Abstract
Purposes:
To assess overall survival and to determine factors predicting outcome after thoracic endovascular aortic repair.
Materials and methods:
A Retrospective analysis was performed on a cohort of 212 consecutive patients (165 men and 47 women; mean age 64 years) who underwent thoracic endovascular aortic repair in a tertiary referral center for aortic disease. Main indications were true thoracic aortic aneurysm (n = 58; 27.6%), traumatic aortic rupture (n = 33; 15.7%), anastomotic pseudoaneurysms (n = 23; 10.9%), chronic type B aortic dissection (n = 22; 10.5%), and symptomatic, acute type B dissection (n = 21; 10.0%). In 79 patients (37.3%), a hybrid procedure, including supra-aortic rerouting, was performed. Kaplan–Meier estimates were used for overall survival and Cox regression models were used for univariable analysis of the association between risk factors and survival.
Results:
Proximal landing zones were predominantly zone 3 (n = 66; 31.3%), zone 2 (n = 63; 29.9%), and zone 1 (n = 38; 18%). In-hospital mortality was n = 18 (8.5%). Overall survival was 79.6%, 65.9%, and 51.1% at 2, 5, and 10 years, respectively; better overall survival was shown for traumatic aortic rupture, anastomotic pseudoaneurysms, and chronic posttraumatic pseudoaneurysms (p < 0.05). Clinical risk factors influencing overall survival include prior coronary bypass surgery, atrial flutter, arterial hypertension, renal failure, chronic obstructive pulmonary disease, and associated abdominal aortic aneurysm (p < 0.05).
Conclusions:
Thoracic endovascular aortic repair is an effective treatment option for various thoracic aortic diseases with highest survival rates for traumatic aortic rupture and anastomotic pseudoaneurysms. Several clinical parameters are identified as risk factors for overall survival.
Keywords
Introduction
Since the pioneering work by Dake et al. 1 , thoracic endovascular aortic repair (TEVAR) has revolutionized the operative management of many thoracic aortic diseases and is considered the principal treatment option compared to open surgical management in many centers owing to the lower associated perioperative morbidity and mortality in selected patients2,3. In addition, present clinical guidelines4 –6 recommend endovascular treatment as a first-line option for some indications like complicated acute type B aortic dissection, mural hematoma, penetrating aortic ulcer, and traumatic aortic rupture, but it is still emphasized that there is a lack of clear evidence 7 .
Several follow-up studies demonstrated encouraging short-term outcomes after thoracic endovascular repair using a variety of commercially available stent-grafts4,8 –11, but long-term results remain sparse. Few studies report good long-term survival in specific indications11 –14, whereas some reported worse long-term survival after initial lower perioperative mortality 15 .
The purpose of this study was to determine early and late complications, reintervention rate, and overall survival, and to assess risk factors affecting the long-term outcome after TEVAR. A retrospective analysis was therefore carried out on a cohort of 212 consecutive patients who underwent TEVAR from August 1998 to July 2017 at the authors’ institution.
Materials and Methods
Study Design
This is a retrospective, monocentric study, using a prospectively maintained electronic database and including a cohort of 212 patients who underwent a TEVAR procedure between August 1998 and July 2017 at an academic tertiary care institution for aortic diseases. The study was approved by the institution’s Ethics Committee (S 61910), and all patients provided informed consent before the TEVAR procedure. Indication for TEVAR, procedural outcomes, early and late complications, reinterventions, and overall survival were gathered from the patients’ electronic medical records and Picture Archiving and Communication System (PACS) (Agfa, Mortsel, Belgium).
Patients and Demographics
From August 1998 to July 2017, 212 consecutive patients including 165 men (77.8%) and 47 women (22.1%) with a mean age of 58.5 years (standard deviation (SD) = 17.9) (Table 1) presenting with various thoracic aortic diseases underwent thoracic endovascular stent-graft placement either alone or in combination with open surgery (hybrid procedure).
Indications for TEVAR.
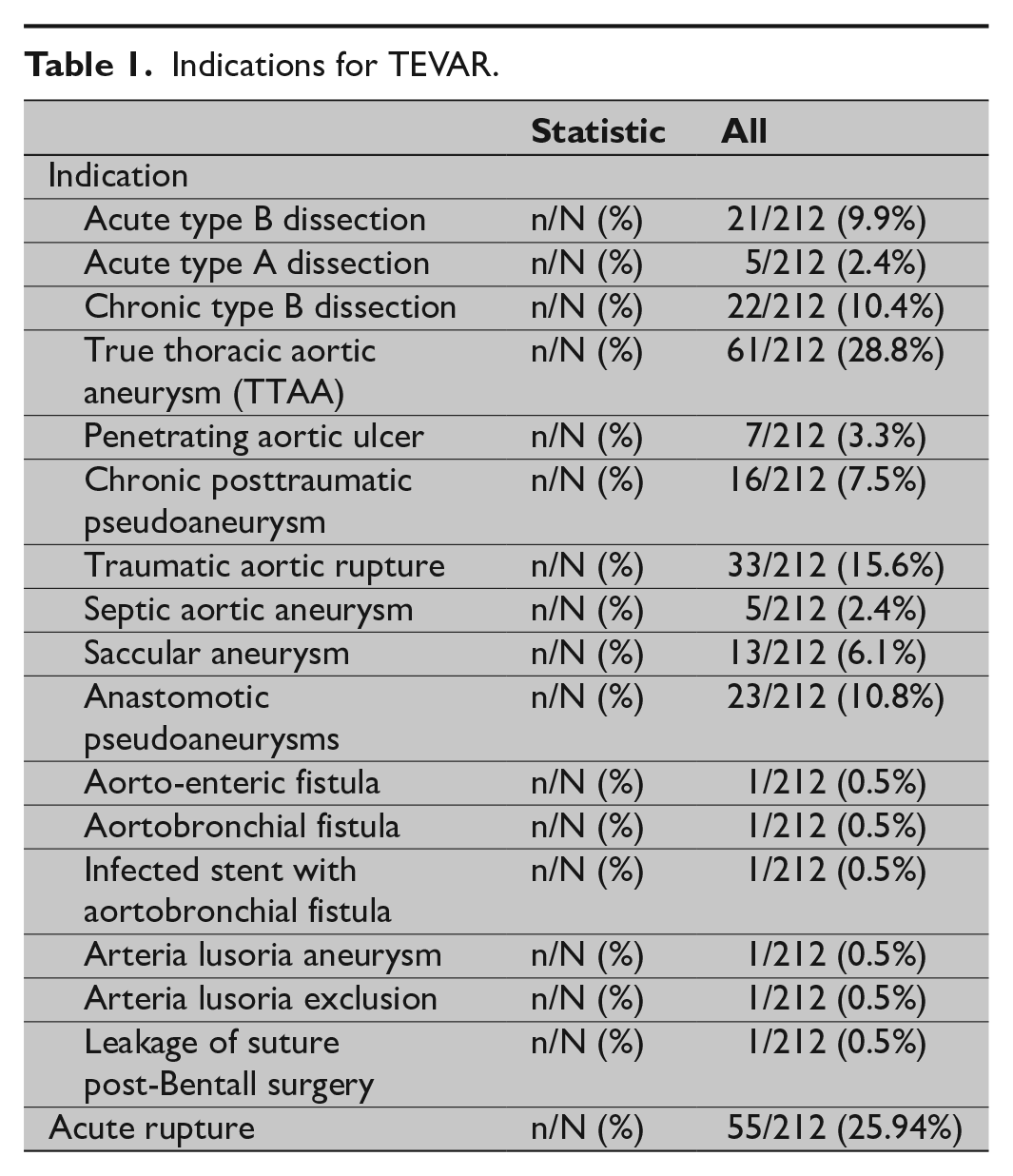
Median days from diagnosis to intervention were 29 days; 64 procedures (30.2%) were performed in an acute setting (less than 48 h after diagnosis) and 48 of those (22.6%) were performed within 24 h of diagnosis. The American Society of Anesthesiologists (ASA) physical status system was determined in 183 patients; the majority of patients (n = 99; 54.1%) being ASA 3 (Table 2). All patients underwent preoperative imaging to assess the feasibility of TEVAR and to plan the stent-graft. Depending on the type and condition of the thoracic aortic disease, various imaging modalities, including computed tomography angiography (CT-A), magnetic resonance imaging (MRI), transthoracic ultrasound (TTU) or transesophageal ultrasound (TEU), and catheter-angiography (CA) were used. Length of proximal and distal landing zones for successful stent-graft deployment was 2 cm and oversizing of the stent-graft depended on the underlying aortic disease—acute dissections and traumatic ruptures: 10%; chronic dissection, degenerative aneurysm, pseudoaneurysm, penetrating ulcer, and others: 10%–20%. The proximal landing zone for TEVAR was classified according to Fillinger et al. 16 and included five groups.
Patients’ characteristics, associated comorbidities, and ASA-status before TEVAR.
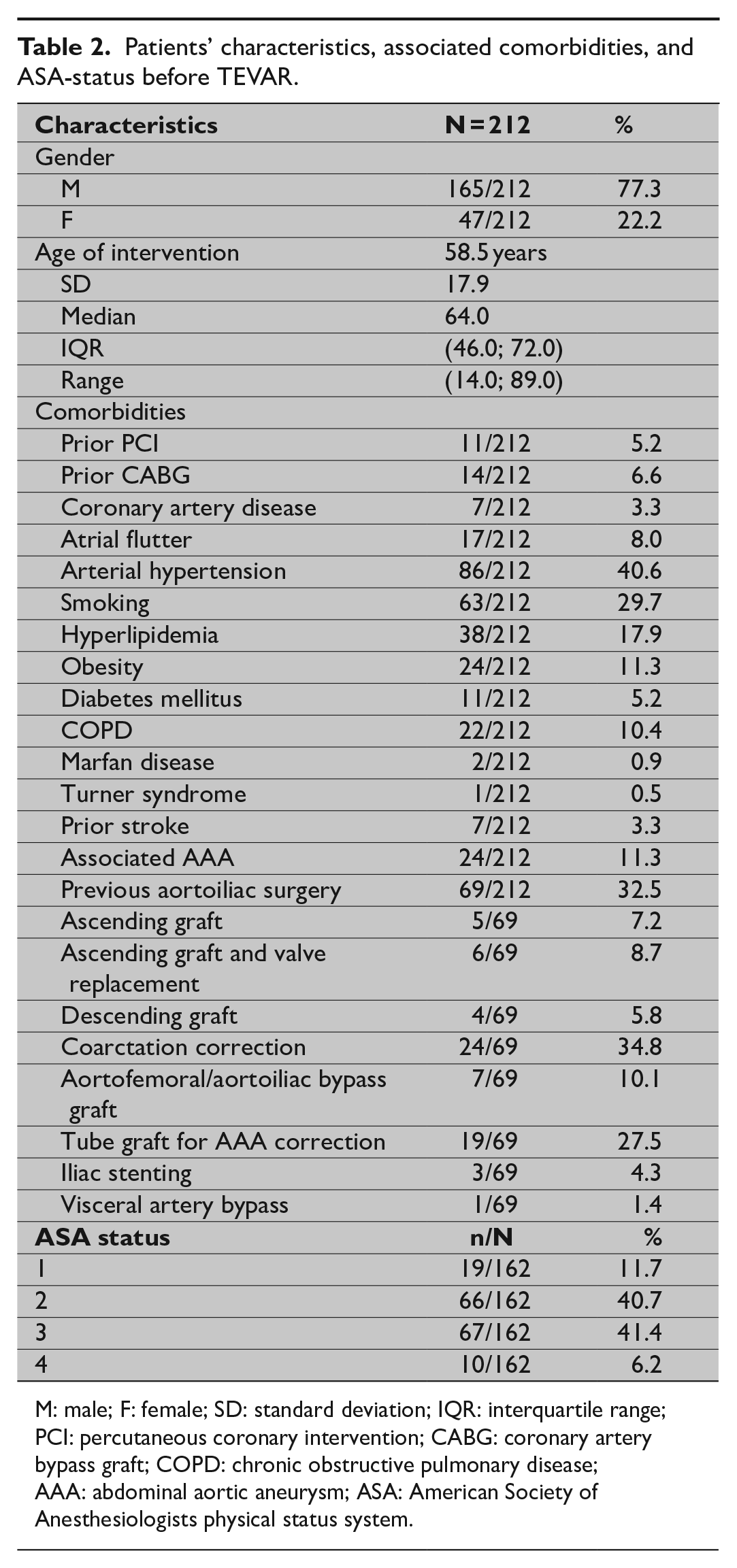
M: male; F: female; SD: standard deviation; IQR: interquartile range; PCI: percutaneous coronary intervention; CABG: coronary artery bypass graft; COPD: chronic obstructive pulmonary disease; AAA: abdominal aortic aneurysm; ASA: American Society of Anesthesiologists physical status system.
Tevar Procedure
TEVAR was performed by a team consisting of a vascular surgeon and an interventional radiologist, both with more than 5 years of experience in aortic endografting. All procedures were performed under general anesthesia in an angiography suite equipped with digital subtraction angiography (Angiostar Plus; Siemens Healthcare, Forchheim, Germany) or in the operating theater with use of a mobile angiography system (Siremobil; Siemens) in case of a hybrid procedure; after 2008, the TEVAR procedure was performed in the operating theater with use of hybrid angiographic equipment (Artis Zeego, Siemens). In case of a hybrid procedure, supra-aortic vessel debranching, performed to achieve a suitable proximal landing zone, was done in the same session as the thoracic stent-graft placement. Technical details of thoracic stent-graft placement have been described previously 17 ; briefly, after surgical cutdown or after gaining percutaneous access to the right common femoral artery using a closure device (Preclose; Abbott, Abbott Park, IL, USA), a 7-French sheath is introduced and a pigtail catheter is inserted into the aortic arch using a steerable hydrophilic guidewire (Glidewire; Terumo Europe, Leuven, Belgium). The latter is exchanged for a stiff guidewire (Back-up Meier; Boston Scientific, Natick, MA, USA or Lunderquist; Cook Medical, Bloomington, IN, USA). The pigtail is reintroduced in a 5-F sheath placed percutaneously in the contralateral common femoral artery or right brachial artery for peri- and postoperative angiographic imaging.
The stent-graft is introduced through the right common femoral artery or through a surgical conduit and deployed after angiographic control imaging. If more than one stent-graft was placed, an overlap of at least 3 cm between two stent-grafts was considered and stent-graft modeling, using a low-pressure balloon, was performed if indicated, except in cases of traumatic rupture or dissection.
Prophylactic drainage of cerebrospinal fluid to prevent spinal cord ischemia was performed if the stent-graft was planned to land at the level of the eighth or lower thoracic vertebra; rapid pacing was performed in cases of a type 0-1 according to Fillinger et al. 16 .
Outcome Criteria and Clinical and Radiological Follow-Up
Patients were followed up clinically by the attending vascular surgeon and radiologically, according to the EUROSTAR-follow-up scheme 18 , by the attending interventional radiologist. Radiological follow-up was performed at 3, 6, 12 months, and yearly thereafter, mainly with use of CT-A or MRI if there were contra-indications for CT-A. Outcome criteria were assessed according to Fillinger et al. 16 ; perioperative, early and late postoperative phase is defined as <24 h after start of the TEVAR procedure, <30 days and >30 after the TEVAR operation, respectively. Reinterventions were performed based on clinical and/or radiological findings and included open surgical and endovascular interventions.
Statistics
Summary statistics are presented as means and SD/medians and interquartile range (IQR) for continuous variables and as frequencies and percentages for categorical variables. Kaplan–Meier estimates are used for overall survival. The log-rank test is used for testing differences in survival between indications.
The association between two categorical variables was tested by chi-square test or Fisher’s exact test, as appropriate. The association between ordinal and categorical variables was tested by the Mann–Whitney U test. Cox regression models are used for univariable analysis of the association between risk factors and survival. Results are presented as hazard ratios (HR) with 95% confidence intervals (CI). All tests are two-sided; a 5% significance level is assumed for all tests. Analyses were performed using SAS software (version 9.4 of the SAS System for Windows).
Results
Indications for Tevar
The most common indications for TEVAR were true thoracic aortic aneurysm (n = 58; 27.4%), traumatic aortic rupture (n = 33; 15.6%), anastomotic pseudoaneurysm (n = 23; 10.8%), acute type B aortic dissection (n = 21; 9.9%), chronic type B aortic dissection (n = 22; 10.4%), chronic posttraumatic pseudoaneurysm (n = 16; 7.5%), and saccular aneurysm (n = 13; 6.1%) and are summarized in Table 1. Emergency operative repair was associated with acute thoracic aortic rupture in n = 55 (25.9%) of the patients.
Preoperative Imaging
Preoperative imaging included CT angiography in n/N = 180/212 (84.9%), MRI in n/N = 23/212 (10.8%), TTU in n/N = 76/212 (35.8%), TEU in n/N = 26/212 (12.3%), and catheter-directed angiography in n/N = 111/212 (52.4%). The number of days between the initial diagnosis and day of TEVAR was a mean of 160 days (median = 29 days; IQR = (1.0; 72.5 days); SD = 595.9 days).
Hybrid Procedures
Vascular access for stent-graft introduction was mainly performed through the right common femoral artery after surgical cutdown (n = 196; 92.4%) or percutaneously (n = 2; 1%). In a minority of patients (n = 14; 6.6%), a surgical conduit was created in order to create access to the diseased thoracic aorta: conduit to the external iliac artery (n = 2), conduit to the common iliac artery (n = 3), conduit to the common carotid artery (n = 1), conduit to the infrarenal abdominal aorta (n = 6), and conduit to the ascending thoracic aorta (n = 2). Rerouting of the supra-aortic vessels was performed in 79 patients (37%) with carotidocarotid bypass being the commonest procedure (n = 36; 45.6%), followed by carotidosubclavian bypass (n = 21; 26.6%) and total arch rerouting (n = 16; 20.3%). In one case, a preoperative coil embolization of the left subclavian artery was performed to avoid potential postoperative type II endoleak. In another 20 patients (9.4%), TEVAR was combined with other vascular surgical procedures, including open repair of thoracic arch (n = 5; 25%), open repair of abdominal aortic aneurysm (n = 4; 20%), endovascular abdominal aortic repair (EVAR) (n = 6; 30%), carotid stenting (n = 1; 5%), iliac stenting (n = 1; 5%), aortobifemoral grafting (n = 1; 5%), and iliaco-mesenteric bypass graft (n = 2; 10%).
Intraoperative Tevar Results
In 210 patients (99.0%), the stent-graft could be deployed in the thoracic aorta. In two patients (1%), the stent-graft could not be positioned in the arch owing to the gothic configuration of the arch and to a sudden cardiac collapse and death of the patient during TEVAR, respectively.
A total of 316 endografts were used with no propensity for types of endografts or for pathologies or zones. Different thoracic stent-grafts used are summarized in Table 3. Proximal stent-graft landing zones mainly included zone 2 (n = 63) and zone 3 (n = 65) according to Fillinger et al (16). In 142 patients (67%), only one endograft was used, two overlapping endografts were used in 45 patients (21.2%), and three or more endografts were used in 25 patients (11.8%). The mean total stent length was 174.4 mm with a mean nominal stent diameter of 34.4 mm.
Procedural parameters of the TEVAR procedure.
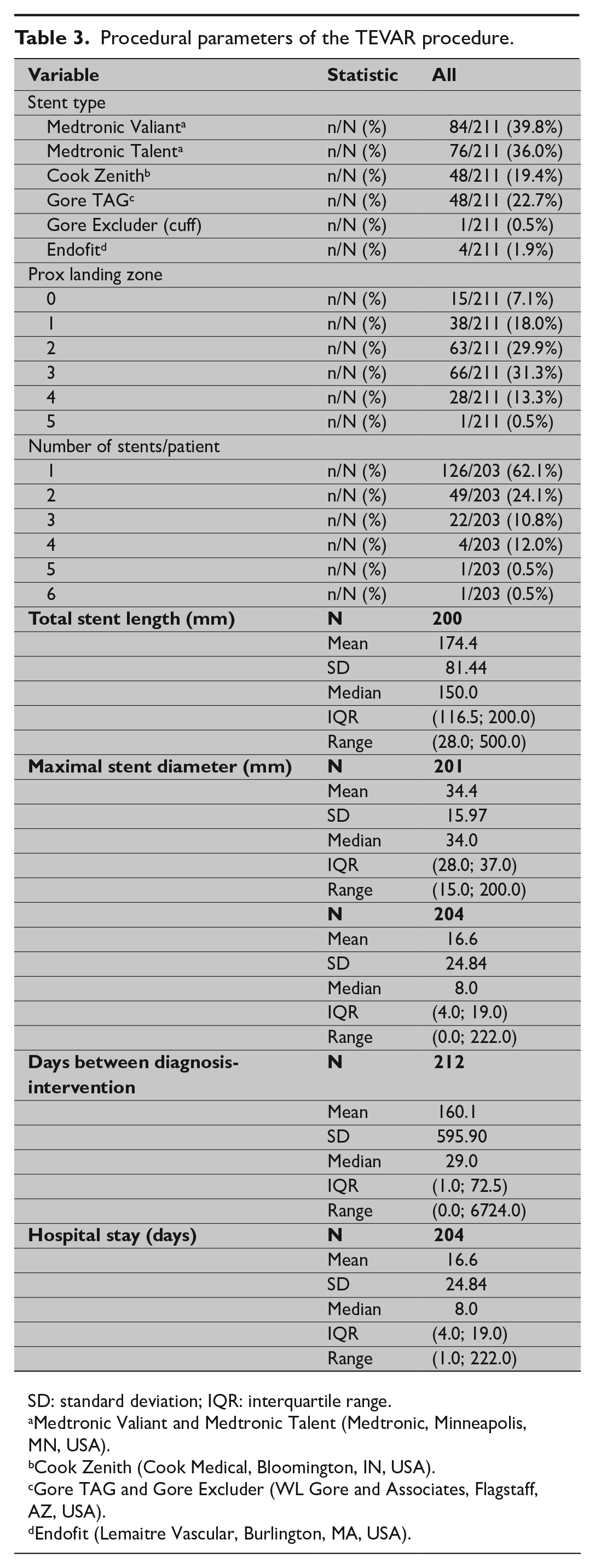
SD: standard deviation; IQR: interquartile range.
Medtronic Valiant and Medtronic Talent (Medtronic, Minneapolis, MN, USA).
Cook Zenith (Cook Medical, Bloomington, IN, USA).
Gore TAG and Gore Excluder (WL Gore and Associates, Flagstaff, AZ, USA).
Endofit (Lemaitre Vascular, Burlington, MA, USA).
Intraoperative, Early, and Late Complications
The mean time from intervention to complication was 21.2 months. Complications were encountered in 84 patients (39%), with endoleak accounting for almost half of them (n = 41; 48%) (Table 4). Two consecutive complications were seen in 13 patients and three consecutive complications were seen in one patient.
Complication rate per indication for TEVAR.
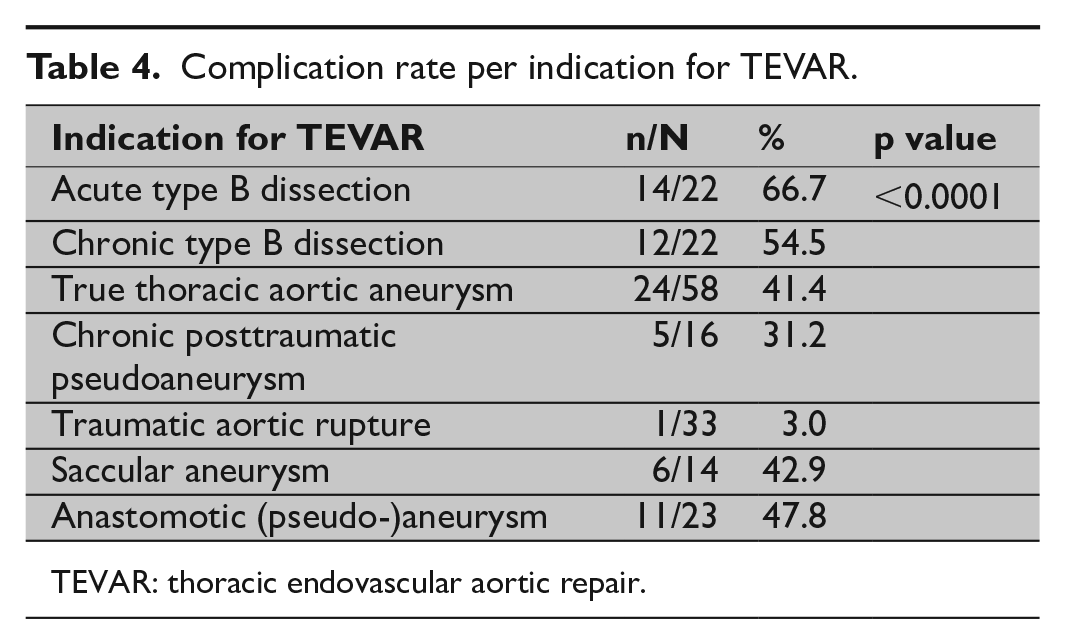
TEVAR: thoracic endovascular aortic repair.
Intraoperative complications were encountered in 11 patients (5.2%), including persistent type Ia endoleak (n = 1), inadvertent covering of the left subclavian artery with subclavian steal syndrome (n = 1), wire perforation of the left ventricle (n = 1), and rupture of the aorta (n = 4), which resulted in perioperative death in four patients (1.9%).
Thirty-one early (<30 days after TEVAR) complications were encountered in 22 patients (10.4%). In the majority of cases (n = 13; 59.1%), an endoleak was identified on follow-up imaging: type Ia (n = 5), type Ib (n = 2), type II (n = 5), and type III (n = 1). The remaining early complications included aortic rupture (n = 3), retroperitoneal hemorrhage (n = 2), retrograde type A dissection (n = 3), esophageal necrosis with aortoesophageal fistula (n = 1), aortic dissection distal to the endograft (n = 1), stroke (n = 4), access artery occlusion (n = 2), and bowel infarction (n = 2).
Fifty-five late (>30 days after TEVAR) complications were identified in 47 patients (22.2%). In nearly half of these cases (n = 27; 49.1%), an endoleak was identified on follow-up imaging type Ia (n = 8), type Ib (n = 4), type II (n = 5), and type III (n = 9); one patient presented with a combined endoleak type Ia, Ib, and III. Other late complications included a retroperitoneal hematoma (n = 2), stent-graft infection (n = 3), esophageal necrosis (n = 1), aortic thrombus distal to the endograft (n = 1), retrograde type A dissection (n = 1), increased aortic diameter distal to the endograft (n = 5), dissection distal to the endograft (n = 1), increased aortic diameter without visible endoleak (n = 8), stenosis of double transposition bypass (n = 1), left arm claudication (n = 2), bowel infarction (n = 2), and paraplegia (n = 1).
Overall complication rate with regard to underlying thoracic aortic disease is summarized in Table 4; overall complication ratio was highest for acute and chronic type B dissection with 66.7% and 54.6%, respectively, and lowest for acute traumatic aortic rupture (3%).
Reinterventions During Follow-Up
Reintervention to manage post-TEVAR complications was performed in 46 out of 84 patients (54.8%) including 30 endovascular procedures, 15 open surgical repairs, and one hybrid procedure (Table 5). Reintervention was indicated for 27 endoleaks (27/41; 65.9%), most commonly treated by additional stent-graft placement (n = 20/27; 74.1%) followed by coiling of the left subclavian artery (n = 4/27; 14.8%). A second complication after management of the first complication was identified in 13 patients (6.1%) and included aortic dissection distal to the thoracic endograft (n = 1), stent-graft infection (n = 1), infolding of the stent-graft (n = 2), aortobronchial fistula (n = 1), aortic thrombus distal to the endograft (n = 1), and increase in aortic diameter without visible endoleak (n = 3); reintervention to manage a second post-TEVAR complication was performed in 7 out of 13 patients (53.9%): additional stent-graft implantation (n = 5) and open repair (n = 2). One patient presented with paraplegia after additional stent-graft implantation for aortic thrombus distal to the endograft.
Reintervention for post-TEVAR complication management.
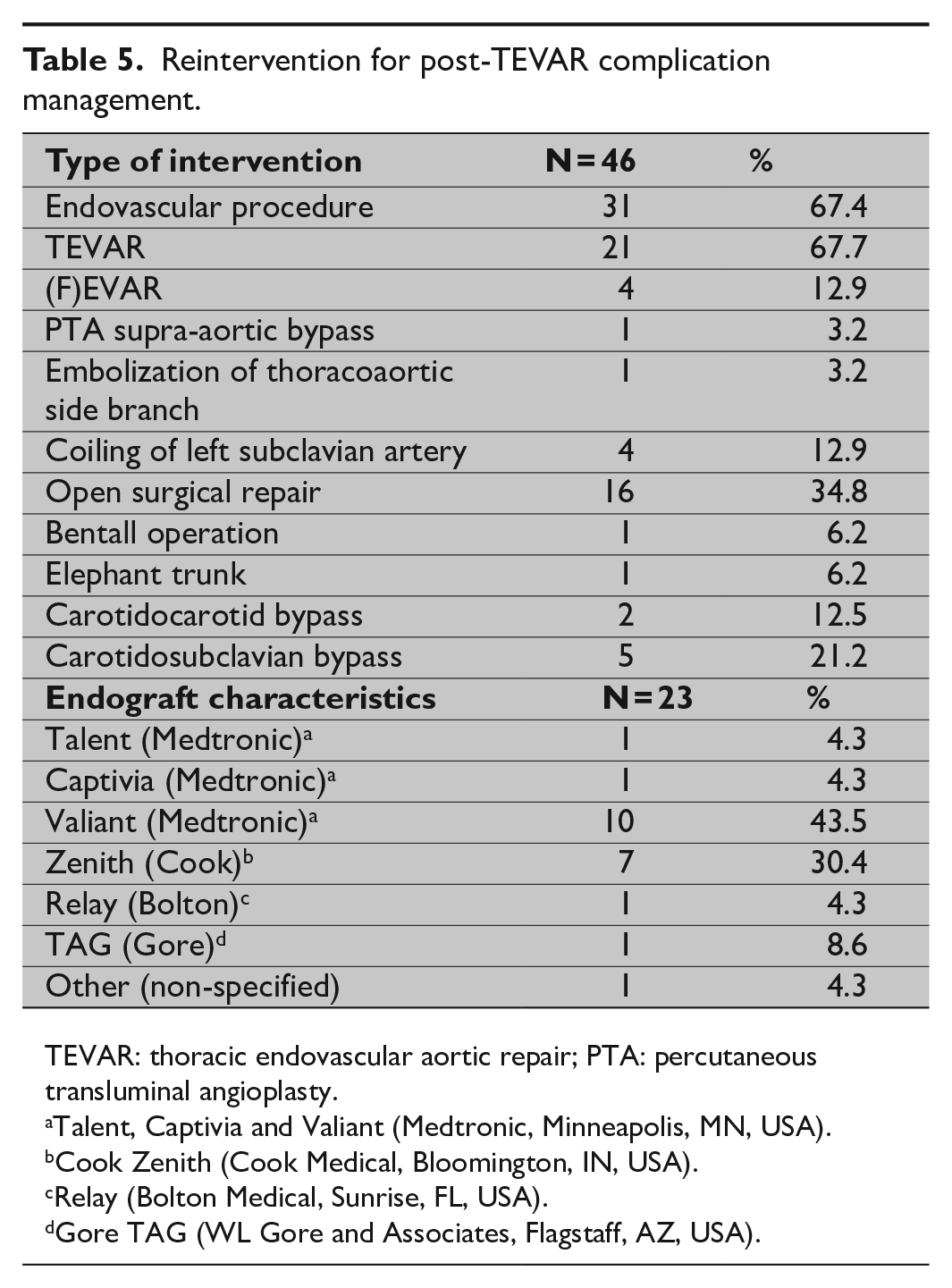
TEVAR: thoracic endovascular aortic repair; PTA: percutaneous transluminal angioplasty.
Talent, Captivia and Valiant (Medtronic, Minneapolis, MN, USA).
Cook Zenith (Cook Medical, Bloomington, IN, USA).
Relay (Bolton Medical, Sunrise, FL, USA).
Gore TAG (WL Gore and Associates, Flagstaff, AZ, USA).
Overall Survival
Mean hospital stay was 16.6 days (SD = 24.8 days; median = 8.0 days; IQR = 4.0; 19 days); mean follow-up was 80.9 months.
Short-term survival: 30-day mortality was 8.5% (n = 18/212) with a 30-day mortality for acute and elective cases of 17.2% (11/64) and 4.7% (7/148), respectively. The in-hospital mortality was n = 8.5% (n = 18/212).
Median overall survival was 11.5 years. Overall survival estimates after 2, 5, and 10 years were, respectively, 79.6%, 65.9%, and 51.1% (Table 6; Fig. 1). The highest 5-year survival rates were seen in traumatic aortic rupture (94%) and anastomotic pseudoaneurysm (83%). Slightly lower 5-year survival rates were seen with saccular aneurysm (71%) and chronic posttraumatic pseudoaneurysm (67%). The lowest 5-year survival rates were seen in acute type B dissection (62%), chronic type B dissection (60%), and true thoracic aortic aneurysm (53%) (Table 7; Fig. 2).
Overall survival estimates.
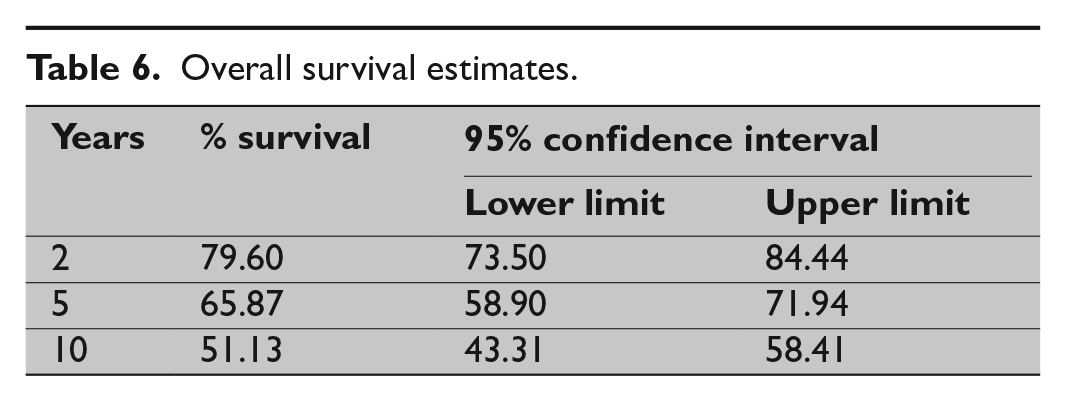
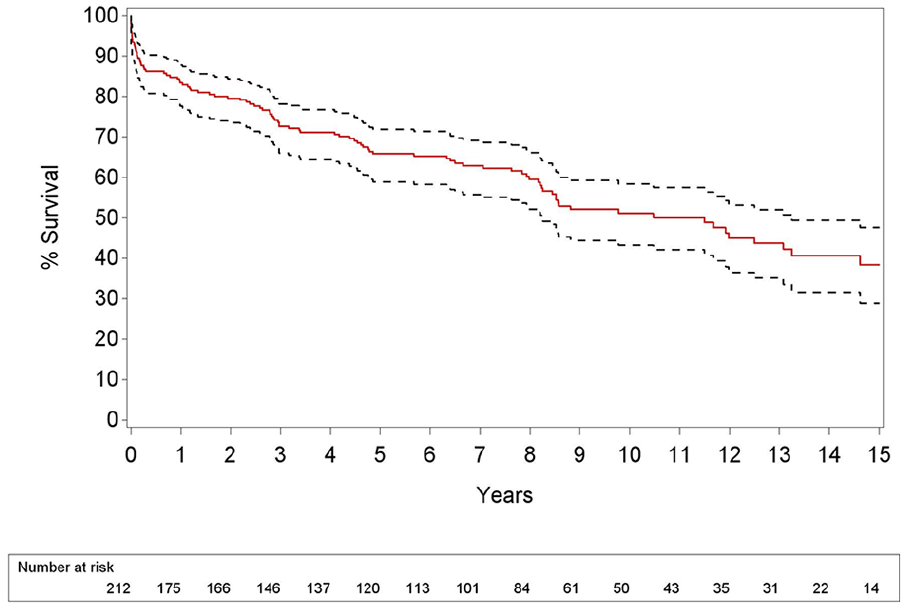
Kaplan–Meier estimates demonstrate an overall survival after thoracic endovascular aortic repair of 79.6%, 65.9%, and 51.1% after 2, 5, and 10 years, respectively.
Overall survival rates by indications.
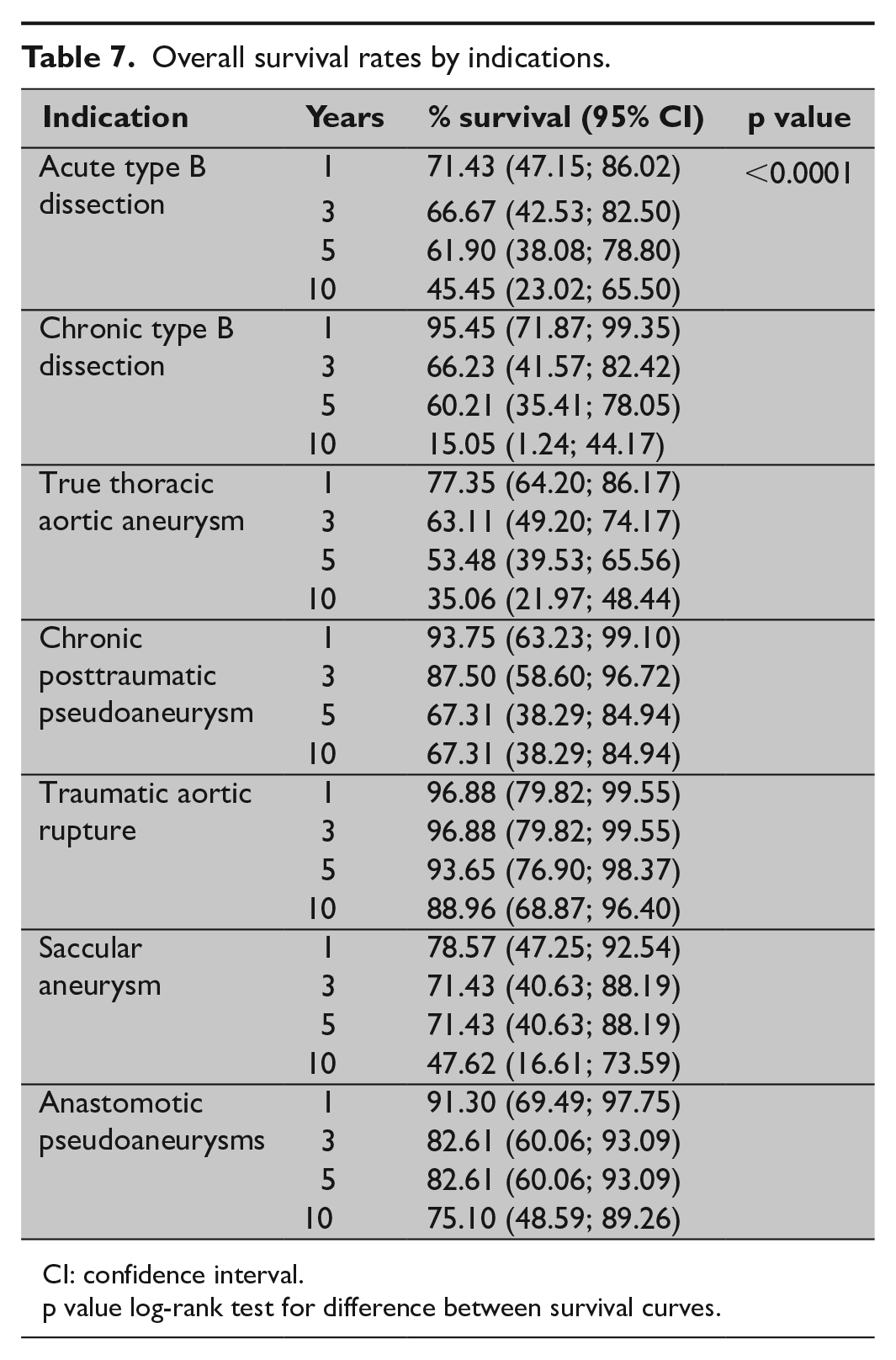
CI: confidence interval.
p value log-rank test for difference between survival curves.
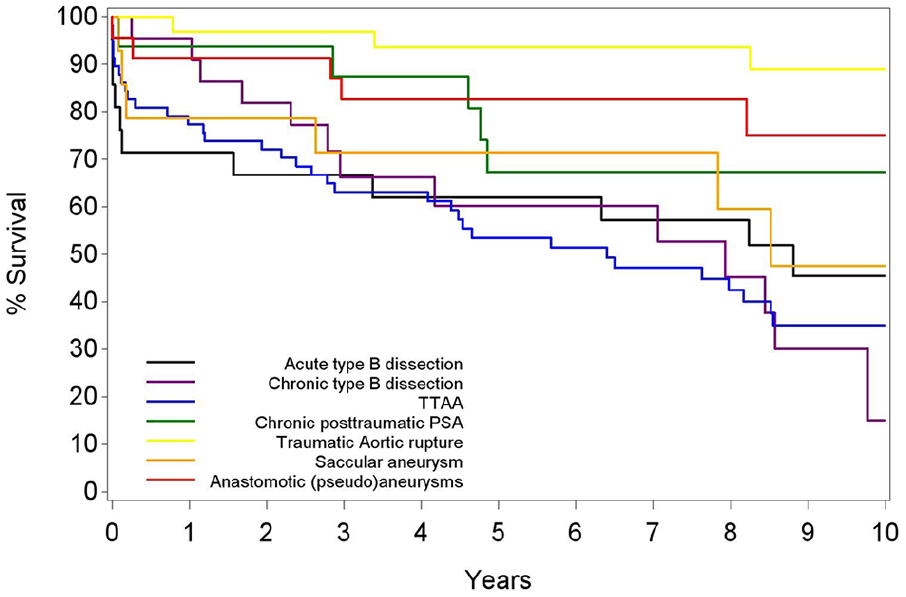
Better overall survival rates are found for traumatic aortic rupture (10-year survival of 88.96 (68.87–96.40) years), anastomotic pseudoaneurysm (10-year survival of 75.10 (48.59–89.26) years), and chronic posttraumatic pseudoaneurysm (10-year survival of 67.31 (38.29–84.94) years).
Risk Factors
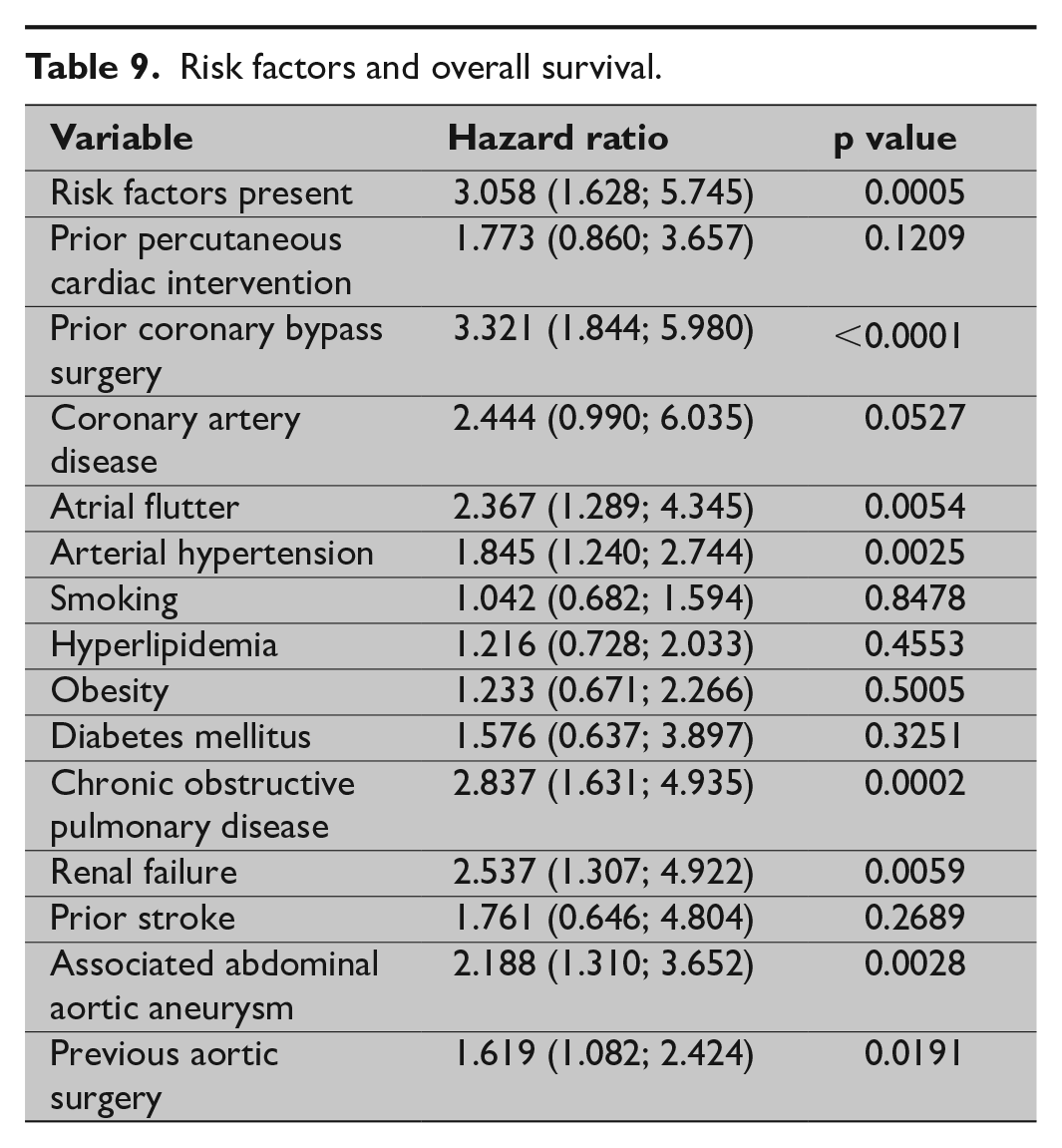
Clinical risk factors are summarized in Table 9. The highest HR were associated with prior coronary bypass surgery (HR = 3.3), chronic obstructive pulmonary disease (HR = 2.8), arterial hypertension, abdominal aortic aneurysm, atrial flutter, and renal failure (Table 9).
TEVAR with (n = 79) or without supra-aortic rerouting (n = 133) was associated with an overall complication rate of n = 42 (53.2%) and n = 42 (31.6%), respectively (p = 0.002). HR for overall survival in patients with rerouting was 0.948 (0.632; 1.423) with a p value of 0.797. No statistically significant difference in overall survival (p = 0.6444) was found between the group of patients treated from 1998 till 2006 (n = 99) versus the group of patients treated from 2007 till 2015 (n = 113) as demonstrated in Table 8 and Fig. 3.
Overall survival rates by time period.
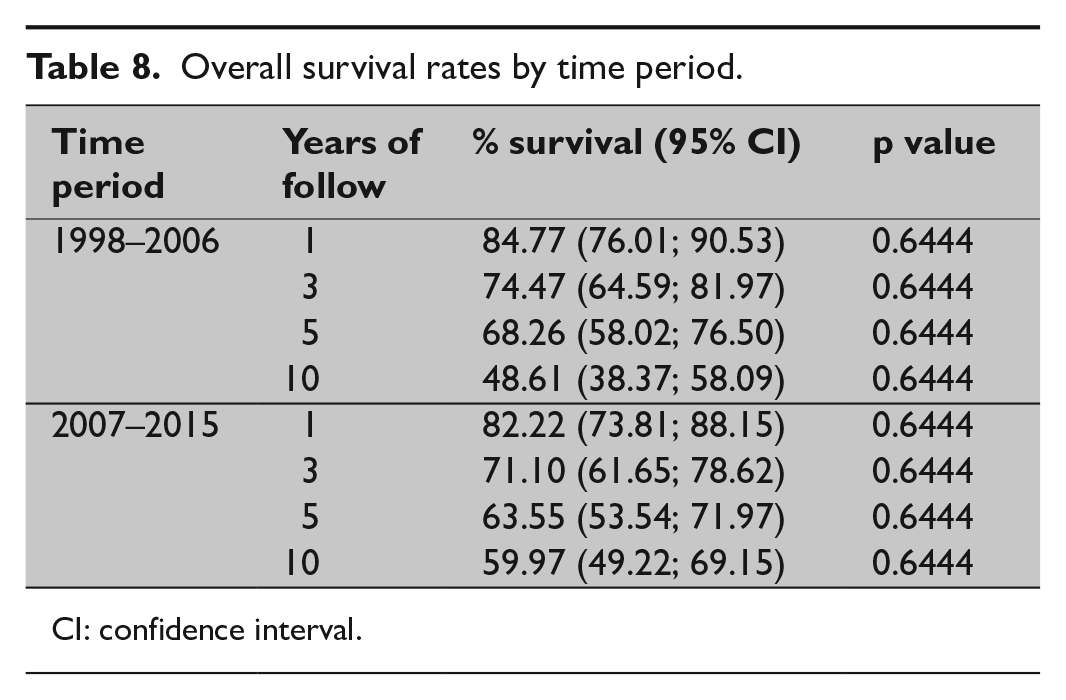
CI: confidence interval.
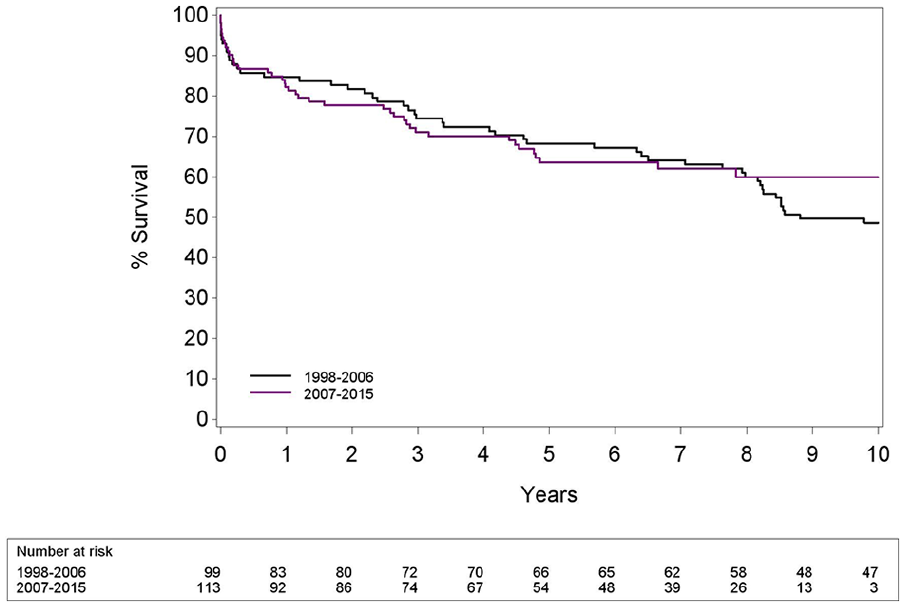
No difference (p = 0.6444) in overall survival between patients treated from 1998 to 2006 and patients treated from 2007 to 2015 (p = 0.6444).
Risk factors and overall survival.
Discussion
Recent international guidelines suggest TEVAR as a valuable, minimally invasive treatment alternative for a variety of thoracic aortic diseases, including acute and chronic trauma, atherosclerotic aneurysm, penetrating ulcer and dissection6,19. However, rates of early and late complications after TEVAR are not negligible. This study reveals an intraoperative mortality rate of nearly 2% and an in-hospital and a 30-day mortality rate of 8.5%, which is in line with other reports from high-volume tertiary referral centers for aortic disease dealing with outcomes after TEVAR for a variety of thoracic aortic pathologies over a long period: 4.8% 20 , 5% 21 , 7.5% 22 , 10.1% 14 , and 11.7% 23 .
Procedure-related stroke (2%) and spinal cord ischemia (1%) are acceptably low and in line with other reports9,11,13 despite a considerable number of patients (37%) who underwent a supra-aortic rerouting procedure prior to TEVAR, including a carotid–carotid bypass (n = 36) or total arch rerouting (n = 16). Nevertheless, additional rerouting procedures are associated with a higher overall incidence of complications (p = 0.002), despite early reports of excellent short-term outcomes 24 .
Endoleak after TEVAR continues to be a cause of concern: this is the most frequent TEVAR-related complication including 40 patients (18.9%) in this series, which is in line with other reports: Wiedemann et al. 22 found 54 endoleaks in 300 patients (18%) and Ziza et al. 23 found 36 endoleaks in 197 patients (18.3%). In contrast, Patel et al. 20 and Etezadi et al. 21 found more endoleaks during follow-up: 138 out of 420 patients (32.9%) and 39 out of 133 patients (29%), respectively. In most of the cases, a type I or type III endoleak is identified on early or late follow-up CT, with various underlying mechanisms, including stent-graft migration, infolding, or disconnection of various components.
Long-term outcome after TEVAR shows a median overall survival rate of 79.6%, 65.9%, and 51.1% after, respectively, 2, 5, and 10 years of follow-up. These results are nearly identical to the report of Wiedemann et al. 22 revealing a median survival of 86%, 63%, and 44% after, respectively 1, 5, and 10 years of follow-up. In addition, the overall survival in patients treated for acute traumatic rupture and anastomotic pseudoaneurysms is significantly better compared to patients treated for true thoracic aortic aneurysms and aortic dissection (p < 0.05). Trauma patients and patients with an anastomotic pseudoaneurysm after coarctation repair 25 are younger and present with less severe comorbidities than patients presenting with an atherosclerotic thoracic aortic pseudoaneurysm. TEVAR for aortic dissections still remains a challenging operation associated with a considerable early and late complication rate of more than 50%, as confirmed by this and other studies26,27.
Other risk factors for mortality after TEVAR include cardiac disease, chronic obstructive pulmonary disease, and renal failure, which were also found in a univariate analysis by Patel et al. 20 and Wiedemann et al. 22 .
Finally, this study has also several limitations. First, the retrospective nature of the study without a control group means that no conclusion can be offered on a subgroup of patients for whom TEVAR might be superior compared to conventional surgery. Second, this is a single-center study based on a cohort of 212 patients presenting with a variety of thoracic aortic pathologies. However, indication, technique, and follow-up did not change significantly over the years, and the large majority of cases were performed with modern and currently available thoracic stent-grafts. Finally, a substantial number of patients were treated by a hybrid approach and outcome results may be more influenced by the surgical part or by the endovascular part of the operation.
In conclusion, this retrospective analysis of the long-term outcome after TEVAR confirms the effectiveness and relative safety of thoracic aortic stent-grafting for a variety of aortic diseases, with better overall survival rates for traumatic aortic rupture and anastomotic pseudoaneurysms. Finally, late complications, mainly endoleaks, still may occur, necessitating long-term imaging follow-up for these patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
