Abstract
Background and Aims:
Minimally invasive esophagectomy is a favored alternative in high-volume centers. We evaluated the introduction of, and transition to, minimally invasive esophagectomy at a medium volume tertiary referral center (10–20 esophagectomies annually) with focus on surgical results.
Material and Methods:
Patients who underwent minimally invasive esophagectomy or open transthoracic surgery for carcinoma of the esophagus or gastroesophageal junction (Siewert I and II) during 2007–2016 were retrospectively studied. Sorted on surgical approach, perioperative data, surgical outcomes, and postoperative complications were analyzed and multivariate regression models were used to adjust for possible confounders.
Results:
One hundred and sixteen patients were included, 51 minimally invasive esophagectomy (21 hybrid and 30 totally minimally invasive) and 65 open resections. The groups were well matched. However, higher body mass index, neoadjuvant chemoradiotherapy, and cervical anastomosis were more frequent in the minimally invasive esophagectomy group. Minimally invasive esophagectomy was associated with less peroperative bleeding (384 vs 607 mL, p = 0.036) and reduced length of stay (14 vs 15 days, p = 0.042). Duration of surgery, radical resection rate, and postoperative complications did not differ between groups. Lymph node yield was higher in the minimally invasive esophagectomy group, 18 (13–23) vs 12 (8–16), p < 0.001, confirmed in a multivariate regression model (adjusted odds ratio 3.15, 95% class interval 1.11–8.98, p = 0.032).
Conclusion:
The introduction of minimally invasive esophagectomy at a medium volume tertiary referral center resulted in superior lymph node yield, less peroperative blood loss and shorter length of stay, without compromising the rate of radical resection, or increasing the complication rate.
Keywords
Introduction
Minimally invasive esophagectomy (MIE) (1) has emerged as a safe and many times favored alternative to open surgery (2). Technical advances and mounting experience have made MIE available to an increasing number of patients with cancer in the esophagus and gastroesophageal junction (GEJ). MIE has been proposed to reduce the risk of serious postoperative pulmonary complications (2, 3) compared to open surgery and studies indicate satisfactory surgical results (4).
When introducing a minimally invasive procedure, or any new medical treatment, it is of utmost importance not to compromise the basic principles of surgery such as radical resection, adequate lymph node clearance, and the safety of the procedure. The vast majority of studies regarding MIE stem from high-volume centers where new techniques are implemented at a quicker pace. With minimally invasive esophageal surgery being increasingly implemented at centers with lower volume, the transition to MIE at such a center benefits from evaluation.
MIE, with the present technique, has been performed since 2013 at our institution. In Sweden, during 2014–2015, 36% of all esophagectomies were performed as minimally invasive procedures, predominantly at three tertiary centers. The aim of this study was to evaluate the introduction of, and transition to, MIE at a medium volume tertiary referral center with focus on surgical results.
Material and Methods
Study Design
Patients who underwent elective, open or minimally invasive, surgery for esophageal (ICD C15.0–15.9) or GEJ cancer, Siewert I and II (ICD C16.0a and 16.0b), at a Swedish university hospital between 2007 and 2016 constitute the cohort for this study. Baseline characteristics, perioperative and procedural data, postoperative complications, pathologic anatomical results from surgical specimens were extracted from patient medical charts and medical administrative systems. Two separate researchers scrutinized the postoperative pathology reports.
Operative Procedures
Open procedures were performed either as an Ivor Lewis esophagectomy through a midline laparotomy and a posterolateral thoracotomy with an intrathoracic stapled circular anastomosis or as a McKeowns esophagectomy through a posterolateral thoracotomy, a midline laparotomy and with a handsewn, left-sided, cervical anastomosis. Transhiatal surgeries were excluded. Minimally invasive procedures were performed either as a hybrid esophagectomy with a thoracoscopic approach and the abdominal part as open surgery with a handsewn, left-sided, cervical anastomosis or as a totally MIE with an intrathoracic linear stapled and handsewn anastomosis. A standardized two-field lymphadenectomy was performed in all patients, including removal of abdominal lymph nodes along the suprapancreatic border and the celiac trunk as well as around the left gastric artery, lesser curvature, and hiatal region. The mediastinal lymphadenectomy involved clearance of the subcarinal lymph nodes and the paraesophageal lymph nodes to meet the abdominal dissection plane.
Neoadjuvant Treatment
The neoadjuvant chemotherapy regime was three cycles of epirubicin and oxaliplatin, in combination with capecitabine, during the study period. For patients receiving neoadjuvant chemoradiotherapy in the early years of the study period, the predominant regime was three cycles of oxaliplatin in combination with 5-fluorouracil and concomitant external radiation to 44–50 Gy. In the latter years of the study period, neoadjuvant chemoradiotherapy was given according to the CROSS-regime with five cycles of carboplatin/paclitaxel and concomitant external radiation to 41.4 Gy.
Ethics
The study was approved by the regional ethics committee in Uppsala, DNR 2017/206.
Statistical Methods
Baseline characteristics and perioperative data were analyzed with descriptive statistics, Wilcoxon rank-sum test or two-sided t-test for continuous variables and Pearson’s χ2 test for ordinal variables. Surgical oncological outcomes as well as postoperative complications were analyzed with multivariate logistic regression models. Potential confounding factors in each model were identified using causal directed acyclic graphs (5) (DAGs). All statistical analysis was performed with STATA© version 14.2 (StataCorp, College Station, TX, USA).
Results
Baseline Characteristics
The study comprised 116 patients (88% men, median age 66 years) in two groups: 51 MIE (21 hybrid and 30 totally minimally invasive) and 65 open (53 Ivor Lewis and 12 McKeowns) procedures. In the MIE group, a cervical anastomosis, neoadjuvant chemoradiotherapy, and positive radiological suspected lymph nodes on preoperative workup were more frequent. A slightly higher body mass index (BMI) in the MIE group was also observed. The groups were well matched in all other aspects. Baseline characteristics are outlined in Table 1.
Baseline data on patients with esophageal or GEJ cancer, 2007–2016, grouped by type of surgery.
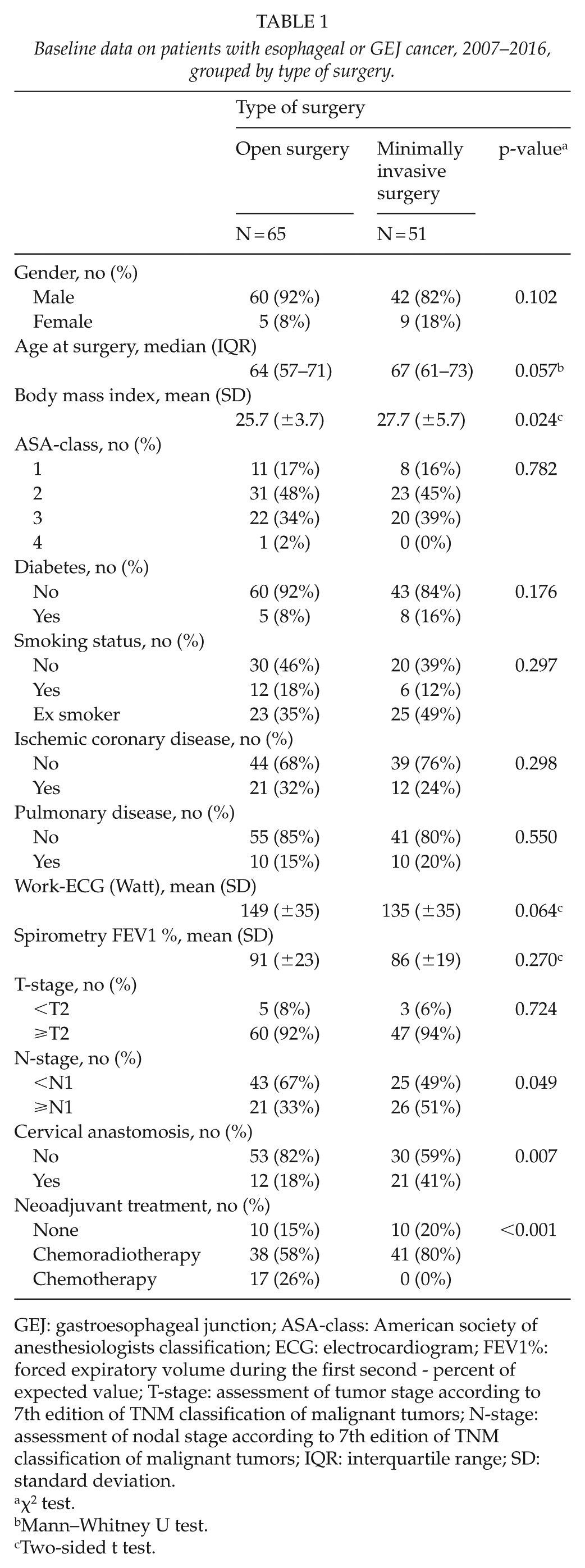
GEJ: gastroesophageal junction; ASA-class: American society of anesthesiologists classification; ECG: electrocardiogram; FEV1%: forced expiratory volume during the first second - percent of expected value; T-stage: assessment of tumor stage according to 7th edition of TNM classification of malignant tumors; N-stage: assessment of nodal stage according to 7th edition of TNM classification of malignant tumors; IQR: interquartile range; SD: standard deviation.
χ2 test.
Mann–Whitney U test.
Two-sided t test.
Perioperative Data
The duration of surgery did not differ between groups (315 ± 63 vs 303 ± 57, minutes, mean ± SD, p = 0.265). MIE was associated with reduced mean peroperative blood loss (384 ± 651 vs 607 ± 481, mL, p = 0.036) and shorter median length of stay in hospital care (14 (10–23) vs 15 (14–21), days, p = 0.042). Total time in a unit with postoperative intensive care did not differ between groups, Table 2.
Perioperative data on patients with esophageal or GEJ cancer, 2007–2016, grouped by type of surgery.
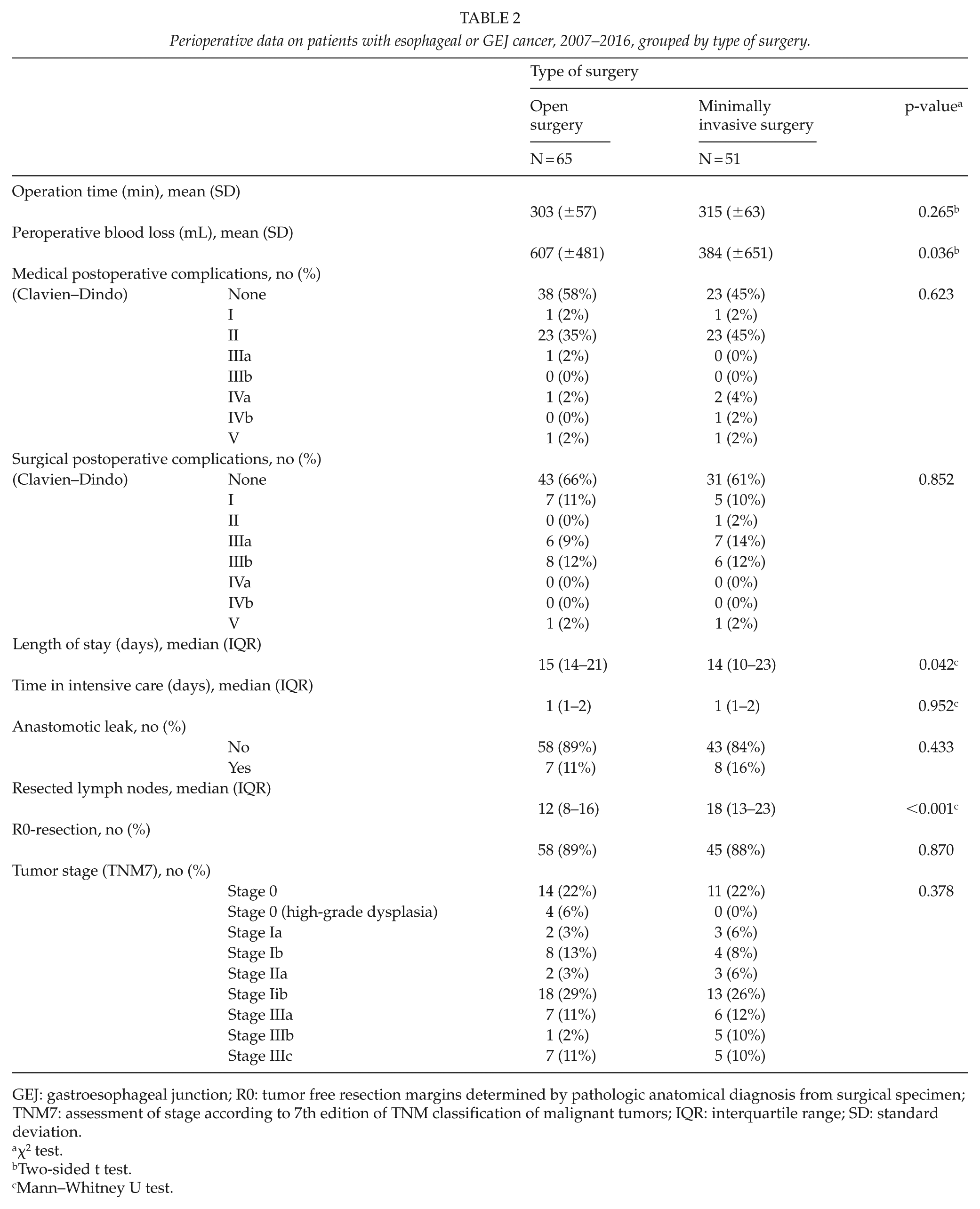
GEJ: gastroesophageal junction; R0: tumor free resection margins determined by pathologic anatomical diagnosis from surgical specimen; TNM7: assessment of stage according to 7th edition of TNM classification of malignant tumors; IQR: interquartile range; SD: standard deviation.
χ2 test.
Two-sided t test.
Mann–Whitney U test.
Lymph Node Yield and Radicality
The number of resected nodes was higher in the MIE group, 18 (13–23), median (interquartile range), compared to 12 (8–16) in the open surgery group, p < 0.001. When adjusting for confounding factors in a multivariate ordinal logistic regression model, MIE remained a positive predictor for a higher lymph node yield (adjusted odds ratio (OR) 3.15, 95% confidence interval (CI) 1.11–8.98, p = 0.032). Neoadjuvant chemoradiotherapy was associated with decreased lymph node yield (adjusted OR 0.28, 95% CI 0.08–0.97, p = 0.045) (Fig. 1).
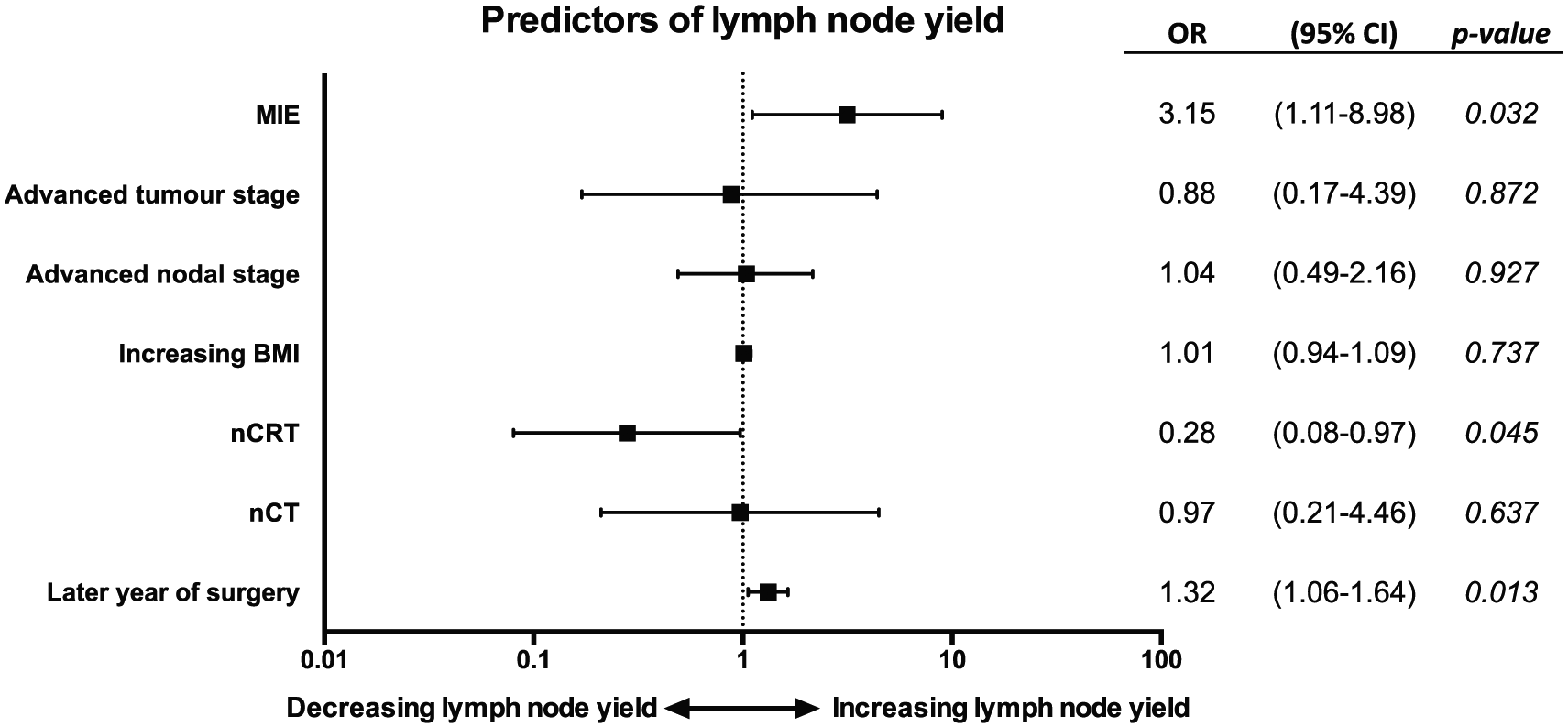
Odds ratios (OR) with 95% confidence interval (CI) and graphic presentation of predictors of lymph node yield from a multivariate ordered logistic regression model in 116 patients undergoing esophagectomy for esophageal or gastroesophageal cancer.
The rate of radical resection, as determined by a tumor free proximal, distal, and circumferential resection margin, did not differ between the groups; 88% (45/51) in the MIE group versus 89% (58/65) in the open surgery group, p = 0.870, Table 2.
Postoperative Complications
There were no significant differences in the rate of anastomotic leakage between groups (16% vs 11%, p = 0.433). The frequency and severity of medical and surgical postoperative complications did not differ between the two groups in an unadjusted comparison, Table 2, or in multivariate logistic regression with adjustments for age, American society of anesthesiologists classification (ASA-class), BMI, neoadjuvant treatment, smoking, and histological tumor type (medical complications: OR 2.16, 95% CI 0.9–5.19, p = 0.085; surgical complications: OR 1.99, 95% CI 0.78–5.07, p = 0.148).
Discussion
Surgical Results
Basic principles in surgical management of gastrointestinal cancer constitute a multi-modal approach with radical resection of the tumor as a key element in a potential cure. Over the last two decades, MIE has increasingly been used for resection of esophageal and GEJ cancers and has been found to reduce postoperative morbidity at high-volume centers (2, 3, 6). Studies regarding oncological outcomes and survival after esophagectomy emphasize the advantages of neoadjuvant chemoradiotherapy (nCRT) (7) or chemotherapy (8) and en-bloc resection of the tumor (9). Extensive lymphadenectomy improves postoperative tumor staging (10, 11); yet there is also evidence in support of the importance of a high lymph node yield for improved survival and loco regional disease control (12, 13). However, the use of nCRT affects the total number of resected lymph nodes (13), as was also the case in the multivariate regression model of the present study. Thus, there is an ongoing debate concerning the usefulness of extended lymphadenectomy in the era of nCRT treatment (14). Until harder evidence in any other direction is presented, a radical en-bloc resection with a high lymph node yield, while minimizing postoperative morbidity, should be strived for.
Learning Curve
The study period involved the first 51 MIE surgeries performed at our center and thus clearly includes the greater part of the learning curve of the MIE operation. The first 21 cases were operated with a hybrid approach and after successful implementation of this technically less demanding technique; the following procedures were totally minimally invasive. Reports propose the learning curve of MIE to be around 30–40 operations (15, 16) to overcome the technical difficulties and even suggested as high as 119 surgeries to surpass the learning curve regarding anastomotic insufficiency (17). The present study of 51 MIE patients operated in a medium volume center proposes similar results to Nilsson et al. reporting on the successful implementation of MIE at a Swedish high-volume center in 173 patients over 5 years (18). In the present study, the duration of surgery was unaffected by the transition to MIE, possibly reflecting the present surgeons’ extensive experience in other advanced laparoscopic procedures. In addition, the peroperative blood loss was reduced, in conformity with a previous study (19).
Lymph Node Yield
The number of resected lymph nodes was greater in the group of patients who underwent MIE and the multivariate logistic regression model addressing potential covariates influencing the number of resected lymph nodes confirmed MIE as a solid positive predictor for increased lymph node yield. A plausible explanation for the increased number of lymph nodes could be improved visual field by magnification of the video-laparoscope and/or the reduced peroperative bleeding. Another possible explanatory factor is if the pathologists have improved their technique or thoroughness (20) thus identifying more nodes in the latter years of the study period, in parallel to the introduction of MIE at our institution. We addressed this issue by including year of surgery (equaling year of pathologic examination) as a potential confounder in the multivariate logistic regression model. Yet the firm association between MIE and increased lymph node yield still remained.
Postoperative Complications
A similar proportion of postoperative surgical complications were registered in the MIE and open group (40% vs 34%), with severe complications (Clavien–Dindo ⩾ 3b, that is, intervention in general anesthesia) occurring in 14% in both groups. Neither the total number of medical complication (55% vs 42%), nor the Clavien–Dindo ⩾ 3b, 8% and 3%, differed significantly between the groups. No firm associations between MIE and surgical or medical postoperative complications were found with multivariate logistic regression. Although a tendency toward increased postoperative complications with MIE, foremost medical, could be discerned from the increased adjusted ORs. This was probably due to that the learning curve of MIE was included in the cohort. Even though cervical anastomoses were more frequent in the MIE group, the rates of anastomotic leakage did not differ between groups (16% vs 11%, p = 0.433), bearing in mind that the study was not powered for this analysis.
Length of Stay
Patients in the MIE group had a shorter overall length of stay in the hospital. The same routines for discharge applied to all patients, regardless of operative approach, where they were not discharged until they could independently manage basal daily activities and basal care. Less postoperative pain could be an important factor facilitating an earlier discharge of patients in the MIE group. Although it can be argued that minimized trauma to the thoracic and abdominal wall results in an eased recovery, the constant strives for reduced in-hospital stay can bias the results as the majority of MIE patients underwent surgery in the latter years of the study period, influenced by enhanced recovery programs. At our center, the introduction of MIE allowed us to monitor the patients overnight in the postoperative recovery room instead of the ICU.
Limitations
The retrospective cohort design is a limitation to this study, as is the absence of randomization. By including all patients undergoing elective surgery during the study period, the results are less likely biased by patient selection. As mentioned above, the minimally invasive procedures were introduced and carried out in the three latter years of the study period. This fact could well influence the results and cloud some associations; thus, we addressed the issue by treating year of surgery as a confounding factor and adjusted accordingly in the appropriate statistical analyses.
Conclusion
The introduction of, and transition to, MIE at a tertiary referral center with medium annual operative volume was successful and in the present setting of approximately 20 esophagectomies annually resulted in superior lymph node yield compared to open surgery. The introduction of MIE, including the entire learning curve, was not significantly associated with more postoperative complications than open surgery. In contrast, MIE was associated with less peroperative blood loss and a shorter length of stay. Even in centers that do not reach the prerequisites for high-volume caseload, MIE is plausible with adequate or even improved surgical outcomes in patients with cancer in the esophagus or GEJ.
Footnotes
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by the regional ethics committee in Uppsala, DNR 2017/206.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Partial financial support for this study was provided by the Bengt Ihre research fund and Swedish Cancer Society (to J.H.) and by the Lennander’s research fund (to G.L.).
