Abstract
Background:
Perihilar cholangiocarcinoma and distal cholangiocarcinoma arise from the same tissue but require different surgical treatment methods. It remains unclear whether these cholangiocarcinoma types have different outcomes, prognostic factors, and/or recurrence patterns.
Methods:
This retrospective study evaluated patients who underwent curative-intent resection for perihilar cholangiocarcinoma or distal cholangiocarcinoma at a tertiary academic hospital during 2000–2015. Survival and prognostic factors were identified using Kaplan–Meier and Cox regression analyses.
Results:
The 90-day mortality rates were 0% for perihilar cholangiocarcinoma (36 patients) and 4% for distal cholangiocarcinoma (47 patients). There were no significant differences between perihilar cholangiocarcinoma or distal cholangiocarcinoma in median overall survival (30.9 vs 40.4 months) or median disease-free survival (14.2 vs 21.4 months). Among perihilar cholangiocarcinoma patients, age > 65 years was an independent predictor of poorer overall survival (hazard ratio: 2.45, 95% confidence interval: 1.07–5.64), while requiring bile duct re-resection was an independent predictor of disease-free survival (hazard ratio: 2.76, 95% confidence interval: 1.01–7.51). Among distal cholangiocarcinoma patients, a pN1 category independently predicted poorer overall survival (hazard ratio: 3.40, 95% confidence interval: 1.14–10.11), while preoperative CA19-9 levels >30 U/mL (hazard ratio: 2.51, 95% confidence interval: 1.09–5.79) and pN1 category (hazard ratio: 2.51, 95% confidence interval: 1.09–5.79) predicted a shorter disease-free survival. Local recurrence was more common with perihilar cholangiocarcinoma (50% of recurrences), while multiple synchronous sites were more common for distal cholangiocarcinoma (41% of recurrences).
Conclusion:
Perihilar cholangiocarcinoma and distal cholangiocarcinoma patients have similar survival outcomes. However, local control appears to be more prognostic for perihilar cholangiocarcinoma patients, while positive lymph nodes are critical prognostic factor for distal cholangiocarcinoma patients.
Introduction
Extrahepatic cholangiocarcinomas (CCAs) are classified as perihilar cholangiocarcinoma (pCCA) or distal cholangiocarcinoma (dCCA) based on the tumor’s location. pCCAs remain a challenge for hepato-pancreato-biliary surgeons because of the tumor’s proximity to vital vessels and the difficulty in achieving adequate tumor-free margins. In contrast, dCCA can be treated via a more simpler pancreaticoduodenectomy and high bile duct resection, although approximately 45% of extrahepatic CCAs are located distally (1). CCA is a relatively rare disease, and while complete tumor resection remains the only potentially curative treatment, most patients present with metastasized or unresectable disease (2); hence, few patients undergo resection with curative intent. Interestingly, despite the differences in the surgical managements of pCCA and dCCA, these tumors arise only a few centimeters apart from the same epithelial tissue. As few studies have examined the differences in prognosis and recurrence between these tumor types (3–5), this study aimed to compare the prognostic factors and patterns of recurrence between pCCA and dCCA.
Methods
This retrospective study evaluated patients who underwent resection with curative intent for extrahepatic CCA at a tertiary hospital during 2000–2015. The study’s retrospective protocol was approved by the institutional review board and the National Institute of Health and Welfare. Patients were identified from prospectively maintained databases that included patients diagnosed with CCA or those who were undergoing pancreaticoduodenectomy. In addition, hospital records were cross-referenced to identify any patients with a CCA diagnosis code (International Classification of Diseases-10th edition—codes C22–C24) during the study period. Patients were excluded if they had undergone non-curative intent, if the final pathological diagnosis was not extrahepatic CCA, or if there was a presence of intrahepatic, ampullary, pancreatic, gallbladder, or papilla of Vater tumors. In addition, four patients were excluded because the CCA was detected in the resected liver after liver transplantation for primary sclerosing cholangitis. The extrahepatic CCA cases were classified as dCCA if pancreaticoduodenectomy was necessary to obtain clear margins; otherwise, they were designated pCCAs. At our institution, pCCAs are managed by liver surgeons, while dCCAs are managed by pancreatic surgeons.
Vascular resections were defined as those involving the porto-mesenteric vein or hepatic arteries, while R0 resection was defined as achieving a microscopic tumor-free margin of ⩾1 mm. Complications that occurred within 30 days post-surgery were classified according to the Clavien-Dindo criteria (6). Specific complications were retrospectively classified according to the International Study Group of Pancreatic Surgery grading for pancreatic fistula and post-pancreatectomy hemorrhage (7, 8), or according to the International Study Group of Liver Surgery grading for bile leakage, post-hepatectomy hemorrhage, and post-hepatectomy liver failure (9–11). Bilirubin and CA19-9 levels were measured simultaneously, and the values obtained closest to the time of surgery were used. The 7th version of American Joint Commission on Cancer staging system was used for TNM-staging.
Follow-up data were obtained by contacting the referring hospital or the hospital to which the patient was referred to after surgery. Only one patient had unavailable follow-up data, because of the hospital’s policy of deleting patient records at 12 years after death. Dates of death for all patients were obtained from the Population Register Centre. Disease-free survival (DFS) was defined as the interval between surgery and the first instance of recurrence or the last follow-up by a healthcare provider. Overall survival (OS) was defined as the interval between surgery and the date of death from the Population Register Centre. Patients were censored at their final follow-up, which was defined as the last contact marked in the patient records. The Mann–Whitney U-test was used to compare continuous variables, while the chi-square or Fisher’s exact test was used to compare categorical variables. Survival outcomes were compared using the Kaplan–Meier method and log-rank test, and predictors of survival were evaluated using univariate and multivariate Cox regression analyses. Factors that were statistically significant in univariate analyses were selected for multivariate analyses, and only factors that remained statistically significant on the final multivariate analysis are presented.
The CA19-9 cut-off was set at 30 U/mL based on the normal laboratory upper threshold (26 U/mL) and median value of CA19-9 in our study samples (33 U/mL). Statistical analyses were performed using SPSS version 24 (IBM, Armonk, NY, USA).
Results
Patient Characteristics
A total of 83 patients were investigated, including 36 patients with pCCA and 47 patients with dCCA. The patients’ demographic characteristics are shown in Table 1 (median age: 65 years, 68% male). Preoperative endoscopic retrograde cholangiography was performed on 95% of the patients and class 4–5 brush cytology was detected in 59%. Although most cases were sporadic (84%), the etiology of approximately 13% patients was primary sclerosing cholangitis.
The patient’s basic characteristics.
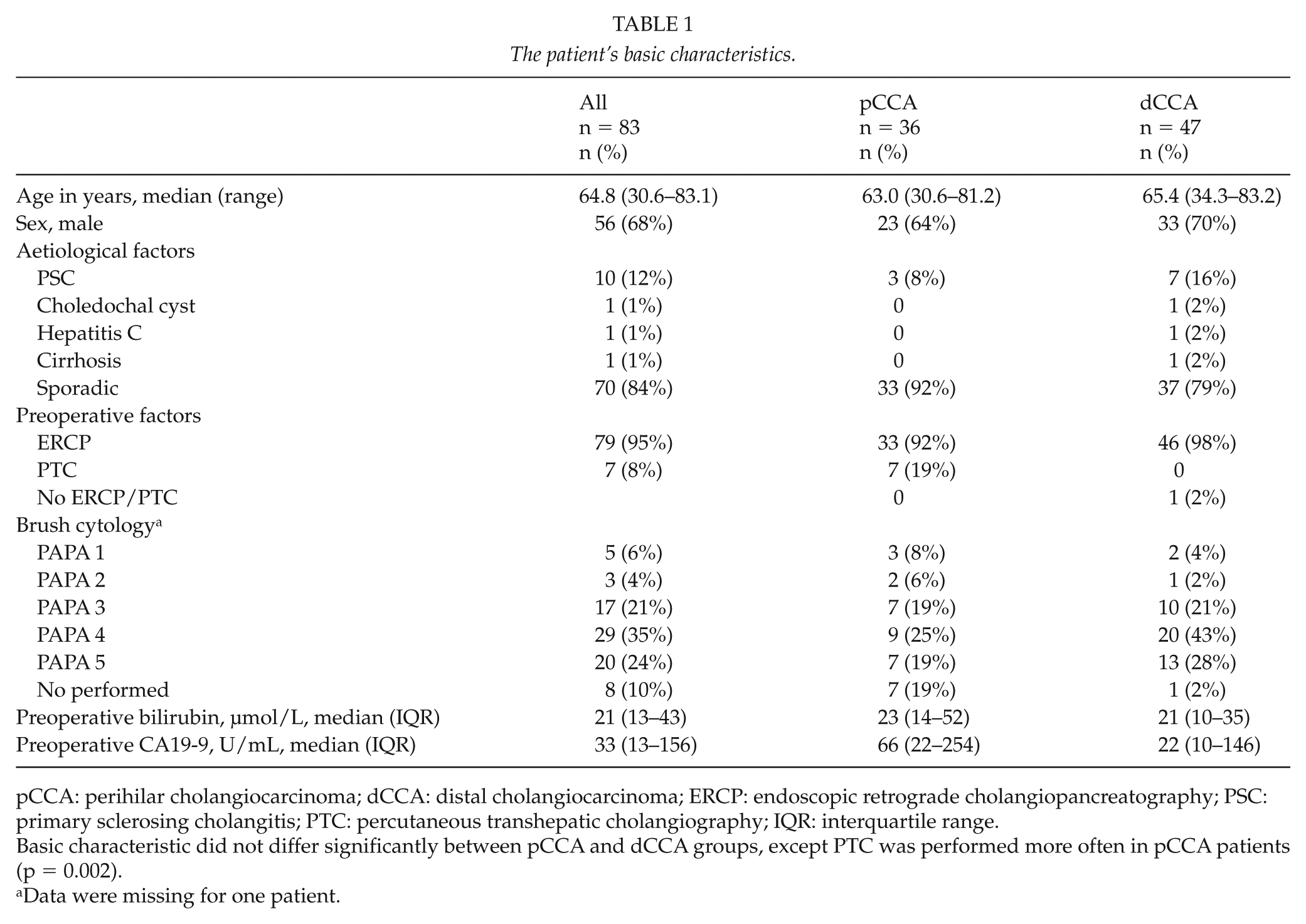
pCCA: perihilar cholangiocarcinoma; dCCA: distal cholangiocarcinoma; ERCP: endoscopic retrograde cholangiopancreatography; PSC: primary sclerosing cholangitis; PTC: percutaneous transhepatic cholangiography; IQR: interquartile range.
Basic characteristic did not differ significantly between pCCA and dCCA groups, except PTC was performed more often in pCCA patients (p = 0.002).
Data were missing for one patient.
Surgical Details
The surgical details are shown in Table 2; 57% of patients underwent pancreaticoduodenectomy with high bile duct resection, 28% underwent hemihepatectomy with extrahepatic bile duct resection, and 16% underwent extrahepatic bile duct resection with or without minor liver resection. There were no patients in whom both hemihepatectomy and pancreatectomy were performed during the same operation, and no preoperative portal embolizations were performed. Multiple (>2) bile duct anastomoses were more commonly required for patients with pCCA than for those with dCCA (22% vs 4%). Vascular resections were more common in patients with pCCA than in those with dCCA (25% vs 13%). Complications were noted in 80% of the patients, and approximately 8% of the patients experienced a Clavien-Dindo grade 4 complication. Patients with dCCA had low rates of pancreatic fistula (grade B: 13%, grade C: 0%) and patients with pCCA had low rates of postoperative liver failure (grade B: 8%, grade C: 0%). None of the patients died within 30 days, although two patients with dCCA who had undergone pancreaticoduodenectomies died within 90 days after the operation. The first patient died suddenly at a community hospital on the 48th postoperative day and was found to have a massive intra-abdominal hemorrhage on autopsy, while the other patient had previously undergone kidney transplantation and developed severe fungal sepsis that led to his death on the 87th postoperative day.
Operative details.
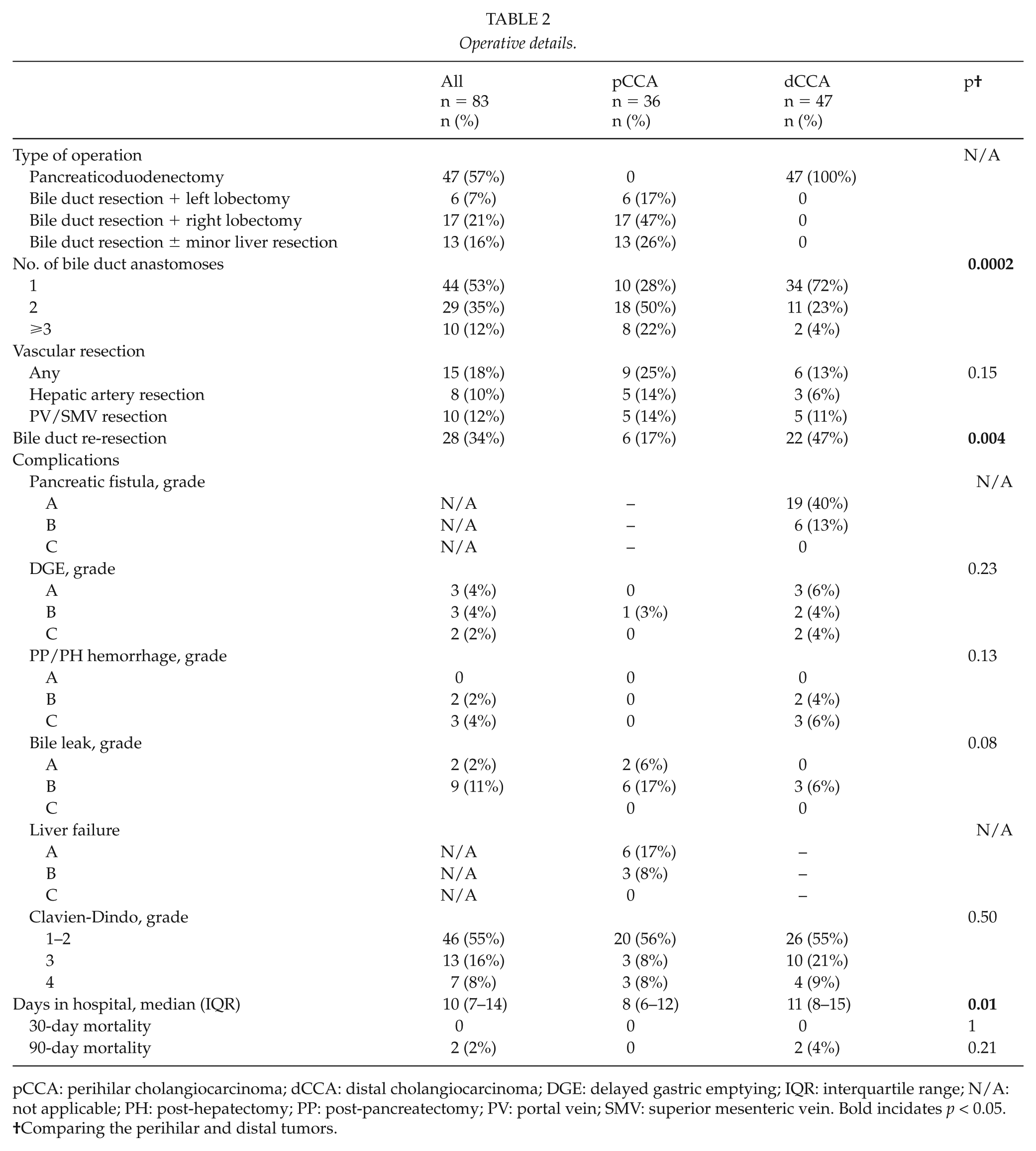
pCCA: perihilar cholangiocarcinoma; dCCA: distal cholangiocarcinoma; DGE: delayed gastric emptying; IQR: interquartile range; N/A: not applicable; PH: post-hepatectomy; PP: post-pancreatectomy; PV: portal vein; SMV: superior mesenteric vein. Bold incidates p < 0.05.
Comparing the perihilar and distal tumors.
(Neo)Adjuvant Therapy
Neoadjuvant therapy using cisplatin plus gemcitabine was administered to one patient with pCCA and none with dCCA. Nine patients (25%) with pCCA received adjuvant therapy (4 with gemcitabine monotherapy, 4 with gemcitabine plus cisplatin, and 1 with chemoradiotherapy using leucovorin plus 5-fluorouracil). Data were missing for one patient. In total, 16 patients (34%) with dCCA underwent adjuvant therapy (5 with gemcitabine monotherapy, 10 with gemcitabine + cisplatin, and 1 with capecitabine monotherapy).
Tumor Histopathology
The histopathological details of the patient samples are shown in Table 3. Patients with pCCA were likely to have lower T and N categories than those with dCCA. However, pCCAs were more likely to involve grade 3 tumors than dCCA cases (44% vs 12%), and more lymph nodes were harvested from patients with dCCA cases (median: 19 vs 3). Patients with dCCA were more likely to achieve R0 resection than those with pCCA (74% vs 47%).
Histopathological details.
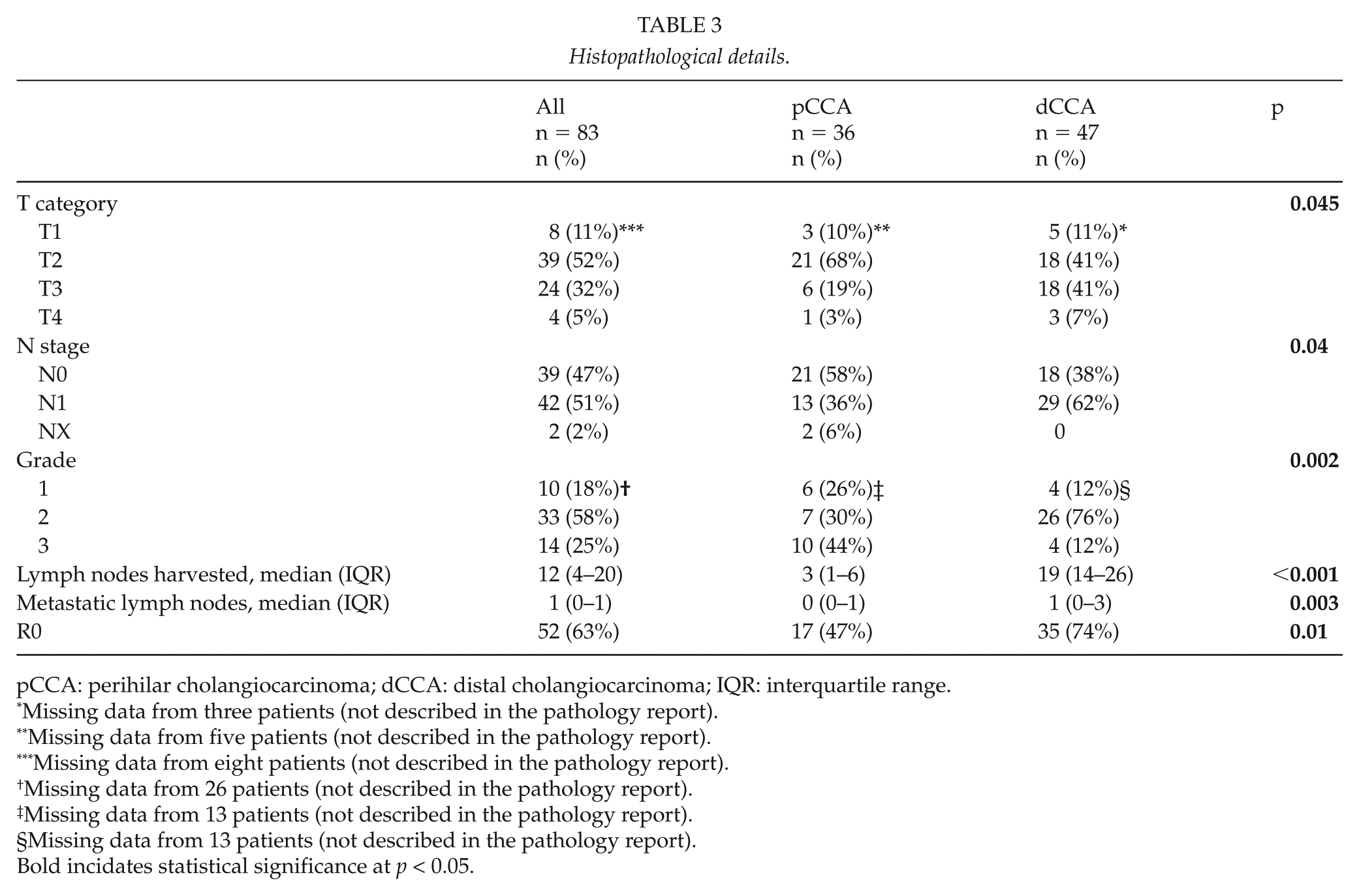
pCCA: perihilar cholangiocarcinoma; dCCA: distal cholangiocarcinoma; IQR: interquartile range.
Missing data from three patients (not described in the pathology report).
Missing data from five patients (not described in the pathology report).
Missing data from eight patients (not described in the pathology report).
Missing data from 26 patients (not described in the pathology report).
Missing data from 13 patients (not described in the pathology report).
Missing data from 13 patients (not described in the pathology report).
Bold incidates statistical significance at p < 0.05.
Os and Dfs
There were no differences in OS or DFS among patients with pCCA vs dCCA (Fig. 1A and B). The estimated median OS for pCCA patients was 30.9 months (95% confidence interval (CI): 17.6–44.3 months), while that for dCCA was 40.4 months (95% CI: 19.1–61.7 months) (p = 0.68). Most deaths were related to metastasis, although two patients died because of postoperative complications and one patient had no known cause of death because of unavailable medical records. The estimated median DFS for pCCA patients was 14.2 months (95% CI: 8.3–20.1 months), while that for dCCA patients was 21.4 months (95% CI: 11.1–31.7 months; p = 0.14).
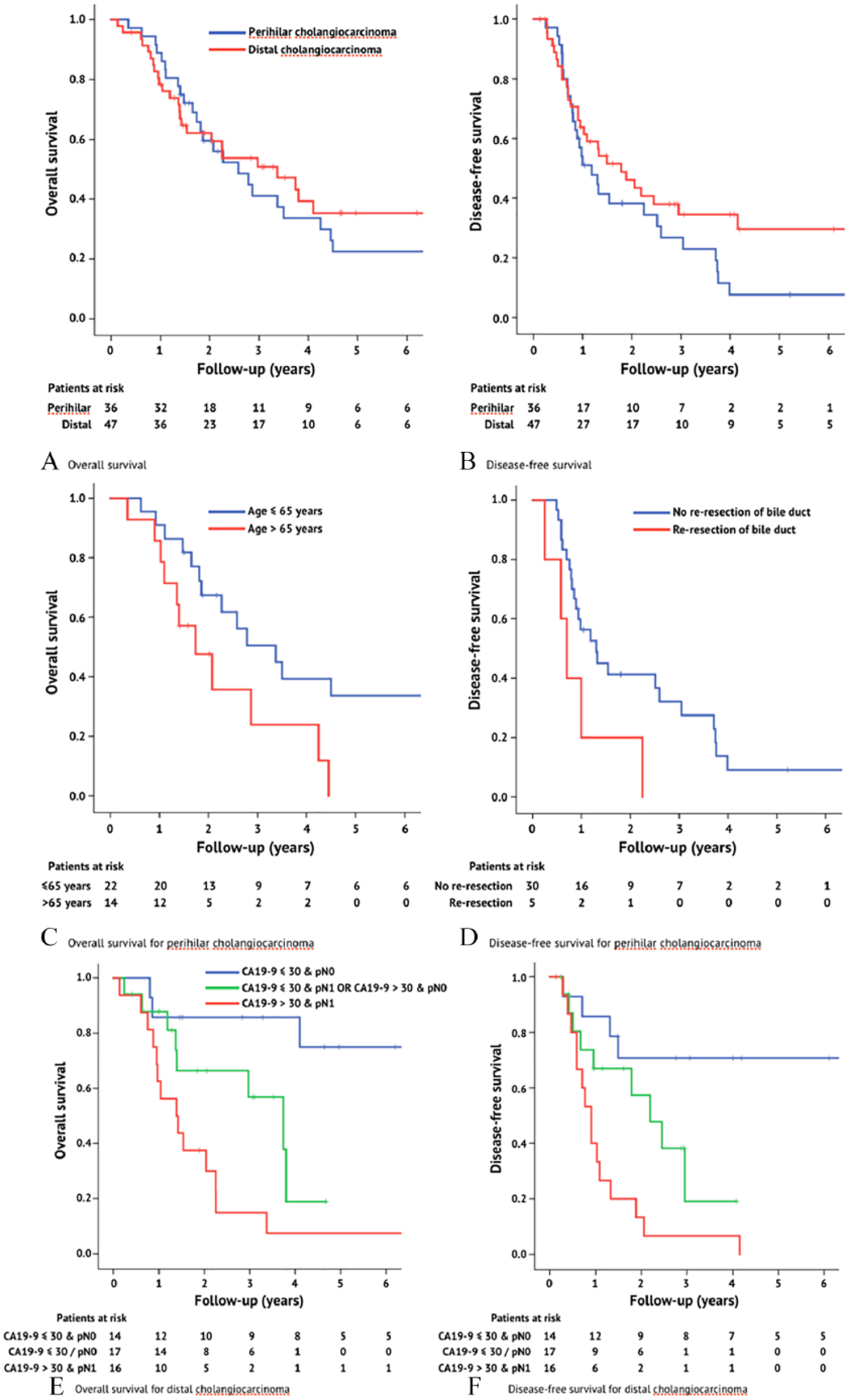
A) Overall survival (log-rank p = 0.68) and B) disease-free survival (log-rank p = 0.14) after resection with curative intent for extrahepatic cholangiocarcinoma. C) Overall survival (log-rank p = 0.03) and D) disease-free survival (log-rank = 0.01) after resection with curative intent for perihilar cholangiocarcinoma stratified according to independent prognostic factors identified on multivariate analysis. E) Overall survival (log-rank p = 0.00009) and F) disease-free survival (log-rank p = 0.001) after resection with curative intent for distal cholangiocarcinoma stratified according to the independent prognostic factors from the multivariate analysis. Patients were censored at their last follow-up (censoring is marked with a vertical line).
The predictors of DFS for patients with pCCA and dCCA differed. Among pCCA patients, poor DFS was associated with the need for bile duct re-resection based on frozen sections, while poor DFS among dCCA patients was associated with preoperative CA19-9 levels of >30 U/mL, R1 resection, and pN1 category (Supplementary Table 1, Fig. 1C to F). Interestingly, R1 resection did not predict DFS among pCCA patients (R0: 15.6 months (95% CI: 9.3–21.8 months) vs R1: 10.6 months (95% CI: 5.9–15.3 months), p = 0.82). On multivariate analyses, preoperative CA19-9 levels of >30 U/mL and pN1 independently predicted shorter DFS among dCCA patients (Table 4, Fig. 1E and F), while R-stage did not.
Multivariate analysis of factors predicting disease-free and overall survival (Cox regression with forward LR selection).
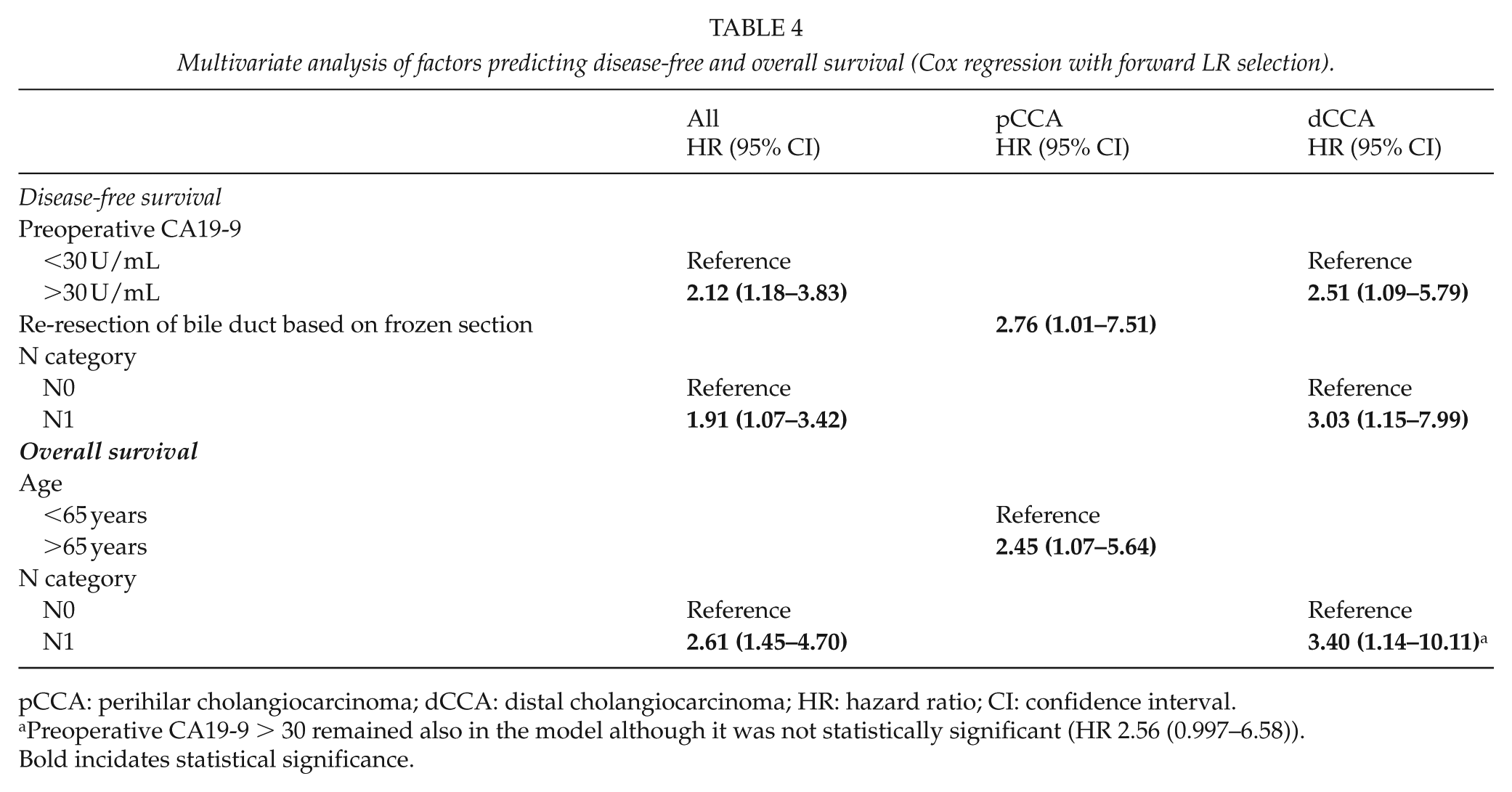
pCCA: perihilar cholangiocarcinoma; dCCA: distal cholangiocarcinoma; HR: hazard ratio; CI: confidence interval.
Preoperative CA19-9 > 30 remained also in the model although it was not statistically significant (HR 2.56 (0.997–6.58)).
Bold incidates statistical significance.
The predictors of OS also differed in patients with pCCA versus dCCA (Supplementary Table 2). Among pCCA patients, only an age of >65 years predicted poorer OS on univariate and multivariate analyses (Supplementary Tables 2 and 4, Fig. 1C and D), although age was not a prognostic factor among dCCA patients. Among pCCA patients, R-stage did not influence OS (R0: 30.9 months (95% CI: 20.2–41.7 months) vs R1: 24.9 months (95% CI: 3.9–45.9 months), p = 0.56). Among dCCA patients, a preoperative CA19-9 level of >30 U/mL, R1 resection, higher pT category, and pN1 category were associated with a poor OS (Supplementary Table 2). On multivariate analyses, only pN1 independently predicted a poorer OS among dCCA patients (Table 4, Fig. 1E and F).
Patterns of Recurrence
The patterns of recurrence among patients with pCCA and dCCA also differed (Fig. 2). Local recurrence was the most common recurrence site among pCCA patients (15 of the 29 patients with recurrence (52%)) Notably, the resection margin (R0 or R1) did not influence the pattern of pCCA recurrence, with local recurrence accounting for 43% of all recurrences after R0 resection and 60% of recurrences after R1 resection. Seventeen percent of patients with pCCA did not experience recurrence after either R0 or R1 resection. In contrast, multiple synchronous recurrences were the most common form in patients with dCCA (41% of recurrences in this group). Only 6% of patients experienced local recurrence after R0 resection of dCCA, while 25% experienced local recurrence after R1 resection. One-half of the patients with dCCA did not experience recurrence during their post-R0 resection follow-up.
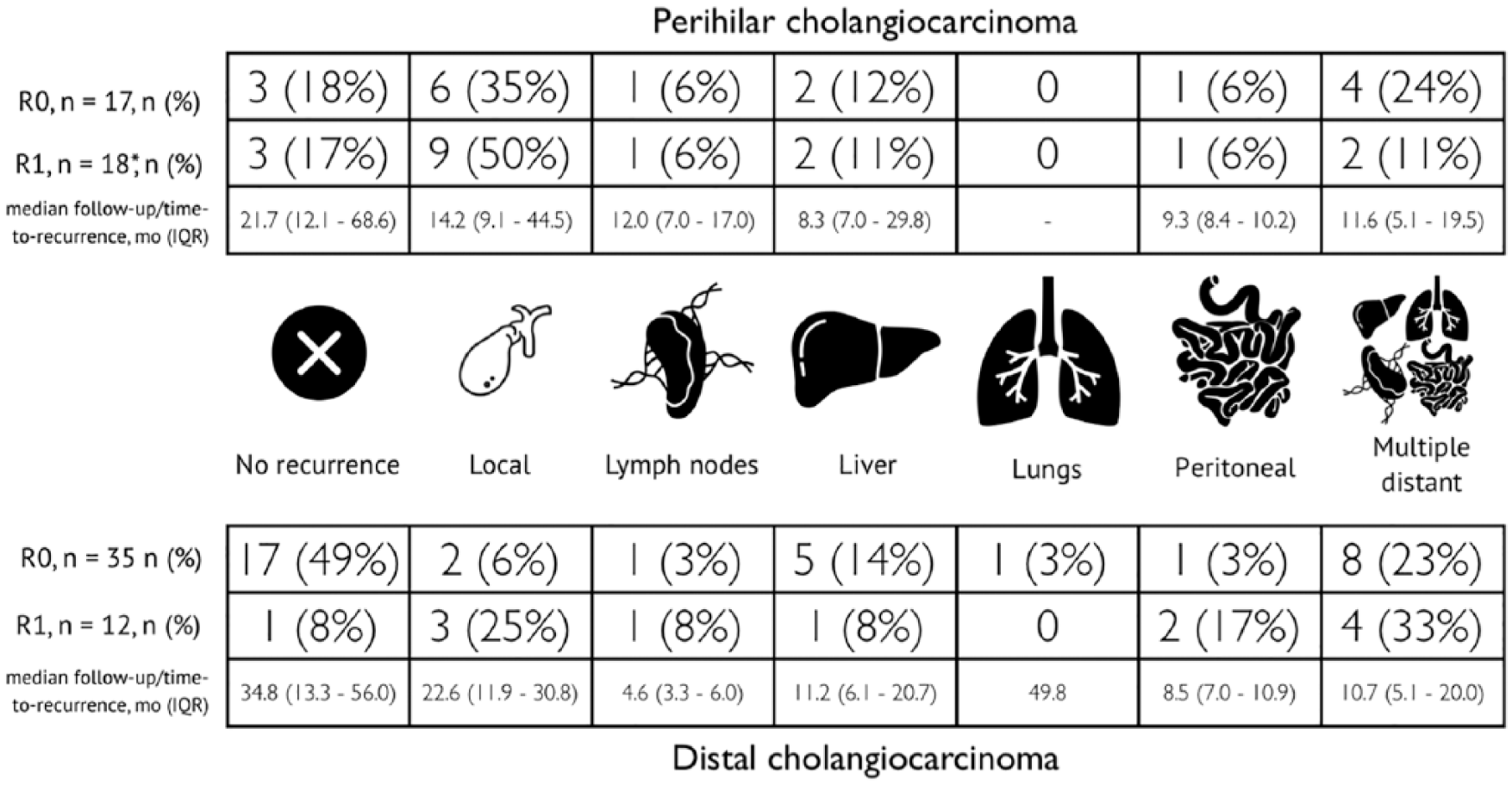
Patterns in the first detected recurrences for perihilar and distal cholangiocarcinomas. The most common recurrence types were local for perihilar cholangiocarcinoma and multiple for distal cholangiocarcinoma.
Discussion
This study evaluated the outcomes of patients who underwent resection with curative intent for extrahepatic CCA at a tertiary center during a 15-year period and revealed several important findings. First, although R0 resection is the desired goal of surgery, it was not found to be a prognostic factor for pCCA on univariate analysis nor was it an independent prognostic factor for dCCA. Second, most patients with pCCA experienced local recurrence, which highlights the importance of local control, although even patients with R0 resection did not necessarily have a lower local recurrence rate. Third, lymph node metastases appear to be more important prognostic factor in patients with dCCA. This is reflected in the recurrence pattern, and recurrence in patients with dCCA most often occurred in multiple synchronous sites. Fourth, the DFS and OS rates were similar in patients with pCCA and dCCA, despite the differences in their recurrence patterns.
In several earlier studies, lymph node metastases were the most widely reported prognostic factor for dCCA (12–16) and that tumor grade and CA19-9 levels were the most common prognostic factors for pCCA (17, 18). Although R0 resection is an important prognostic factor for pancreatic ductal adenocarcinoma, it appears to have a smaller role in CCA. Consistent with our findings, a large multi-center study revealed that R0 resection was not an independent prognostic factor for disease-specific survival among dCCA patients (19). Furthermore, although lymph node metastases play a major role in prognosis, the benefit of lymphadenectomy for patients with pCCA remains unclear. A few studies have revealed better survival among patients with positive lymph nodes who underwent resection than among patients whose tumors were not resected (20, 21). However, it remains unclear whether the survival benefit is due to resection or selection bias.
The overall recurrence rates for pCCA in previous large series were 55% and 78% (17, 22), which are lower than our recurrence rate of 83%. Nevertheless, local control remains an important challenge, as the rate of local recurrence among our patients was 43%; larger series have revealed somewhat lower numbers (26% in Groot Koerkamp et al.’s (17) and 28% in Dumitrascu et al.’s (23) studies). Some authors suggested that more extensive resection is required to reduce the recurrence rates, which could involve en bloc resection of the portal vein along with extended right hepatectomy or hepatopancreaticoduodenectomy (24, 25). However, such an approach produced similar local recurrence rates (29%–45%) albeit with greater morbidity rate than in previous studies (24, 25). Therefore, it is unclear whether more extensive surgery can provide better local and distant control. In our study, 20% of patients with pCCA underwent ⩾3 cholangiojejunostomies and 33% of patients underwent vascular resection. More extensive resection would likely increase the risk of postoperative complications, which must be balanced with the anticipated benefits of this approach. While other high-volume centers report mortality rates of approximately 10% after resection for pCCA (22, 26), our 90-day mortality rate of 0% likely indicates a different risk/benefit balance that influenced patient selection.
Our study revealed a local recurrence rate of 11% after dCCA resection, which is lower than the previously reported rates of 14%–24% (12, 15). It is easier to achieve clear resection margins in patients with dCCA because, in contrast to pCCA, the anatomical structures around the tumor can usually be resected. Furthermore, dCCA required fewer vascular resections than pCCA in our study (13% vs 25%) as well as fewer cholangiojejunostomies (>2 anastomoses: 4% vs 22%). Moreover, the recurrence patterns appeared to differ between pCCA and dCCA in a manner indicating that local control is an important issue for patients with pCCA, while distant control remains a challenge for dCCA.
Active surveillance is widely implemented after resection with curative intent, although the optimal duration and expected benefit remain unclear. In this study, all recurrences occurred within 5 years post-resection, and only one patient had an initial isolated lung metastasis. Unfortunately, there are few options after recurrence, even though the majority of pCCA recurrences were local. Re-resection is rarely possible, and chemotherapy has few benefits. Thus, some of our patients underwent regular follow-up with imaging, while other patients only underwent imaging when they developed symptoms. However, recent results from the BILCAP trial indicate that capecitabine might be an effective adjuvant therapy that could be used during a more intensive follow-up (27).
This study has several important limitations, such its single-center retrospective design and the small sample size that limit the power of the multivariate and survival analyses. However, these issues may be unavoidable given the fact that we included patients with resectable extrahepatic CCA who were treated at a tertiary hospital during a 15-year period.
In conclusion, this study demonstrated that, despite the close proximity and similar prognoses of pCCA and dCCA, these two tumor types have different risk factors and recurrence patterns.
Supplemental Material
Supplementary_Table_1 – Supplemental material for Differences in Prognostic Factors and Recurrence Patterns after Curative-Intent Resection of Perihilar and Distal Cholangiocarcinomas
Supplemental material, Supplementary_Table_1 for Differences in Prognostic Factors and Recurrence Patterns after Curative-Intent Resection of Perihilar and Distal Cholangiocarcinomas by V. Sallinen, J. Sirén, H. Mäkisalo, T. E. Lehtimäki, E. Lantto, A. Kokkola and A. Nordin in Scandinavian Journal of Surgery
Supplemental Material
Supplementary_Table_2 – Supplemental material for Differences in Prognostic Factors and Recurrence Patterns after Curative-Intent Resection of Perihilar and Distal Cholangiocarcinomas
Supplemental material, Supplementary_Table_2 for Differences in Prognostic Factors and Recurrence Patterns after Curative-Intent Resection of Perihilar and Distal Cholangiocarcinomas by V. Sallinen, J. Sirén, H. Mäkisalo, T. E. Lehtimäki, E. Lantto, A. Kokkola and A. Nordin in Scandinavian Journal of Surgery
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the institutional review board and the National Institute of Health and Welfare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Helsinki University Hospital VTR-research funds, the Mary and Georg Ehrnrooth’s Foundation, and the Vatsatautien Tutkimussäätiö Foundation. The funding sources had no role in the study design, data acquisition, data analysis, manuscript preparation, or the decision to publish the results.
Informed Consent
Informed consent was not required owing to the retrospective nature of the study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
