Abstract
Background and Aims:
T2 gallbladder cancer requires lymph node dissection for curative resection, whereas simple cholecystectomy is adequate to treat T1 gallbladder cancer. Hence, this study aimed to develop an accurate scoring system to preoperatively predict pT2 in gallbladder cancer.
Material and Methods:
We retrospectively assessed data from 57 patients with suspected gallbladder cancer who underwent curative resection between September 2003 and May 2017. Six with apparent invasion of adjacent organs on preoperative images were excluded. We evaluated preoperative computed tomography, magnetic resonance and endoscopic ultrasonographic images, blood biochemistry, and the maximum standard uptake value in fluorodeoxyglucose-positron emission tomography images. We analyzed whether correlations between preoperative findings and the depth of tumor invasion could predict pT2.
Results:
The pathological diagnosis was gallbladder cancer in 30 (58.8%) patients, of whom 21 (69.9%) had pT2 or worse. Multivariate analyses selected carcinoembryonic antigen and tumor diameter as independent predictors of pT2 or worse (odds ratios = 1.741 and 1.098, respectively; 95% confidence intervals = 1.004–3.020 and 1.008–1.197, respectively). A regression formula was created using carcinoembryonic antigen and tumor diameter to calculate pT2 predictive scores. The area under the receiver operating characteristics curve of the pT2 predictive score was 0.873.
Conclusion:
We created a scoring system to predict pT2 in gallbladder cancer using carcinoembryonic antigen and tumor diameter. The present findings suggested that carcinoembryonic antigen is important for the preoperative evaluation of gallbladder cancer.
Keywords
Introduction
The surgical procedure for gall-bladder cancer (GBC) differs depending on cancer progression (1, 2). Since lymph node metastasis is absent when a tumor is confined to the mucosa or muscle layer (T1), simple cholecystectomy is adequate for treating T1 GBC. Tumors that invade the subserosal layer (T2) require extended cholecystectomy with a tumor-free margin and lymph node dissection, because 40%–50% of patients have lymph node metastasis (1–7). Therefore, a preoperative diagnosis of whether a GBC is T1, or T2 or worse is important. However, to accurately diagnose the preoperative depth of GBC invasion is difficult even with repeated preoperative imaging (2, 8). The reported accuracy of a preoperative diagnosis of pT2 is only 33% even when endoscopic ultrasonography (EUS) is combined with computed tomography (CT) (8). Furthermore, although EUS is reportedly useful to preoperatively diagnose GBC tumor depth (9, 10), limitations include poor accessibility, technical complexity, and difficulties associated with ultrasonographic evaluation by surgeons (11). This study aimed to develop an accurate means of predicting the depth of pathological T2 (pT2) or worse (⩾pT2) GBC using simple, objective factors to ensure that patients undergo optimal surgical procedures.
Material and methods
Patients and study setting
We retrospectively reviewed the records of 57 patients with suspected GBC who underwent curative resection at Ehime University Hospital between September 2003 and May 2017. Suspected GBC was preoperatively defined as a broad-based mass including focal or diffuse thickening of the wall, or a tumor ⩾1 cm with a tendency to increase in size. After excluding six patients with apparent invasion to adjacent organs on preoperative images, we analyzed data from 51 patients. The median age of the patients was 71 (range = 36–85) years and 27 (52.9%) were male. We evaluated correlations between preoperative findings and the pathological tumor depth of invasion as, benign/T1 GBC or ⩾T2 GBC. The depth of tumor invasion was defined using the TNM Classification of Malignant Tumors published by the Union for International Cancer Control (UICC), Seventh Edition (12).
Preoperative Imaging
Tumor diameter, morphology, localization, and the depth of tumor invasion were preoperatively diagnosed using (contrast-enhanced) CT, magnetic resonance imaging (MRI), trans-abdominal ultra-sonography (US), and EUS. Tumor diameter was defined as the longest diameter on the clearest of the above images. The diameters of the tumor occupying the circumferential wall of the gallbladder were defined as the straight length from the fundus to the medial terminus of wall thickening, and not the curvature line along the gallbladder wall. Tumor morphology was classified as pedunculated or broad based (including wall thickening). The findings of tumor type determined by EUS are described as A, B, C, and D according to Fujita et al. (9), where type A is a pedunculated mass with a nodular surface, type B is a broad-based mass or wall thickening with a regular surface, type C is a broad-based mass or wall thickening with an irregular surface and an irregular outer hyperechoic layer of the wall, and type D is a broad-based mass or wall thickening with an irregular surface and an outer hyperechoic layer of the adjacent wall disrupted by a mass. Considering correlations between pT2 and preoperative images, we defined tumors diagnosed as ⩾T2 from CT, MRI, US, or EUS images as Image ⩾T2. The diagnostic criteria for ⩾T2 on contrast-enhanced CT and MRI images comprised a contrast-enhanced, transmural broad-based mass or gallbladder wall thickening, and those on US or EUS images were defined as hypoechoic masses that had invaded the outer hyperechoic layer or beyond (types C, D, and some B). We evaluated pancreaticobiliary maljunctions using magnetic resonance cholangiopancreatography (MRCP), drip infusion cholangiographic–computed tomography (DIC-CT), or endoscopic retrograde cholangiopancreatography (ERCP). We also assessed the presence or absence of cholecystolithiasis using imaging modalities.
Fluorodeoxyglucose–Positron Emission Tomography
We evaluated maximum standard uptake values (SUVmax) of gallbladder tumors using fluorodeoxyglucose-positron emission tomography (FDG-PET) when patients had no co-morbid uncontrollable diabetes.
Blood Biochemistry
We evaluated carcinoembryonic antigen (CEA), carbohydrate antigen 19-9 (CA19-9), C-reactive protein (CRP), and neutrophil-to-lymphocyte ratios (N/L ratio) as potential predictors of tumor progression.
Statistical Analysis
Continuous variables were compared using Mann-Whitney U tests and are presented as medians with ranges. Categorical variables compared using chi-square or Fisher exact tests are presented as numbers with ratios (%). Longitudinal trends between continuous variables were calculated using Spearman ρ correlation. Statistically significant variables that correlated with ⩾pT2 in univariate analyses were included in multivariate analyses with logistic regression using forward selection and likelihood tests. Regression models were calibrated using Hosmer-Lemeshow tests. Scores for a regression formula calculated from the outcomes of logistic regression analyses were defined as predictive of ⩾pT2 and were evaluated using receiver operating characteristics (ROC) curves and area under ROC curves (AUC). Statistical significance was defined as p < 0.05. All data were statistically analyzed using SPSS statistical software version 24.0 (IBM Corp., Armonk, NY, USA).
Ethical Consideration
The institutional review board at Ehime University Hospital approved this study (Approval number: 1712021), which proceeded in accordance with the ethical standards established in the Declaration of Helsinki in 1995 (revised, Brazil 2013). Written informed consent was obtained using the opt-out principle. The study and the right of refusal to participate were disclosed to the public online. None of the authors have any conflict of interest to disclose regarding this study.
Results
Table 1 shows the preoperative features of the patients determined by imaging. We pathologically diagnosed GBC in 30 (58.8%) patients, of whom 21 (69.9%) had ⩾pT2 tumors (Table 2). Lymph node metastasis was pathologically positive in 11 patients who had ⩾pT2 tumors. In total, 51 (100%), 47 (92.2%), and 39 (76.5%) of the patients enrolled in this study were respectively assessed by preoperative contrast-enhanced CT, MRI, and EUS. The tumor diameters were 16 (7–47) mm and 27 (14–46) mm for patients who were assessed or not by EUS, respectively. To determine tumor depth using images from 7 (17.9%) of 39 patients who underwent EUS was problematic because intestinal gas, tumor location in the cystic duct, scope positioning instability, and intra-gallbladder gas caused by prior ERCP interfered with identifying tumors. Tumor depth in these 7 patients was determined by trans-abdominal US (2) and CT (5), and mainly by EUS in the remaining 32 patients. Of the 32 patients who were diagnosed using mainly EUS, 16 were preoperatively diagnosed as having ⩾T2 (Table 3). Among them, 15 and 1 had types C and B tumors, respectively. The positive and negative predictive values for patients with tumor depth determined by EUS were 56.3% (9/16) and 87.5% (14/16), respectively. Of the 17 patients with tumor depth diagnosed mainly by CT, 12 were preoperatively diagnosed as having ⩾T2 (Table 3). GBC was difficult to differentiate from chronic cholecystitis or xanthogranulomatous cholecystitis (XGC) in 9 of these 12 patients. Four and five of these nine patients were pathologically diagnosed with ⩾T2 GBC, and with chronic cholecystitis or XGC, respectively. The positive and negative predictive values were 66.7% (8/12) and 60% (3/5), respectively, among patients diagnosed mainly using CT.
Preoperative imaging findings (n = 51).
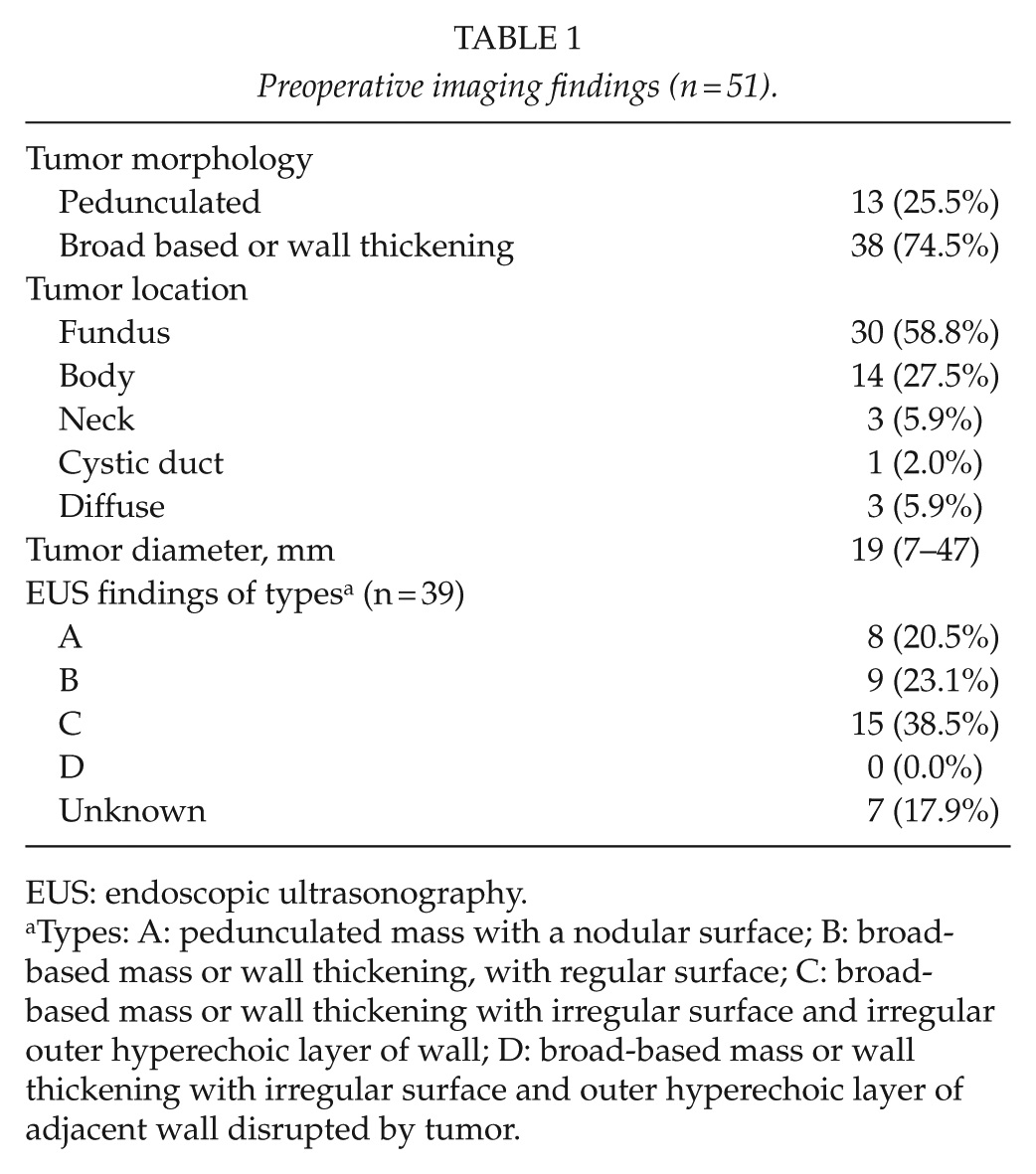
EUS: endoscopic ultrasonography.
Types: A: pedunculated mass with a nodular surface; B: broad-based mass or wall thickening, with regular surface; C: broad-based mass or wall thickening with irregular surface and irregular outer hyperechoic layer of wall; D: broad-based mass or wall thickening with irregular surface and outer hyperechoic layer of adjacent wall disrupted by tumor.
Pathological findings (n = 51).
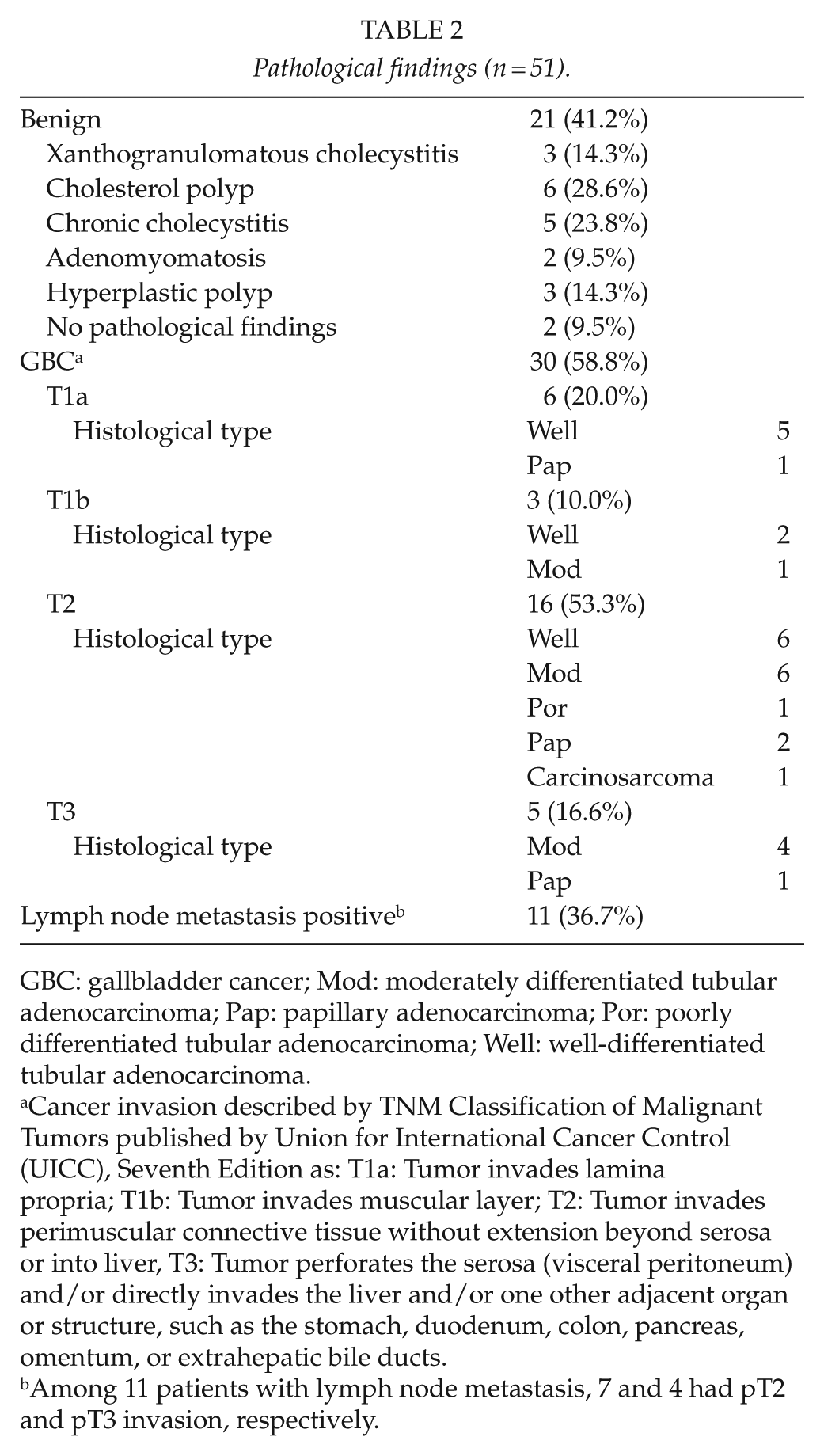
GBC: gallbladder cancer; Mod: moderately differentiated tubular adenocarcinoma; Pap: papillary adenocarcinoma; Por: poorly differentiated tubular adenocarcinoma; Well: well-differentiated tubular adenocarcinoma.
Cancer invasion described by TNM Classification of Malignant Tumors published by Union for International Cancer Control (UICC), Seventh Edition as: T1a: Tumor invades lamina propria; T1b: Tumor invades muscular layer; T2: Tumor invades perimuscular connective tissue without extension beyond serosa or into liver, T3: Tumor perforates the serosa (visceral peritoneum) and/or directly invades the liver and/or one other adjacent organ or structure, such as the stomach, duodenum, colon, pancreas, omentum, or extrahepatic bile ducts.
Among 11 patients with lymph node metastasis, 7 and 4 had pT2 and pT3 invasion, respectively.
Comparison of preoperative and pathological tumor depth among imaging modalities.
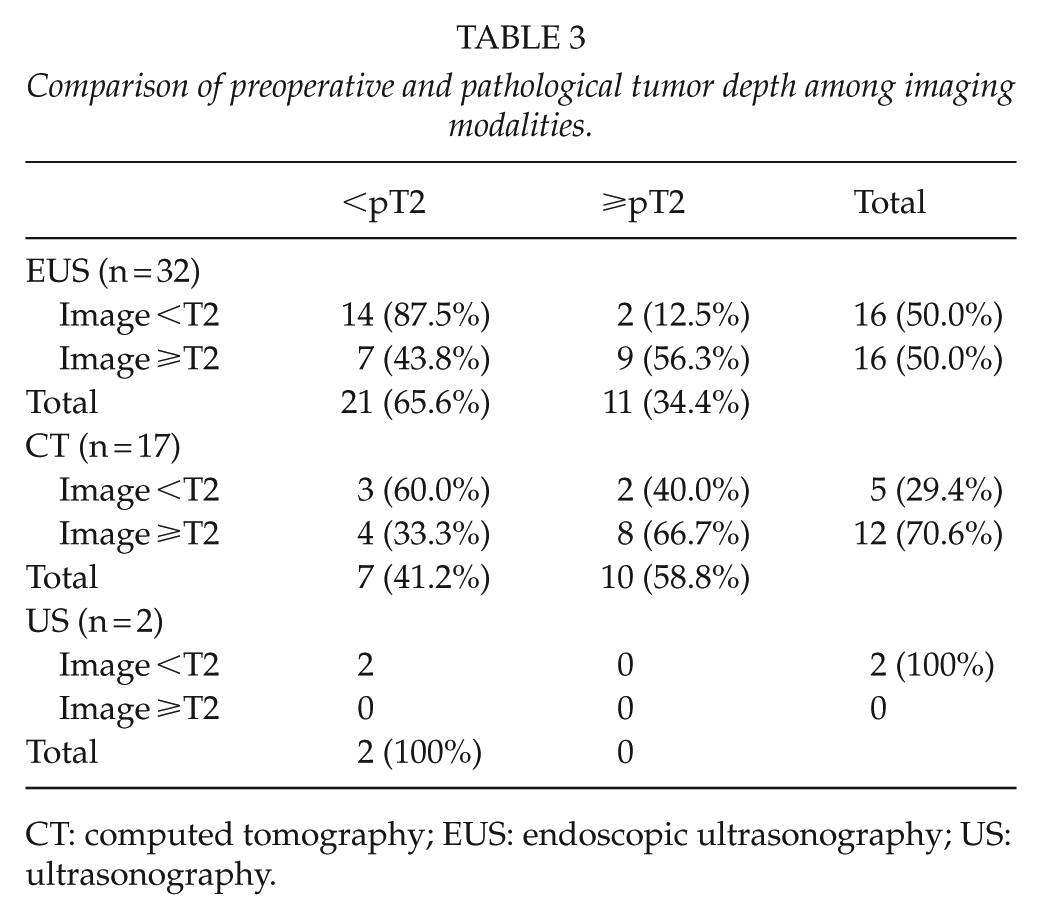
CT: computed tomography; EUS: endoscopic ultrasonography; US: ultrasonography.
Preoperative tumor variables were compared between patients with benign or <pT2 and ≥pT2 tumors. The results of univariate analyses showed that image ≥T2, tumor diameter, CEA, CA19-9, and SUVmax significantly differed between the two groups (Table 4). A total of 24 (47.1%) patients had missing FDG-PET data due to the severe comorbid diabetes or FDG-PET was judged unnecessary by physicians. Tumor diameter and CEA were independent predictive factors for ≥pT2 in multivariate analyses (Table 4). Tumor diameter correlated with CRP and SUVmax (r = 0.741 and 0.391, respectively), whereas CEA did not correlate with other variables (data not shown).
Uni- and multivariate analyses of predictive factors for ⩾T2.
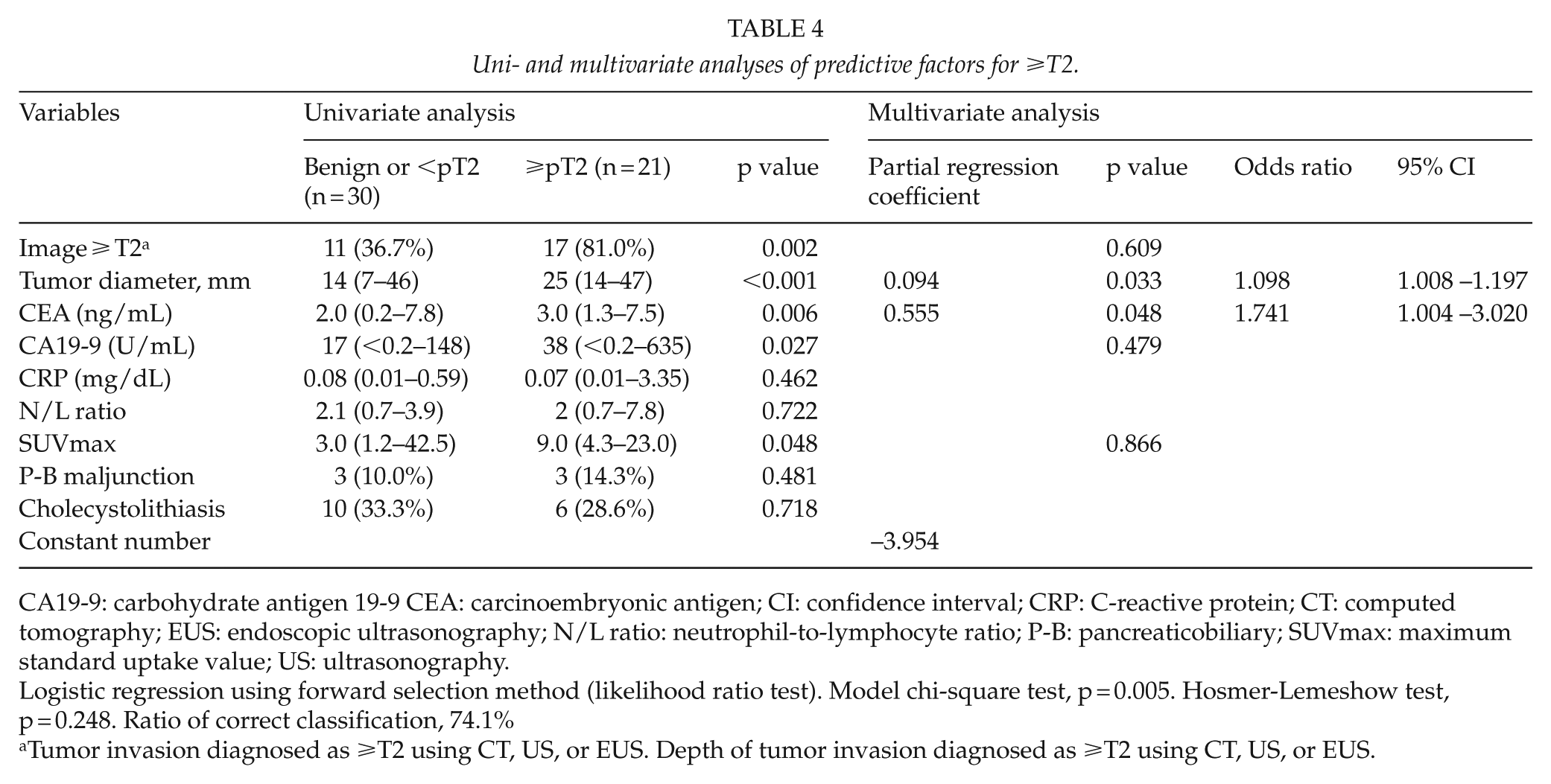
CA19-9: carbohydrate antigen 19-9 CEA: carcinoembryonic antigen; CI: confidence interval; CRP: C-reactive protein; CT: computed tomography; EUS: endoscopic ultrasonography; N/L ratio: neutrophil-to-lymphocyte ratio; P-B: pancreaticobiliary; SUVmax: maximum standard uptake value; US: ultrasonography.
Logistic regression using forward selection method (likelihood ratio test). Model chi-square test, p = 0.005. Hosmer-Lemeshow test, p = 0.248. Ratio of correct classification, 74.1%
Tumor invasion diagnosed as ⩾T2 using CT, US, or EUS. Depth of tumor invasion diagnosed as ⩾T2 using CT, US, or EUS.
The regression formula to calculate pT2 predictive scores was
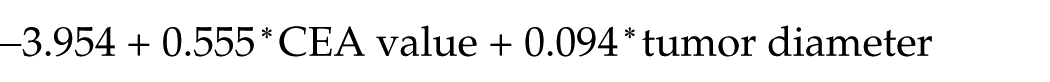
The AUC determined from scores calculated using the formula was 0.873 (Fig. 1). The cut-off that became the closest coordinate to the upper left of the ROC curve was −0.584, and the sensitivity and specificity derived from the cut-off were 85.0% and 87.1%, respectively.
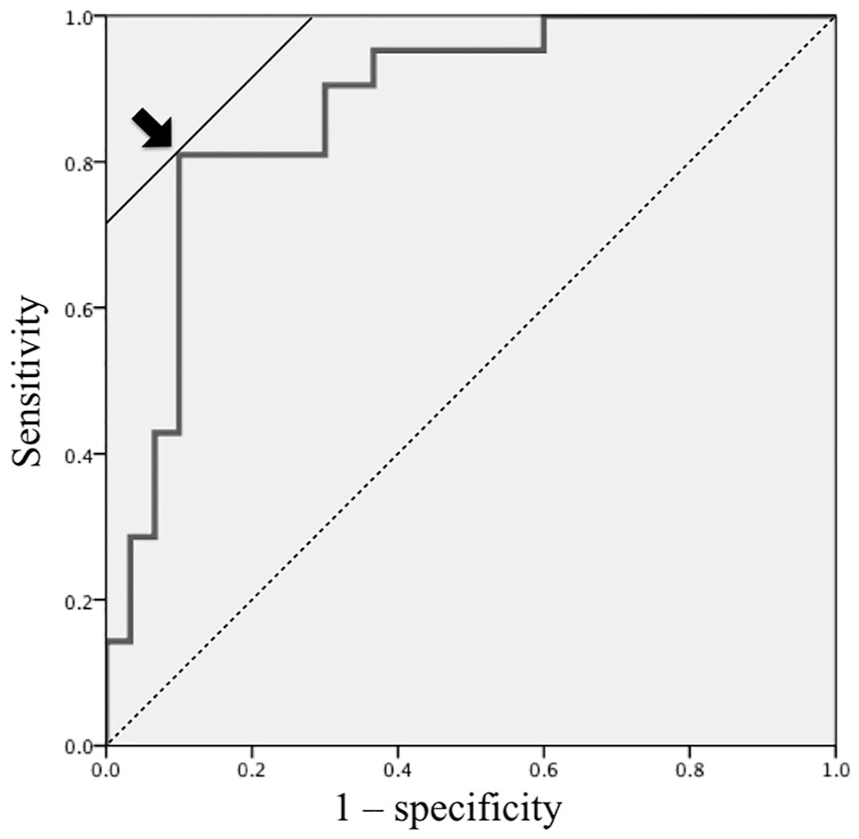
ROC curve generated using our scoring system.
Case Presentation
Figs 2 and 3 show typical patients. Fig. 2 shows a patient with pathological T1a that was suspected as being T2 from preoperative images. However, based on a tumor diameter of 25 mm and a preoperative CEA value of 1.6 ng/mL, the calculated pT2 predictive score was ‒0.716, indicating <pT2. Fig. 3 shows a patient with a preoperative diagnosis of <T2. However, based on a tumor diameter of 30 mm and a preoperative CEA value of 5.7 ng/mL, the calculated pT2 predictive score was 2.058, indicating ⩾pT2.
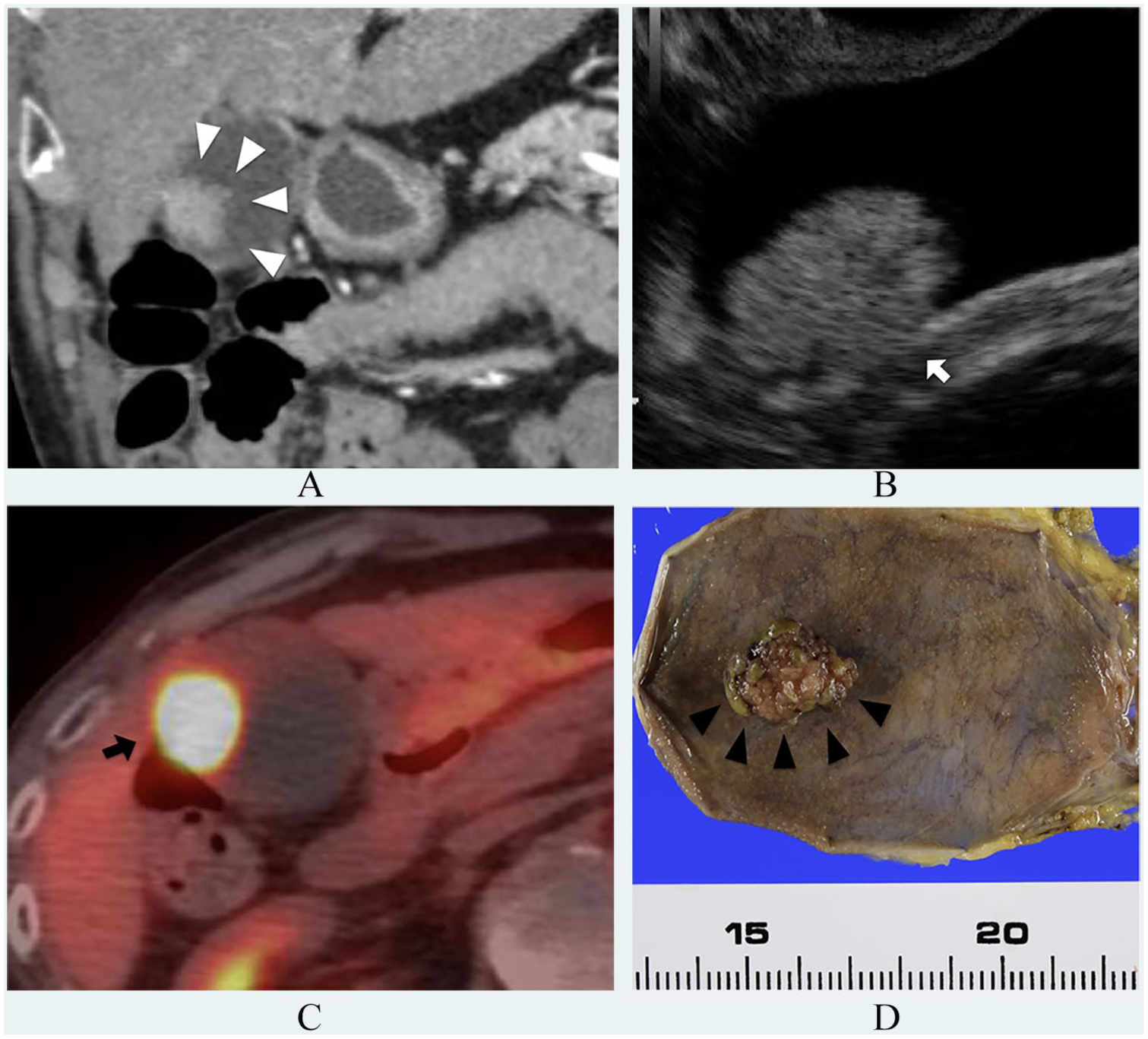
Patient preoperatively diagnosed with T2 that was pathologically T1a. (A) CT findings. Broad-based tumor with constricted wall at gallbladder body (white arrowhead). (B) EUS findings. Diameter of tumor located at gallbladder body is 25 mm. Outer hyperechoic layer is irregular (type C, white arrow). (C) FDG-PET findings. SUVmax of tumor located at gallbladder body, 42.5 (black arrow). (D) Gross and pathological findings of resected specimen. Tumor protrudes from fundus to gallbladder body. Tumor invasion is confined to mucosal layer (black arrowhead).
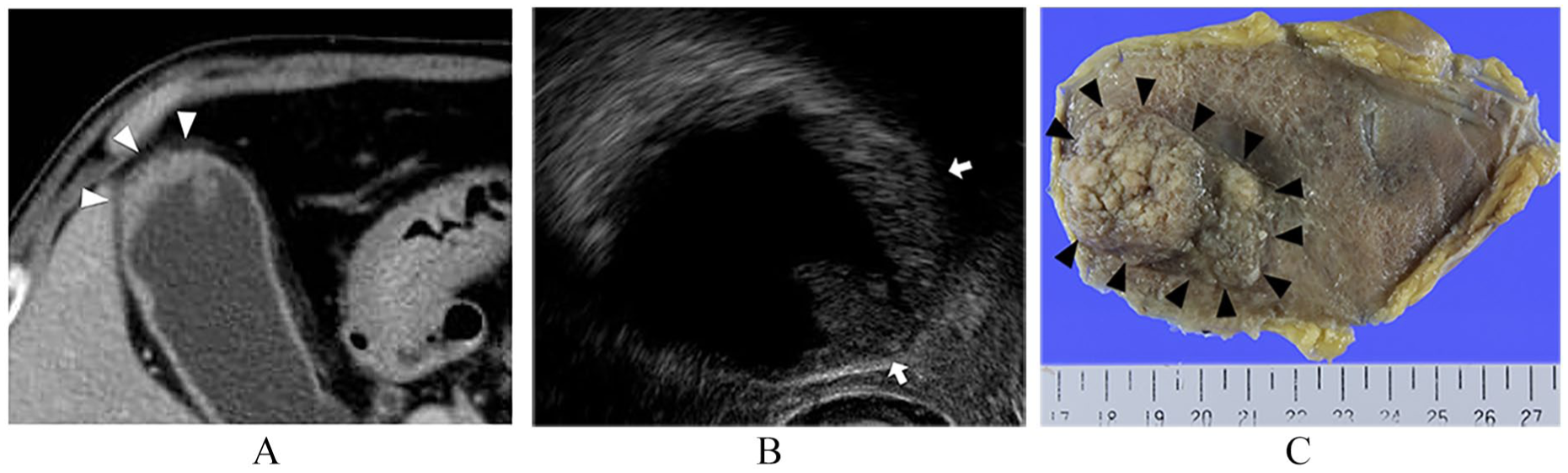
Patient preoperatively diagnosed with T1 GBC accompanied by adenomyomatosis that was pathologically T2. (A) CT findings: Lesion with wall thickening 30 mm in diameter with papillary tumor at fundus of gallbladder (white arrowhead). (B) EUS findings: Lesion with wall thickening lesion and spotty hyperechoic area at fundus beside papillary tumor. Outer hyperechoic layer is smooth (type B, white arrow). Gallbladder cancer combined with adenomyomatosis is suspected. (C) Gross and pathological findings of resected specimen. Lesion with rough surface wall thickening from fundus to gallbladder body (black arrowhead). Subserosal invasive adenocarcinoma along Rokitansky–Aschoff sinus mixed with ordinary serosal invasive adenocarcinoma.
Discussion
A preoperative diagnosis of tumor invasiveness is essential for selecting an appropriate strategy to treat pT2 GBC, as it requires cholecystectomy with an appropriate surgical margin and lymph node dissection. Because pT2 GBC includes patients that were preoperatively diagnosed with benign disease and vice versa (13), we aimed to identify ⩾pT2 among patients with suspected GBC including those with benign diseases.
Some studies have attempted to predict GBC tumor depth (14–16), but none have used tumor markers or FDG-PET. Since factors to predict the depth of GBC tumor invasion that were previously evaluated comprised mainly imaging features that might be open to different interpretations by physicians, radiologists, and surgeons, we attempted to create a more objective predictive method. Regarding imaging features, almost all patients were assessed by contrast-enhanced CT, whereas 39 patients with tumors that were relatively smaller than those of the other 12 patients were assessed by EUS. The positive predictive value for image ⩾T2 was lower for patients assessed by EUS than for those who were not. However, we did not consider that the diagnostic accuracy of EUS was low because the negative predictive value was 87.5%. Furthermore, to preoperatively diagnose smaller tumors is generally more difficult. We presently recommend EUS and CT for patients who have at least suspected GBC in addition to trans-abdominal US which are mainly used for screening. In addition, almost all patients underwent MRCP because preoperative assessment of biliary anatomy and finding a pancreaticobiliary maljunction are important for treating GBC (17, 18). If MRI was not informative, DIC-CT was added. Although we did not consider MRI useful with respect to determining tumor depth, diffusion-weighted MRI might support a preoperative diagnosis of GBC (19–21). However, even using diverse imaging modalities, the accuracy of a preoperative diagnosis of pT2 in our series, namely, a positive predictive value of “Image ⩾T2,” was not very high, at 17 (61%) of 28 patients (Table 3). Therefore, we combined biochemical data, FDG-PET, and conventional imaging findings to create a more accurate preoperative system. We found that tumor diameter and preoperative CEA values were independent predictors of ⩾pT2 GBC and we used them to create a scoring formula. The representative examples shown in Figs 2 and 3 indicate the accuracy of this formula.
Tumor diameter is important factor when measuring gallbladder polyps because larger polyps can become malignant (22–25). Although a correlation between tumor diameter and depth of tumor invasion has not been reported, larger tumors without pedunculated polyps are commonly considered to infiltrate deeper layers. Tumor diameters ranged from 7 to 47 mm in this study. Among 6 of the 51 patients with tumor diameters >36 mm, which automatically exceeded the cut-off, 4 had >pT2 GBC and 2 had XGC. Because XGC is often detected as diffuse thickening of the gallbladder wall, the diameter of such lesions is essentially larger. The diameters of the XGC in three patents were 46, 39, and 18 mm. Of these three, <pT2 could be diagnosed in only one patient using our scoring system. Thus, applying our scoring system to patients with diffuse wall thickning caused by XGC or diffuse chronic cholecystitis remains controversial, so our scoring system might be useful for tumors < 30 mm of which the depth of invasion can be difficult to preoperatively diagnose from images. In addition, other reports have associated GBC with XGC, which further complicates this controversy (26, 27). However, the CEA value might help to preoperatively diagnosis XGC because the preoperative CEA values of all three patients with XGC were <2.5 ng/mL. This was relatively low considering the tumor diameter. Further investigation into the preoperative diagnosis of XGC is required.
Biochemical tumor markers were predictors of ⩾pT2 in univariate analysis, whereas CRP and the N/L ratio, that are prognostic factors for several malignancies (28–30), were not significant for GBC tumor depth in our series. Both CEA and CA19-9 are serum tumor markers for GBC (31), but a correlation with tumor depth has not been reported. Although CA19-9 is the standard tumor marker for pancreatic adenocarcinoma and biliary tract tumors, CA19-9 can become elevated even under benign conditions such as cholangitis and pancreatitis (32, 33). However, CEA is important to the survival of patients with cholangiocarcinoma and GBC (33–35). Therefore, CEA might have reflected GBC progression more sensitively than CA19-9. The range of CEA values among the six patients who were excluded from this study due to apparent invasion of adjacent organs was 1.2–312 (median = 3.8) ng/mL, and their predictive scores exceeded the cut-off range of 1.812–172.59 ng/mL.
The depth of GBC tumors might be a concern not only in terms of the need for lymph node dissection but also the choice of surgical approach. Even GBC can now be treated by laparoscopic radical resection thanks to recent advances in surgical techniques and instrumentation (13). Although laparoscopic lymph node dissection might be problematic, at least laparoscopic cholecystectomy or whole-layer cholecystectomy with gentle handling to prevent intraoperative perforation of the gallbladder has the potential to become a standard procedure for general surgeons. Therefore, the accurate prediction of tumor depth might enable patients with a high probability of benign disease or <pT2 GBC to be treated using a laparoscopic approach, and only patients with a high probability of ⩾pT2 GBC will need to be treated by an open approach. More specifically, patients with suspected GBC accompanied by low scores in our system could be treated by laparoscopic whole-layer cholecystectomy, whereas those with high scores could undergo lymphadenectomy and extended cholecystectomy with an open approach. Of course, if a target lesion is located at the hepatic side of the gallbladder, combined gallbladder bed resection must be considered after intraoperative US regardless of predictive scores.
This study is limited by a small patient cohort, the data were derived from patients with limited ethnicity in a single institution, and data for some patients were incomplete. However, the accumulation of data from more patients and verification of the validity of our scoring system should overcome these limitations. Including preoperative CEA values in our novel scoring system accurately predicted tumor depth and will enable the selection of optimal surgical procedures for patients with suspected GBC.
In conclusion, we created a novel scoring system using CEA and tumor diameter to predict the tumor depth of GBC. We also found that CEA values are important for the preoperative evaluation of GBC.
Footnotes
Acknowledgements
The authors thank Taiji Tohyama, Jota Watanabe, Taro Nakamura, Tetsuya Mizumoto, Kei Tamura, Masahiko Honjo, and Hisato Yamamoto for assistance with preliminary data analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The institutional review board at Ehime University Hospital approved this study (Approval number: 1712021), which proceeded in accordance with the ethical standards established in the Declaration of Helsinki in 1995 (revised, Brazil 2013).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Written informed consent was obtained using the opt-out principle. The study and the right of refusal to participate were disclosed to the public online.
