Abstract
Background and Aims:
Pancreatic cysts are increasingly diagnosed, mainly during abdominal imaging performed for other reasons. Between pancreatic cystic neoplasm, intraductal papillary mucinous neoplasms are the most common pre-malignant entities. Intraductal papillary mucinous neoplasms involving side branches overall harbor a low risk of malignancy, and in the recent past, a progressively more conservative approach has been consolidated. Purpose of this report is to summarize the evidence supporting the current practice for the management of branch duct intraductal papillary mucinous neoplasm and to offer a useful practical guide from first observation to post-operative follow-up.
Materials and Methods:
Review of the most important scientific literature on intraductal papillary mucinous neoplasms was made. In this review article, we also report the experience of a high volume center in managing Pancreatic cystic neoplasms.
Results:
The correct management during surveillance still is a matter of debate, since many guidelines have been published suggesting different clinical approaches. Recently, follow-up discontinuation has also been proposed in selected cases.
Conclusion:
Despite significant improvements made by the increase of evidence, selecting surgical candidates because of an increased risk of malignant progression remains an unsolved issue and a hot topic for pancreatologists.
Keywords
Introduction
Since the first description in 1982 by Ohashi and colleagues (1), knowledge about intraductal papillary mucinous neoplasms (IPMNs) of the pancreas has increased and improved overall. IPMNs can be defined as preinvasive, intraepithelial, mucin-producing neoplasms that grow within the pancreatic ductal system (2) and belong to a more heterogeneous group of cystic neoplasms (pancreatic cystic neoplasm (PCNs)), with a broad spectrum of biological behaviors ranging from indolent, slowly progressive epithelial dysplasia to rapidly progressive invasive cancer.
IPMNs were initially described as rare entities, but the actual estimated prevalence ranges from 2.6% to 15%, reaching 37% in patients older than 80 years (3, 4). The widespread use of abdominal imaging, such as ultrasounds and, in particular, contrast tomography and magnetic resonance imaging, is the main cause of this phenomenon since most of these neoplasms are incidentally discovered. Symptoms directly related to the presence of a cyst are in fact quite rare (5). Among incidentally discovered PCNs, IPMNs account for at least 80% of presumed diagnoses (3).
Almost 40 years after the first description of IPMNs, some issues remain unsolved. Despite increased awareness about PCNs, diagnostic accuracy is still low (47%–78%) (6, 7, 8, 9), which is responsible for potential clinically relevant errors (8). The vast majority of PCNs are known to be branch duct intraductal papillary mucinous neoplasms (BD-IPMNs) (3). Even if the risk of malignant progression is extremely low overall, BD-IPMNs can evolve into cancer. Current guidelines provide help in their management with high sensitivity but low specificity for cancer detection. Therefore, the natural history of these neoplasms remains poorly understood.
IPMN Classification and Biology
IPMNs are the expression of a disease that extensively involves the ductal epithelium of the pancreas. As proof of this genetic “field defect,” IPMNs are frequently multifocal, and patients are at risk of developing metachronous IPMNs or distinct pancreatic ductal adenocarcinoma (PDAC) (9, 10).
According to the 2010 World Health Organization (WHO) classification, only cysts >10 mm should be considered as presumed IPMNs (11). At radiological evaluation, communication with the ductal system of the pancreas is crucial to rule out an alternative diagnosis even if it can sometimes be difficult to demonstrate.
Depending on the type of involvement of the ductal system, IPMNs are classified into BD-IPMNs, main duct IPMNs (MD-IPMNs), and mixed-type IPMNs (MT-IPMNs) if a cystic dilatation of the branch ducts is associated with a dilatation of the main pancreatic duct. There are several lines of evidence that converge toward an increased risk for malignancy in the case of MD-IPMN. In particular, the rate of high-grade dysplasia (HGD) or invasive IPMN in surgical series is reported to be between 57% and 92% in these cases (12). On the contrary, the rate of malignancy in the case of operated BD-IPMNs ranges from 6% to 46% (13, 14). These data, however, must be interpreted with caution due to a very frequent selection bias, as this evidence is inferred from surgical series without considering patients under surveillance.
IPMN heterogeneity goes far deeper. Four different histologic epithelial subtypes have been identified: gastric, intestinal, oncocytic, and pancreatobiliary. These subtypes have different carcinogenetic pathways, cancer progression rates, immunophenotypes, and molecular characteristics (2, 15). Moreover, different histologic subtypes can coexist in the same IPMN or in the same pancreas (2). Finally, progression to invasive cancer occurs with different histologic characteristics, namely, ductal/tubular, colloid/muco-nodular, and oncocytic.
Intestinal-type epithelium harbors a high rate of progression to carcinoma, particularly toward the colloid type. Of note, this type of adenoma-to-carcinoma sequence is most common in MD-IPMNs (10, 15, 16). The gastric/foveolar type has a lower rate of progression to invasive carcinoma, which typically has a ductal/tubular histotype. This latter adenoma-to-carcinoma sequence is most common in BD-IPMNs (17, 18).
The pancreatobiliary and oncocytic subtypes are quite rare. The former mainly represents a high-grade transformation of the gastric subtype (15) and has the highest rate of invasive progression, with ductal/tubular differentiation. In the latter, invasive carcinoma is extremely rare and can occur many decades after initial diagnosis (10).
Guidelines
The increased clinical relevance of PCNs has led to the creation of different algorithms for their management.
The International Association of Pancreatology (IAP) published the first international guidelines on PCNs in 2006 (19). These guidelines introduced the concept of high-risk stigmata (HRS) for mucinous neoplasms of the pancreas, highlighting those features related to an increased risk for malignancy. Surgical resection was advocated if one or more HRS was present for a cyst larger than 30 mm or for symptoms related to the IPMN (19). The so-called Sendai positive cysts, however, harbored malignancy only in 29.9% of surgical cases (20–23). Despite good values of sensitivity and negative predictive value, the Sendai criteria showed low specificity, increasing the risk of overtreatment. Sendai guidelines achieved the goal of avoiding fatal errors in the case of potentially malignant PCNs but also led to 75% of potentially unrequested resections (20, 24, 25).
The IAP updated these criteria in 2012 during the Fukuoka Consensus by introducing the concept of worrisome features (WFs). Unlike HRS, WFs do not represent an absolute indication for surgery but call for further assessment with endoscopic ultrasounds (EUS) (26). The last update of these criteria was published in 2016 (27) and is in line with a progressively more conservative approach, aimed to reduce the rate of false-positive results for malignancy and unrequested surgery (28). An important novelty of these guidelines is the concept of the cyst growth rate as a predictor of possible neoplastic degeneration, rather than the absolute value of the cyst’s dimensions, as confirmed by several further studies (29). Moreover, the presence of mural nodules was additionally stratified since only enhanced mural nodules >5 mm are now considered one of the HRS that would serve as the strongest predictor of malignant transformation of IPMNs (27, 30). According to these latest IAP guidelines, surgery is indicated if one or more HRS is present or, in case of suspicious aspects found on second level exams (EUS) in a fit-for-surgery patient. Those IPMNs not fulfilling these criteria are advised for surveillance with abdominal imaging, and follow-up intervals are established according to the cyst’s dimensions (see Table 1) (27).
Comparison of principal guidelines for the management of IPMNs.
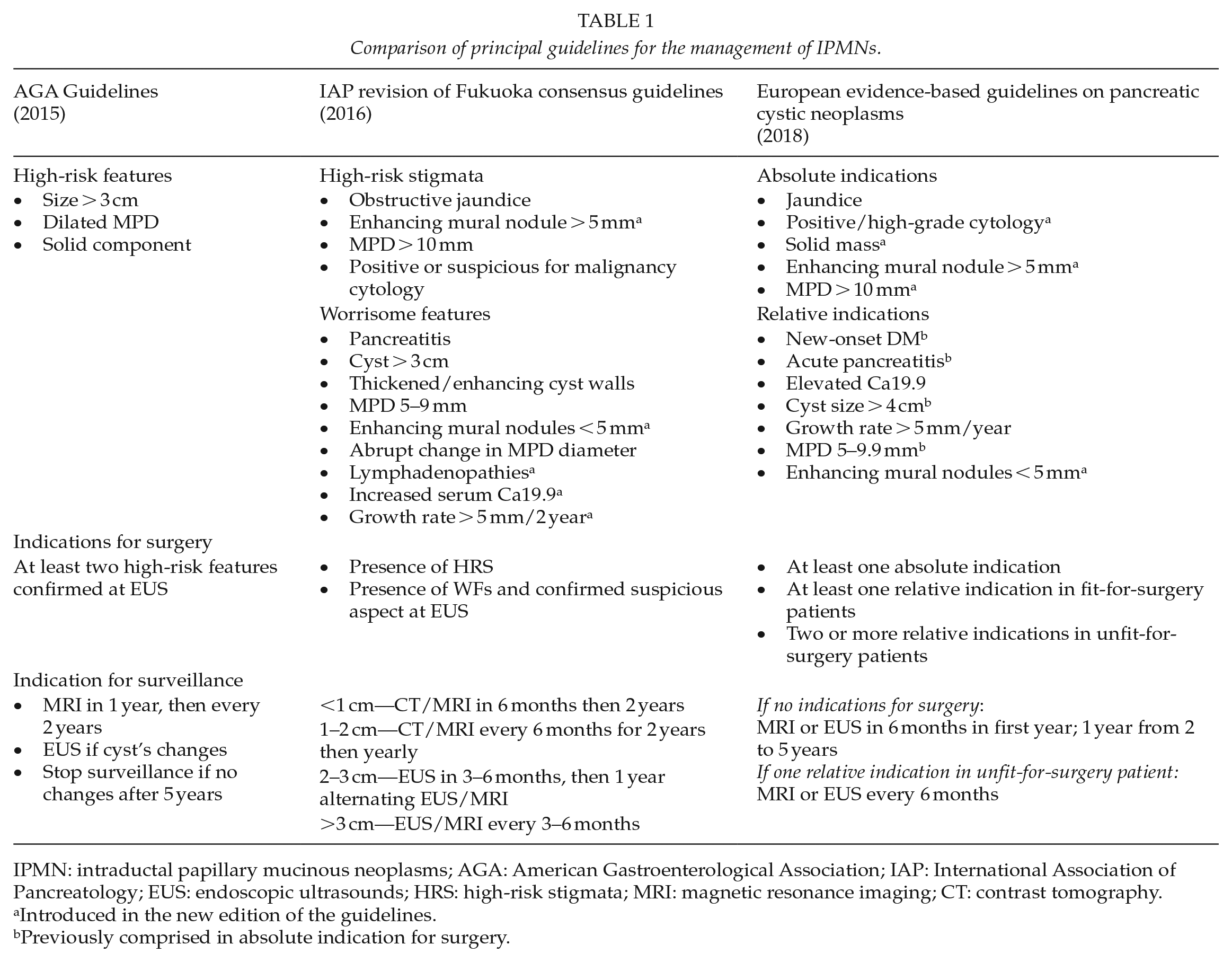
IPMN: intraductal papillary mucinous neoplasms; AGA: American Gastroenterological Association; IAP: International Association of Pancreatology; EUS: endoscopic ultrasounds; HRS: high-risk stigmata; MRI: magnetic resonance imaging; CT: contrast tomography.
Introduced in the new edition of the guidelines.
Previously comprised in absolute indication for surgery.
The European Study Group on Cystic Tumors of the Pancreas published the first European guidelines in 2012 (31). An evidence-based update was then published in 2018 (32). European guidelines distinguish absolute and relative indications for surgery, mimicking the concepts of WFs and HRS established by the IAP. In the absence of indications for surgical resection, a 6-month surveillance is suggested for the first year. Clinical evaluation is then suggested yearly, regardless of the cyst’s dimensions (32).
Recently, the American Gastroenterological Association (AGA) published its guidelines for the management of asymptomatic neoplastic cysts of the pancreas. The AGA proposed a far more conservative approach, suggesting surgery only if at least two HRS are present. Interestingly, the AGA guidelines introduced the proposal of follow-up discontinuation for patients without radiologic and clinical changes after 5 years of follow-up (33). This recommendation raised significant concerns, as it does not consider many studies reporting a possible malignant degeneration or PDAC development beyond such a 5-year threshold (34–36). Nevertheless, the issue of follow-up discontinuation should strongly be taken into account since the prevalence of IPMNs is rapidly increasing. The cost-effectiveness of lifelong surveillance should be assessed since not all patients affected by a BD-IPMN have an increased risk of malignant degeneration. In the future, identifying a subpopulation of patients at the lowest risk of developing malignancy as a potential target for follow-up discontinuation will be mandatory.
Only the European Study Group guidelines are evidence-based. However, most of the studies on BD-IPMN used to draft the European guidelines refer to surgical series and show a low level of evidence. For this reason, we believe that the guideline suggestions must always be tailored to each clinical case.
BD-IPMN in the Era of Observational Series
Most of the knowledge on the natural history of IPMNs is derived from surgical series. However, considering only resected lesions, malignancy rates are unavoidably overestimated due to selection bias. Nevertheless, conservative management has been affirmed in recent years for low-risk lesions, such as most presumed BD-IPMNs (26, 27). Moreover, the risk related to a major pancreatic resection must balance the risk of surveillance.
The understanding of the natural history of BD-IPMNs through clinical observational series has been one of the main gastrointestinal (GI) topics of the last 5 years, most likely triggered by the release of the AGA guidelines (see Table 2). Some studies focused on low-risk cysts at diagnosis (34–37), while others focused on high-risk cysts in patients not fit for surgery (37–39). The former studies showed how BD-IPMNs frequently present radiological changes during their natural history (44%–48%) (34, 36, 37). However, the rates of development of WFs (13%–18%) or HRS (4%–9%) are quite low (34, 36). Therefore, the rate of patients crossing-over from surveillance to surgery is low. Survival analysis in surveilled BD-IPMNs revealed excellent outcomes, as the 5-year disease-specific survival is approximately 100% (37). Considering only BD-IPMNs, 5-year disease-specific survival and overall survival are approximately 97% and 86.4%, respectively, as other causes of death are much more frequent than IPMN-related mortality (37, 38, 39). Moreover, the final pathology of crossing-over BD-IPMNs usually shows low rates of malignancy (34). The overall risk of developing cancer in this cohort remains low (1%–2%) (28, 35, 40). The Massachusetts General Hospital group has published data suggesting an extremely low 5-year risk of developing cancer (0.9%) in patients with cysts <15 mm (35). These results stressed the concept that the risk of developing malignancy in BD-IPMN is low and almost comparable (if not smaller) to the post-operative mortality of high volume centers (41). An indiscriminate surgical treatment would result in a harmful overtreatment. Surgery for BD-IPMN represents an act of prevention that could cause morbidity, mortality, and long-term sequelae, including diabetes and exocrine insufficiency, which could negatively impact the patient’s life expectancy more than the BD-IPMN itself. An important topic in the immediate future would therefore be to identify sub-sets of patients, targeting the risk of developing a malignancy (42) and therefore offering a tailored follow-up program or prompt surgical treatment because of a high risk of cancer.
Most recent observational series reporting the “natural history” of presumed BD-IPMN.
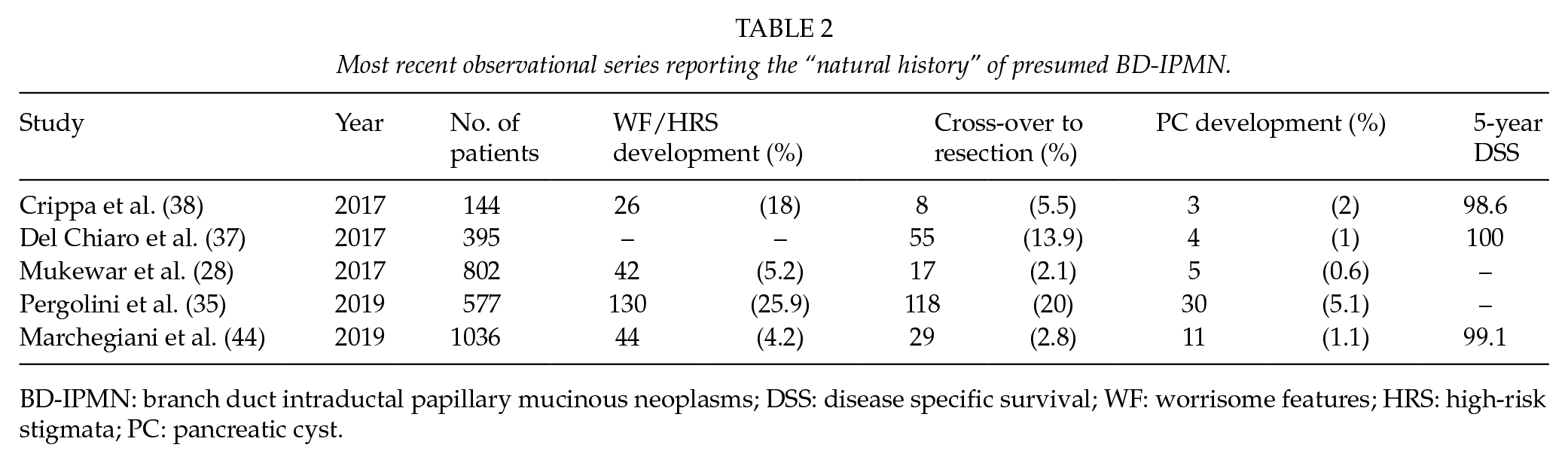
BD-IPMN: branch duct intraductal papillary mucinous neoplasms; DSS: disease specific survival; WF: worrisome features; HRS: high-risk stigmata; PC: pancreatic cyst.
Our institution has recently published an observational series including 1036 presumed BD-IPMNs under surveillance without WFs/HRS at diagnosis. WFs/HRS developed in 4.2% of patients, and 1.1% developed pancreatic cyst (PC) after a median of 62 months. The main predictor of PC development was a growth rate ⩾2.5 mm/year. In patients aged >65 years, with no change in cyst features after 5 year surveillance, the risk of developing PC appeared not to be increased compared to the age-matched general population. Our results confirmed the safety of conservative management of low-risk BD-IPMN. The rate of PC development still remains comparable to surgical mortality (43, 44). Furthermore, we identified a possible target for follow-up discontinuation (44). Today, surveillance can be discontinued only when a patient is no longer fit for surgery. Future studies will provide the evidence needed to identify a population of patients who can safely discontinue surveillance.
Surgical Indication in BD-IPMN: Choose a Strategy
Even if BD-IPMNs can be considered indolent entities, surgery is mandatory in those patients in which the risk of malignancy is considerably high. The correct goal of surgery is not well established; however, we believe that HGD is the perfect target of resection, as the presence of invasive cancer would represent a treatment delay.
The IAP, AGA, and European guidelines delineate absolute and relative indications for surgery in BD-IPMN (Table 1). Given the precise goal of surgery, a standard oncological resection with lymphadenectomy is recommended for BD-IPMN. Parenchyma-sparing surgeries are discouraged because of inadequate oncological benefits. Some experts suggest atypical resections only in very limited cases, namely, in young patients with very low-risk IPMN who show a strong will to be operated on (27, 31, 32, 45). Surgeons must face two main issues in selecting the surgical strategy for BD-IPMN: the multifocality of the disease and the management of the pancreatic stump at the time of surgery. The various guidelines suggest analogous management for unifocal and multifocal disease since each BD-IPMN carries an independent risk of malignancy and malignancy risk is not affected by multifocality (46, 47). If multifocal cysts are limited to a specific pancreatic region, segmental oncological resection should be able to remove the entire disease. In other cases, complete excision of the disease could not be reached without total pancreatectomy. In these cases, it is reasonable to remove the lesion with the highest risk of malignancy and perform surveillance for the pancreatic remnant (Table 1).
The extent of resection must be guided by routine use of intraoperative frozen sections since preoperative assessment is not accurate in determining the actual disease extent (48). This issue is crucial in IPMN involving the MPD. A wider MPD involvement should always be considered, even if it is not suspected preoperatively through imaging (49). Moreover, the definition of “margin clearance” is not well defined in the current literature (50). Guidelines suggest that the presence of HGD or invasive cancer at the pancreatic margin is an indication for further resection with possible extension to total pancreatectomy. If LGD is reported, no further resection is recommended since it does not appear to affect the recurrence rate after resection (27, 32). The absence of epithelial cells in the frozen section is reported as “denuded epithelium and inflammation.” Denudation may prove the presence of an adjacent tumor, and it has been reported as a predictor of recurrence (10, 51, 52). For this reason, further resection must be strongly considered in cases of denuded epithelium (27).
After Surgery: A New Surveillance
Surveillance after resection should be scheduled on the basis of the type of surgery performed, the presence of synchronous lesions in the pancreatic remnant, and the final pathology including resection margin status. In the case of partial pancreatectomy, surveillance is indicated because of the genetic “field defect” linked to IPMNs. Metachronous development of new IPMNs (8%) (10, 46, 53–57) and/or PDAC in the remnant pancreas is well described, even in cases of resection for non-malignant tumors.
Even if malignant, IPMNs with an invasive component have a better prognosis compared with conventional PDAC (58, 59). For BD-IPMNs with low-grade dysplasia, the reported 5-year overall survival is excellent (77%–100%) (10, 57). Some studies report local or distant recurrence in these patients, which may be attributable to the development of metachronous IPMNs or PDAC in the remnant pancreas. Different grades of dysplasia are directly associated with recurrence rates (57). Finally, HGD in resected MD-IPMNs was reported to be predictive of recurrence in the pancreatic remnant and a risk factor for subsequent development of PC (60).
Surveillance in patients with invasive IPMN or IPMN-associated PDAC should be the same as for PDAC. In fact, however, their prognosis is dismal (53, 57). Recurrence of resected invasive IPMN occurs in nearly half of the cases, even many years after resection of the primary tumor. If a resectable local recurrence occurs, completion pancreatectomy is a feasible option with a good prognosis (10, 56). Interestingly, the epithelial subtype seems to play a major role in the pattern of recurrence (10). The role of the resection margin status is much more debated since literature data are contrasting. Some studies report that even HGD at the resection margin is not a predictor of recurrence in the pancreatic remnant (56). Other surgical series define the margin status as the main predictor of recurrence and OS. Of these, the Memorial Sloan-Kettering group reported how dysplasia of any degree at the resection margin is an independent predictor of any type of recurrence (61).
A positive family history for pancreatic malignancies represents another widely explored risk factor, possibly contributing to personalized surveillance. Once it has been found to be an independent predictor of the development of new or progressive IPMN (56, 62), a closer surveillance has been advocated for these individuals. Based on these considerations, the IAP guidelines suggest surveillance as long as the patient remains fit for surgery, with a closer interval (6 months) for high-risk patients (positive family history, non-intestinal subtype, positive surgical margin). In all other conditions, a 6–12 month interval is suggested (27).
Future Perspectives
Adjuvant Therapy for Invasive IPMN
Nearly half the patients affected by IPMN with an invasive component have nodal involvement (63). In addition, either local or distant recurrence is frequently observed. Despite this, the role of systemic therapies is not well established since high-quality data are lacking.
One of the largest retrospective cohorts was published by McMillan and colleagues and included 1220 patients with invasive IPMN. Adjuvant treatment was associated with a survival benefit in patients with stage II–III disease, nodal disease, positive resection margins, and poorly differentiated tumors. In this subgroup of patients, chemoradiotherapy had better outcomes than chemotherapy alone. No benefit on survival was noted in patients with stage I and/or node-negative disease (63). Similar results have been shown by Marchegiani and colleagues, suggesting that chemotherapy could confer a survival benefit mainly in patients with node-positive disease of the tubular subtype (64).
Although these data seem to encourage adjuvant treatment for invasive IPMN, there is a large lack of consensus on chemotherapy regimens. Most patients are treated with gemcitabine alone, while only a few patients are treated with combined drug regimens (64). Moreover, variability in chemotherapy effects could be represented by different histologic subtypes of the invasive component. Further studies are needed to better clarify the real benefit of adjuvant treatment in this disease, hopefully tailoring treatment according to specific pathological features.
Molecular Markers in IPMN
Determining the actual risk of malignancy for a specific cystic lesion of the pancreas remains the hardest challenge. An important additional source of information could be gathered from cyst fluid analysis since radiological and clinical criteria have shown low specificity in predicting malignancy. Fine needle aspiration cytology demonstrated low accuracy and an incomplete clinical application (65). Instead, cyst fluid analysis is becoming increasingly of interest. Molecular profiling of a pancreatic cyst could help distinguish high- from low-risk IPMN. Furthermore, molecular analysis could help to reveal apparently harmless cysts with an impending malignant potential and otherwise biologically innocuous cysts showing HRS, bridging the gap in the sensibility and specificity of current guidelines.
Several studies have investigated the association between protein expression, miRNA, RNA, DNA, cytokines, and IPMN malignancy (66, 67). Recently, Maker and colleagues built a gene expression-based assay including multiple IPMN biomarkers into a molecular signature potentially stratifying low- and high-risk IPMNs. A combination of IL-1β, MUC4, and PTGES2 in cyst fluid had 86% accuracy in detecting high-risk IPMN, far more accurate than the current guidelines (68). The integration of clinical and molecular data derived from cyst fluid analysis was recently developed in a cohort of surgically resected patients. This combined test performed better than clinical and radiological criteria alone, avoiding resection in 60% of mucin-producing cysts potentially amenable to conservative management (69).
The molecular signature of cyst fluid remains the most promising field of research on IPMNs.
Footnotes
Acknowledgements
The authors acknowledged E. Secchettin and D. Bonamini, General and Pancreatic Surgery, The Pancreas Institute, University of Verona Hospital Trust, Verona, Italy.
Author Contributions
Andrea Caravati, Stefano Andrianello, Tommaso Pollini, Marco Biancotto, and Alberto Balduzzi conducted the literature review; Andrea Caravati, Stefano Andrianello, Giovanni Marchegiani, Giuseppe Malleo, and Roberto Salvia contributed to the manuscript drafting and revision.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
