 6.9%, 82.5% ± 11.5%, and 66% ± 17.4%, respectively. One case was a 91-year-old male considered unfit for open surgery, without visible endoleak after the procedure; the rupture occurred 3 months after EVAS and the patient died. The other rupture occurred 1 year after EVAS. Minor type IA endoleak had been followed without sac enlargement; then, once ruptured, it was converted to open surgery and the patient recovered. The last one had a rupture after almost 3 years; the prosthesis was explanted, but the patient died of multi-organ failure during hospitalization. We registered only one case (6.6%) of limb occlusion 2 years after treatment which required a femoral–femoral crossover bypass for acute limb ischemia and one case (6.6%) of sac growth without need for reintervention and only followed up with DUS.
Endoleak type I rate was about 30% (three cases) during follow-up. Freedom from endoleak rate at 1, 2, and 3 years of follow-up was 92.9% ± 6.9%, 85.1% ± 9.7%, and 74.5% ± 13.1%, respectively. In all cases with endoleak or rupture, an iatrogenic reason could be found. One of these cases was particularly challenging: a sac enlargement was detected before by DUS at 6 months and then confirmed by CTA, without evidence of endoleak (Fig. 3). The only particular finding was a progressive separation between the endobags. The aneurysm remained stable for at least other 3 months, and then the patients came with a ruptured aneurysm, significantly increased in diameter with a remarkable separation between the endobags, 1 year and 5 months after EVAS procedure.
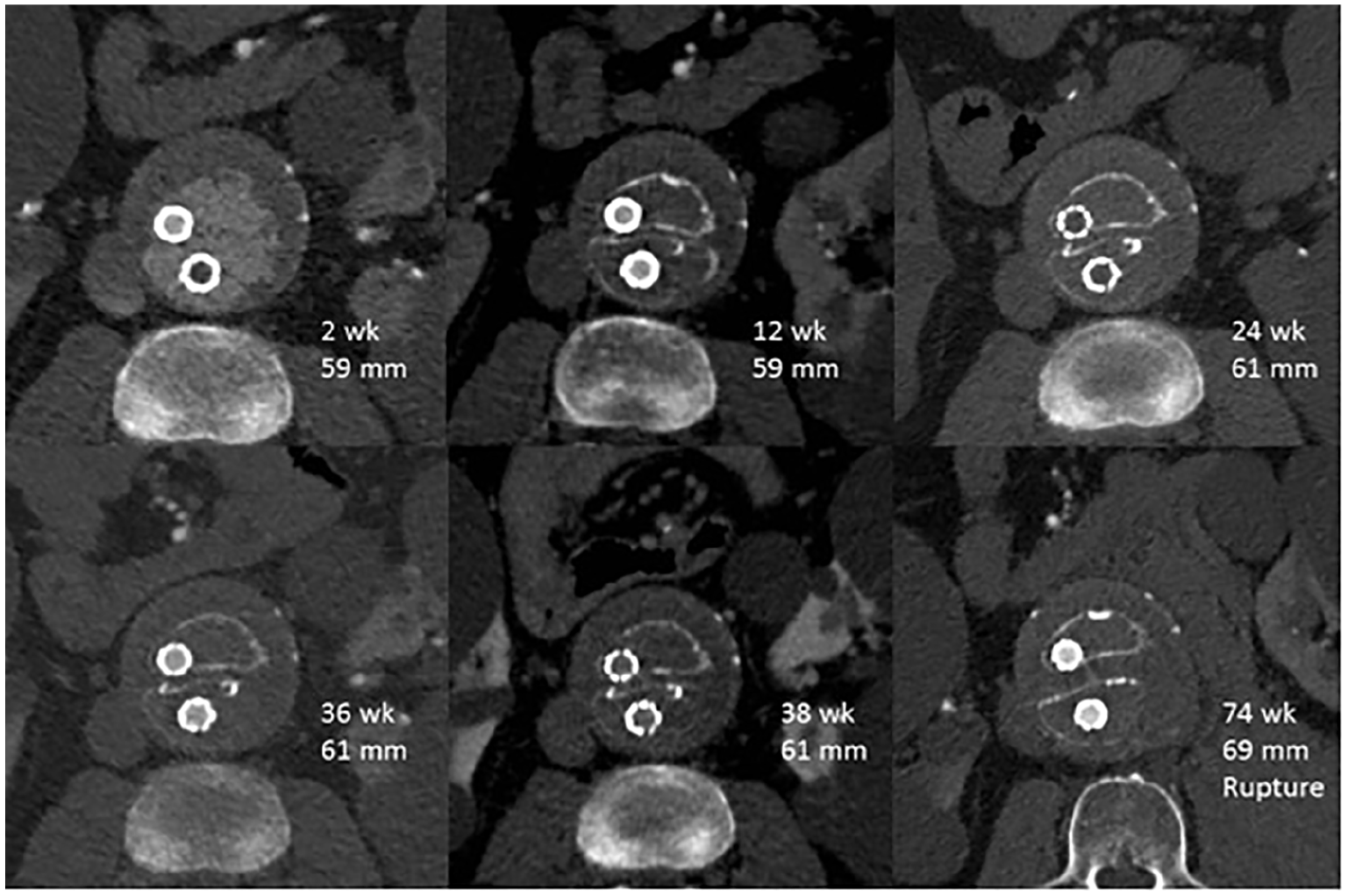
Here is shown a case in which a sac enlargement was detected with CTA without any evidence of endoleak and with a progressive widening of the space between the endobags. The aneurysm remained stable for at least 38 weeks, but at 74 weeks, the patient was admitted to emergency department for a ruptured aneurysm, as shown in the last picture of the series.
Altogether, seven patients (47%) needed reintervention. Freedom from reintervention rate at 1, 2, 3, and 4 years was 86.7% ± 8.8%, 79.4% ± 10.6%, 70.6% ± 12.6%, and 37.1% ± 15.9%, respectively (Fig. 4). Among these, three open surgery conversions were needed, two for suspected prosthetic infection and one for ruptured AAA. Freedom from conversion rate at 1, 2, 3, and 4 years was 93.3% ± 6.4%, 93.3% ± 6.4%, 83% ± 11.3%, and 66.4% ± 17.4%, respectively.
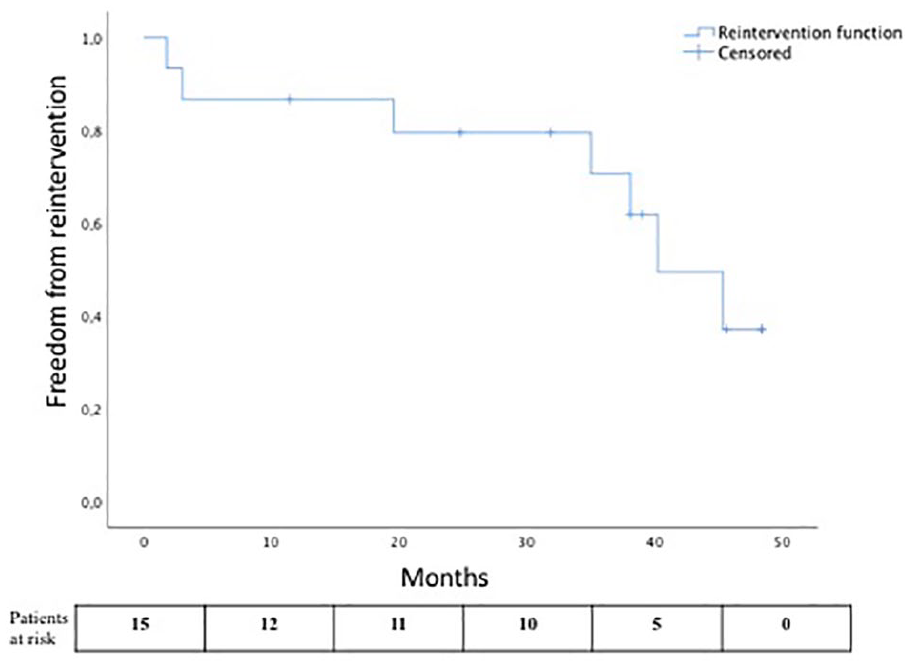
The Kaplan–Meier curve showing freedom from reintervention rate during follow-up.
Results by IFU
We made a further analysis of data dividing patients into two groups depending on the IFU under which they were treated (2013 and 2016). Eleven patients (73.3%) were treated under the 2013 IFU, while four patients (26.7%) under 2016 IFU. The endoleak rate was 25% and 27.2% for the 2013 group and 2016 group, respectively, without any significant difference (p = .9). No statistically significant difference was found also for rupture and reintervention rates between the two groups (p = .51 and p = .56, respectively).
Despite these results, only five patients (four in 2013 group and one in 2016 group; 33.3%) effectively responded to IFU. Analyzing these other two groups (inside IFU and outside IFU), we were able to see how all endoleaks were detected in follow-up of the patients outside IFU, and none of them were found in the inside IFU group (p = .05), and these data are valid for ruptures (p = .5) and AAA-related death as well (p = .42). Complications were equally spread among the two groups (two cases for each group), without significant difference (p = .56). Reintervention rate was 20% and 60% during follow-up of the two groups, but was not statistically relevant (p = .28).
Discussion
The EVAS represented a completely new idea in the treatment of AAA and it showed very good short-term results and almost zero endoleak rate (17). These successful outcomes led to a widespread use of this device in many centers worldwide. We have to emphasize that, in our center, Nellix was used in cases without any other options of treatment for their aneurysms.
Our perioperative results were very promising regarding mortality (no events registered at 30 days) and medical or surgical complications, with a very satisfying technical success (93.3%) and only one case of on-table type IA endoleak, though only a third of the population responded to IFU. Regarding follow-up results, almost a half of the population died (47%), with a survival rate at 3 years of 59.3% and an aneurysm-related mortality of 20%. We reported freedom from reintervention and freedom from conversion rates of 70.6% and 83% at 3 years, respectively. Looking at these results, EVAS seemed to offer very good immediate- and short-term results, with an increase in complications and mortality during follow-up, which underlines the importance of accurate evaluation of long-term outcome before new technology is adopted. In this particular cohort, the patients were mostly unfit for open surgery without any other possible endovascular solution. As the annual rupture rate in large aneurysms is considerable, this solution was seen as a better alternative than conservative treatment, which carries a substantially higher mortality risk for the patient.
The EVAS technique was adopted in many centers as the short-term results were excellent. However, long-term results are still few (18). The mid-term experiences have been reported by several authors. The technical success rate has been 98%, the short-term endoleak rate has been less than 1%, and the 1-year reintervention rate has been 3%–4% (8, 10, 13). One-year AAA-related mortality and conversion rates have been 1.3% and 0.7%, respectively (8, 13). One-year endoleak rates have been around 1.5% (8, 13). Gossetti et al. (13) reported a 1-year reintervention rate of 4.7%, including four conversions; freedom from aneurysm-related intervention was 98.3% at 1 month and 94.7% at 12 months.
In 2018, Zoethout et al. (19) reported results at 2 years of follow-up, specifying the outcomes also on the basis of the new 2016 IFU. Technical success was very similar for both groups (98.2% for 2013 group and 97.9% for 2016 group); freedom from reintervention rate was 89.7% and 95.7%, respectively. Endoleak rate was 5.4% for the 2013 group and 2.1% for the 2016 group, but the difference was not statistically significant; migrations of the graft requiring intervention occurred only in the 2013 group in 7.1% of cases. This study showed that probably the more strict IFU of 2016 reduced the rate of complications during the follow-up, but so far there are no statistically significant data on that.
A recent review of the literature about EVAS this year (2019) by Choo et al. (17) combines results of 14 single-arm observational studies, with 1510 patients recruited and a mean follow-up of 11.6 ± 5.4 months. The rupture rate at 30 days was 0.7% and 0.8% during the overall follow-up. Early and late endoleak rate reported was 2.8% and 1.9%, with 2.1% of device migration rate and 3.5% of late reintervention rate. The AAA-related death rate during follow-up was about 1%. In their publication, Choo et al. concluded that reported outcomes are acceptable, in particular during the short-term period, although the risk of important and life-threatening complications is undeniable during follow-up. In conclusion, all the publications report mid-term results which are relatively good. However, our results show that a significant increase in reintervention rate is seen after 30-month follow-up, indicating that with this device the true problems do not show up during the 2–3 first years, but thereafter (18).
In EVAS, the sealing does not take place in the infrarenal neck, but the endobags seal the aneurysm itself. This different concept has allowed the treatment of aneurysms with short neck with EVAS that would have not been suitable for EVAR exactly for the infrarenal anatomic features. Indeed, the sealing in EVAS is obtained in the aneurysmal sac and not in the neck, and although the short-term morbidity using this technique is low, long-term follow-up has shown that the endobags migrate causing IA endoleak and eventually rapid sac increase. After its exclusion, the aneurysmatic sac goes usually toward to shrinkage, and this could undermine the purpose of the EVAS device due to perpendicular forces to blood flow that could be responsible for graft instability and migration (9). If type IA endoleak is not promptly corrected, it could be responsible for the rupture of the aneurysm, with high rate of mortality and morbidity.
When the IFU for Nellix were revised, the most important adjustments were made to the diameter of proximal neck (the maximum was reduced from 32 to 28 mm) and the definition of aortic neck length was updated to diameter change of 10% versus the previous 20%. Moreover, a ratio between the maximum diameter of the aneurysm and the diameter of the effective aortic blood lumen was introduced. The revision of the IFU in 2016 made the inclusion criteria for treatment more severe and the treatment more addressed to the right patients. Some published studies (10, 20, 21) have shown how working under IFU revealed to be crucial for the outcome of the procedure in time, both for EVAR and for EVAS: the more IFU are violated, the more failure is possible, and along years, we have learned that patient selection is one of the most important moments of decision-making before surgery.
In our experience, we treated the majority of our patients under the availability of the 2013 IFU, but nevertheless the cases we treated were for a great part outside of the IFU. Despite this, our short- and long-term results were not statistically different neither for the patients treated inside IFU nor for those treated after the adjustment of the IFU.
The median follow-up in our study was about 3 years, and literature lacks such long-term results yet. For this reason, there is still poor knowledge of what should be the proper times and methods for the follow-up of patients who received Nellix. In our clinical practice, we follow our EVAR patients with a CTA 3 months and 1 year after the procedure, a DUS at 6 months, and thereafter once a year with DUS in case of a regular decursus. In case of suspicion of sac increase, computed tomography (CT) is performed. Due to the high risk of late complications, patients with Nellix device may need more intensive follow-up with CT. Furthermore, we have learnt that some phenomena related to changes in endobags configuration are special to EVAS technique. For example, we witnessed a particular case in which no endoleak was detectable at the imaging; the only clue we had for detecting what was going wrong was the increasing distance between the endobags. In the end, this patient had secondary aneurysm rupture. This finding was useful for us to understand that these patients should be followed up closely, and paying attention to details may be not so commonly evaluated in traditional EVAR cases. We used Nellix only in very special cases, and after the first signals of late problems, we completely stopped using it. As a consequence of “Nellix-story,” new European Society for Vascular Surgery (ESVS) guidelines on the treatment of AAA (22) state that new techniques such as EVAS are not recommended.
This study has several limitations. The cohort of patients is small, and thus no proper comparisons or subgroup analysis can be made. Moreover, they were all patients with moderate to high cardiologic risk and almost all unfit for open surgery. These features could have influenced the mortality and the reintervention rates. On the contrary, we have comprehensive surveillance of all patients, which makes the results we present here reliable.
Conclusion
Endovascular aortic sealing system is a good example of the challenges related to the adoption of new technology at a phase where long-term follow-up is not yet available. Results in the short term seem to be encouraging, with very satisfying technical success, mortality, and morbidity rates. The main issue remains the safety and effectiveness during time, because this system is still burdened by high rates of reintervention and conversion for type IA endoleaks and secondary aneurysm ruptures. Our study, although has a small number of patients, gives important information on the behavior of Nellix device and may give some beneficial information for the surveillance of these patients. Further studies with long-term follow-up are needed to define outcomes and to assess a proper surveillance protocol for these patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
