Abstract
Background:
Acute pancreatitis is a common disease, the incidence of which is 75–100/100,000/year in Finland. The worldwide incidence of acute pancreatitis is increasing. The identified mildcases usually show rapid recovery with conservative treatment allowing early discharge. Severe cases need early intensive care to reduce the risk of serious complications such as multi-organ failure. The revised Atlanta classification of acute pancreatitis was introduced in 2012–2013. A recurrent acute pancreatitis is defined as two or more well-documented separate attacks of acute pancreatitis with complete resolution in between. Alcoholic pancreatitis is the most common recurrent acute pancreatitis type.
Methods:
In this review current severity classifications and literature on the prevention of recurrent acute pancreatitis are analyzed.
Results:
The severity of the disease is classified as mild, moderately severe, and severe acute pancreatitis. Novel entities include acute peripancreatic fluid collections in mild acute pancreatitis and acute necrotic collections in necrotizing acute pancreatitis lesser than 4 weeks after the onset and pancreatic pseudocyst in mild acute pancreatitis and walled-off necrosis in necrotizing acute pancreatitis more than 4 weeks after the onset of the disease. After the first attack of alcohol-induced acute pancreatitis, 46% of the patients develop at least one recurrence within 10- to 20-year follow-up. With repeated intervention against alcohol consumption, it is possible to reduce the recurrences. Removing the gall bladder after biliary pancreatitis is the key preventing recurrences. In mild cases, even during the index admission; in severe cases, it is recommended to wait until the inflammatory changes have resolved. Of total, 59% of the idiopathic pancreatitis had sludge of stones in the gall bladder. In other etiologies, addressing the etiological factor may prevent recurrent acute pancreatitis.
Conclusions:
This review describes current use of novel severity classifications and also different possibilities to prevent recurrent acute pancreatitis with different etiologies including idiopathic.
Acute pancreatitis (AP) is a common disease, the incidence of which is 75–100/100,000/year in Finland (1). It was the most frequent principal discharge diagnosis in gastrointestinal disease in the United States in 2009 (2). Furthermore, the worldwide incidence of AP is increasing.
Classification of AP
AP needs to be classified as accurately and as early as possible because the identified mild cases usually show rapid recovery with conservative treatment allowing early discharge, and thus saving intensive care resources. In contrast, moderate and severe cases need early intensive care to reduce the risk of serious complications such as multi-organ failure. Furthermore, designing therapeutic trials necessitates proper and generally accepted classification of the severity of the disease.
The revised Atlanta classification (RAC) of AP was introduced in 2012–2013 (3, 4). The types of AP are interstitial edematous pancreatitis (IEP) and acute necrotizing pancreatitis (ANP). The severity of the disease is classified as mild, moderately severe, and severe AP. Early phase of AP is less than a week from the onset of symptoms. Late phase involves cases with more than a week from the onset of symptoms and occurs only in patients with moderately severe or severe AP. In mild AP, there is no organ dysfunction or failure nor local or systemic complications. Spontaneous recovery usually takes place during the first week. New category in RAC is moderately severe pancreatitis. It involves organ dysfunction (transient organ failure) resolving in 48 h and/or local or systemic complications (e.g. peripancreatic fluid collections) without organ failure. In severe or necrotizing form of AP, there is a persistent organ failure (lasting for longer than 48 h). Organ failure assessment can be accomplished by the modified Marshall multiple organ dysfunction score (MODS), which is included in RAC or sequential organ failure assessment (SOFA) score (5, 6). The type of necrosis in severe pancreatitis can be either sterile or infected, and its location can be pancreatic and/or peripancreatic or peripancreatic only. The location and the amount of necrosis can typically be assessed in the contrast-enhanced computed tomography (3, 4).
In the past, the nomenclature of pancreatic fluid collections was very diverse. In the RAC, the fluid collections have been classified according to the severity of the AP as well as to time after the onset of the disease (<4 weeks and >4 weeks). Fluid collections can be either sterile or infected. Novel entities include acute peripancreatic fluid collections (APFC) and acute necrotic collections (ANC). Patients with moderately severe AP can, according to RAC, have pancreatic and/or peripancreatic necrosis. Furthermore, patients with severe pancreatitis can develop walled-off necrosis (WON) more than 4 weeks after the onset of the disease (4).
Determinant-based classification (DBC) of AP was developed and published almost simultaneously with RAC (7). In DBC, the disease is classified as mild, moderate, severe, or critical depending on the presence of necrosis (sterile or infected) and/or organ failure (transient/persistent). Both RAC and DBC accurately classify the severity of AP in subgroup of patients (8). RAC reflects the early dynamic nature of AP and is suggested to be clinically more relevant than DBC. RAC has gained more popularity and is currently more often used in studies and clinical work than DBC.
Severity Assessment of AP
Several methods for assessing the severity of AP have been proposed. Clinical findings alone have been shown to lack the required sensitivity and specificity.
Multiple Scoring Systems
Several multifactorial scoring systems, such as Ranson’s score, modified Glasgow/Imrie score, bedside index for severity in AP (BISAP), harmless acute pancreatitis score (HAPS), Acute Physiology and Chronic Health Evaluation II (APACHE-II score), Panc 3, and the Japanese severity score have been introduced over the years (9–12). Most of them are, however, complex and time-consuming, which limit their routine clinical use in the emergency units. Furthermore, comparison of existing clinical scoring systems in the prediction of persistent organ failure in patients with AP also suggested limited clinical use (13). The BISAP and HAPS scores represent the simplest scoring systems that can be used in the clinics in spite of the limitations (14).
According to the International Association of Pancreatology (IAP)/the American Pancreatic Asso-ciation (APA) guidelines, systemic inflammatory response syndrome (SIRS) criteria (2 or more positive, such as body temperature > 38 °C or 36 °C; heart rate > 90/min; respiratory rate > 20/min or PaCO2 < 4.3 kPa; and leukocyte count > 12 × 109/L or <4 × 109/L or 10% immature forms) are the best predictor of severe AP at admission and at 48 h (15).
The development of organ dysfunction and failure can predict the severity of AP (Table 1). As previously mentioned, along with RAC, organ failure assessment can be accomplished by MODS or SOFA score. Mentula et al. (16) showed that also interleukin (IL)-10 or calcium values can be used in predicting organ failure in severe AP. Severe AP and persistent organ failure can also be predicted with soluble CD73, IL-6, and hepatocyte growth factor (HGF) (17, 18).
Classification of the severity of acute pancreatitis (7).
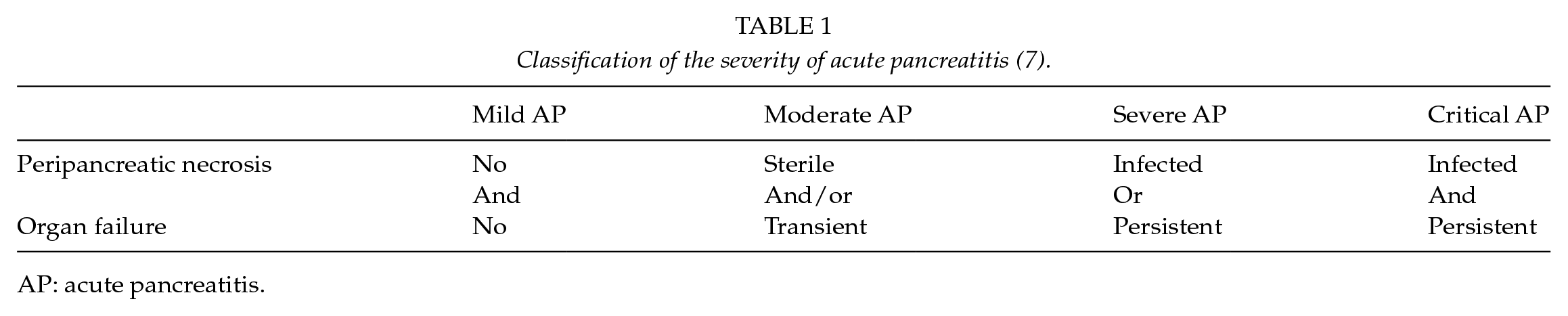
AP: acute pancreatitis.
Single Markers
Numerous individual markers have been studied for assessing the severity of AP. They include C-reactive protein (CRP), hematocrit, creatinine, blood urea nitrogen (BUN), procalcitonin, trypsinogen activation peptide (TAP), trypsinogen-2, and inflammatory markers such as IL-6 and soluble urokinase-type plasminogen activator receptor (suPAR) (19–24). Most frequently used single marker of the severity of AP is CRP despite of the delay in the increase in the values even in severe AP (19). Several single markers are in only limited routine clinical use due to various limitations (22–24).
Imaging Studies
The contrast-enhanced tomography (CT) is able to detect necrosis in the pancreas. Computed tomography severity index (CTSI) was shown to be a good predictor of both mortality and AP severity (14). However, CT has limitations for the identification of necrosis on CT early in the course of the disease. Thus, the routine use of CT in diagnosing AP is not recommended by the APA guidelines (25).
Recurrent Acute Pancreatitis
The patient is considered to have a recurrent acute pancreatitis (RAP) according to a new mechanistic definition if there have been two or more well-documented separate attacks of AP with complete resolution for more than 3 months between the attacks (26). The frequency of RAP after the first attack of AP is 17%–30% (26–29). Defining the etiology of the acute attack is the key to prevent further AP attacks. Different etiologies are presented in Table 2.
Possible etiological and concomitant factors of acute pancreatitis.
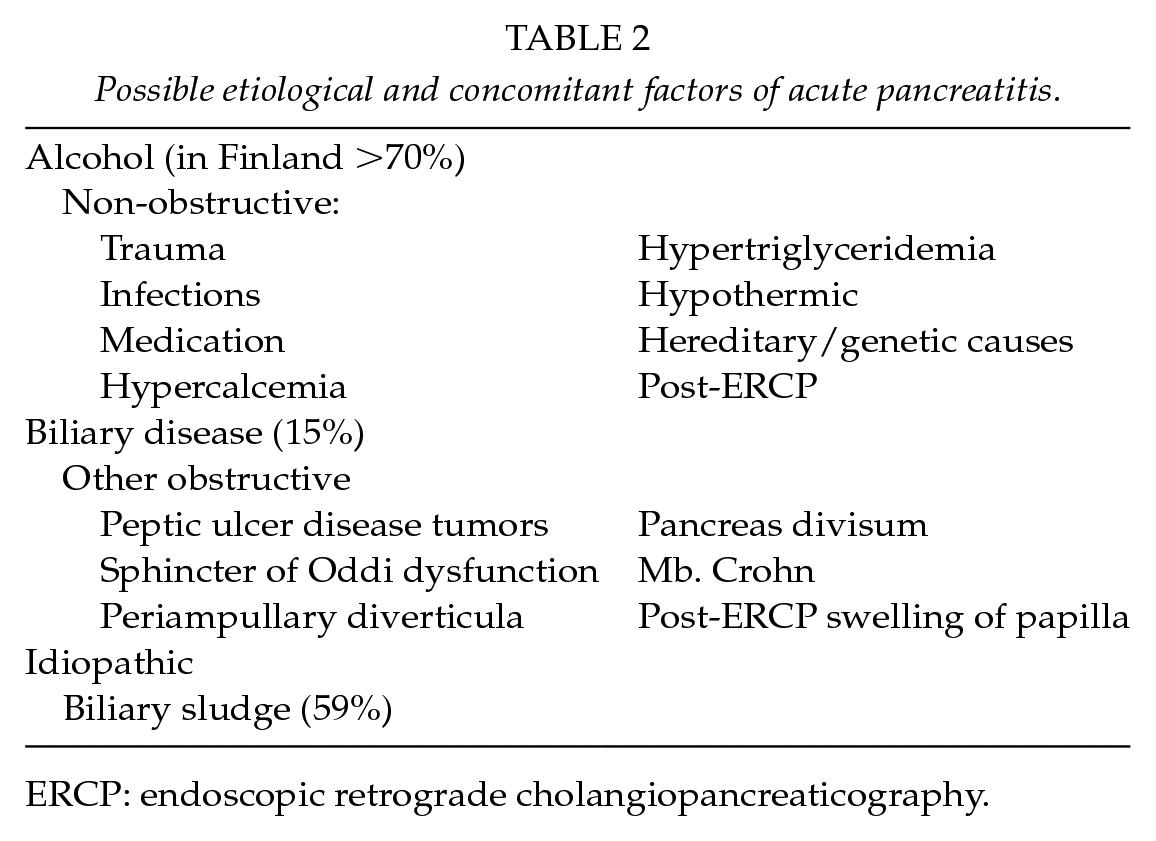
ERCP: endoscopic retrograde cholangiopancreaticography.
Alcoholic Pancreatitis
Only 5%–10% of heavy alcohol consumers develop AP attacks (30). In Finland, 70% of AP patients consume alcohol heavily (1). After the first attack of alcohol-induced AP, 46% of the patients develop at least one recurrence within 10- to 20-year follow-up (31); 80% of the patients who develop recurrence do so within 3–5 years (31). Individual alcohol consumption level did not associate with RAP nor did the type of alcohol beverage (32). Smoking and obesity are also concomitant risk factors for alcohol-induced RAP (27, 33).
If alcohol consumption continues after the first alcohol-induced AP, there is an increased risk for recurrent attack or chronic pancreatitis (CP) (32, 34, 35). In a Finnish study, no patient with alcohol abstinence after the first AP developed RAP (32, 36). In a randomized trial, supportive intervention against continuous alcohol consumption every 6 months reduced the RAP rate. In a repeated intervention (every 6 months) group, 8% of patients recurred within 2 years. In a single intervention group, 21% recurred. The single intervention was done at the hospital during the first acute attack. The patients who refused to take part to the study had the highest recurring rate of 28% (36).
Biliary Pancreatitis
To prevent biliary pancreatitis from recurring, the gall bladder removal is necessary. When the acute biliary pancreatitis attack is mild, the gall bladder is recommended to be removed during the hospital stay of the first acute attack (37–39). In reality, the gall bladder removal is often delayed (35, 37). When the AP attack is severe, it is recommended to wait until the patient has recovered and local signs of inflammation have resolved (40, 41). If the patient is not fit for surgery, endoscopic sphincterotomy should be performed (42). If the AP recurs even after cholecystectomy, endoscopic sphincterotomy should be performed to remove possible residual or later developed stones from the bile duct (41).
Metabolic RAP
Patients with hypercalcemia may develop AP, and it may recur unless the calcium levels can be normalized. In many cases, hyperparathyroidism is behind elevated calcemic concentration (43). The treatment of hypercalcemia is essential for the prevention of RAP. In addition, elevated serum triglyceride levels (>500–1000 mg/dL) have been associated with AP (26, 44, 45). Elevated triglycerides may be associated with alcohol-induced pancreatitis (46). High triglyceride levels may also be part of metabolic syndrome, and treating the initial cause may help to prevent RAP. There may also be other attempts to treat hypertriglyceridemia such as insulin, heparin, fibric acids, omega-3 fatty acids, plasmapheresis, but no guidelines for the treatment exist (47). Coffee drinking has been associated with protective effect against RAP (48).
Inherited RAP
There are several mutations that are associated with RAP such as cationic trypsinogen (PRSS1), pancreatic secretory trypsin inhibitor (PSTI/SPINK1), and cystic fibrosis transmembrane conductance regulator (CFTR). Especially in the Finnish population, the prevalence of SPINK1 mutation is 3.4%, 7.6% in alcohol-induced pancreatitis patients, and only 1.0% in heavy alcohol consumers without pancreatitis (49). In a multicenter study of 155 children with RAP, 48% had some of the most common mutations. SPINK1 mutation was found in 13% of the patients (50). Among 134 patients with no known etiologic factor and the first AP episode under 35 years of age, a pathologic genetic variant was found in 58% (51). The role of genetic testing can be able to give the patients information to avoid other risk factors (26, 52).
Idiopathic RAP
It has been considered if the etiology behind most idiopathic pancreatitis could be biliary microlithiasis undetectable by conventional methods or sphincter of Oddi dysfunction. Ursodeoxycholic acid (12 mg/kg) treatment with or without endoscopic biliary sphincterotomy preventing RAP is still not proven (26, 42, 53). In a Finnish multicenter randomized study, 85 patients were considered as idiopathic pancreatitis after conventional exclusion of other etiologies. Thirty-nine patients had laparoscopic cholecystectomy and the RAP rate was 20.5%, whereas in the follow-up group, the RAP rate was 50%, which is significantly higher. Inside the removed gall bladder, stones or biliary sludge was present in 59% of the patients. The remaining 41% of the patients did not have genetic testing (54). The true frequency of idiopathic pancreatitis is reduced, the more extensive the search for less common etiologies. Etiologic diagnosis of unexplained RAP is presented in Fig. 1.
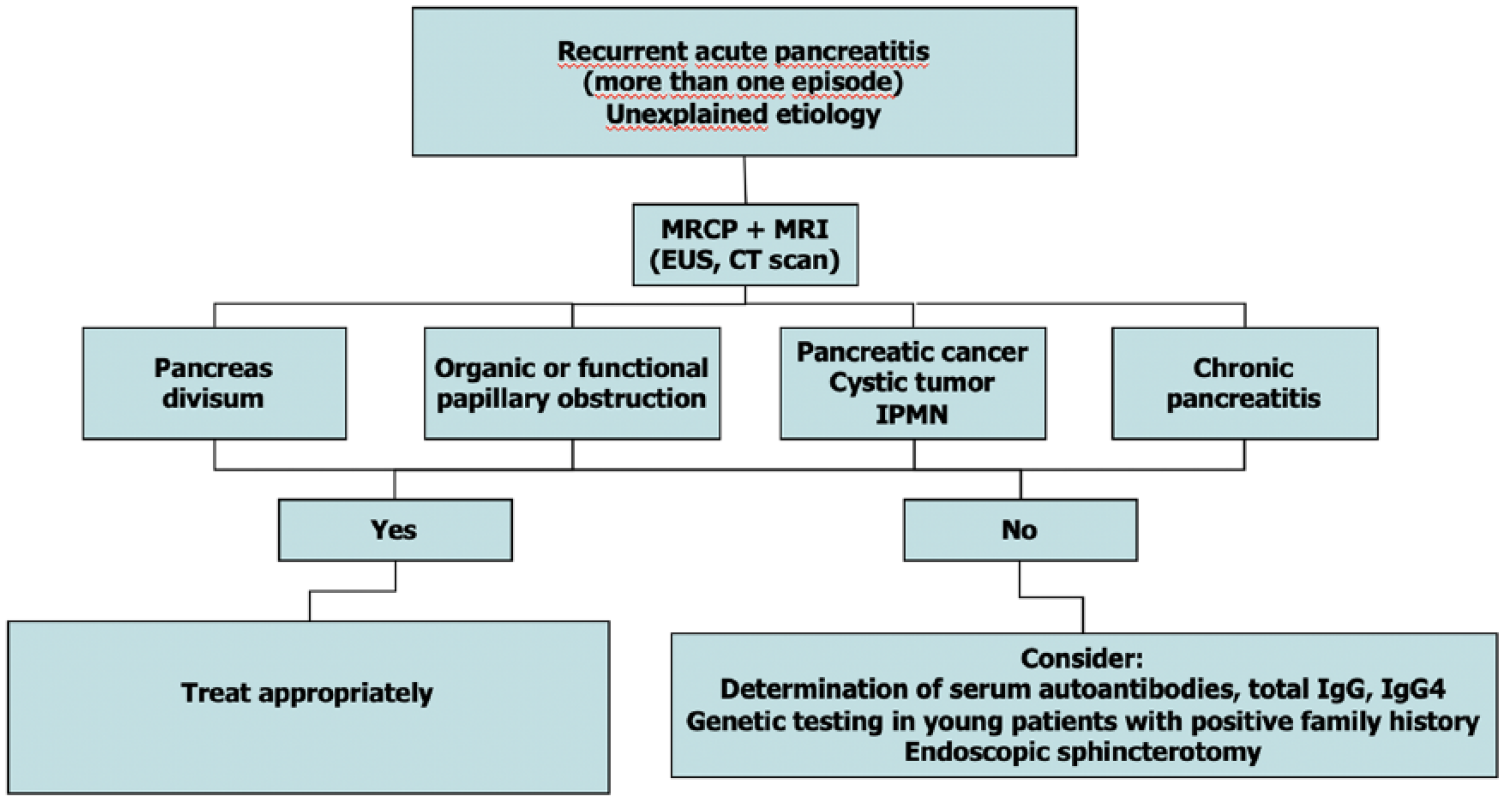
Etiologic diagnosis of recurrent unexplained attack of acute pancreatitis (55).
Other Risk Factors
Type 2 diabetes increases the risk for AP 1.8–2.9 times, particularly high risk, in young patients. Excess risk reduced by antidiabetic drugs (56). Pancreas divisum may also cause obstruction and pancreatitis. Stenting through minor papilla can reduce re-hospital admissions to one-third at least in short term (57). In addition, smoking has been associated as a risk factor for RAP after the first AP episode, but no randomized studies have been done whether cessation would prevent from recurrences (58).
Medications Associated with AP
There are several pharmaceutical agents that have been associated with AP. The most common agents and probabilities of associations to AP are presented in Table 3. Discontinuing the probable causing medication with follow-up may be helpful for the patient.
The most typical medical agents to associate with recurrent acute pancreatitis.

Some medications have also been used to prevent RAP such as antioxidant supplementations, octreotide, pancreatic enzymes, ursodeoxycholic acid, and statins. High-quality and convincing data supporting the medical therapy for recurrent AP are lacking. Many times, the knowledge is based on case reports only (26).
Holistic Prevention of RAP
Primary, secondary, and tertiary prevention strategies should be systematically employed in order to lessen the effect of AP and its sequelae (29). Holistic approach includes the education of general population and patients with prior AP attack, effective in-hospital management to prevent repeated attacks, and screening of high-risk patients. Prevention involves public health specialists, primary care physicians, gastroenterologists, surgeons, and endocrinologists (29).
Conclusion
The RAC assesses mild, moderate, and severe pancreatitis. Furthermore, peripancreatic fluid collections were given new nomenclature. To prevent most of the RAP episodes, it needs alcohol abstinence and repeated interventions, and removal of the gall bladder in biliary AP and in 59% with supposed idiopathic AP. The aim is to create specific treatment including the holistic prevention principles.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
