Abstract
Background and Aims:
Intraoperative localization of pathologic parathyroid glands is of major importance for the hyperparathyroidism treatment. Based on the small size and the anatomic variability, the localization can be very challenging. The current practice is to compare preoperative ultrasonography with Technetium-99m sestamibi scintigraphy (MIBI) and plan the resection accordingly. In this study, we implemented indocyanine green angiography for the intraoperative localization of parathyroid glands.
Materials and Methods:
This is a retrospective analysis of 37 patients with primary, secondary, or tertiary hyperparathyroidism who were operated using indocyanine green angiography for the intraoperative localization of pathological parathyroid glands. An indocyanine green solution of 2.5 mg was were intravenously administered for parathyroid gland visualization. Different fluorescence scores were correlated with changes in postoperative parathyroid hormone levels.
Results:
Patients were divided into two groups depending on the presence of uniglandular or multiglandular disease. Sixty-four lesions were resected, and the final histopathologic analysis confirmed the parathyroid origin in 62 of them (96.8%). None of the patients with uniglandular disease developed postoperative hypoparathyroidism, whereas three patients in the multiglandular group developed temporary hypoparathyroidism symptoms. Indocyanine green imaging had higher sensitivity for the intraoperative detection of parathyroid glands compared with ultrasonography and MIBI (p < 0.001).
Conclusion:
Indocyanine green angiography indicated high sensitivity for the intraoperative identification of pathologic parathyroid glands leading to a resection rate of 95.16%. The modality was useful, especially in cases of revisional surgery or ectopic parathyroid glands. Randomized trials have already proven the value of indocyanine green imaging in predicting postoperative hypocalcemia. Our results support the regular use of this method during parathyroid surgery.
Keywords
Introduction
Parathyroidectomy is the treatment of choice in patients with primary hyperparathyroidism (pHPT) and tertiary hyperparathyroidism (tHPT) but also in those with secondary hyperparathyroidism (sHPT) resistant to pharmacological treatments (1). Resection of all hyperfunctioning parathyroid glands (PGs) is the essence of successful parathyroidectomy. However, identification of PGs can be challenging, even for experienced surgeons, mainly because of their variability in number and anatomy (2).
Despite the improvements in nuclear (MIBI) and ultrasonography (US), the reported sensitivity widely ranges, from 50%–80% and 40%–85%, respectively (3 –9). Moreover, concomitant thyroid disease influences considerably scintigraphic and ultrasonographic detection of pathological PGs. Furthermore, these preoperative findings are difficult to correlate with the intraoperative anatomy.
Intraoperative adjuncts in order to support PG identification are even more limited. Fluorescence imaging using near-infrared fluorophores is a useful technology for this purpose. Currently, available fluorophores for clinical use include methylene blue (MB), aminolevulinic acid (ALA), and indocyanine green (ICG). Several study groups have attempted to utilize MB; however, the risk of toxic metabolic encephalopathy and blue staining of the surgical field has not led to a widespread use (10, 11). ALA has been successfully tested in small clinical studies, but its efficacy is confined by the need to photosensitize patients, administer the compound 4–5 h prior to surgery, and shield them from direct light exposure for further 48 h after surgery (2). ICG was found to be useful in localizing PGs in dog models in a preclinical trial (12). Intraoperative indocyanine green angiography (ICGA) can be used to assess and predict PG function after thyroidectomy (13, 14), to identify PGs during parathyroidectomy, and to avoid hypoparathyroidism in subtotal parathyroidectomy (15). This hypothesis has been recently confirmed by a randomized trial, which entrenched the ICG imaging role in parathyroid surgery (PS) ( 16 ).
The aim of this study was to assess the intraoperative ICG fluorescence angiography in localizing PGs and to compare it with US and MIBI in patients with uniglandular and multiglandular disease.
Materials and Methods
The study was performed in accordance with the Declaration of Helsinki and was approved by the local ethics committee (2015-854R-MA). Due to the retrospective nature of the study and the fact that all data presented here were not collected during a formal prospective trial, the local ethics committee waived the requirement for individuals’ informed consent.
Thirty-seven consecutive patients undergoing surgery for biochemically proven, symptomatic HPT between February 2016 and March 2018 at the University Hospital of Mannheim were eligible for enrollment in the study. Patients with known iodine allergy were excluded.
All patients presented to routine preoperative workup, including measurement of serum electrolytes, cell blood count, and coagulation tests. Prior to surgery, all patients underwent US and MIBI for preoperative planning, except the patients with pHPT, due to a single parathyroid adenoma precisely and accurately localized by US. The number and the location of PGs detected by each method were recorded. We also recorded the following parameters: age, gender, surgery date, preoperative diagnosis, preoperative and postoperative day 1 (POD1) calcium, preoperative and POD1 parathyroid hormone (PTH), PG greatest dimension, ICG uptake, and histopathology. Patients were divided into two groups: uniglandular disease—undergoing a one-gland resection (U-group)—and multiglandular disease—undergoing a four-gland surgery (total or subtotal parathyroidectomy, M-group), regardless of the HPT type.
Surgical Procedure
Surgery was performed under general anesthesia. Bilateral exploration was performed for patients with multiglandular disease, whereas a minimally invasive open approach was applied for those with uniglandular disease. Microsurgical technique and loupe magnification were routinely utilized. Visual identification and recurrent laryngeal nerve (RLN) monitoring were systematically carried out (NIM 30®; Medtronic, Dublin, Ireland). In case of multiglandular disease, PTH was measured after completing the resection. If the intraoperatively measured value had decreased over 90% of the preoperative PTH, we did not search for supernumerary PGs. Otherwise we continued the search for ectopic PGs. Search for ectopic glands was performed in superior mediastinum if an inferior PG was missing and in the lateral retroesophageal area if a superior PG was missing. Subtotal parathyroidectomy was performed by applying non-absorbable stitches or clips onto the parathyroid remnant. Criteria, such as anatomic localization, vascularization, and morphologic aspect of PGs, were used to select the gland for subtotal resection (remnant).
Intraoperative ICG Fluorescence Angiography
An ICG/aqua solution of 2.5 mg was administered intravenously through a peripheral venous line. The injection was repeated, if needed, without reaching the far higher maximum daily dose of 5 mg/kg (17, 18). Operating lights were turned off, and real-time fluorescence imaging was performed using the laparoscopic charge-coupled PINPOINT® camera (Novadaq, Ontario, Canada) immediately after the ICG injection. Each PG visualized by naked eye was assessed for ICG uptake using a feasible and easy-to-use scoring system recently described; ICG0: the PG appears black after ICG injection, meaning that it is not vascularized; ICG1: gray or heterogeneous PG, suggesting its partial vascularization; and ICG2: white PG, meaning that it is well vascularized ( 13 ). An additional ICG dose was administered to assess the remnant perfusion in patients undergoing four-gland surgery.
All suspected lesions were resected during surgery, and histology of the frozen sections was conducted. Definitive histology served as the gold standard for parathyroid identification. In addition, confirmation of surgical success was attained by a PTH drop of >50% at 10 min after excision compared to the preoperative baseline serum concentration (Miami criteria) (1, 19, 20).
Postoperative Management
Postoperative patients were monitored cautiously, and hypocalcemic symptoms such as numbness, facial paraesthesia, Chvostek’s signs, and muscular spasm were recorded as soon as they occurred. Hospital stay was at least 24 h. Serum calcium and PTH levels were measured on POD1, and routine oral supplementation with calcium and 25-hydroxyvitamin-D was administered in case of hypocalcemia. Adjustment of total calcium for albumin was always obtained. Reference values are 2.2–2.6 mmol/L and 12–88 ng/L, for serum adjusted calcium and PTH, respectively. Hypocalcemia was defined as an adjusted calcium value <2.0 mmol/L within 24 h from surgery.
Statistic
Continuous data are presented as mean, standard deviation (SD), and range (min–max). To compare qualitative variables, we used the chi-square test of independence to test for equality of proportions between groups. The Kruskal–Wallis test was applied to test continuous variables. A p value of 0.05 was used as the cut-off for statistical significance. Data analysis was performed using Stata 12 (StataCorp LP, College Station, TX, USA). Univariate and multivariate analysis was performed, assessing predictors of the ICG score. Sensitivity analysis was additionally conducted to determine differences among the three used modalities (US, MIBI, and ICGA).
Results
Patients
Thirty-seven patients (19 females and 18 males, mean age 59 ± 12 years) underwent PS using ICG imaging. Four patients with pHPT had a mediastinal ectopic PG. In one of the cases, the ectopic gland was located in the aortopulmonary window and was resected thoracoscopically under fluorescence guidance. In order to improve the understanding of the perfusion patterns of the pathological PGs, we created two groups based on the number of PGs which were affected; patients with only one PG resected were allocated in the U-group (uniglandular disease, n = 28), and patients with more than one PG resected formed the M-group (multiglandular disease, n = 9). A flow chart of the study is shown in Fig. 1.
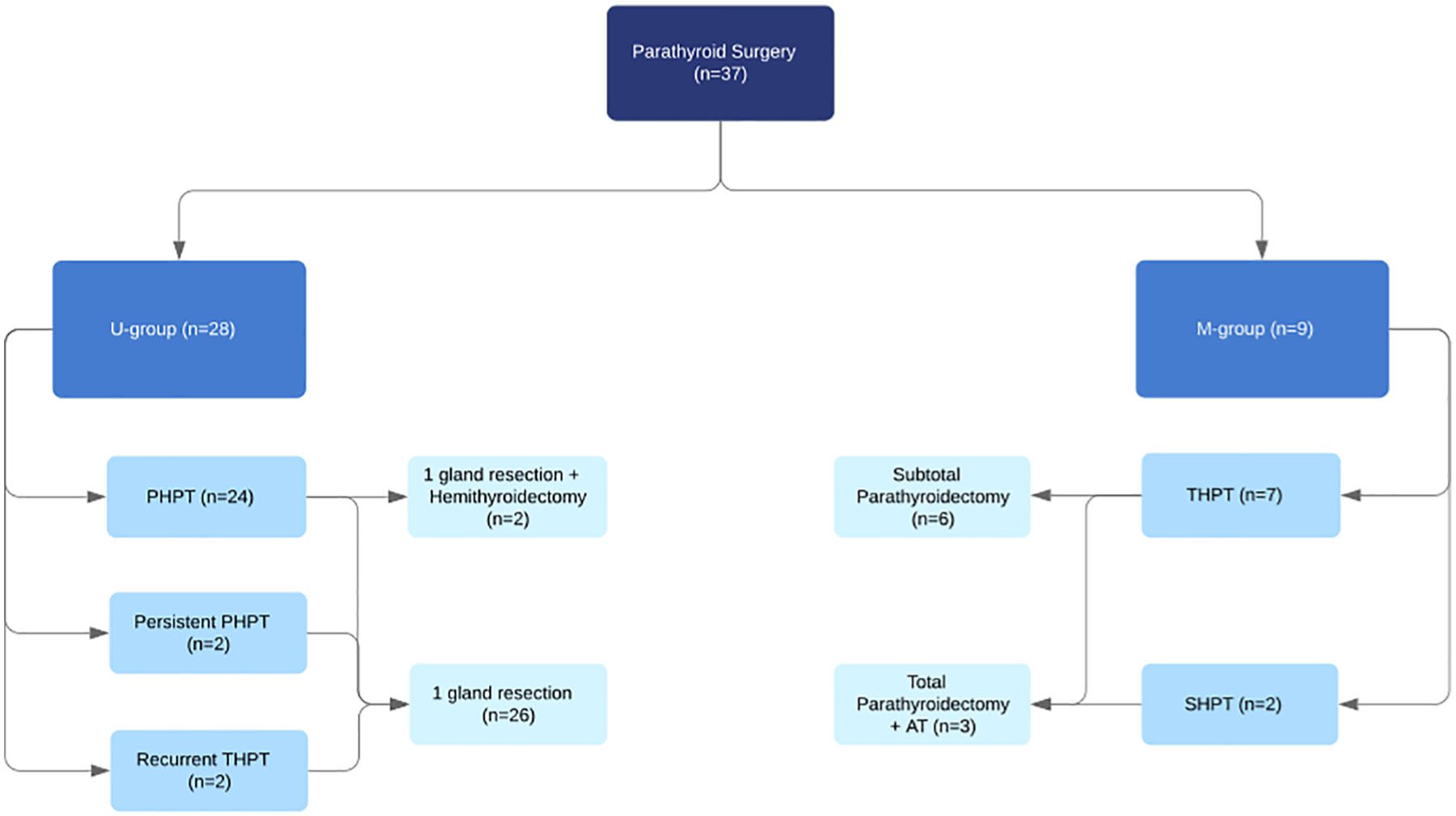
Flowchart of the study presenting the allocation of the patients to the groups and the type of surgery performed.
Mean preoperative and POD1 PTH was 584.3 ng/L (SD 712.9, range 70.2–3943.9) and 21.0 ng/L (SD 25.2, range 0.3–98.2), respectively, showing a mean PTH drop of 563.3 ng/L (SD 710.6, range 64.0–3939.4) after surgery. Likewise, mean preoperative and POD1 adjusted serum calcium was 2.7 mmol/L (SD 0.25, range 2.2–3.4) and 2.3 mmol/L (SD 0.2, range 1.7–3.0), respectively, with a calcium drop of 0.4 mmol/L (SD 0.2, range 0.1–1.1). Mean operative time was 91.1 ± 53.2 min in the U-group and 140.3 ± 52.2 min in the M-group. No ICG adverse reactions were recorded.
In the U-group, 26 of 28 patients were operated for pHPT caused by a single adenoma (2 of them for persistent pHPT after an initial unsuccessful parathyroid exploration), and 2 patients were operated for recurrent tHPT after a previous subtotal parathyroidectomy. Three patients with pHPT in the U-group underwent ipsilateral hemithyroidectomy during the same surgical procedure due to suspicious thyroid nodules.
Seven patients in the M-group were operated for tHPT and two patients for sHPT. Among the M-group, four patients had 3½ glands removed, two patients had three glands removed, and three patients had total parathyroidectomy with autotrasplantation.
Overall 64 lesions were resected. Frozen section was always performed, and the final histopathology confirmed the parathyroid origin in 62 of them (96.8%). In the other two cases, a highly fluorescing thyroid nodule was mistaken for PG and resected. Frozen section, in conjunction with the absence of PTH drop, led to the resection of the true PG. According to the postoperative pathology, 46.8% of the resected PGs were hyperplastic, 45.2% were adenoma, and 5 were normal PGs (8.1%).
At POD 1, mean PTH levels were 21.9 ng/L (SD 25.9, range 0.3–98.2) and 17.4 ng/L (SD 20.8, range 1.7–64.7) in the U- and M-group, respectively, while corrected calcium levels were 2.4 mmol/L (SD 0.2, range 1.7–3.0) and 2.2 mmol/L (SD 0.2, range 1.9–2.5), respectively. In the U-group, all patients had PTH values at POD1 under the upper reference level except for the one patient who had a PTH of 98.2 mmol/L, decreased by 75% of its preoperative value, with a corrected calcium of 2.61 mmol/L. PTH could be measured in all patients in the M-group at POD1, proving that the remnant or the transplanted parathyroid was well functioning, and the HPT was healed.
Intraoperative ICGA
Intraoperative ICGA successfully identified 59 of 62 PGs (95.16%). In 7 of 62 cases, the ICGA primarily guided the resection since the PG could not be visually identified clearly, in order to perform the resection. The ICG score was 0 in 3 PGs (5.08%), 1 in 8 PGs (13.56%), and 2 in 51 PGs (86.44%). All resected lesions with ICG2, and 80% of those with ICG1, were truly identified as PGs, whereas the remaining resected lesions with ICG1 were thyroid nodules.
The ICG uptake became visible at 30–60 s after intravenous injection and persisted up to 20 min after injection. Uptake was additionally noted in PGs after resection (Fig. 2). Fig. 3 shows an example of intraoperative fluorescence signal in PGs.

Ex vivo ICG uptake.
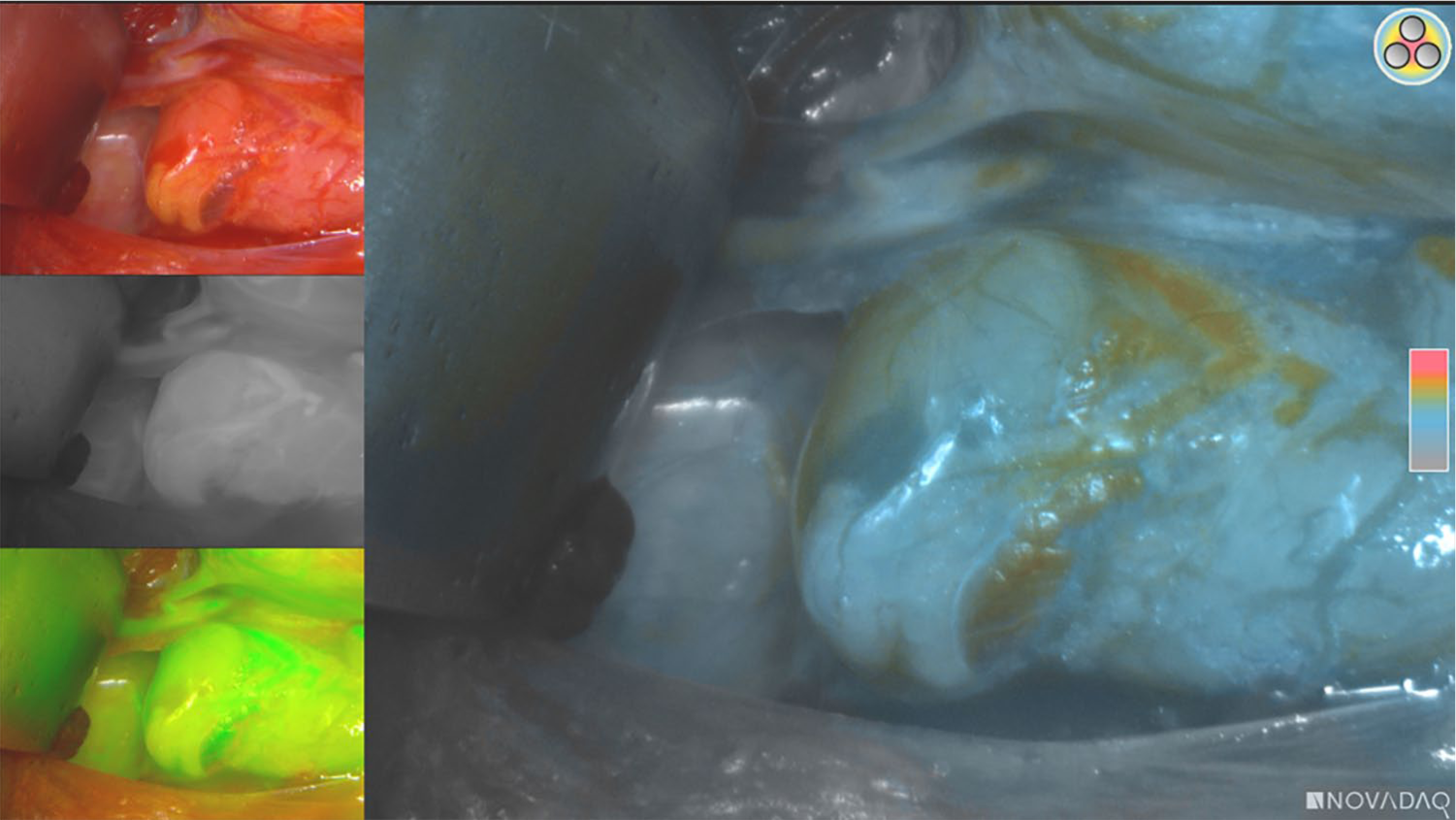
In vivo ICG uptake.
In patients undergoing subtotal (3.5 or 3 glands) parathyroidectomy, an ICG2 was considered a satisfactory remnant perfusion. The ICGA was performed for the identification of the parathyroids and was repeated in order to confirm the perfusion of the remnant after or before placing the clip. Only parathyroids with an ICG score of 2 were chosen to be preserved as remnants in order to assure adequate function. Only one of those patients exhibited postoperative symptomatic hypoparathyroidism (1/6). When the ICG score of the most normal-looking PG chosen to be preserved was 0 (n = 1) or 1 (n = 2), a total parathyroidectomy with autotransplantation into the sternocleidomastoid muscle was performed. Two of the three patients undergoing this surgical procedure experienced hypoparathyroidism symptoms.
Comparison of ICG Imaging with other Preoperative Imaging
US successfully localized 42 of 62 PGs (67.74%), 25 (40.32%) in the U-group and 17 (27.42%) in the M-group. MIBI was not performed in seven patients of the U-group because of the precise localization using the US. A total of 72% of PGs showed ICG2, 14% ICG1, and 14% ICG0. Thirty-five of 62 (56.45%) PGs surgically removed were seen on the preoperative MIBI, 17/21 (81%) in the U-group and 18/34 (52.9%)in the M-group. In the U-group, all the PGs with a negative US showed ICG2. Similarly, all the PGs with a negative MIBI had ICG2. Furthermore, in the M-group, all sonographically undetected PGs had ICG2, while among MIBI undetected PGs, 69% showed ICG2 and the others, ICG1. Among patients with ectopic PGs (three in the U-group and one in the M-group), US expectedly detected only one of the four ectopic PGs, while in two of the three patients, who underwent MIBI, the mediastinal PG was recognized preoperatively. All mediastinal PGs had ICG2.
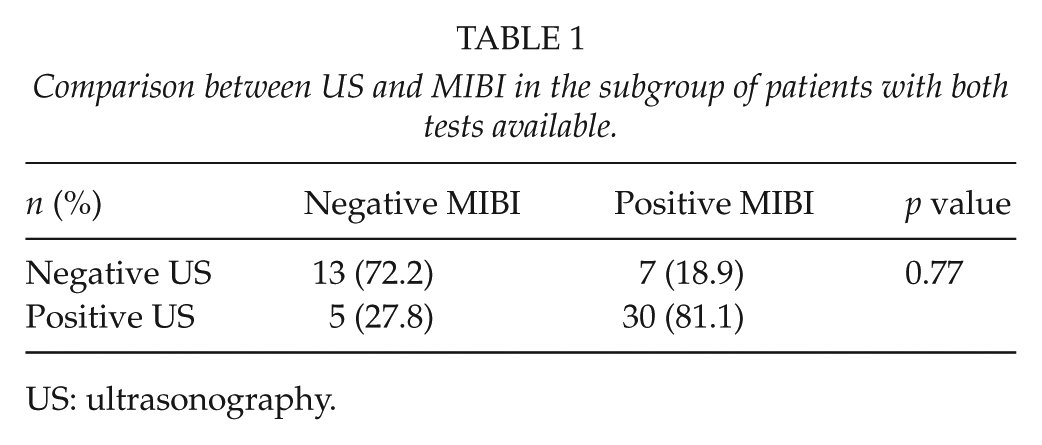
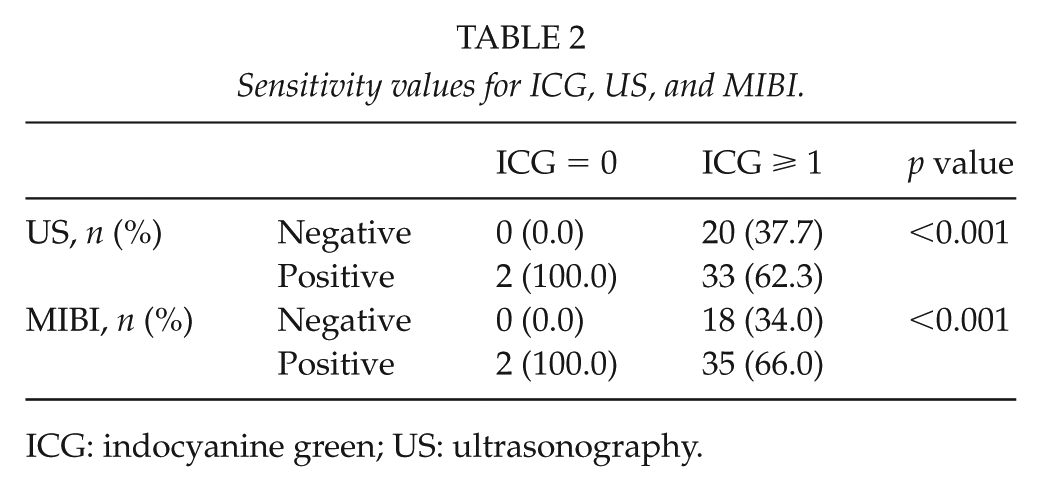
We were unable to run receiver operating characteristic curve (ROC) analysis in order to compare the area under the ROC curve of the three tests, so we included a subanalysis only with patients who had all tests available (30 patients, 55 PGs). Sensitivity for ICG, US, and MIBI was 96% (53/55), 55% (35/55), and 63% (37/55), respectively (Tables 1 and 2). The McNemar chi-square test for paired observations confirmed superiority of ICG compared with both US (<0.001) and MIBI (<0.001).
Comparison between US and MIBI in the subgroup of patients with both tests available.
US: ultrasonography.
Sensitivity values for ICG, US, and MIBI.
ICG: indocyanine green; US: ultrasonography.
Univariate and Multivariate Analysis of Preoperative Characteristics According to the ICG Score
Age, sex, preoperative calcium and PTH, size, and US positivity were not associated with the ICG score. However, positive MIBI increased 5 times the probability of positive ICGA according to the univariate analysis and 10 times according to the multivariate analysis.
Discussion
Although parathyroidectomy is commonly performed as a curative procedure in over 95% of cases, intraoperative identification of PGs can be challenging even for experts causing surgical failure (21). In our study, we analyzed the efficiency of ICGA to detect PGs, distinguished them from thyroid and surrounding tissue, and compared it to preoperative imaging.
MIBI is a radionuclide imaging, which has higher sensitivity for hyperfunctional PGs than normal glands, based on the preferential sestamibi uptake in the mitochondria-rich parathyroid cells. MIBI sensitivity in pHPT reaches approximately 90% (22). Furthermore, MIBI sensitivity and specificity in multiglandular disease have been reported as 58% and 93%, respectively (4, 23). In our subanalysis, MIBI sensitivity was 63%.
US sensitivity depends on the lesion size and contrasts the difference from adjacent tissues. However, some anatomical areas are not accessible to US, like the retrotracheal or retroesophageal space, as well as ectopic PGs in the mediastinum. Moreover, US sensitivity in multiple and single PG disease widely ranges from 30% to 80% and 76% to 87%, respectively (4, 24). In our subanalysis, US sensitivity (uniglandular and multiglandular disease) was 55%.
An ideal agent for the intraoperative localization of PGs should be safe, quick to use, trustworthy, and immediately available without the need for preoperative or postoperative preparation or care. ICG preferential accumulation in PGs has spread its use in thyroid and parathyroid surgery. The actual mechanism that mediates the higher ICG uptake of PGs than thyroid or surrounding tissue is still unknown. Apart from the ICG-supported fluorescence intensity of the PGs, the glands themselves seem to exhibit higher autofluorescence signals than the surrounding tissue. A recent study which applied near-infrared spectrometry has shown increased autofluorescence spectra, regardless of the underlying pathology, compared with the thyroid, muscles, trachea, and other neighboring structures (25). Nevertheless, the mainly accepted hypothesis correlates with a high metabolic activity and proportionally with a higher number of mitochondria in the parathyroids than other tissues (26, 27). On the other hand, other tissue cells (e.g. fat, muscle, and lymph node) can rapidly clear out ICG and appear dark, while ICG has delayed excretion due to the disarranged architecture of hyperplastic PGs (26). The dense vascularization of the parathyroids combined with the very small volume of the organ and the superficial course of the supplying vessels on the surface of the organ could also play a role in the high uptake of ICG. We noted ICG uptake in 95% of all PGs evaluated. In the sensitivity analysis, ICG sensitivity reached 96% which was significantly higher than both US (<0.001) and MIBI (<0.001). As already suggested by Zaidi et al. (15), it is possible to hypothesize that abnormal PGs would exhibit increased ICG signal; however, our results showed that hyperplasia bore no difference in fluorescence.
These results suggest that the near-infrared imaging can be performed successfully with a satisfying outcome. Although in our study, over 90% of PGs showed fluorescence, there were two cases in the U-group who had an initial resection of a thyroid nodule, demonstrating a significant ICG uptake and a subsequent finding of one non-fluorescent PG during the same surgery. Thus, these PGs presented as false-negative ICGA suggest both that concomitant thyroid ICG uptake could limit the ability to distinguish PGs and that fluorescence imaging combined with careful visual inspection is still necessary. Even though we have made some notable progress in research of intraoperative fluorescence imaging indeed, thyroid background signal still remains a potential pitfall using this technique. Another possible explanation for the false-negative fluorescence is that we could have devascularized the PGs during the preparation for the resection of the thyroid nodule.
ICGA proved to be extremely helpful in cases of patients requiring four-gland surgery for sHPT or tHPT. ICGA allowed reliable assessment of the perfusion of the remnant and guided the autotransplantation choice in those PGs whose vascular supply was unsatisfying. None of the patients who underwent subtotal resection showed permanent hypoparathyroidism. This fact supports the idea that a well-perfused remnant will produce measurable PTH levels and therefore avoid long-term postoperative hypoparathyroidism.
In conclusion, ICGA is a very valuable technical support in PS. Despite the superiority in identifying pathological PGs in primary, secondary, or tertiary disease compared with the other available modalities, ICGA allows for a precise localization of the altered PG, can guide surgery in cases of ectopic disease, and measure the adequacy of the perfusion of the transplanted tissue in cases of subtotal resections. Several surgical departments have fluorescing laparoscopic units which can be used in different fields of general surgery, making the costs for an additional use in PS non-significant. Previous studies of our group have showed that the use is not associated with increased operating time (28). Recent randomized controlled trials (RCTs) (15) have demonstrated the value of the ICGA in predicting hypocalcemia after PS. Our results, in accordance with the other published trials, support the idea of routine implementation of ICGA in PS centers.
Limitations
This retrospective analysis encompasses all well-known bias risks of retrospective studies including selection bias, recall bias, and so on. Concerning the sensitivity analysis, it is clear that the comparison includes methods which are designed for preoperative use and the ICGA which is an intraoperative tool. However, this method presents a very sensitive modality compared with both ultrasonography and scintigraphy and, after acquisition of certain expertise, could probably replace them in the routine PS.
Footnotes
Acknowledgements
The authors would like to thank Ms. Eleni Karampini for performing the language editing.
Authors’ Contributions
G.D.M. was involved in the management, collection, process, and assessment of the data and drafted the manuscript. I.K. was involved in the planning of the study and critically revised the manuscript for important intellectual content. A.G. was involved in the planning of the study and processing of the data. A.L. has critically revised the manuscript for important intellectual content. S.P. performed the statistical analysis. K.N. planned the study, collected, processed, and evaluated the data and revised the manuscript.
Declaration Of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Kai Nowak serves as an external consultant for Stryker. All other authors have no conflicts of interest relevant to this work to declare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
