Abstract
Background and Aims:
Additive manufacturing or three-dimensional printing is a novel production methodology for producing patient-specific models, medical aids, tools, and implants. However, the clinical impact of this technology is unknown. In this study, we sought to characterize the clinical adoption of medical additive manufacturing in Finland in 2016–2017. We focused on non-dental usage at university hospitals.
Materials and Methods:
A questionnaire containing five questions was sent by email to all operative, radiologic, and oncologic departments of all university hospitals in Finland. Respondents who reported extensive use of medical additive manufacturing were contacted with additional, personalized questions.
Results:
Of the 115 questionnaires sent, 58 received answers. Of the responders, 41% identified as non-users, including all general/gastrointestinal (GI) and vascular surgeons, urologists, and gynecologists; 23% identified as experimenters or previous users; and 36% identified as heavy users. Usage was concentrated around the head area by various specialties (neurosurgical, craniomaxillofacial, ear, nose and throat diseases (ENT), plastic surgery). Applications included repair of cranial vault defects and malformations, surgical oncology, trauma, and cleft palate reconstruction. Some routine usage was also reported in orthopedics. In addition to these patient-specific uses, we identified several off-the-shelf medical components that were produced by additive manufacturing, while some important patient-specific components were produced by traditional methodologies such as milling.
Conclusion:
During 2016–2017, medical additive manufacturing in Finland was routinely used at university hospitals for several applications in the head area. Outside of this area, usage was much less common. Future research should include all patient-specific products created by a computer-aided design/manufacture workflow from imaging data, instead of concentrating on the production methodology.
Keywords
Introduction
Additive manufacturing (AM) or three-dimensional printing (3DP) is a production methodology where, instead of subtractive manufacturing like milling or drilling, or formative manufacturing like forging or bending, 3D products are built by the addition of material, usually in successive layers (1). While specific AM technology names may differ between producers of 3D printers, the recent International Organization for Standardization (ISO)/ASTM International standard indicates seven groups of technologies: vat photopolymerization, material jetting, binder jetting, material extrusion, powder bed fusion, sheet lamination, and direct energy deposition (1). For a review on 3DP technologies, medical applications, and the required image processing, see Mitsouras et al. (2).
During the past decades, AM has found an increasing number of applications in medicine. For the first time, AM has allowed the reproduction of complex patient geometry in models for surgical planning and education. AM has also been used for producing patient-specific implants (PSIs), tools such as saw guides, and to some degree custom-made supports and splints. While bioprinting (i.e. the printing of biological tissues or organs) has potentially revolutionary clinical implications, at present it is not in clinical use (3).
The literature on AM still mainly consists of case reports and small case series. Published applications illustrating the scope of possibilities include orbital reconstruction with a PSI (4), an endovascular aortic aneurysm repair facilitated by a 3D printed hollow model (5), a printed gynecological training simulator (6), and creating a printed death mask to cover the face of a facial allotransplant donor (7). However, reports of single, specific cases typically only offer proof of concept and provide weak evidence for their general practicality and efficiency. In order to impact daily practice, AM applications must be clinically and economically justifiable, providing better outcomes or less costly operations, for example through reducing time spent in the operating room. To date, there has not been any randomized trials comparing AM methodologies with traditional approaches; the existing studies usually only report subjective surgeon satisfaction with the technology (8). Recently, several reviews have been published that concern AM in a specific medical specialty, such as otorhinolaryngology—head and neck surgery (8), spine surgery (9), urology (10), and plastic surgery (11). However, the true scale of utilization and the impact of AM technology on clinical practice to date have not been studied.
In this study, we attempt to characterize the clinical adoption and use of medical AM technologies and their impact on medical practice in Finland during 2016–2017. Our aim was to identify the fields—if any—where medical AM has reached routine status, and best practices have been established, which could potentially be transferred to other applications. Furthermore, we sought to identify the fields where AM application has been met with problems, which could provide opportunities for further research and problem solving. We chose to focus on non-dental applications of AM, though dental use is addressed in the “Discussion” section (Fig. 1).
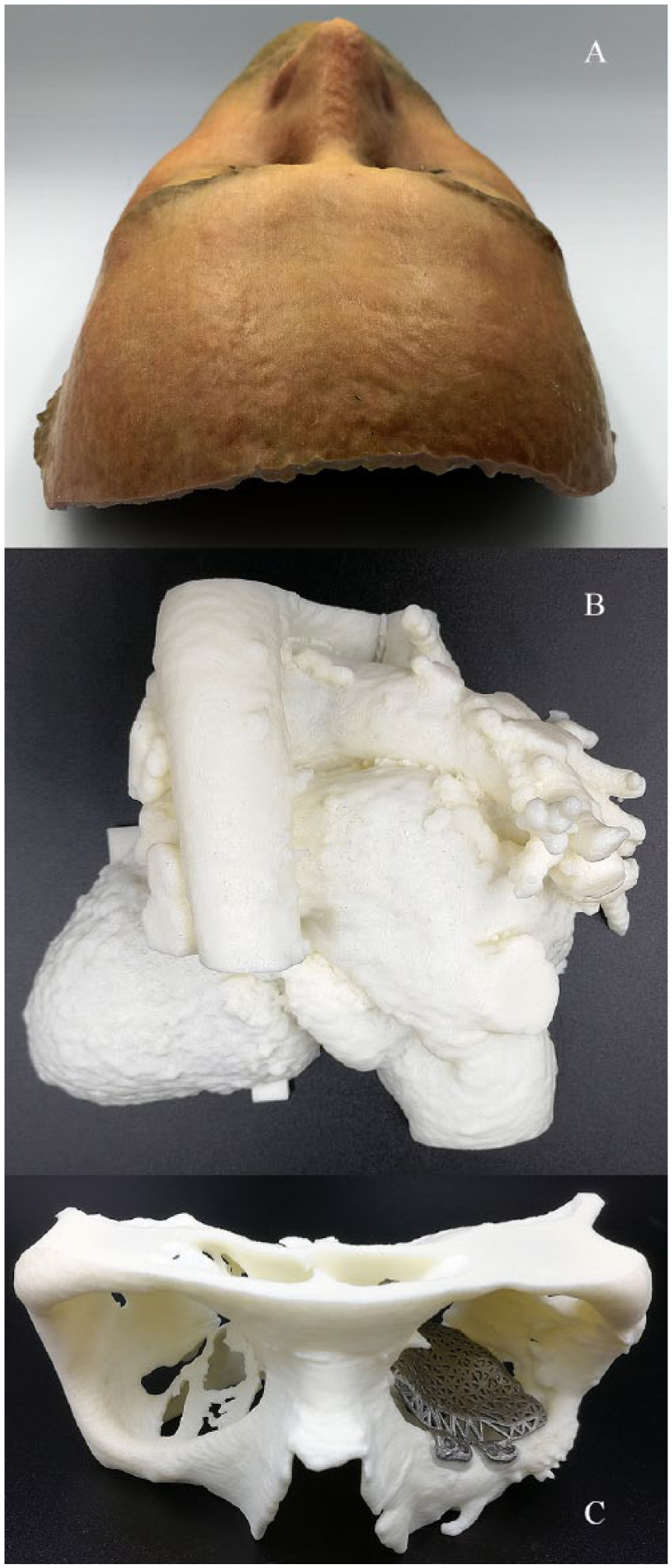
Previously realized medical AM products from collaborations between Aalto University and University of Helsinki: (A) prosthetic mask to cover the face of a facial allotransplant donor (training model based on volunteer), (B) heart with congenital malformation and previous repair, and (C) model of orbital floor fracture and custom-made implant, both manufactured by AM.
Materials and Methods
A structured questionnaire consisting of five questions (Table 1) was sent by email to all operative (i.e. surgery and associated subspecialties, orthopedics, ear, nose and throat diseases (ENT), gynecology etc.), oncologic, and radiologic departments at each of the five university hospitals in Finland. The questionnaire was addressed to the head of department or professor, as determined by the hospital’s web pages or by contacting the department secretary. In addition, a number of individual clinicians at university hospitals who had previously expressed interest in using AM were contacted with the same questions. One of the questions asked was whether the respondent knew of anybody else using AM at their hospital, and these persons were in turn sent the same questionnaire until a saturation point was reached and no further users were identified.
List of questions asked in the questionnaire.
If the person contacted did not reply to the email, a reminder was sent, after which contact by telephone was attempted. Respondents who had used AM more than once were sent additional individualized, open-ended questions; these results are addressed in the “Discussion” section.
Results
The questionnaire was sent to 115 people, of whom 68 (59%) eventually answered, either by email or telephone. Excluding those who only referred to another person, we recorded 58 responses (50% of the people to whom the questionnaire was sent), some of which were from different people in the same departments. Based on their reported usage, we broke down the responses into three categories: non-users, previous users and experimenters, and heavy users (Table 2). The frequencies are presented in Fig. 2.
Answers by specialty.

AM: additive manufacturing.
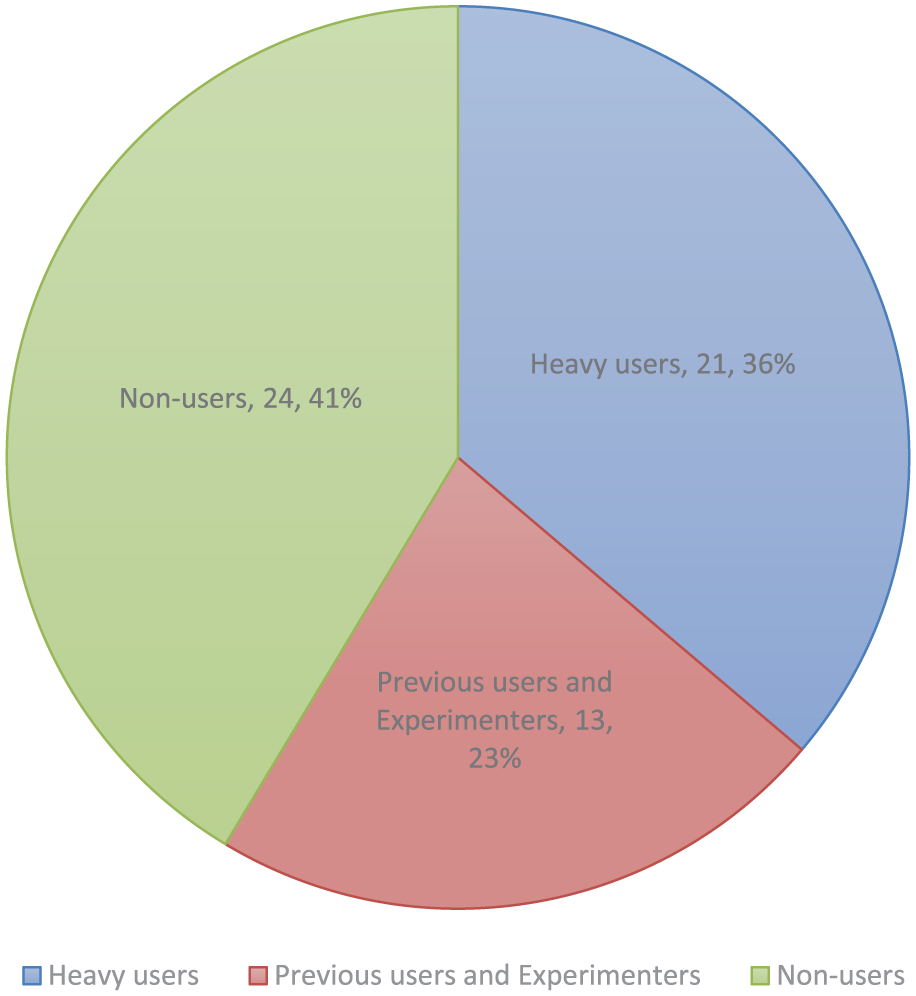
Breakdown of respondents by AM usage.
Non-Users
This group, who had never used AM, included all respondents from general/gastrointestinal (GI) surgery, urology, vascular surgery, and gynecology.
The majority had not used any patient-specific products and had no plans for doing so in the foreseeable future. One gynecological department used manually produced vaginal dilatators that could potentially be manufactured by AM in the future. In addition, one urological department indicated that AM-produced preoperative models could perhaps be useful as planning aids.
Three responders from radiation oncology departments used various patient-specific products, namely, positioning devices, radiation shields, and so on, which were either commercial (thermoplastic, water cured, or vacuum-based systems) or manufactured by on-site Computer Numerical Control (CNC) milling of foam. Using AM for these applications in the future was viewed as a possibility.
Previous Users and Experimenters
Respondents in this group had some AM experience but were not active users. Combining answers referring to the same usage, nine distinct usages were identified. The represented specialties were bone tumor, pediatric and lower limb orthopedics, cardiothoracic surgery, and interventional cardiology.
Two orthopedic surgeons from different bone tumor units had previously used PSIs and patient-specific instrumentation (saw guides) for scapular and pelvic tumors. However, they were disappointed with the results and switched to modular prosthetics—in one case combined with virtual planning and intraoperative navigation. The number of implants previously used was undetermined.
Two pediatric cardiothoracic surgeons had ordered a single model of a child’s heart with a congenital malformation, and another cardiothoracic surgeon had modeled a sternum to test the possibilities of AM applications.
One interventional cardiologist had ordered two models of hearts with complex congenital deformities, which were used to plan and practice a catheter-based endovascular repair.
Six orthopedic surgeons from three different lower limb units reported previous experience with AM. One department had printed two preoperative models of a foot and a leg, another department printed one model of a leg, and the third department printed one model of a foot. In addition, one pediatric orthopedic surgeon reported having experimented with preoperative models for bone deformities. All cases were especially complex deformities, either congenital or the result of malunited fractures.
Excluding the bone tumor users, all cases were experimental and ordered for especially complex and unique cases in order to determine if a printed preoperative model would be helpful. The models were seen as moderately useful but ultimately not worth the investment for further routine use.
Heavy Users
Heavy usage largely applied to the head region, with users specialized in maxillofacial surgery, ENT, plastic surgery, pediatric surgery, orthopedics, and neurosurgery (Table 3).
Estimated yearly volumes of AM products used per specialty.
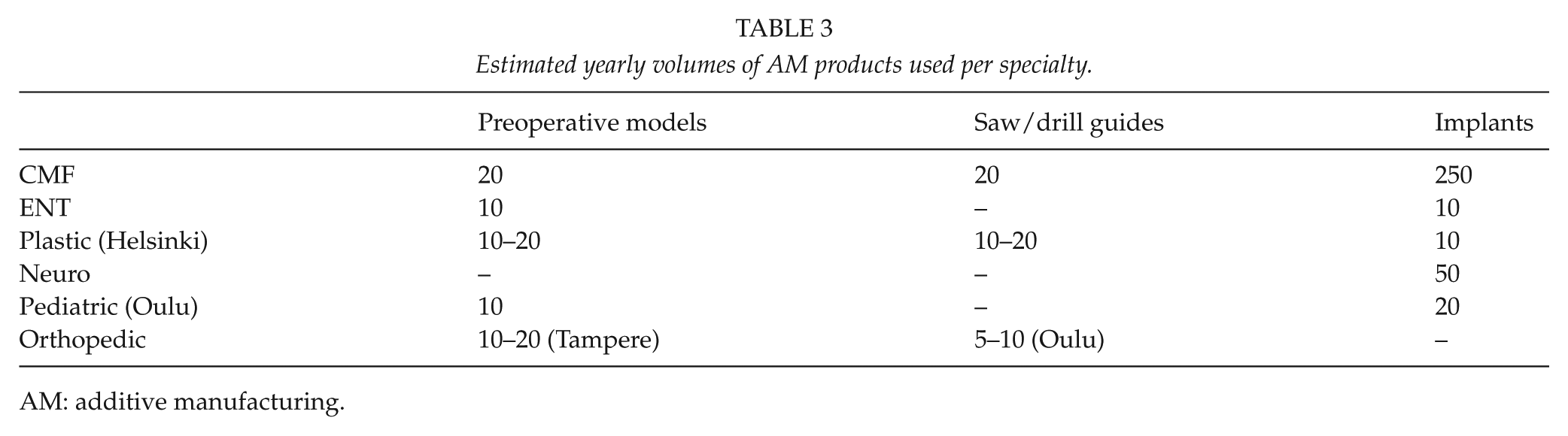
AM: additive manufacturing.
Six neurosurgeons from four different departments reported using patient-specific skull reconstruction implants for hemicraniectomy repair. These were ordered from various companies, which in turn use AM at least as a part of their manufacturing process. The cumulative volume of PSIs ordered was estimated to be around 50. The process of ordering PSIs had strongly modified common practice, in that neurosurgical departments had stopped freezing autologic bone grafts, since the results with synthetic implants were superior.
Four craniomaxillofacial (CMF) surgeons from three different departments, five ENT surgeons from four different departments, and two plastic surgeons from one department reported extensive use of AM for preoperative planning models, osteotomy saw guides, and implants. Cumulatively, the CMF surgeons used about 250 PSIs, 20 planning models, and 20 saw guides per year. The use of printed planning models for CMF applications had diminished because of a transition toward virtual planning. The listed benefits of AM were shortened operative times and improved precision, especially in mandibular surgery, where surgeons credited PSIs with ability to produce truly precise results for the first time. Fields of use were tumor resection, orthognathic surgery, and trauma. The fields of ENT and CMF surgery overlap to some extent, and the largest user of AM—a CMF department that used about 150 PSIs/year—reported operating on about 50 orbital fractures per year, using PSIs for all cases, that is, even for simple blow-out type fractures. While this contributed to their high volumes, the interpretation is complicated by the fact that some of these orbital floor implants were produced by milling and some by AM, and the company from which these were sourced did not divulge the specific ratio.
The plastic surgeons estimated using 10 to 20 planning models and saw guides and somewhat fewer PSIs per year, mainly in cleft palate reconstruction and tumor surgery. The ENT surgeons reported using cumulatively about 10 planning models and a similar number of PSIs per year.
One pediatric surgeon specialized in skull deformity repair reported using both printed planning models and PSIs. In this case, about 10 models and 20 implants were used per year. This usage was also mentioned by another responder (a physicist from the radiology department who operated the university’s own 3D printer).
Outside of the head region, one upper limb orthopedic surgeon reported using 10 to 20 planning models per year, mainly for planning corrective operations of fracture malunions. This user found these models very helpful in planning complex operations and preferred them over virtual planning. Another orthopedic surgeon specializing in shoulder surgery estimated using about 5 to 10 commercial drill guides per year for positioning certain shoulder joint replacements. These drill guides include a model of the glenoid that they are modeled to fit and in theory make positioning of the implant foolproof. However, in practice, the user found that the results varied. While the surgeon ordered drill guides for only some cases, virtual planning was used on all patients.
All PSIs and saw guides were ordered from commercial companies. In addition, almost all planning aids were ordered from commercial companies, with two exceptions: the pediatric surgeon identified as a heavy user produced preoperative models on a university-owned printer that was operated by the radiology department, and a maxillofacial department—the largest user of AM—reported owning a printer that they used for some preoperative models.
Discussion
To our knowledge, this is the first nationwide survey concerning the clinical adoption of medical AM. We attempted to characterize the clinical use of medical AM in Finland occurring in 2016–2017, via email questionnaires and telephone interviews. Our results indicate that AM had found routine use at university clinics with specialties relating to the head, that is, neurosurgery, ENT, plastic, and maxillofacial surgery. Outside of the head region, usage declined sharply; orthopedic surgery had some routine usage by a limited number of clinicians, and most of the other usage was experimental. The vast majority of prints (planning models, saw guides/drill guides, and PSIs) were ordered from commercial providers.
Many advanced forms of treatment are centralized to one or more of Finland’s five university hospitals. Because of this centralization, we assumed that the majority of non-dental AM applications would currently be found there. This was later confirmed by cross-referencing with providers of AM in Finland, who reported no major medical customers outside of tertiary-level hospitals.
The current study has several limitations. First, the nature of the study questions, limited and biased number of responders, and non-random sampling preclude a meaningful quantitative analysis. Therefore, this study aims to characterize the adoption of AM technology in clinical care in a qualitative manner. Second, despite our efforts to follow the “web” of AM users, there may be individuals or groups that have been missed by this study because of our focus on tertiary-level hospitals and the bias of the authors’ connections. Third, since none of the reported numbers are based on reliable registries or statistics but rather estimates or recollections, there is a possibility of significant over- or underestimation.
Barriers to Adoption
A common theme in the interviews with heavy users was that AM is irreplaceable for certain applications, even though no good studies exist to prove this. However, in the setting of rising cost pressures in health care, widescale adoption of new clinical technology requires evidence of efficacy. Many applications in the head area, such as cancer and complex trauma, are such that comparison trials between AM-assisted and traditional approaches are impossible because of strong clinician preference for AM. However, traditional means are still being used for many other applications such as prosthetic dentistry and orthopedics, and therefore comparative studies of efficacy would seem feasible.
A number of respondents reported that ordering AM products was difficult and slow, hence presenting a barrier to large-scale adoption. Since very few users produced and planned products in-house, design and ordering were usually done through online collaboration with engineers from commercial 3D printing vendors. For the heaviest users of AM, the imaging, design, production, and delivery of an implant and patient-specific guides could be done within 24 h if needed, based on an optimized and familiar workflow and co-location with the actual producer. However, depending on the provider, a 24-h timescale may not be possible for most applications.
A viable alternative for further integrating AM into clinical work and reducing turnaround time is to bring the design and production in-house (12). In several hospitals outside of Finland, the design is done either by hospital-employed design engineers or the clinicians themselves, and many institutions also have their own 3D printers, especially for applications other than implants. While somewhat time-consuming, many clinicians view this as a rewarding, more educative, and ultimately even faster process than using outside providers. In Finland, Oulu University has its own 3D printer for producing medical models but cannot serve other hospitals due to different regulations applied to production for in-house versus outside customers.
Regulation of medical AM is also a complex issue internationally, and currently it remains the clinical end user’s responsibility to ascertain that the producer of their prints has the required certification. In Europe, even preoperative planning models are equated with medical devices and thus are required to meet several criteria, such as registration of the producer and a quality control system.
Am in of-the-Shelf Production and other Invisible Uses
Numerically, the largest impact of AM on medical care has consisted of off-the-shelf implants—namely, components for orthopedic joint replacement—rather than patient-specific products. As this non-disruptive change in manufacturing technology has been invisible to clinicians, it was also not recognized by the current study. While the exact percentage is unknown, several of the largest vendors are moving toward (or are already) producing parts of their joint replacement products by AM. Components currently produced by AM include tibial components of knee prosthetics, cup components of hips, and augments for hip revision. Over 22,000 hip or knee joint replacements were performed in Finland in 2015 (13), and similar numbers are estimated to have continued throughout the study period. Hence, the volumes of components are several orders of magnitude larger than those of PSIs.
Another noteworthy invisible usage is hearing aids, as the majority of device shells are produced by AM for purely economic reasons, while earpieces are individually printed based on scanned moldings of patients’ ears (8). Similarly, increased usage of AM for non-customized products is also seen in other industries (e.g. aerospace), with AM being used more frequently for mass production and less for prototyping and planning (14). Medical companies have adopted AM for the same reasons as other industries, namely, economics when producing limited runs of a product, and feasibility of geometries and structures (for instance, controlled surface porosity) not achievable by traditional methodologies.
Dental Usage of Am
The dental sector in Finland is highly decentralized, making characterization by our chosen methodology impossible. However, most dental laboratories buy their supplies from two large distributors who also offer milling and AM services, both of which use the same subcontractor for metallic AM. We interviewed a representative of this company, and a dentist well known for his use of a 3D printer at his own practice.
Based on these data, we estimate that during the study period, metallic AM was mainly used in substructures for fixed prosthetic devices—a component difficult to produce by traditional methodologies. Approximately 4 to 15 cobalt-chromium substructures were produced weekly by the domestic service provider, which amounts to around 500 per year. For other prosthetics in general, AM was still not a mainstream technology, with only about 20 bridges and 1 to 2 individual crowns produced per year. One reason for this is that these products are often produced from ceramics like zirconia, and printing these types of materials is currently not as successful as that of metals (15). Moreover, many laboratories are already heavily invested in computer-aided design/manufacture (CAD/CAM) milling technology for prosthetic and restorative devices, making them reluctant to switch to AM.
A commercial orthodontic product that uses a series of printed corrective dental films is available through certain private dentists, but most orthodontic work is done through traditional means. Usage of plastic drill guides and planning models is not mainstream, but some dentists do use them extensively. In dental prosthetic work, preoperative imaging is usually available, work may be spread out over many sessions with ample time for producing prints, and prints may be produced at a low cost using “prosumer”-level vat photopolymerization devices. Therefore, the biggest obstacle for widescale adoption seems to be lack of proven benefit, either in results, operative time, or cost.
Physical Preoperative Planning Aids Versus Virtual Planning
The most well-known and earliest application of medical AM is the production of physical models for planning complex procedures. This usage was recognized by our study, but the opinions about its usefulness varied. For example, some respondents found printed models useful, but many others felt that they did not get any added value from them. Some respondents felt that the initial 3D printed models of complex pathologies had helped them understand the topology, but after having associated the models with certain imaging findings, they did not need prints of individual cases anymore.
Virtual planning using reconstructed computed tomography (CT) images is another option for preoperative planning (16, 17). It may be extended to the operating theater through intraoperative navigation, or even assistive robotics such as those currently commercially available for joint replacement operations (18), which can be seen as the virtual planning-alternative to printed saw guides. Augmented reality (AR) applications are currently in development. Compared to physical models, virtual models have certain benefits. For example, they do not require waiting for print completion, and they offer the ability to zoom, section, and manipulate at will, to try several destructive approaches like cutting the model without requiring a new printed object, and computer-assisted planning such as structural integrity optimization using Finite-Element Modeling. A virtual model may also change over time, thus being more of a “4D model” and potentially better at approximating real-world organs such as the heart, which constantly changes its geometry during the cardiac cycle. Drawbacks of virtual models, on the other hand, include inability to test tools or devices that have not been modeled, or the influence of the actual physical dexterity of the operator. There are studies suggesting a better grasp of proportions and relationships between structures, and especially a quicker ability to grasp these concepts, when using physical planning models compared to virtual 3D reconstructions or unprocessed CT slices (19). Current AR solutions are still quite cumbersome and do not approach the level of authenticity of a printed model. In our study, some respondents were enthusiastic about the possibilities of virtual planning, but others did not even like looking at 3D renderings of CT images, preferring just traditional CT slices and envisioning the 3D anatomy by themselves.
Concentrating on Am Versus Patient-Specific Products in General
Today, the lay public has a general concept of what 3D printing is, often associating it with material extrusion-type devices. However, they may not fully understand machining or foundry technologies, or their capabilities. Indeed, our study revealed that many clinicians thought certain patient-specific products (metallic orbital floor implants) were 3D printed when in fact they were often produced through machining.
From an engineering point of view, AM is just one of many production methodologies (although a very unique one) available for producing patient-specific products, which uses CAD/CAM. AM allows the creation of unique geometries that are not achievable through other means, and complex internal and surface structures are available “for free” (i.e. no extra machining cost). Using AM, implants may be constructed to include additional novel features, such as mechanisms for site-specific drug delivery and radio-frequency identification (20). However, when none of these properties are required, AM may not always be the most economical or otherwise preferable methodology. The mechanical properties of AM-produced components may differ from those produced by traditional means, and a significant part of the AM literature is concerned with the influences of production methodology on factors such as osseointegration, fatigue strength, and biocompatibility.
From a clinical perspective, clear evidence favoring one methodology over another is lacking. However, the underlying manufacturing details as well as the associated engineering and biomedical issues remain of minor importance compared to cost, ease of planning, ordering, usage, and the changes caused to clinical practice/operative technique. Therefore, patient-specific products may be seen as the novel and disruptive technology regardless of the underlying production process. As such, future studies would benefit from including all individualized products based on clinical imaging through a CAD/CAM workflow, instead of focusing only on 3D printed products.
In conclusion, we found that while medical AM has become routine for certain applications concerning the head area, most specialties did not utilize it in any way. Orthopedic surgery had the largest variation in terms of usage and perceived benefit between clinicians, and thoracic surgery and cardiology had some experimental use with unique challenges. Instead of technological issues such as lack of geometric fidelity, the largest obstacles for adopting AM into new applications were either the complexity of the ordering process or lack of perceived benefit. Further research into the clinical adoption of these novel technologies should choose whether to concentrate on the production methodology of AM, including off-the-shelf components, or on CAD- or CAM-produced patient-specific products regardless of whether they use more traditional methodologies.
Footnotes
Acknowledgements
The authors wish to thank all the respondents and interview subjects for offering their time and insights. In addition, they wish to thank the funders for the financial support provided for this work.
Authors’ Note
A. A. Mäkitie is also affiliated with Research Programme in Systems Oncology, Faculty of Medicine, University of Helsinki, Helsinki, Finland.
Declaration of Conflicting Interests
The authors declared that there is no conflict of interest, except for Vallittu P: shareholder and Member of the Board of Skulle Implants Corporation.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Helsinki University Hospital Research Fund, the Viipuri Tuberculosis Foundation, the South Karelia Medical Association, and the Eksote research fund.
