Abstract
Background and Aims:
The most effective dissection technique for raising the flap in abdominoplasty is still controversial, particularly in high-risk massive-weight-loss patients. LigaSure Impact™ vessel-sealing system (Medtronic, Dublin, Ireland) is an energy device commonly used among different surgical specialties to reduce morbidity and implement outcomes. The aim of this study was to investigate the effectiveness of LigaSure Impact in abdominoplasty compared with the conventional techniques, diathermia.
Material and Methods:
Patients underwent primary abdominoplasty after massive weight loss at a single center from 1 January 2008 to 31 May 2015 and were retrospectively reviewed. A total of 94 patients eligible for the study were divided into two groups on basis of the dissection technique: LigaSure Impact group (29 patients) and the conventional technique group (65 patients). Total intraoperative blood loss was the primary endpoint. Duration of the operation, perioperative complications and re-operation were recorded as secondary endpoints.
Results:
Baseline characteristics were well balanced between the groups. Significant differences were found in intraoperative blood loss favoring LigaSure Impact group (259.6 ± 198.8 mL vs 377.9 ± 190.0 mL, p = 0.004) and blood transfusion rates (13.8% vs 35.4%, p = 0.047). In contrast, operative time was significantly longer in LigaSure Impact group (168.6 ± 121.2 vs 179.7 ± 57.6 min, p = 0.005), while a tendency to shorter hospital stay was found in LigaSure Impact group (3.6 ± 1.1 days vs 4.6 ± 3.2 days, p = 0.081). Overall complications occurrence, Clavien–Dindo grade II (24.1% vs 55.4%) and grade III (13.8% vs 30.8%) complications were significantly lower in LigaSure Impact group (respectively, p = 0.005, p = 0.007, p = 0.016). Late (>30 days) re-operation rate was significantly lower in the LigaSure Impact group (6.9% vs 27.70%, p = 0.0028). Specific wound complications showed no significant difference.
Conclusion:
LigaSure Impact vessel-sealing system may be beneficial in improving abdominoplasty outcomes in massive-weight-loss patients because it might reduce blood loss, need for transfusions, complications, and re-operations.
Keywords
Introduction
Abdominoplasty is the most commonly performed plastic surgical procedure after massive weight loss, especially due to the increase of bariatric surgery (1). Benefits of this operation include improved quality of life, body satisfaction, self-esteem, physiological well-being, and better long-term weight control after bariatric surgery (2–4).
Compared with other esthetic surgery operations, abdominoplasty has a high rate of complications, particularly in massive-weight-loss patients (5). Most frequently observed complications include seroma, hematoma, infection, wound dehiscence and healing problems, partial skin necrosis, fat necrosis, lateral thigh cutaneous nerve damage, hypertrophic scarring, and systemic complications (5–16). Complication rates range from 4.2% to 50.7% and re-operation rate up to 43% (7–16).
Different technical approaches have been described to reduce complications and improve the outcomes: Scarpa fascia preservation (10, 16, 17), selective flap undermining (7, 18), combined liposuction (15, 19), use of tissue glues (20, 21), internal fixation techniques at closure (13, 19, 22), and postoperative compression dressings (23). Different studies have showed contradictory results when comparing the technique for raising the abdominal flap, mainly focusing on scalpel versus diathermy (8, 14, 24).
LigaSure Impact (LS, Medtronic, Dublin, Ireland) is an electrosurgical bipolar vessel sealing device which fuses and divides tissues with mechanic pressure and energy. It seals vessels up to 7 mm of diameter and delivers the right amount of energy when used (25). Various studies have proved it to be safe, effective, and to reduce intraoperative blood loss and operative time in urological, gynecological, colorectal, and general surgery procedures when compared with scalpel dissection and clips, conventional diathermy, and other energy-based instruments (26–29). However, LS application is described only in a few publications from the field of plastic surgery. Although described to be effective also in plastic surgery procedures (30, 31), there are no previous studies on LS use for abdominoplasty.
We hypothesized that the use of LS can reduce intraoperative blood loss and complications in abdominoplasty after massive weight loss, resulting in better outcomes. Thus, the aim of this study was to assess the impact of LS compared with conventional dissection techniques in abdominoplasty.
Material and Methods
This is a retrospective review of prospectively collected data. From 1 January 2008 to 31 May 2015, 94 consecutive patients underwent a primary abdominoplasty after massive weight loss at Turku University Hospital. The patients were identified through the hospital surgery registry. This was a retrospective study of prospectively collected data. Ethical approval was obtained from the institutional ethics committee. Exclusion criteria were as follows: body lift or belt lipectomy, secondary procedures, revisions or operations with other energy devices, and follow-up less than 6 months.
The aforementioned 94 consecutive patients eligible for the study were stratified into two groups (LS group and control group) on the basis of the dissection technique used for the abdominal flap: LS and conventional techniques (CTs) containing both steel scalpel and diathermy (on coagulation mode and average voltage of 35–45 V with regular tip). The indication for the use of LS was at the discretion of the individual surgeon.
Endpoints
The primary endpoint was the total intra-operative blood loss, considered as the total amount of blood lost from skin incision to skin closure. Blood was measured as collected in the suction containers. Swabs were squeezed, and their contents were suctioned and added to the collected fluid.
Secondary endpoints comprised the duration of the operation and the occurrence of postoperative complications such as seroma, hematoma, infections, wound dehiscence, fat necrosis, and blood transfusions using the Clavien–Dindo classification to document their severity (32). Hospitalization length, late complications (over 30 days), and re-operation were also recorded.
Patients
LS group comprised 29 patients (28 female and 1 male; mean age ± standard deviation (SD): 43.0 ± 9.9 years), whereas control group included 65 patients (56 female and 9 males; mean age ± SD: 46.6 ± 10.1 years).
Patient charts were analyzed for general characteristics including age, sex, body mass index (BMI), medical history, bariatric procedures, the history of smoking, and the amount of weight loss. Patient smoking less than 4 weeks preoperatively was considered as a smoker, and patient taking a diabetic medication was graded as a diabetic.
Definitions
A wound infection was any surgical wound requiring an antibiotic treatment for superficial infection; emergency drainage or hospital admission for deep infection. Wound dehiscence was a wound breakdown with full-thickness skin separation extending >0.5 cm with or without infection, resulting delayed healing longer than 2 weeks or demanding specialist dressing care. Fat necrosis was a palpable firmness 1 cm or greater in diameter that persisted beyond 3 months postoperatively. Seroma was defined as a collection of serous fluid or blood between tissues, identified through a clinical examination or imaging and requiring percutaneous or operative drainage.
Hematomas included hemorrhages requiring blood cell transfusion (Clavien–Dindo grade II) or emergency exploration (Clavien–Dindo grade II). Intra-, peri-, and postoperative blood transfusions given to treat hemorrhaging or anemia during hospitalization were recorded. Blood cell transfusion was usually performed for intraoperative or postoperative hemoglobin value <9 g/dL. Any re-operations, general system complications, and late complications were also tracked. The follow-up was determined as the last plastic surgeon contact or recorded weighing.
Surgical Technique
All patients in the series underwent a full abdominoplasty under general anesthesia with the same surgical team and preoperative care. Patients were marked preoperatively in a standing position and abdominoplasties were performed in a method previously described (33). A single intravenous dose of 1500-mg cefuroxime or 600-mg clindamycin was administered intraoperatively.
Briefly, the abdominal skin was perforated sharply to the level of premuscular fascia, and the dissection of the cutaneous abdominal flap up to xiphoid process was carried out either with LS or CT (Fig. 1). The hemostasis was achieved with bipolar electrocoagulation, besides LigaSure Impact in LS group; otherwise the operation technique between the groups was identical. After removing the excess skin, abdomen was closed in layers and compressive dressings were used. Generally, two closed suction drains were placed bilaterally to the low abdomen. Prophylactic enoxaparin 40 mg subcutaneously was given during the hospital stay. Intravenous antibiotic was continued postoperatively until the next day, followed by a perioral antibiotic for one week or until the drain removal for the majority of patients. Postoperative pain control was based on a multimodal approach including NSAIDs and opioids. Some patients (32%) received a pain pump catheter equipped with an elastomeric continuous infusion pump (bupivacaine or ropivacaine). Based on the output (< 50ml/day), drains were removed during hospital stay or afterwards at local health care center.
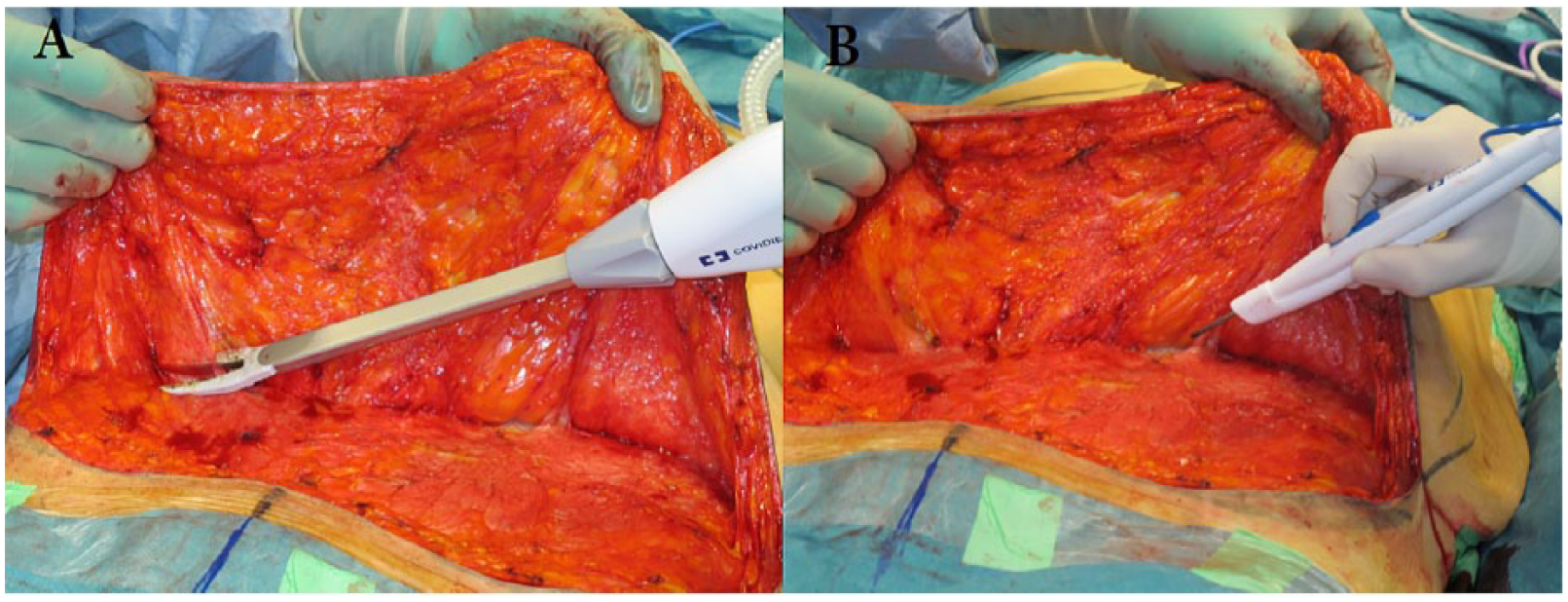
Abdominal flap dissection using (A) LigaSure Impact and (B) conventional diathermia.
Statistical Analysis
The results of parametric and nonparametric data were expressed as mean ± SD, and SPSS statistical software (SPSS 23.0, Chicago, IL, USA) was used for all statistical analyses. Comparisons between both groups were determined using the chi-square test or Fisher’s exact test when appropriate. Continuous variables were compared using the analysis of variance test. Univariate and multivariate logistic regression models were used to determine the associations between the occurrence of postoperative complications and patient and operative variables. A stepwise model-selection method was used to fit a multivariate regression model. Confidence intervals were set at 95%. A two-sided p-value of 0.05 was considered as statistically significant.
Results
The two groups were well balanced and mostly comparable (Table 1). Significant differences were found in decreased blood loss (259.60 ± 198.80 mL for LS group vs 377.85 ± 190.04 mL for the control group, p = 0.004) and in the operative time (179.73 ± 57.64 vs 168.59 ± 121.23 min, p = 0.005) favoring LS group, whereas no significant difference was detected in resection weight (1818.72 ± 702.43 vs 1818.00 ± 1356.21 g, p = 0.036; Table 2). Although not statistically significant, the result demonstrated a trend among the LS group toward reduction of hospital stay (3.6 ± 1.1 vs 4.6 ± 3.2 days, p = 0.081). The follow-up was significantly longer in the control group (42.38 ± 36.78 vs 8.21 ± 9.53 months, p = 0.001).
Demographics of patients at time of study.
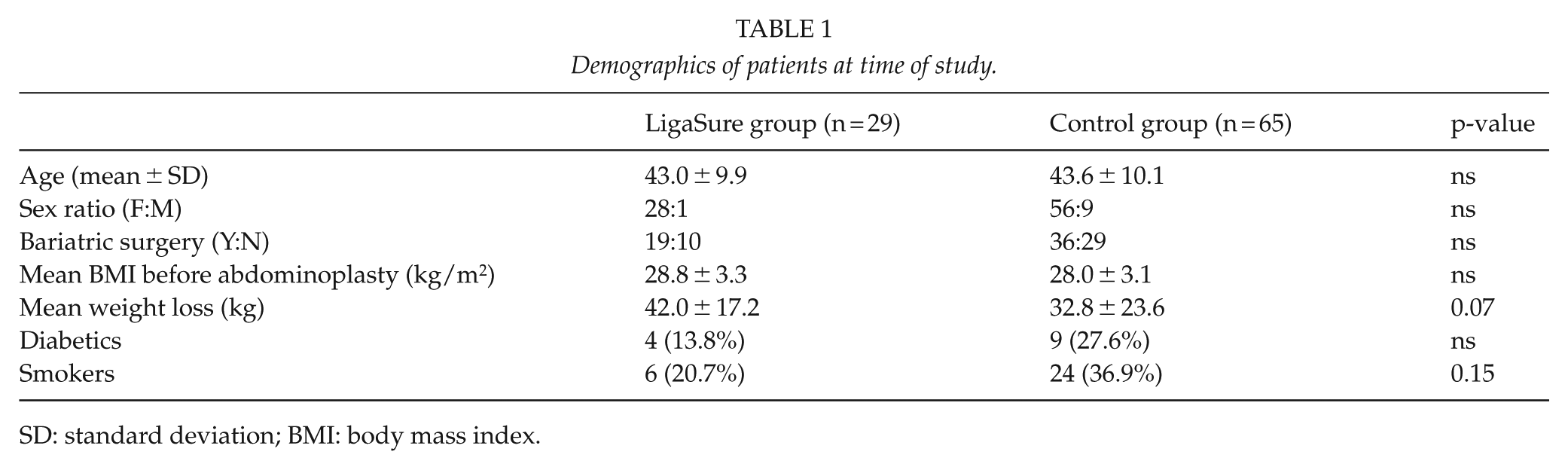
SD: standard deviation; BMI: body mass index.
Comparison of perioperative parameters in the two groups of patients.
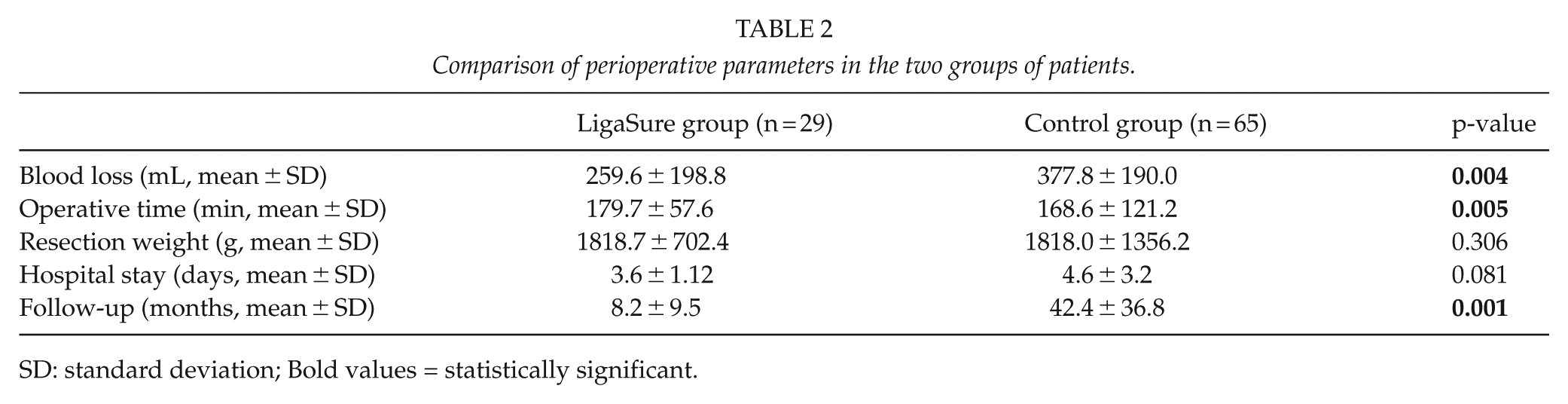
SD: standard deviation; Bold values = statistically significant.
The occurrences of patients with complications were 34.48% in the LS group and 47.69% in the control group. The LS group showed significantly lower overall early complication occurrence and Clavien–Dindo grade II and III complications occurrences (p = 0.005, p = 0.007, p = 0.016, respectively; Table 3). Particularly, the LS group had a significantly reduced need for blood transfusion (13.79% vs 35.38%, p = 0.0472; Table 3). The occurrence of seromas was higher in the control groups than the LS group (10.34% vs 20%, p = 0.375), whereas the occurrence of postoperative hematomas, superficial and deep wound infections, wound dehiscence, and fat necrosis did not significantly differ between the groups.
Postoperative complications < 30 days.
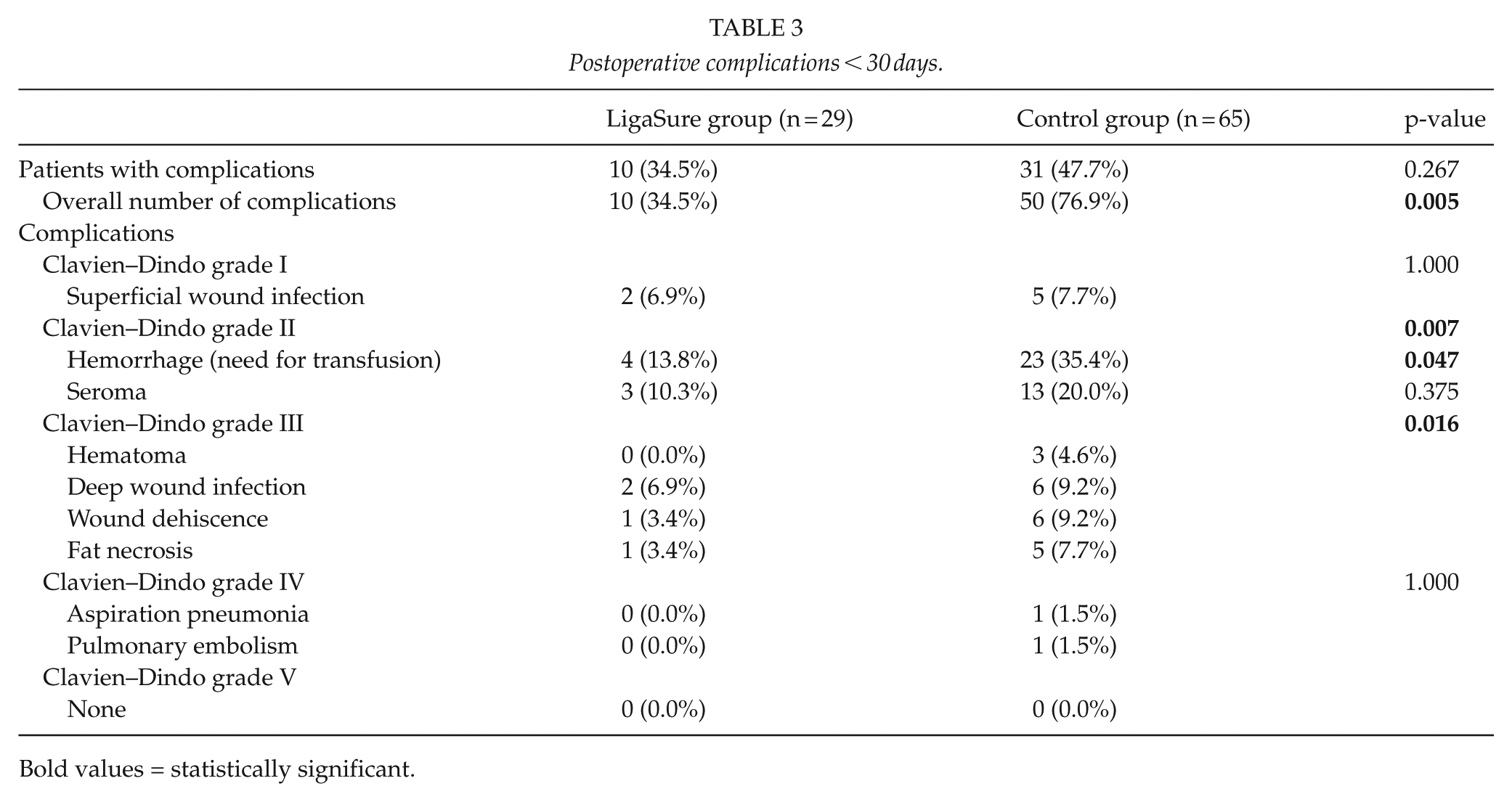
Bold values = statistically significant.
Two Clavien–Dindo grade IV complications occurred: one aspiration pneumonia demanding intensive care unit and one massive bilateral pulmonary embolism requiring cardiac care unit, both in the control group and without further consequences (Table 3). There were no Clavien–Dindo grade V complications.
Long-term complications are outlined in Table 4, with significantly higher re-operation rate in the control group (6.9% vs 27.7%, p = 0.028). However, the majority of the re-operatives were due to esthetic reasons. Two re-operations occurred in the LS group: one in consequence of a deep infection and another to correct hypertrophic scarring.
Late complications (>30 days) requiring re-operation.
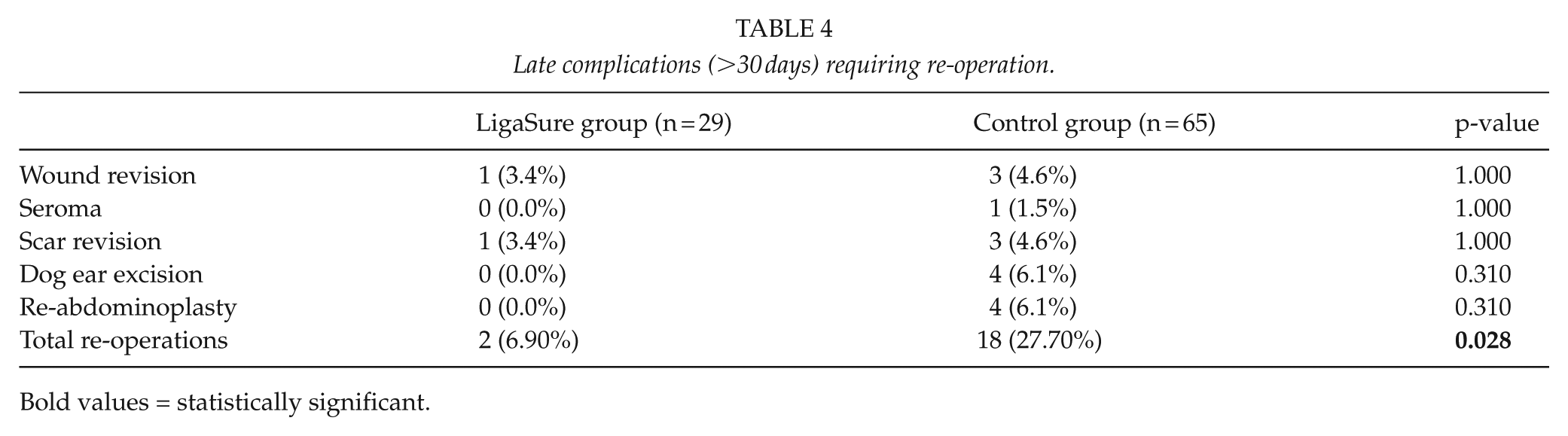
Bold values = statistically significant.
Univariate analysis showed increased intra-operative blood loss, increased massive weight loss, previous bariatric surgery, advanced age, any comorbidity, and higher BMI to be associated with postoperative complication occurrence. These factors were then analyzed with multivariate logistic regression which confirmed increased intra-operative blood loss, increased massive weight loss, and advanced age to be independent predictors of postoperative complication occurrence (Table 5).
Univariate variables put into a multivariate model for any occurrence of postoperative complication.
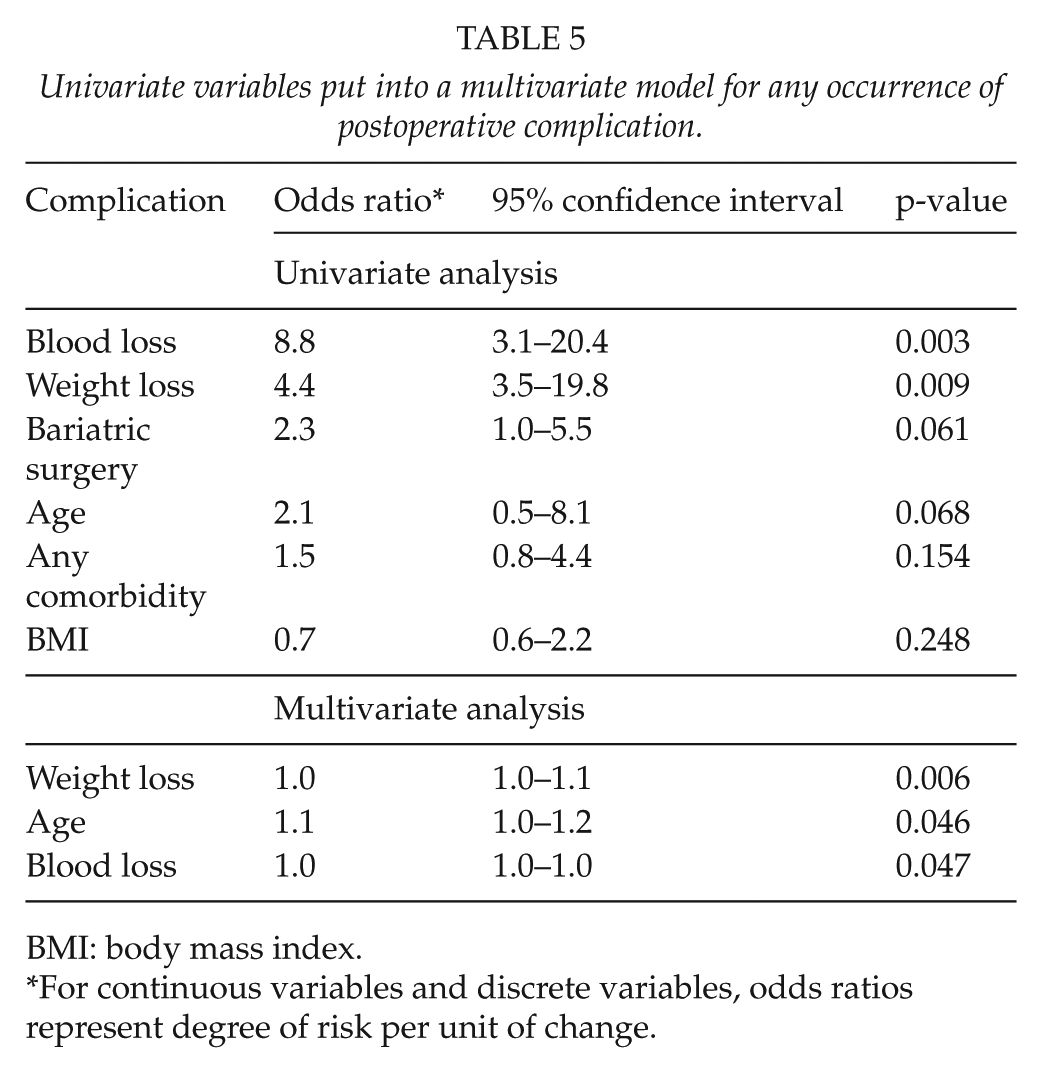
BMI: body mass index.
For continuous variables and discrete variables, odds ratios represent degree of risk per unit of change.
Discussion
The use of LS compared with conventional dissection techniques in abdominoplasty decreased intra-operative blood loss; blood transfusions; and the occurrences of Clavien–Dindo grade II, grade III, and overall complications. In contrast, the operative time was increased in the LS group. Although not significant, a tendency to reduce the length of hospital stay was noticed favoring the LS group. Specific wound complications showed no significant difference, although there were slightly less seroma occurrence using LS.
To the best of our knowledge, this is the first study on the use of LS or similar electrothermal bipolar vessel sealers in abdominoplasty. Only few studies evaluate the effectiveness of LS in plastic surgery with a small series of patients. Güven et al. (31) harvested endoscopically the latissimus dorsi-flap, reducing bleeding, operative time, and drain output, as well as transfusions and hospitalization length. Similar results were found by Konno and Kishi (30) in neurofibroma removal comparing LS with electrocautery.
The superiority of LS in controlling the bleeding has been demonstrated in comparative studies to CTs from various surgical fields (26–30). Correspondingly, using LS, we showed a significantly decreased blood loss by an average of 120 mL, which is about one-third of the total. The improved hemostasis is likely due to LS vessel sealing properties.
Operative time is generally demonstrated to be shortened with LS, as the need for other mechanic hemostatic methods to control bleeding is reduced (26–29, 31). However, our operative time was significantly about 10 min longer than LS. The increase may be resulted because each cutting and sealing cycle with LS takes 2–4 s, whereas dissection with CTs is faster and hemostasis is still achieved without other time-consuming methods. Longer operative time increases complications, especially wound infections, in addition to expanding operative theater costs (34).
Complications in abdominoplasties occur rather often with rates between 4.2–50.7% (7–15), although the lack of uniform definitions for complications hampers precise comparing between individual studies. In our study, there were overall significantly less complications with LS. Particularly, although not significant difference, the occurrences of patients with complications were 34.5% with LS and 47.7% with CT. Our complication rates, despite being coherent with literature, are relatively high, which may be consequent of our patient material comprising mostly overweight (BMI between 25 and 30 kg/m2) and massive weight loss patients (5, 11, 35). Indeed, since all the patients had a postoperative control at our hospital, our awareness of the occurred complications was high.
We graded our complications further according to Clavien–Dindo system (32) for better comparison between the groups: there were significantly less grade II and grade III complications with LS. Clavien–Dindo grade II complications require blood transfusions or drugs other than antiemetics, antipyretics, analgesics, diuretics, or electrolytes, and grade III complications need surgical, endoscopic, or radiological intervention (32).
We detected that the need for transfusions was significantly decreased with LS, with a 61.0% reduction, from 35.4% with CT to 13.8% with LS. Data regarding transfusions in abdominoplasties is limited with few recent studies representing rates as low as 0%–1.3% up to 26.6% in post-bariatric circular lipo-abdominoplasty (8, 9, 16, 20). Recently, a large cohort study of 20,310 post-bariatric abdominoplasties in the United States found transfusion rate of 9.3%, but less complex panniculectomies were also included (36). Our high rates may be partly due to the widely varying threshold of giving perioperative transfusions. For example, our transfusion practice, where transfusions are usually given when hemoglobin value (Hb) is below approximately 9 g/dL, is somehow more liberal than the guide-lined thresholds between Hb 6–10 g/dL by the American Society of Anesthesiologists (37). Furthermore, the base-line anemia and comorbidities, which were frequently present among our patients with massive weight loss and previous bariatric procedures, increase transfusions rates (38, 39). This issue has its importance because, although considered generally safe, transfusions can cause immunological responses, including severe hemolytic reactions. Moreover, perioperative transfusions are associated with complications, higher mortality, longer hospitalization and cardiovascular events (36, 39).
Although not significant, there were less seromas with LS (10.3% versus 20.0% with CT) comparable to 0.1%–60% of previous studies (5, 7–22, 24). Seroma formation is assumed to be multifactorial, and it is associated with damaged lymphatic flow, inflammatory response, surgically created dead space, and the use of electrosurgery (8, 14, 22). We did not perform ligation of the perforators during the abdominal flap raising (40). In our study, the lower seroma rate using LS is possibly due its lower energy release.
According to literature, re-operation and revision rates range from 5% to 43%, depending on differing definitions, operative techniques, and follow-up times (7–9, 11, 13, 15). We found late (>30 days) re-operation occurrence of 6.9% with LS is significantly lower than 27.7% of the CT group. However, the majority of re-operations were for esthetic reasons. Therefore, this reduction is unlikely to be solely due to the less complications requiring surgical intervention with LS. Possibly, the postoperative wound complications had an esthetically negative effect to the outcomes, reflecting the decreased overall complications with LS on the re-operation rates.
The length of hospital stay was 1 day shorter with LS, but significant difference to CT was not achieved. The fewer transfusions and other early complications with LS are likely to lead to a better postoperative recovery. Nevertheless, LS reportedly results less postoperative pain, allowing earlier mobilization (29).
Finally, we identified in the multivariate analysis that not only increased blood loss and preoperative weight loss but also advanced age amounts to be predictive of postoperative complications. This fact may be taken into consideration for patient selection in abdominoplasty after massive weight loss (43).
A disposable LS costs about 400 euros (41) with additional expenses due to possible longer operative theater time, but it can be considered cost-saving as it shortens the length of hospitalization and causes less complications and re-operations leading to re-visits and hospital re-admissions. Secondary savings may also occur from reducing patients’ work leaves. Further cost-saving effect using LS might result due to the decrease of transfusions: it has been estimated that the real cost of one red cell unit transfusion to a surgical patient is about 700$ in Switzerland, when adjusted to inflation (42).
The increasing demand for particularly post-bariatric abdominoplasties and the awareness of the high complication rates make important to find the most effective techniques in terms of safety, esthetic outcomes, and economic aspects.
Despite the interesting findings, this study has several limitations, and the major drawback is due to the retrospective nature and small number of participants. The patients were mostly massive-weight-loss patients with higher complication risks, and LS group presented an average of 10 kg greater preoperative weight loss, which may have affected the results between the groups (35).
In addition, the higher complication rate reported in the conventional diathermy group might be influenced by the higher number of smokers in the same group although not statistically significant in demographics, neither in the multivariable analysis. Nonetheless, another potential bias is the shorter follow-up time of the LS group which may influence the late re-operation rates. Larger, prospective randomized studies on this topic are needed to further clarify the re-operation rate and the cost-effectiveness of LS.
Conclusions
Our results suggest that performing abdominoplasty with LS is beneficial in reducing intra-operative blood loss, transfusions needs, and the occurrence of complications and re-operations. LS might decrease the length of hospital stay than CT, even though it increases the operative time.
