Abstract
Background and Aims:
Traumatic pancreatic injury is associated with high morbidity and mortality rates, and the management strategies associated with the best clinical outcomes are unknown. Our aims were to identify the incidence of traumatic pancreatic injury in adult patients in the United States using the National Trauma Data Bank, evaluate management strategies and clinical outcomes, and identify predictors of in-hospital mortality.
Materials and Methods:
We retrospectively analyzed National Trauma Data Bank data from 2007 to 2011, and identified patients ⩾14 years old with pancreatic injuries either due to blunt or penetrating trauma. Patient characteristics, injury-associated factors, clinical outcomes, and in-hospital mortality rates were evaluated and compared between two groups stratified by injury type (blunt vs penetrating trauma). Statistical analyses used included Pearson’s chi-square, Fisher’s exact test, and analysis of variance. Factors independently associated with in-hospital mortality were identified using multivariable logistic regression.
Results:
We identified 8386 (0.3%) patients with pancreatic injuries. Of these, 3244 (38.7%) had penetrating injuries and 5142 (61.3%) had blunt injuries. Penetrating traumas were more likely to undergo surgical management compared with blunt traumas. The overall in-hospital mortality rate was 21.2% (n = 1776), with penetrating traumas more likely to be associated with mortality (26.5% penetrating vs 17.8% blunt, p < 0.001). Unadjusted mortality rates varied by management strategy, from 6.7% for those treated with a drainage procedure to >15% in those treated with pancreatic repair or resection. Adjusted analysis identified drainage procedure as an independent factor associated with decreased mortality. Independent predictors of mortality included age ⩾70 years, injury severity score ⩾15, Glasgow Coma Scale motor <6, gunshot wound, and associated injuries.
Conclusions:
Traumatic pancreatic injuries are a rare but critical condition. The incidence of pancreatic injury was 0.3%. The overall morbidity and mortality rates were 53% and 21.2%, respectively. Patients undergoing less invasive procedures, such as drainage, were associated with improved outcomes.
Introduction
Pancreatic trauma is rare, occurring at an incidence ranging from <1% (1) to 12% of all abdominal trauma (2). When present, pancreatic injury can be a devastating insult with significant morbidity and mortality. Reported rates of mortality have varied widely, ranging from 2% to 40% (3, 4), while morbidity rates are as high as 60% (5, 6, 7). Mortality is highly associated with exsanguination from damage to critical vascular structures located near the pancreas (3). The average length of stay (LOS) after pancreas injury has been estimated to be 2–4 weeks (4, 8).
Recent trends in management have demonstrated a shift toward initial non-operative (medical) management or avoidance of major pancreatic resection at the time of initial exploratory laparotomy (4), and existing studies have not found a benefit of early major pancreatic resection (4, 9, 10). In fact, the current management algorithm for pancreatic injury from the 2013 Western Trauma Association (WTA) guidelines recommend less invasive interventions such as drainage alone for most low-grade pancreas injuries and delaying definitive repair in hemodynamically unstable patients (11); however, rigorous data to support these recommendations remains limited. The ideal management of pancreatic trauma associated with the best outcomes is not known (2).
There is a paucity of well-designed and/or large sample-sized studies evaluating the management of pancreatic injuries in trauma patients and morbidity and mortality rates. The 2016 Eastern Association for the Surgery of Trauma (EAST) guidelines on the management of pancreatic injury are based off few very low-quality evidence studies, most of which were comprised of small patient sample sizes or limited institutional samples (2). In addition, there are few database studies that comprehensively examine the management, outcomes, and predictors of mortality of pancreatic trauma (3, 4, 10, 12). It is difficult to make generalized recommendations regarding the optimal management of pancreatic injuries.
To our knowledge, this is the largest National Trauma Data Bank (NTDB) study evaluating the US adult pancreatic trauma patient population. The major aims of this study were to (1) estimate the incidence of traumatic pancreatic injury, (2) describe the patient characteristics and injury-associated factors, (3) describe management strategies, (4) determine morbidity and mortality rates, and (5) identify predictors of mortality in US adult patients with abdominal trauma presenting with pancreatic injuries as identified by the NTDB from the years 2007 to 2011.
Materials and Methods
This retrospective cohort database study was approved by the institutional review board at Duke University Medical Center. This manuscript observes the pertinent guidelines of the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement.
Study Population
We queried the NTDB between the years of 2007 and 2011, for trauma patients who were ⩾14 years of age and had significant abdominal injury defined as an Abbreviated Injury Scale (AIS) abdominal score of ⩾2. Only patients with pancreatic injury codes as defined by the International Classification of Disease, Ninth Revision, Clinical Modification (ICD-9-CM; Appendix 1) were included for further analysis. This included patients who had concomitant pancreatic and hepatic injury. Patients who sustained both blunt and penetrating trauma and any mechanisms of injury (i.e. motor vehicle accident (MVA), assault, fall, etc.) were included in the analysis.
Patient demographic data, which included age, gender, race, and health insurance information, were collected. The injury profile incorporated the injury severity score (ISS), AIS for non-abdominal body region injuries, pancreatic injury location, injury mechanism and intent, shock (defined as systolic blood pressure (SBP) < 90 mmHg), total and motor Glasgow Coma Scale (GCS), and presence of major traumatic brain injury (TBI).
ICD-9-CM procedure codes (Appendix 2) were used to identify the types of procedures performed in patients with pancreatic injuries, which included non-pancreatic operations as well as pancreatic-related operations and procedures. Pancreatic injury management strategies were divided into five categories: (1) non-operative, (2) abdominal operation without pancreatic intervention, (3) pancreatic drainage, (4) pancreatic repair, and (5) pancreatic resection. Although patients may have undergone surgical procedures unrelated to their pancreatic injuries (i.e. exploratory laparotomy for splenectomy, thoracotomy, etc.), those who had concomitant surgical management of pancreatic injuries were include in our analysis.
Outcomes
Our primary outcome was in-hospital mortality. Secondary outcomes included incidence of pancreatic injury, major complications (i.e. acute renal failure (ARF), acute respiratory distress syndrome (ARDS), wound infection, deep vein thrombosis (DVT), pulmonary embolism (PE), myocardial infarction (MI), pneumonia, stroke, and sepsis), hospital LOS, intensive care unit (ICU) LOS, total number of ventilator days, and discharge disposition.
Statistical Analysis
Descriptive statistics were used to present all patient variables and they were pronounced in the overall pancreatic injury population and by type of injury (blunt vs penetrating trauma). We reported categorical data as frequencies and percentages. We used medians and interquartile ranges (IQRs) to report continuous data.
In-hospital mortality rates and secondary outcomes were then compared by injury type (blunt vs penetrating). In-hospital mortality rates were also compared by intervention strategy (non-operative, non-pancreas operation only, pancreatic drainage, pancreatic repair, and pancreatic resection) and stratified by overall, blunt, and penetrating pancreatic injury. In this analysis, we excluded patients who expired in the emergency department (ED) within 24 h of admission. Comparisons between categorical variables was performed using Pearson’s chi-square or Fisher’s exact test. The Wilcoxon–Mann–Whitney test was used to compare continuous variables.
Multivariable logistic regression was used to identify factors independently associated with in-hospital mortality, after adjusting for confounders such as injury and demographic characteristics. The following variables were included: age, treatment strategy, gender, race, pancreatic injury location and injury severity grade, ISS, associated injuries with AIS severity ⩾3, shock, GCS motor <6, presence of TBI, and mechanism of injury. Missing data were handled using complete case analysis. The p values <0.05 were considered significant, and type I error was controlled at the level of the comparison. Statistical analyses were performed using Stata (version 12.0; Stata Corp, College Station, TX) and R version 3.01 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Incidence and Patient Demographics and Injury Characteristics
There were 2.8 million trauma patients who were ⩾14 years old identified in the NTDB search between 2007 and 2011. Of these patients, 230,533 sustained abdominal injuries. The incidence of pancreatic trauma was 0.3% (n = 8386). This included patients with concomitant pancreatic and hepatic injuries (3420, 0.1%). Table 1 reported the demographic, mechanism of injury, and associated injuries data for patients with pancreatic injury, stratified by blunt versus penetrating trauma. Of the patients with pancreatic trauma, 61.3% (n = 5142) were involved in blunt trauma, while the remaining cases involved penetrating trauma (n = 3244, 38.7%) (Table 1).
Patient demographic and injury characteristics stratified by injury type (blunt vs penetrating).
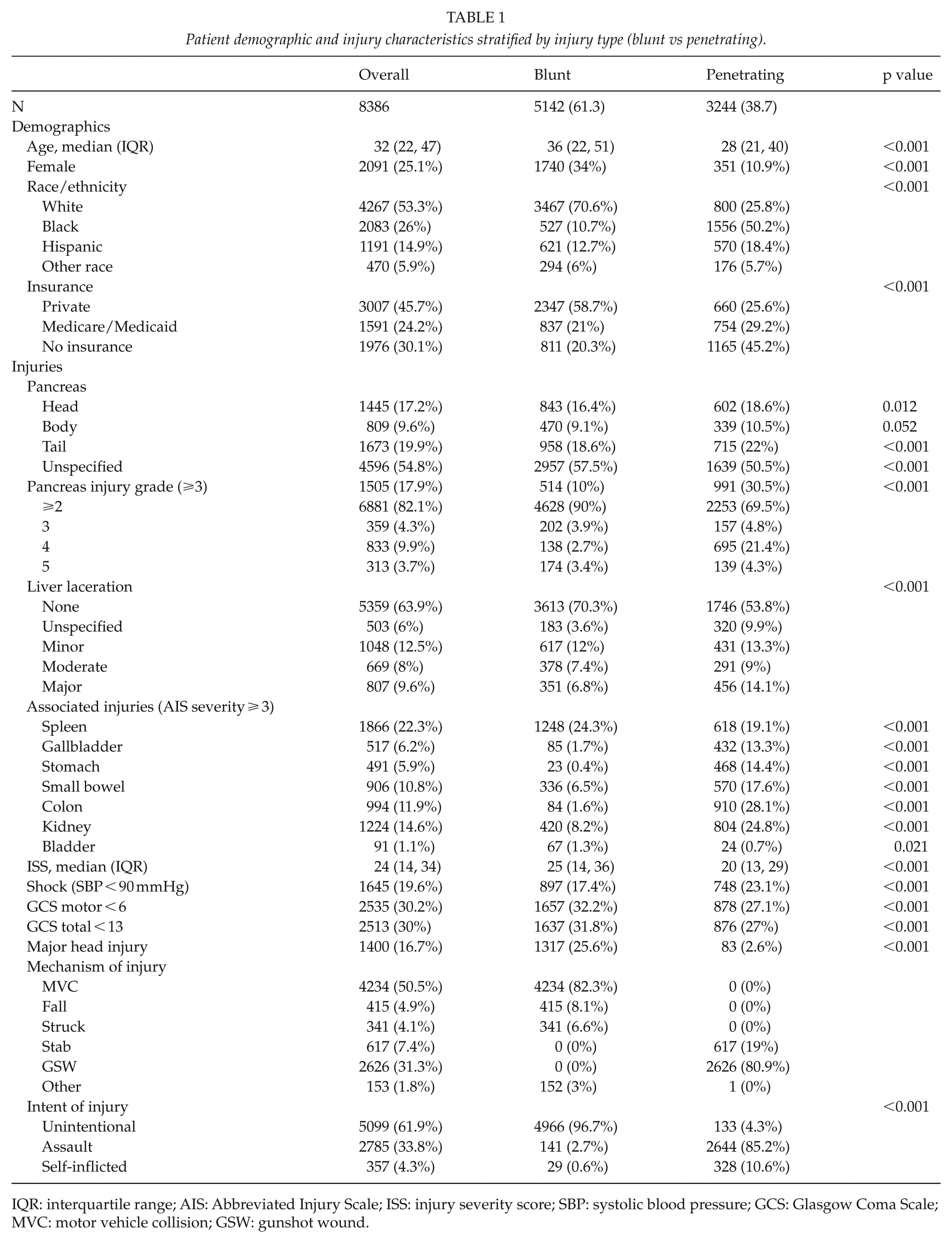
IQR: interquartile range; AIS: Abbreviated Injury Scale; ISS: injury severity score; SBP: systolic blood pressure; GCS: Glasgow Coma Scale; MVC: motor vehicle collision; GSW: gunshot wound.
Overall, patients with pancreatic trauma had a median age of 32 years, were male (n = 6295, 74.9%), were White (n = 4267, 53.3%), and had private health insurance (n = 3007, 45.7%) (Table 1). There were 31% (n = 2524) of patients with a concomitant liver laceration injury, and 1505 (17.9%) had a pancreas injury severity grade of ⩾3. The median ISS was 24 (IQR = 14–34). The majority of injuries were unintentional (61.9%, n = 5099), with the main mechanism of injury being an MVA (50.5%, n = 4234).
When comparing characteristics of pancreatic trauma patients by type of injury, patients with penetrating trauma were more likely to be younger (median age 28 vs 36 years; p < 0.001), male (89.1% vs 66.0%; p < 0.001), Black (50.2% vs 10.7%; p < 0.001), and uninsured (45.2% vs 20.3%; p < 0.001), as compared with those with blunt trauma. Penetrating trauma victims were also more likely to have grade 3 or higher pancreatic injury severity, and lower median ISS scores (20 vs 25, p < 0.001) (Table 1). The most common mechanisms of injury for penetrating trauma were gunshot (n = 2626, 81%) and stab wounds (n = 617, 19%).
Primary Outcome
Overall, in-hospital mortality was 21.2% (n = 1776; Table 2). Patients with penetrating trauma had higher in-hospital mortality rates (26.5% vs 17.8%; p < 0.001) compared with blunt trauma (Table 2).
Outcomes after pancreatic injury, stratified by injury type (blunt vs penetrating).
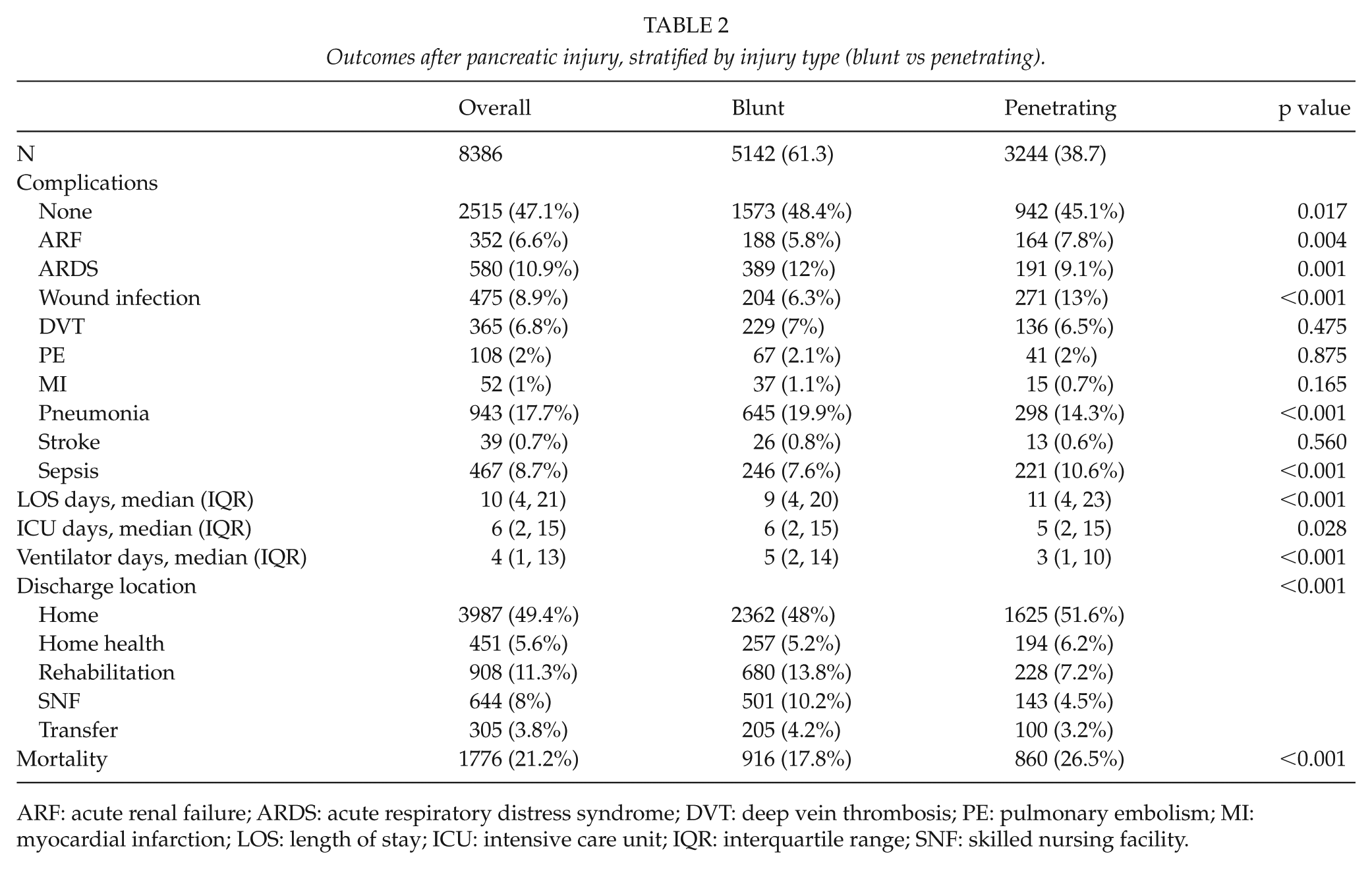
ARF: acute renal failure; ARDS: acute respiratory distress syndrome; DVT: deep vein thrombosis; PE: pulmonary embolism; MI: myocardial infarction; LOS: length of stay; ICU: intensive care unit; IQR: interquartile range; SNF: skilled nursing facility.
Secondary Outcomes
Table 3 reported procedure-related data for pancreatic injury patients as stratified by blunt and penetrating trauma. Overall, 36.2% of patients (n = 3034) with pancreatic injury were medically managed, while 34.8% of patients (n = 2918) underwent a non-pancreatic operation (Table 3). A total of 2434 patients (29%) underwent a pancreatic-related procedure. Of these patients, 4.1% (n = 344), 8.4% (n = 707), and 16.5% (n = 1383) underwent pancreatic drainage, repair, or resection, respectively. Blunt pancreatic trauma was more likely to be managed with non-operative management compared with penetrating trauma (50.7% vs 13.2%, p < 0.001). Penetrating trauma victims were more likely to undergo surgical management for their pancreatic injury compared with blunt trauma, which included pancreatic resection (26.5% vs 10.2%; p < 0.001) and pancreatic repair (15.6% vs 5.6%, p < 0.001) (Table 3).
Management of pancreatic injury stratified by injury type (blunt vs penetrating).
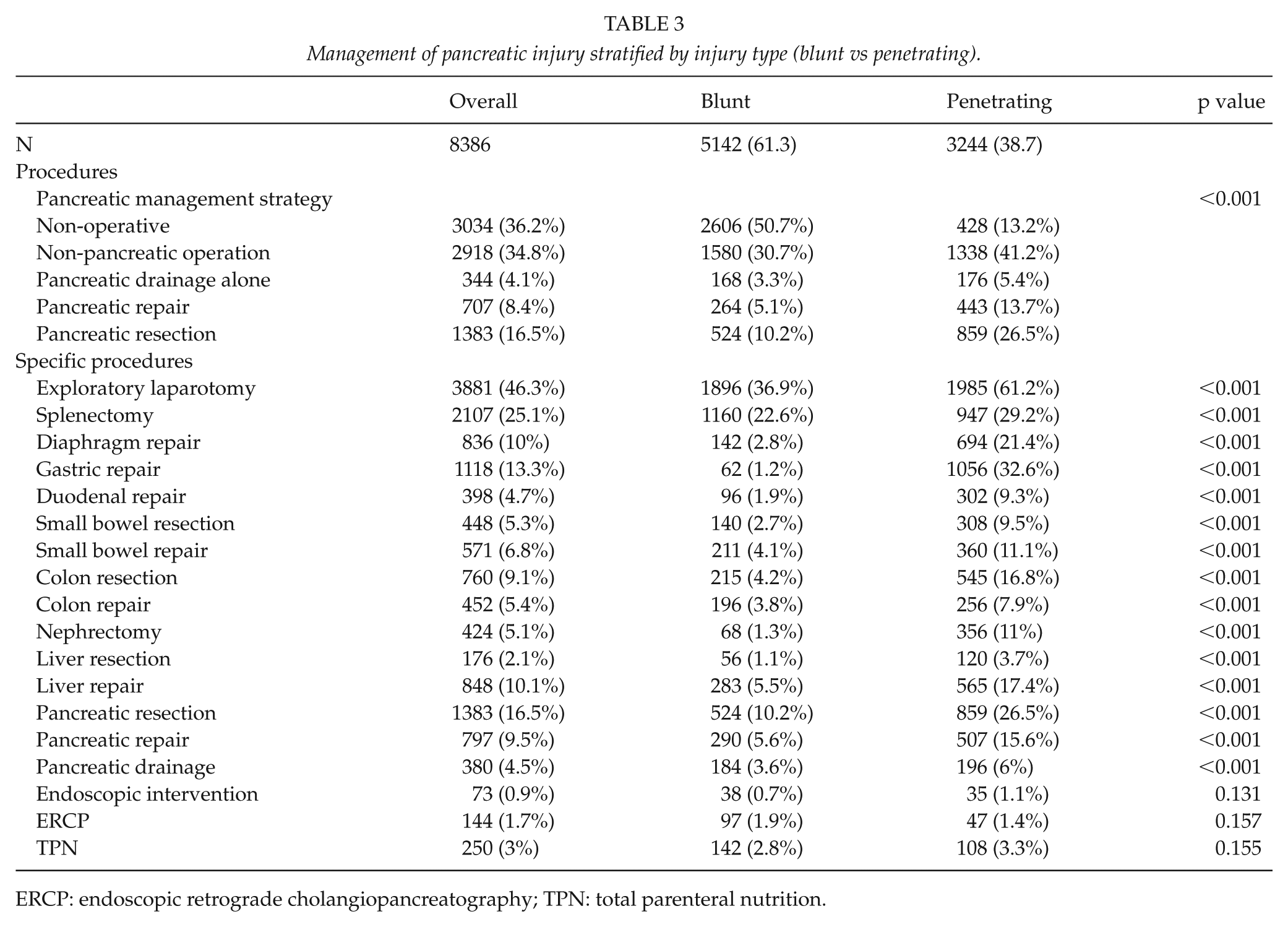
ERCP: endoscopic retrograde cholangiopancreatography; TPN: total parenteral nutrition.
Overall, 53% of patients experienced complications, with penetrating trauma having 54.9% complications compared with 51.6% in blunt trauma. Blunt trauma patients were more likely to have no complications compared with penetrating trauma patients (48.4% vs 45.1%, p = 0.017). ARF, wound infection, and sepsis were more likely in penetrating trauma patients, while ARDS and pneumonia occurred with a higher frequency in blunt trauma patients (Table 2). The overall median ICU and hospital LOS were 6 and 10 days, respectively. Median hospital LOS was longer for pancreatic injury patients with penetrating trauma (11 vs 9; p < 0.001); however, the median ICU LOS was longer for patients with blunt trauma (6 vs 5; p = 0.028). In addition, blunt trauma patients had more ventilator days (5 vs 3 days, p < 0.001) than penetrating trauma patients (Table 2).
Morbidity and Mortality Stratified By Management
When comparing operative and non-operative management, patients undergoing non-operative management had the lowest morbidity, with 40% experiencing a complication. These patients also had the shortest LOS (7 days; IQR = 3–15 days). Complication rates for those undergoing a procedure were as follows: pancreatic drainage, 57.5%; pancreatic repair, 63.0%; pancreatic resection, 57.5%; and abdominal surgery without pancreatic procedure, 60.2%. Patients undergoing pancreatic drainage, repair, and resection had hospital LOS of 16 (IQR = 9–27), 14 (7–26), and 14 (8–28) days, respectively.
Patients undergoing abdominal surgery without a pancreatic procedure were associated with the highest mortality rates in the overall, blunt, and penetrating trauma populations (23.8%, 20.7%, and 27.4%, respectively). Conversely, patients undergoing only a pancreatic drainage procedure were associated with the lowest mortality rates in these groups (6.1%, 4.9%, and 7.2%, respectively).
Multivariable logistic regression demonstrated that compared with non-operative management, abdominal operation without pancreatic procedure was associated with an adjusted odds ratio (AOR) of mortality of 1.25 (95% confidence interval (CI) = (1.02–1.52)), while pancreatic drainage was associated with lower AOR = 0.32 (95% CI = (0.19–0.54)). Mortality rates for pancreatic repair were similar to non-operative management, while pancreatic resection had a lower AOR = 0.65 (95% CI = (0.49–86)).
Risk Factors For In-Hospital Mortality
Multivariable logistic regression also identified several factors associated with in-hospital mortality, which are presented in Fig. 1 and included the following: older age, male sex, higher ISS, shock, presence of major TBI, gunshot wound, more severe pancreatic injury (grade ⩾ 4), and non-pancreatic operation (Fig. 1).
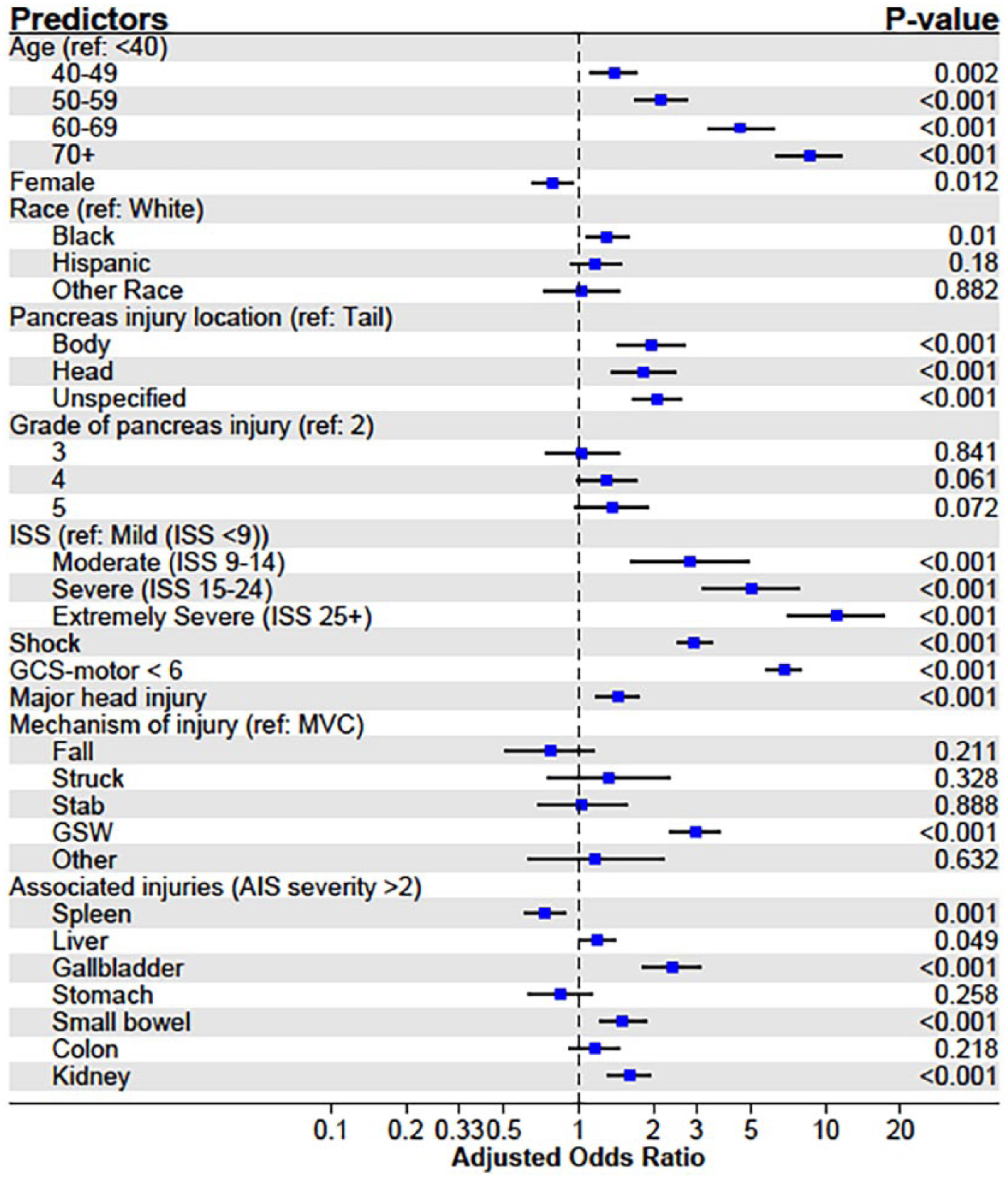
Forest plot of predictors of mortality from multivariable logistic regression.
Discussion
To our knowledge, we performed the largest US NTDB analysis of adult patients with pancreatic trauma injuries. Compared with other US databank studies (3, 4, 10, 12, 13), we included both blunt or penetrating trauma mechanisms, all pancreatic injury grade severities, and all management types. We performed a more thorough evaluation and assessment of this patient population with respect to demographics, injury-associated characteristics, management strategies, and effects on outcomes as stratified by blunt and penetrating injury, as well as in-hospital mortality and other clinical outcomes related to the intervention strategy.
We determined the incidence of pancreatic injuries in US trauma patients ⩾14 years of age to be 0.3% of all trauma injuries and 3.6% of all abdominal traumas reported in the NTDB during 2007–2011; the incidence of pancreatic injury without hepatic injury was 0.2%. Our results support the previously reported pancreatic injury incidence of 0.2%–2% of all trauma (14, 15, 16, 17) and 0.2%–12% of all abdominal trauma (2, 3, 13, 15–19). Although penetrating pancreatic injuries were reported to be more commonly seen in adult patients (18) and in US patients with a reported incidence of 48%–81% (5, 9, 10, 15, 20, 21), we reported that blunt trauma was the most common mechanism of injury in pancreatic trauma (61% compared with 39%, respectively). Outside the United States, blunt trauma is the more common mechanism of injury in pancreatic trauma patients (1, 15, 16). Our study provides a robust estimate of the incidence of pancreatic trauma in the United States, and while this estimate is only generalizable to centers participating in the NTDB, it is likely broadly generalizable to other US trauma centers.
MVAs were the most common cause of blunt pancreatic injuries, while penetrating pancreatic injuries were most commonly due to gunshot wound. Similar observations were reported in other US databank studies (3, 4, 12, 13). Overall, patients with pancreatic injuries were likely to be male and young (median age of 32 years), which is a finding consistent with reports from both United States and international literature (3, 4, 10, 12, 13, 16, 17, 22). Patients in our population were likely to be White, have more severe ISS (⩾15), and have concomitant intraabdominal organ injuries with AIS severity ⩾3 (i.e. spleen, kidney, colon, etc.). Compared with penetrating injuries, blunt pancreatic trauma patients were more likely to be older (median age 36 vs 28 years), male, White, have private insurance, have distal pancreas injury, have less severe pancreas injury (<3), have concomitant major TBI, have higher median ISS, and were less likely to have an associated liver laceration. Penetrating trauma patients also were more likely to have more intraabdominal organ injuries with AIS ⩾ 3. Previous studies have indicated the high proportion of concomitant organ injuries in cases involving pancreatic injury (17). In our study, over 90% of patients had an additional abdominal injury, and 60% of patients had an additional abdominal organ injury with AIS ⩾ 3. The presence of pancreatic injury at evaluation clearly necessitates a thorough search for other abdominal as well as thoracic injuries.
We reported an overall in-hospital mortality rate of 21.2%. Mortality rates for blunt and penetrating pancreatic trauma were 17.8% and 26.5%, respectively. Overall, 53% of patients had complications; this was similar for both the blunt and penetrating trauma groups. Ragulin-Coyne et al. examined over 27,000 patients with pancreaticoduodenal injury using the National Inpatient Sample (NIS) and reported morbidity and mortality rates of 47% and 11.5%, respectively (4). We reported morbidity and mortality rates almost double those reported by Ragulin-Coyne et al., which may be due to the different patient populations and population sizes being examined. Ragulin-Coyne et al. examined a larger population of patients with pancreaticoduodenal injuries (pancreas only 58% (n = 15,812), pancreas + duodenum 8.4% (n = 2286)), while we examined a smaller population of patients with primarily pancreatic injuries (n = 8385). In addition, we analyzed a large group of penetrating trauma (nearly 40%) patients, which may have varied from the patient population in the Ragulin-Coyne study (they did not specify the percentage of patients sustaining penetrating trauma) (4). The NTDB is a heterogeneous group of trauma centers and other hospitals, but is likely to have a more acute population than that seen in the NIS.
We identified the following factors to be independently associated with in-hospital mortality: age > 40 years, male sex, proximal pancreatic injury, ISS (⩾9), pancreatic injury severity grade ⩾ 3, shock, GCS motor score < 6, major TBI, concomitant injury of the gallbladder, small bowel, colon, and kidney (AIS ⩾ 3), non-pancreatic operation, and gunshot wound. Similarly, Ragulin-Coyne et al. identified age > 40 years, penetrating trauma, combined pancreaticoduodenal injury, being uninsured, and undergoing any surgery (but not pancreatic-specific surgery) as predictors of mortality (4). Phillips et al.’s analysis of 777 NTDB patients with penetrating pancreatic injuries, identified older age, lower GCS in stab wounds, higher heart rate, and pancreatic injury severity grade 5 as predictors of mortality (12). Finally, an analysis of 1155 UK trauma patients with pancreaticoduodenal injury identified age, ISS, shock, and non-operative management as predictors of mortality. Number of associated intra- and extraabdominal injuries, sex, and mechanism of injury were not associated with mortality prediction (16). An NTDB analysis of isolated blunt pancreatic injuries reported age, operative intervention, and ISS as independent predictors of mortality; male sex and pancreatic injury severity grade were not predictors (13).
Recent studies have called for the use of non-operative management when possible and damage control surgery if necessary (1, 2, 4, 10, 11, 13, 21, 23, 24), although the management strategy resulting in the best outcomes is not known (2). Furthermore, management may be dictated by the grade of organ injury severity, mechanism of trauma (blunt vs penetrating) (13) and specific pancreatic structure injuries (i.e. pancreatic duct injury) (16). Ragulin-Coyne et al. demonstrated trends over time toward non-operative management, with a slight decrease in mortality rates, but unchanged complication rates. However, the mortality rates among patients who underwent surgical interventions did not change. Patients who underwent surgical or pancreas-specific surgical interventions had an increase in complication rates (4). Similarly, Antonacci et al. showed a trend toward improved mortality associated with less invasive pancreatic procedures after controlling for trauma-specific factors, and reported better outcomes associated with shorter operation times and damage control surgery rather than definitive pancreatic surgery (24). Even in patients with high grade pancreatic injuries (grade 3 and 4), non-operative management may be preferred (3, 10). In addition, pancreatic resections are reported to be associated with longer lengths of stay (1, 3, 13, 21, 23), and surgical interventions are associated with increased mortality (13, 23).
Several treatment algorithms have been proposed for the management of pancreatic injury, with an emphasis on non-operative management for hemodynamically stable patients or drainage after exploration for unstable patients with either proximal or grade 1 and 2 injuries The 2016 EAST guidelines recommend non-operative management for grade 1 and 2 injuries, resection for grade 3 and 4 injuries, and cannot provide recommendations on the optimal management of grade 5 pancreatic injuries (2). Our results demonstrate that most patients underwent non-operative management and patients requiring pancreatic drainage only had decreased mortality rates (6.1%) compared with non-operative management (13.3%), surgery without pancreatic procedure (23.8%), and those undergoing pancreatic repair or resection (15.7% and 15.2%, respectively). While the comparison of treatment strategy undoubtedly lacks sufficient data to control for all confounding variables associated with outcomes, these results suggest that less invasive management such as pancreatic drainage alone, may be associated with improved outcomes.
It is important to note that the level of skill and expertise of the trauma surgeon may influence patient outcomes in the management of pancreatic trauma. Surgical volume (25) and learning curve studies (26) have demonstrated that surgeon experience and the volume of surgical cases a surgeon performs may influence outcomes and complication rates, especially for complex surgeries. Given the rare nature of pancreatic trauma, even for major level I trauma centers, early pancreatic surgeries or resections attempted by trauma surgeons, who do not routinely perform these procedures, may potentially lead to adverse outcomes. Future prospective studies on the optimal management of pancreatic trauma and associated outcomes are warranted.
Study Limitations
This study has clear limitations related to its retrospective nature, including the inability to compare our patients to a control group and the inability to control for unmeasured confounders. Although the NTDB has data from over 900 US trauma centers, there are numerous limitations associated with its data. The NTDB is not a population-based sample; therefore, these results are only generalizable to NTDB-participating centers, which usually are larger centers accustomed to treating more severe traumatic injuries. Data are submitted voluntarily and are subject to selection bias, inconsistent measurement of specific data variables, inter-hospital differences, and information bias (i.e. variability in the AIS versions among hospitals). The data quality may be questionable based on the data submitted by individual hospitals and how well national trauma data standards are used by the individual hospitals; there may be missing or incomplete data, and data entry errors. The NTDB may be missing data on patient factors such as imaging, laboratory results, long-term follow up information, and medical comorbidities (although the young age of the study population likely mitigated this limitation).
Due to the large sample size, our data are susceptible to type I errors. In addition, the age cut-off value we selected (⩾14 years of age) is another source of selection bias and may have confounded our results. There are no clear age definitions for adult patients that will send them exclusively to an adult trauma center, and different institutions have various age cut-offs. The lowest age cut-off for adult trauma centers that we are aware of is 14 years of age and we chose the most inclusive age range to justify excluding patients who may be treated by adult trauma surgeons. The age choice is a source of selection bias which may confound our results.
We did not evaluate for isolated pancreatic injuries or compare outcomes between isolated pancreatic injury or pancreatic injuries with minor coexisting injuries to more severe coexisting injuries. The decision to avoid isolated pancreatic injuries was mainly because isolated pancreatic injuries are extremely rare, and commonly occur with other injuries. In selecting for isolated pancreatic injuries, our sample size would be considerably smaller and make our results less generalizable, as most pancreatic injuries present with other coexisting injuries. The coexisting intraabdominal and extraabdominal injuries may influence the mortality rates, outcomes, and procedures required. Furthermore, patients with concomitant liver injury were included in analysis. Patients with hepatic injuries may be severely ill, so there may be a difference in outcomes if patients have isolated pancreatic injuries. In addition, we did not stratify outcomes and intervention strategies by injury severity or by other coexisting injuries.
As previously mentioned, the expertise of the surgeon is important in outcomes and management. We did not have access to such information, and this is another limiting factor. We also did not compare management strategies between various trauma centers (level I vs level II). Our study is limited by selection bias, as we could not obtain crucial information which could have influenced a surgeon’s operative management decision. Intraoperative findings and the hemodynamic status of the patient over time would both be helpful factors in attempting to control for treatment decisions. We could not extrapolate information on whether the surgery was a damage control procedure or the timing of surgery; definitive surgery versus interval surgery, as well as early versus delayed surgery, could have very different effects on patient outcomes. Previous studies have indicated higher rates of drainage after pancreatic injury than we reported in our study (18). As the NTDB relies on ICD-9-CM procedure codes for procedural details, the use of pancreatic drains and stents, particularly in cases with other major abdominal procedures, may have been underreported. In addition, the involvement and extensiveness of pancreatic duct injury was not reported, and this may have contributed to variability in management strategies and patient outcomes.
We were unable to compare early (with 48 h of injury) versus late diagnosis of pancreatic injury and the impact on outcomes, nor look at potential risk factors contributing to early versus late mortality. We did not evaluate whether or not patients had associated vascular injuries, which seem to be an important predictor of mortality. In addition, we did not assess when the patient died, cause of death, or procedure-related complications (i.e. development of pancreatic fistula), and data on whether surgeries were done in stages was not available. These are all valid points to address in future studies using more recent database data.
Conclusion
We report an incidence of pancreatic injury in adult trauma patients of 0.3% within the NTDB, with a majority of injuries being due to blunt trauma. Traumatic pancreatic injuries were associated with high morbidity and mortality rates of 53% and 21%, respectively. Non-operative management, specifically pancreatic drainage procedures, appear to be associated with decreased mortality. Future prospective studies are needed to establish well-founded treatment recommendations.
Footnotes
Appendix
International Classification of Disease, Ninth Revision, Clinical Modification (ICD-9-CM) diagnosis codes.
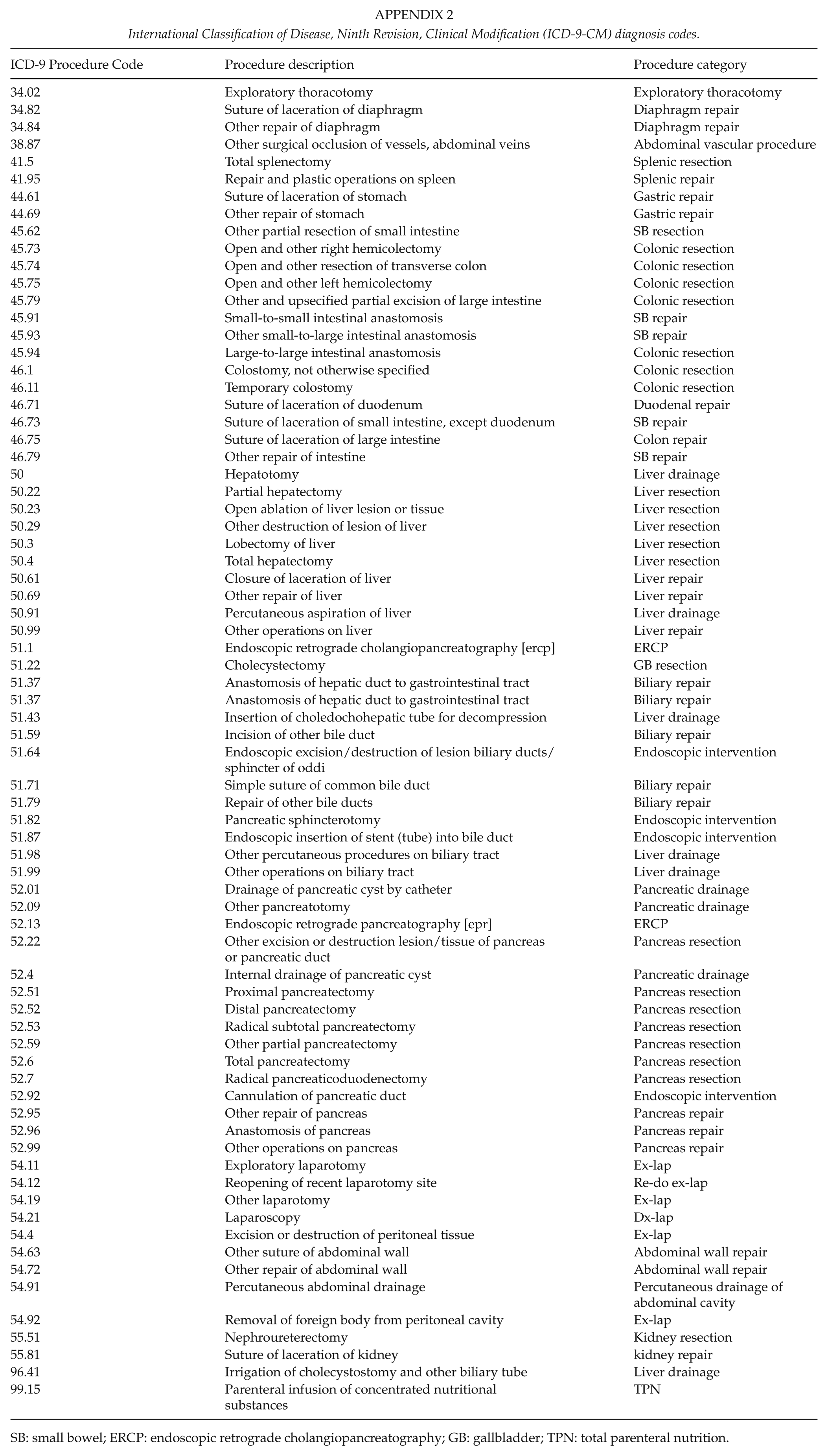
| ICD-9 Procedure Code | Procedure description | Procedure category |
|---|---|---|
| 34.02 | Exploratory thoracotomy | Exploratory thoracotomy |
| 34.82 | Suture of laceration of diaphragm | Diaphragm repair |
| 34.84 | Other repair of diaphragm | Diaphragm repair |
| 38.87 | Other surgical occlusion of vessels, abdominal veins | Abdominal vascular procedure |
| 41.5 | Total splenectomy | Splenic resection |
| 41.95 | Repair and plastic operations on spleen | Splenic repair |
| 44.61 | Suture of laceration of stomach | Gastric repair |
| 44.69 | Other repair of stomach | Gastric repair |
| 45.62 | Other partial resection of small intestine | SB resection |
| 45.73 | Open and other right hemicolectomy | Colonic resection |
| 45.74 | Open and other resection of transverse colon | Colonic resection |
| 45.75 | Open and other left hemicolectomy | Colonic resection |
| 45.79 | Other and upsecified partial excision of large intestine | Colonic resection |
| 45.91 | Small-to-small intestinal anastomosis | SB repair |
| 45.93 | Other small-to-large intestinal anastomosis | SB repair |
| 45.94 | Large-to-large intestinal anastomosis | Colonic resection |
| 46.1 | Colostomy, not otherwise specified | Colonic resection |
| 46.11 | Temporary colostomy | Colonic resection |
| 46.71 | Suture of laceration of duodenum | Duodenal repair |
| 46.73 | Suture of laceration of small intestine, except duodenum | SB repair |
| 46.75 | Suture of laceration of large intestine | Colon repair |
| 46.79 | Other repair of intestine | SB repair |
| 50 | Hepatotomy | Liver drainage |
| 50.22 | Partial hepatectomy | Liver resection |
| 50.23 | Open ablation of liver lesion or tissue | Liver resection |
| 50.29 | Other destruction of lesion of liver | Liver resection |
| 50.3 | Lobectomy of liver | Liver resection |
| 50.4 | Total hepatectomy | Liver resection |
| 50.61 | Closure of laceration of liver | Liver repair |
| 50.69 | Other repair of liver | Liver repair |
| 50.91 | Percutaneous aspiration of liver | Liver drainage |
| 50.99 | Other operations on liver | Liver repair |
| 51.1 | Endoscopic retrograde cholangiopancreatography [ercp] | ERCP |
| 51.22 | Cholecystectomy | GB resection |
| 51.37 | Anastomosis of hepatic duct to gastrointestinal tract | Biliary repair |
| 51.37 | Anastomosis of hepatic duct to gastrointestinal tract | Biliary repair |
| 51.43 | Insertion of choledochohepatic tube for decompression | Liver drainage |
| 51.59 | Incision of other bile duct | Biliary repair |
| 51.64 | Endoscopic excision/destruction of lesion biliary ducts/sphincter of oddi | Endoscopic intervention |
| 51.71 | Simple suture of common bile duct | Biliary repair |
| 51.79 | Repair of other bile ducts | Biliary repair |
| 51.82 | Pancreatic sphincterotomy | Endoscopic intervention |
| 51.87 | Endoscopic insertion of stent (tube) into bile duct | Endoscopic intervention |
| 51.98 | Other percutaneous procedures on biliary tract | Liver drainage |
| 51.99 | Other operations on biliary tract | Liver drainage |
| 52.01 | Drainage of pancreatic cyst by catheter | Pancreatic drainage |
| 52.09 | Other pancreatotomy | Pancreatic drainage |
| 52.13 | Endoscopic retrograde pancreatography [epr] | ERCP |
| 52.22 | Other excision or destruction lesion/tissue of pancreas or pancreatic duct | Pancreas resection |
| 52.4 | Internal drainage of pancreatic cyst | Pancreatic drainage |
| 52.51 | Proximal pancreatectomy | Pancreas resection |
| 52.52 | Distal pancreatectomy | Pancreas resection |
| 52.53 | Radical subtotal pancreatectomy | Pancreas resection |
| 52.59 | Other partial pancreatectomy | Pancreas resection |
| 52.6 | Total pancreatectomy | Pancreas resection |
| 52.7 | Radical pancreaticoduodenectomy | Pancreas resection |
| 52.92 | Cannulation of pancreatic duct | Endoscopic intervention |
| 52.95 | Other repair of pancreas | Pancreas repair |
| 52.96 | Anastomosis of pancreas | Pancreas repair |
| 52.99 | Other operations on pancreas | Pancreas repair |
| 54.11 | Exploratory laparotomy | Ex-lap |
| 54.12 | Reopening of recent laparotomy site | Re-do ex-lap |
| 54.19 | Other laparotomy | Ex-lap |
| 54.21 | Laparoscopy | Dx-lap |
| 54.4 | Excision or destruction of peritoneal tissue | Ex-lap |
| 54.63 | Other suture of abdominal wall | Abdominal wall repair |
| 54.72 | Other repair of abdominal wall | Abdominal wall repair |
| 54.91 | Percutaneous abdominal drainage | Percutaneous drainage of abdominal cavity |
| 54.92 | Removal of foreign body from peritoneal cavity | Ex-lap |
| 55.51 | Nephroureterectomy | Kidney resection |
| 55.81 | Suture of laceration of kidney | kidney repair |
| 96.41 | Irrigation of cholecystostomy and other biliary tube | Liver drainage |
| 99.15 | Parenteral infusion of concentrated nutritional substances | TPN |
SB: small bowel; ERCP: endoscopic retrograde cholangiopancreatography; GB: gallbladder; TPN: total parenteral nutrition.
Acknowledgements
The authors would like to thank all centers participating in the National Trauma Databank (NTDB) and all personnel responsible for reporting and maintaining data to the registry.
Author Contributions
C.M.K. contributed to the literature search, data analysis, data interpretation, writing, and critical revision. S.A.H. contributed to literature search, data collection, data analysis, writing, and critical revision. B.R.E. contributed to literature search, data collection, data analysis, data interpretation, and writing. A.M.G. contributed to data analysis, data interpretation, and critical revision. P.J.S. contributed to data analysis, data interpretation, and critical revision. J.E.S. contributed to data interpretation, critical revision, and writing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Research Ethics
This study was approved by the institutional review board at Duke University Medical Center.
