Abstract
Background and Aims:
Primary aldosteronism is a common cause of secondary hypertension. Primary aldosteronism is caused by an aldosterone-producing adenoma or bilateral hyperplasia that in some cases is asymmetrical with one adrenal dominating aldosterone secretion. Most patients with aldosterone-producing adenoma are biochemically cured by unilateral adrenalectomy, but patients with bilateral hyperplasia have a significant risk of residual or recurrent disease. Here, immunohistochemistry of CYP11B1 and B2 was used to investigate whether these markers could aid in the diagnostic workup of primary aldosteronism patients.
Materials and Methods:
A total of 39 patients with primary aldosteronism who underwent unilateral adrenalectomy for a presumed adenoma during 2013–2016 were included. Immunohistochemistry using monoclonal antibodies identifying the enzymes CYP11B1 and B2 was part of routine histopathological workup in 6 cases; in 33 cases, it was applied retrospectively. The hyperplasia diagnosis was suggested when there was no dominating nodule but immunoreactivity for CYP11B2 was seen in several nodules, which were also seen on routine sections. To distinguish between adenoma and hyperplasia, a ratio between the largest and second largest CYP11B2-positive nodules was calculated.
Results:
In all, 22 patients had an aldosterone-producing adenoma, while 13 patients were judged to have hyperplasia. In four cases, a final diagnosis could not be established, thus these were judged equivocal. Among the 33 cases investigated retrospectively, the primary histopathological diagnosis was altered from hyperplasia to aldosterone-producing adenoma in 9 cases (27%) after immunohistochemistry, and the immunohistochemically rectified adenoma group displayed improved clinical cure rates compared to the routine H&E-diagnosed cohort. Moreover, the B2 ratio was significantly higher in adenoma than in hyperplasia and equivocal cases.
Conclusion:
Immunohistochemistry detecting CYP11B1 and B2 expression is of great help in establishing a final histopathological diagnosis in patients with primary aldosteronism. This procedure should be part of the histopathological routine in all operated primary aldosteronism patients.
Introduction
Primary aldosteronism (PA) is the most frequent cause of secondary hypertension, with an estimated prevalence of 5%–10% in hypertensive patients (1). Patients with primary aldosteronism have a higher prevalence of cardiovascular and cerebrovascular morbidity (2) compared to patients with primary hypertension, matched for age, sex, and blood pressure (3, 4). There is also evidence that the excess risk is reduced with specific treatment, such as mineralocorticoid receptor blockers or adrenalectomy (5–7). Therefore, it is important to identify this condition and offer appropriate treatment. Traditionally, patients with unilateral disease caused by an aldosterone-producing adenoma (APA) are recommended for adrenalectomy, while bilateral adrenal hyperplasia (BAH) is preferably treated medically with mineralocorticoid receptor blockers. The term unilateral adrenal hyperplasia denotes the histological finding of hyperplastic nodules in an adrenal gland without bilateral involvement, but this entity is poorly characterized.
Postoperatively, the functionality of adrenal lesions has not been possible to evaluate, as hematoxylin–eosin staining used in routine histological assessment does not reveal the site of aldosterone production. This field has changed dramatically with the advent of in situ hybridization (8) and the subsequent development of specific antibodies targeting the cytochrome P450 family 11 subfamily B members 1 and 2 (CYP11B1 and CYP11B2), two enzymes responsible for synthesizing cortisol and aldosterone, respectively (9).
The introduction of specific and sensitive antibodies targeting CYP11B1 and B2 has provided new information concerning identification of aldosterone-producing sites (10, 11) as well as the panorama of somatic mutations in the various nodules with visualized aldosterone production (12). More specifically, it allows the practicing pathologist to better distinguish whether the aldosterone production is unifocal (as in APA) or multifocal (as in BAH) using conventional immunohistochemistry (IHC). It is obvious that this new knowledge will have an impact on the routine histopathological evaluation in PA patients (11, 13), as a correct diagnosis is paramount for the management of PA patients. Patients with an APA have low risk of recurrence, while patients with hyperplasia have a higher risk of persistent or recurrent disease.
The aim of this study was to evaluate the contribution of CYP11B1 and B2 IHC to the final pathology assessment in a well-characterized cohort of patients with PA and to correlate the histopathological results to clinical outcome using the Primary Aldosteronism Surgical Outcomes (PASO) criteria (14). This study is original in the sense that we report the clinical use of these markers in an accredited pathology laboratory setting and show how the diagnosis is changed for a subset of cases when antibodies are applied compared to when the diagnosis is put forward using routine H&E staining only.
Materials and Methods
Patients
During 2013–2016, 41 patients with PA were operated with adrenalectomy at the Karolinska University Hospital, Stockholm, Sweden. Two cases were excluded because the formalin-fixated paraffin-embedded material was missing, and the remaining 39 cases were included. Patient records were studied in retrospect, and when data were missing, patients were contacted for complementary information about blood pressure and antihypertensive medication. Summarized patient characteristics are shown in Table 1.
Characteristics of the primary aldosteronism cohort.
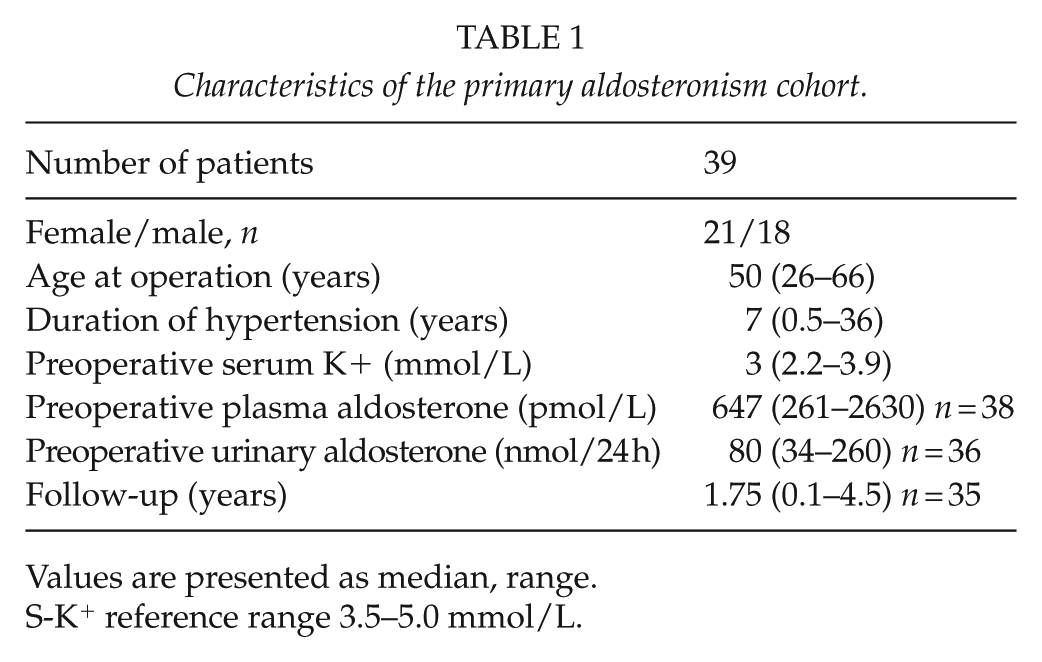
Values are presented as median, range.
S-K+ reference range 3.5–5.0 mmol/L.
The PA diagnosis was established following the Endocrine Society guidelines (15). Patients were investigated as far as possible without medication interfering with the renin–angiotensin–aldosterone system. A positive screening test, that is, an elevated aldosterone to renin ratio, was followed by a confirmatory test, either with the measurement of urinary aldosterone after oral salt load or with a suppression test with intravenous saline or fludrocortisone. PA was diagnosed either at a urinary aldosterone level >60 mmol/24 h together with urinary sodium >200 mmol/24 h or after a fludrocortisone suppression test, urine aldosterone >35 mmol/24 h with urinary sodium >200 mmol/24 h or P-aldosterone 220 pmol/L. Further investigation included a computed tomographic (CT) scan of the adrenals. Lateralization of the aldosterone production was assessed with adrenal vein sampling (AVS), which was performed with sequential catheterization without ACTH stimulation. The selectivity index, that is, the ratio between the adrenal and the peripheral cortisol values, was 3. The lateralization index, that is, the ratio between the aldosterone to cortisol ratios from the right and the left side, was above 4 except in three patients where AVS was not optimal. All patients including these three were discussed at a multidisciplinary conference before the decision on adrenalectomy. At postoperative follow-up, we used the PASO criteria to evaluate the outcome (14). The PASO criteria are standardized outcome criteria, suggested to be applied in the assessment of success of adrenalectomy for PA at follow-up. Both biochemical cure (cure from PA) and clinical cure (cure from hypertension) are evaluated. The study has been approved by the ethical committee of Karolinska Institutet, and consent was obtained from all patients.
Histopathology
All adrenals were examined grossly by an experienced endocrine pathologist and subsequently routine fixated in formalin and processed for further microscopic examination using paraffin-embedment. Slides were stained in hematoxylin–eosin for routine evaluation, and visualized adrenocortical nodules were investigated further using CYP11B1 and B2 IHC. For 33 samples, the IHC was retrospectively performed as a part of a re-investigation of previous pathology reports (without CYP11B1/2 antibodies), whereas for six cases, the stainings were performed as a part of the primary evaluation. For the 33 cases re-analyzed with CYP11B1 and B2 with a previous diagnosis based on routine H&E sections only, the pathologist (C.C.J.) was not blinded—as he was aware of the original report when conducting his second opinion analyses—which was necessary for selecting the appropriate sections for IHC.
IHC was performed using monoclonal CYP11B1 and CYP11B2 rat and mouse antibodies, respectively, in the routine pathology laboratory setting using an automated Ventana Benchmark Ultra system (Ventana Medical Systems, Tucson, AZ, USA). Both antibodies (clones hCYP11B1-80-2-2 and hCYP11B2-41-17B) were kindly provided by Dr Carolina Velarde Miranda, University of Mississippi Medical Center, University of Alabama (Birmingham, AL, USA) and have been previously characterized (9). Later badges of the antibodies were bought from Merck Millipore (Burlington, MA, USA). We performed several control experiments with anonymized APA and cortisol-producing adenoma before a final protocol for our Ventana system was applied. Antigen retrieval using citrate buffer was performed for 72 min. Primary antibody concentrations were 1:50 and 1:500 for CYP11B1 and CYP11B2, respectively, and the primary antibodies were incubated for 20 and 32 min, respectively. The DAB OptiView detection system was used for both antibodies. Since the CYP11B1 antibody is derived from rat, we relied on cross-reactivity from the secondary anti-mouse antibody included in the OptiView kit. As of this, we applied a specific amplification step for this antibody (OptiView Amplification Kit, Ventana Medical Systems) to obtain sufficient signal in our control material (data not shown).
Routine H&E slides as well as slides from immunohistochemical analyses were scanned. Digital slide scanning was used to get the best low-power magnification and overview and to be able to compare the H&E-stained slides and the CYP11B1 and CYP11B2 stainings. Stained slides were scanned at ×20 magnification using a Hamamatsu NanoZoomer XR equipped with an objective ×20/0.17. The viewing software used was NDP, NanoZoomer Digital Pathology, Version 1.1.27. Evaluation for all cases was done in consensus both by light microscopy and the computer, including the six cases who were diagnosed in clinical routine (Table 2).
Diagnosis and cure in patients operated for primary aldosteronism.
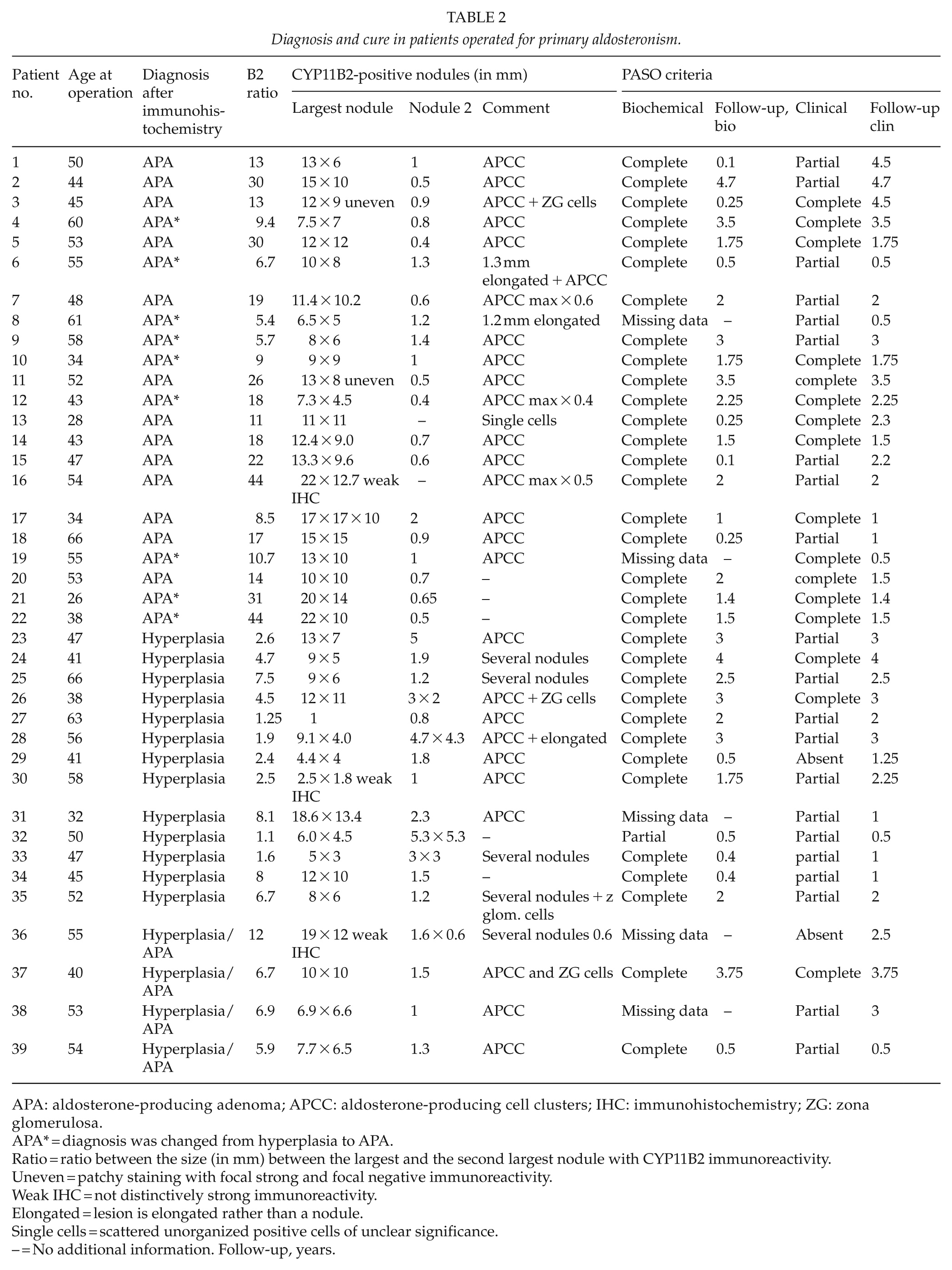
APA: aldosterone-producing adenoma; APCC: aldosterone-producing cell clusters; IHC: immunohistochemistry; ZG: zona glomerulosa.
APA* = diagnosis was changed from hyperplasia to APA.
Ratio = ratio between the size (in mm) between the largest and the second largest nodule with CYP11B2 immunoreactivity.
Uneven = patchy staining with focal strong and focal negative immunoreactivity.
Weak IHC = not distinctively strong immunoreactivity.
Elongated = lesion is elongated rather than a nodule.
Single cells = scattered unorganized positive cells of unclear significance.
– = No additional information. Follow-up, years.
The diagnosis of an APA was established when a dominating tumor showed strong immunoreactivity for CYP11B2, and only small cell clusters in the remaining parts of the adrenal were CYP11B2 positive (denoted as aldosterone-producing cell clusters (APCCs)). A subset of cases was annotated with a “weak” staining if the nodules were heterogeneously stained with variable immunoreactivity. We defined APCC as small, nodular CYP11B2-positive lesions, usually below 1 mm in size, and not visible with routine histological investigation.
The hyperplasia diagnosis was suggested when there was no dominating nodule but immunoreactivity for CYP11B2 in several nodules which were also seen on routine sections. In an attempt to distinguish between adenoma and hyperplasia, a ratio between the largest and the second largest CYP11B2-positive nodules (termed “B2 ratio”) was calculated, to study whether there exists a ratio for the nodule size that clearly defines adenomas with associated APCC from bona fide hyperplasias.
Statistical Analyses
Statistical analyses were made with IBM SPSS Statistics 24 (IBM, Armonk, NY, USA). Non-normal distribution was assumed for all data, applying the Kruskal–Wallis and Mann–Whitney U tests for comparison between groups. p < 0.05 was considered as statistically significant.
Results
General Characteristics
The cohort consisted of 18 male and 21 female patients with an age span of 26–66 years (Table 1). All patients had a diagnosis of PA and had a history of hypertension of median 7 years (range 0.5–36). The median follow-up time was 1.75 years (range 0.1–4.5). Individual follow-up data are shown in Table 2 and are presented according to the PASO criteria (14). It was unfortunately not possible to get a complete follow-up according to PASO criteria on all patients and available follow-up data are shown in Table 2.
Histopathological Evaluation
The histopathology and IHC data are shown in Table 2 and Figs 1 to 4. Using IHC, the final diagnosis was changed in 9 of 33 patients (Table 2). All nine patients with an altered diagnosis were initially judged to have hyperplasia based on routine histology alone, whereas the interpretation after CYP11B2 IHC changed to APA. No cases initially diagnosed as an adenoma were re-classified as hyperplasia. Of these nine cases with changed diagnosis from hyperplasia to adenoma, seven had a complete biochemical response. For two cases, these data were missing.
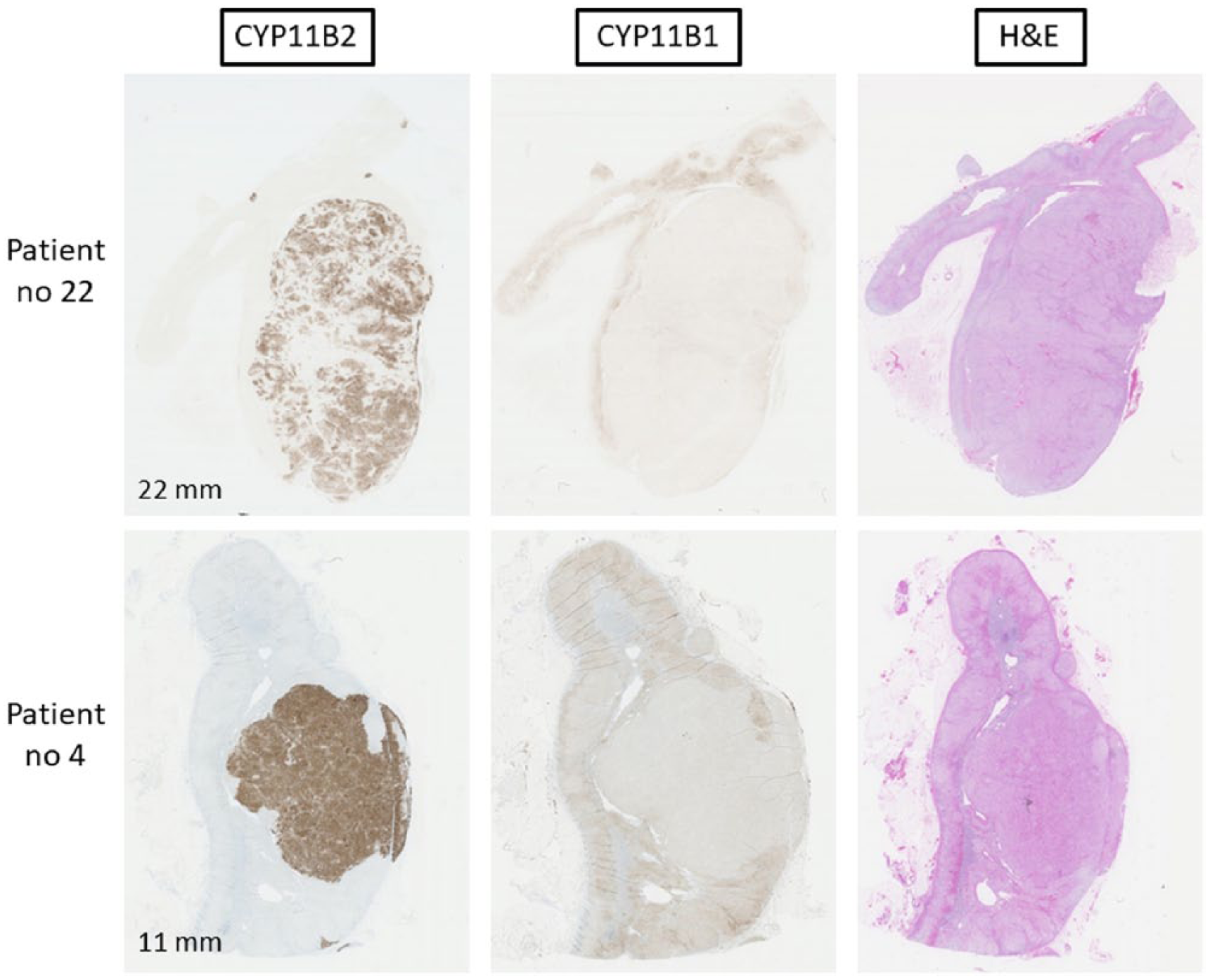
Adrenals with aldosterone-producing adenoma (APA) in patients 22 and 4. Sections were processed for immunohistochemistry using antibodies specific for CYP11B2 and B1 as well as stained with hematoxylin–eosin (H&E). The largest diameter in mm is shown.
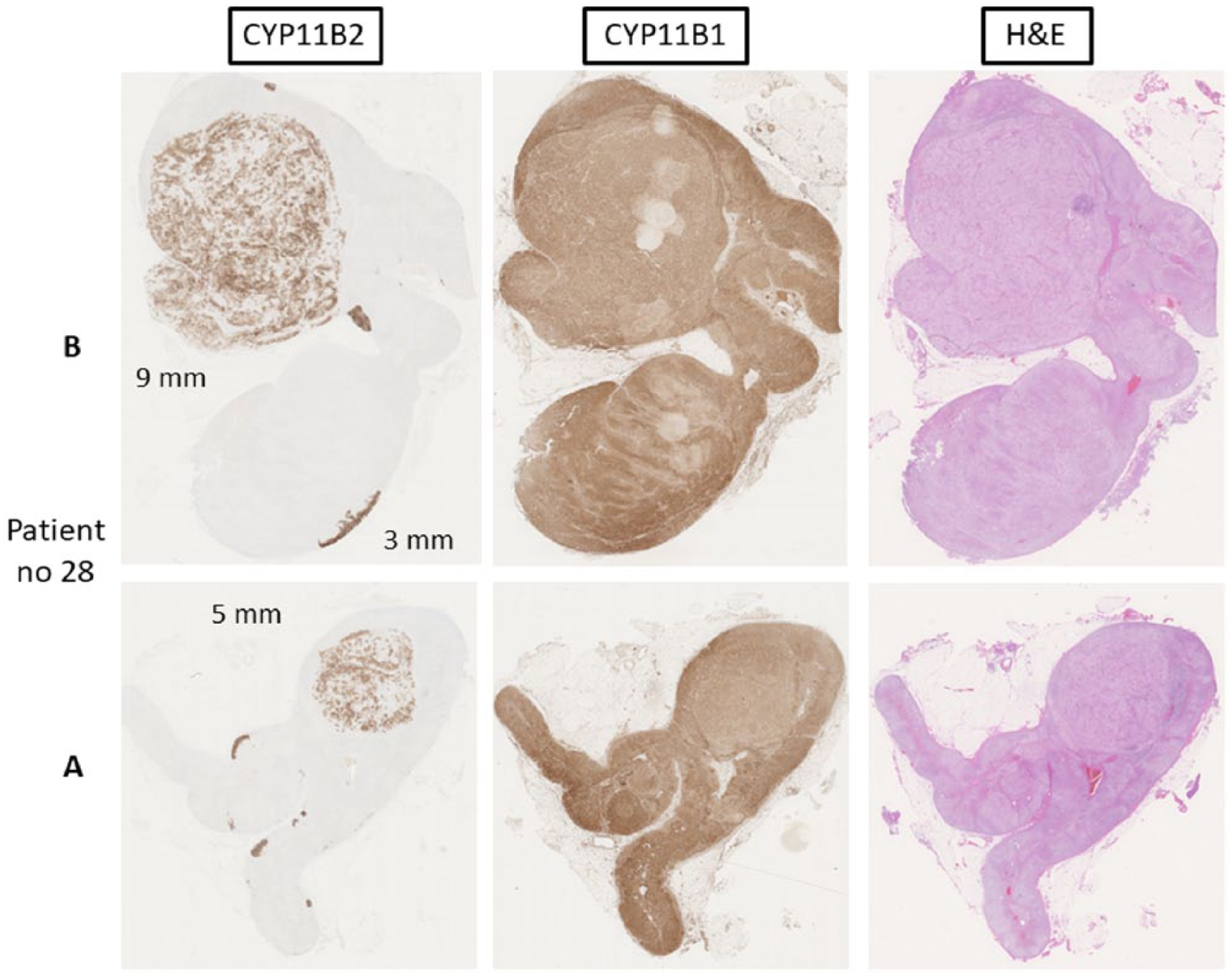
Adrenal with aldosterone-producing hyperplasia from patient 28. Sections were processed for immunohistochemistry using antibodies specific for CYP11B2 and B1 as well as stained with hematoxylin–eosin (H&E). Sections are from different parts of the same adrenal gland (A and B) and the nodular sizes in mm are shown.
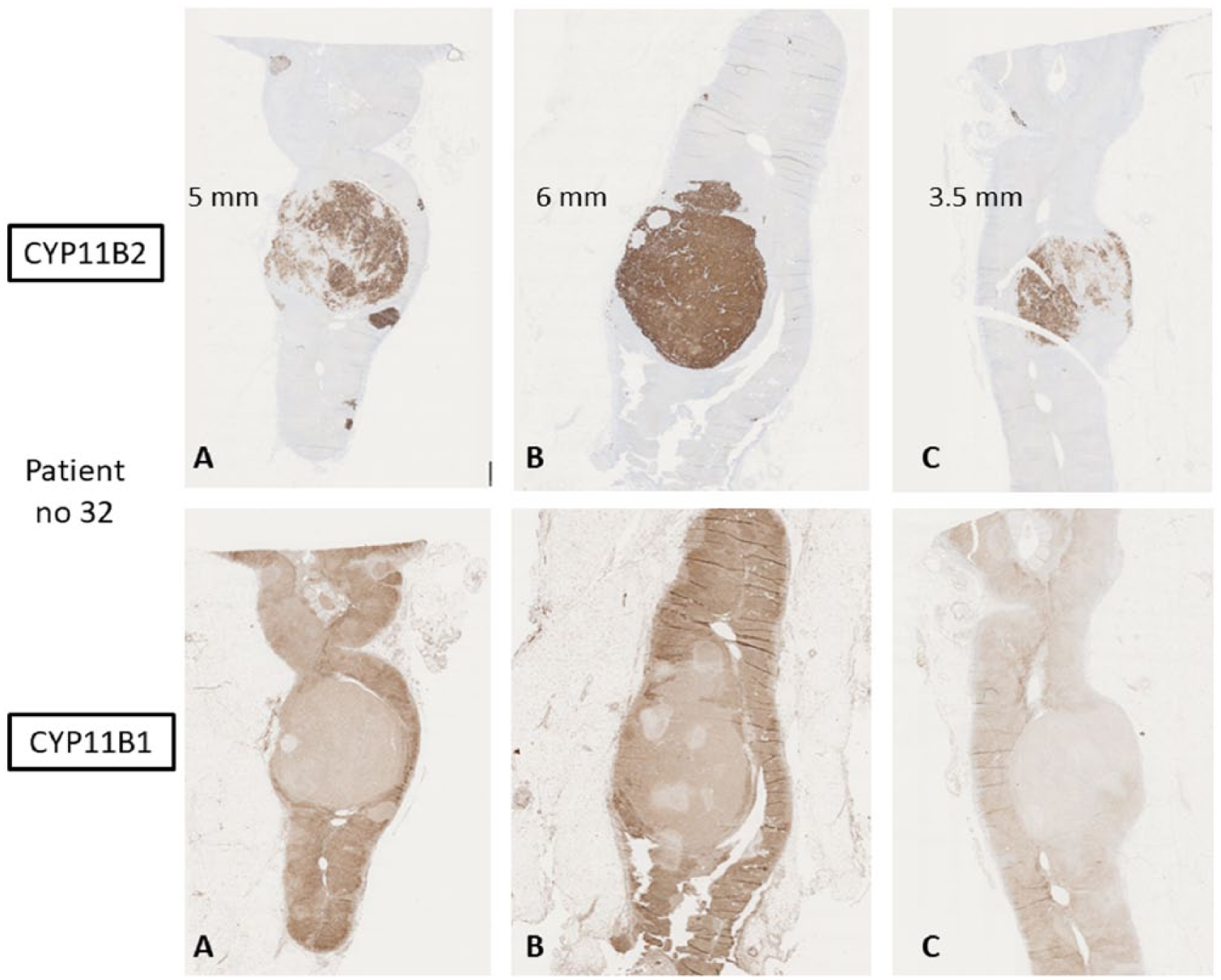
Adrenal with aldosterone-producing hyperplasia from patient 32. Sections were processed for immunohistochemistry using antibodies specific for CYP11B2 and B1 as well as stained with hematoxylin–eosin (H&E). Sections are from different parts of the same adrenal gland (A, B, and C) and the nodular sizes in mm are shown.
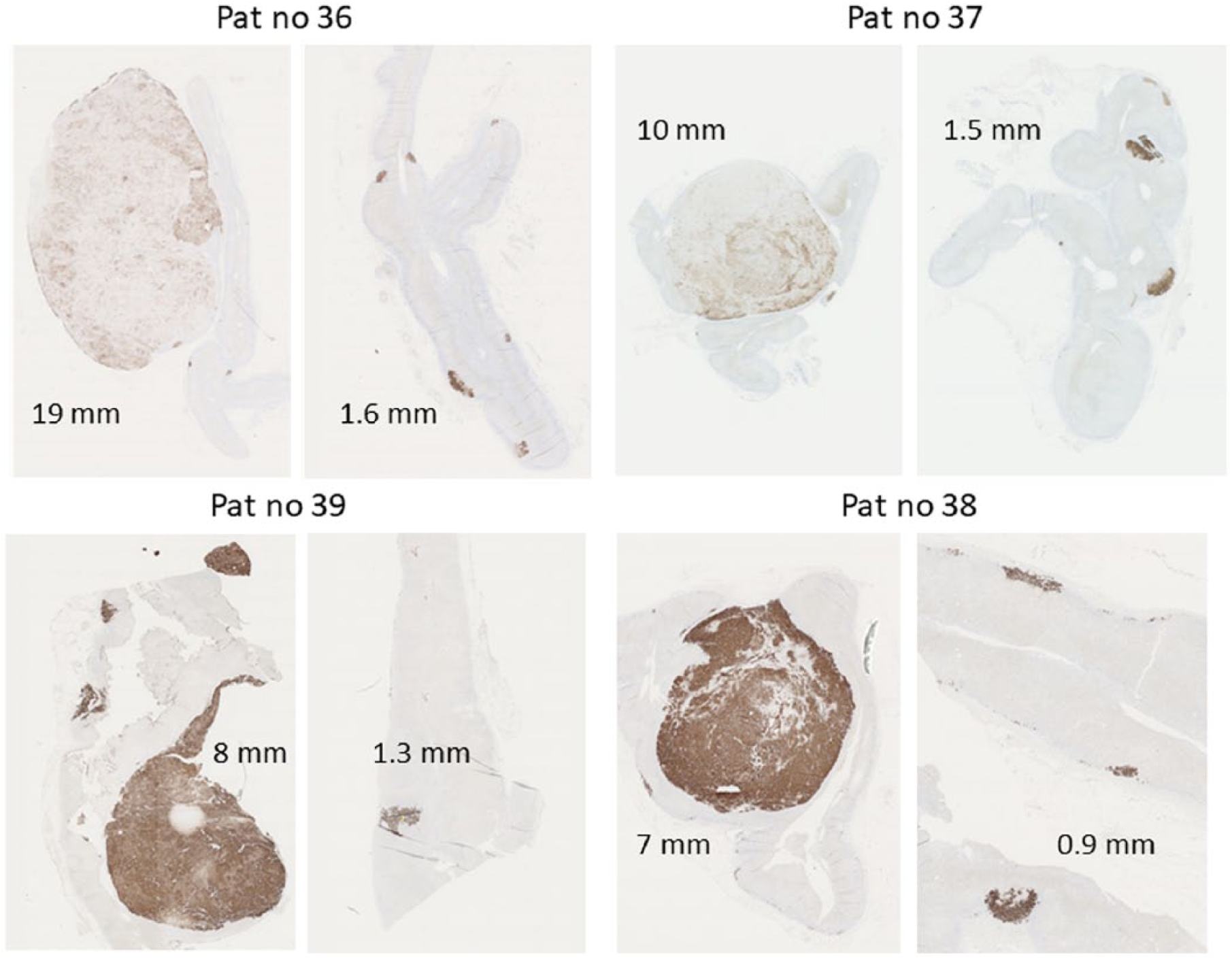
Adrenals from patient 36, 37, 38, and 39. Sections were processed for immunohistochemistry using an antibody specific for CYP11B2. Nodular sizes in mm are shown. It was not possible to safely assign these lesions to either the adenoma or hyperplasia category due to weak staining and/or the small sizes.
In the majority of cases, it was obvious whether the lesions should be classified as adenoma or hyperplasia as adenomas were identified by single lesions with distinct CYP11B2 immunoreactivity, whereas hyperplasias stained positive for CYP11B2 in several nodules. However, in four equivocal cases, it was difficult to decide on a definitive diagnosis (Fig. 4).
In addition to the morphological examination, the ratio between the largest and the second largest CYP11B2-positive lesions was calculated (Table 2). This B2 ratio range for adrenals judged to have hyperplasia was 1.1 to 8.1 (median 2.6). Adenomas had a range between 5.4 and 44 (median 15.5). The four equivocal cases exhibited either a major nodule with only partial or weak CYP11B2 immunoreactivity, alternatively the B2 ratio between the largest and the second largest nodules was fairly low, between 5.9 and 12 (median 6.8), making the distinction between adenoma and hyperplasia difficult.
Overall, the B2 ratio was significantly higher in the adenoma group versus the hyperplasia and equivocal case groups (Kruskal–Wallis test, p < 0.0001, Supplemental Figure 1), which was further supported by a direct comparison between adenomas and hyperplasias (Mann–Whitney U test, p < 0.0001). APCCs were detected in 25 cases, in 16/20 adenomas, in 6/13 hyperplasias and in 3/4 equivocal cases.
Correlation To Patient Follow-Up
Of the 22 patients who were diagnosed with APA after IHC, 20 displayed complete biochemical cure (and 2 cases with missing data) according to the PASO criteria. When consulting PASO for clinical cure, 13 were judged as having complete clinical cure and 9 as having partial clinical cure. In the hyperplasia group (n = 13), 11 displayed complete biochemical cure and 1 showed only partial biochemical cure (as well as 1 case with missing data). When considering clinical cure, 2 cases displayed complete cure, 10 cases presented with partial cure, and 1 case showed signs of absent cure.
When comparing number of cases with complete biochemical cure (compared to partial or absent cure) in the two groups, the difference between adenomas and hyperplasias was non-significant (p = 0.375, Fisher’s exact test). However, when considering clinical cure, the difference between complete and non-complete cure was significant (p = 0.0158, Fisher’s exact test), indicating that the CYP11B2-verified adenoma patients display complete clinical cure to a larger extent than the CYP11B2-verified hyperplasia patients, which therefore supports our diagnostic approach.
If the same analyses were to be performed using the “original” diagnoses (i.e. 9 adenomas in Table 2 would be thought of as hyperplasias based on the original pathology reports lacking CYP11B2 data), the cohort would consist of 13 adenomas and 22 hyperplasias. In this aspect, seven adenomas would display complete clinical cure and six cases would show partial clinical cure. In contrast, eight hyperplasias would display complete clinical cure, 13 cases would show partial clinical cure and 1 case absence of clinical cure. This difference between complete and non-complete cure would not reach statistical significance (p = 0.481, Fisher’s exact test). These results combined indicate that the CYP11B2 IHC correctly re-allocated a number of hyperplasias to the adenoma group, as indicated by the improved clinical cure rates in the immunohistochemically rectified adenoma group compared to the routine H&E-diagnosed cohort.
Correlation To Preoperative Parameters
Preoperative CT-appreciated sizes of the largest nodule per case (in millimeters) were retrieved. Information was found for all cases; however, a few cases were denoted as “0 mm” based on the lack of radiological findings, and for case 27, a 10-mm nodule was found on the contralateral adrenal, whereas the AVS directed surgery toward the adrenal without a radiologic finding, and therefore this case was excluded from calculations. In the “original” cohort (reassigning nine adenomas as hyperplasias), the adenomas displayed size range between 6 and 23 mm, average size 17.5, median 20, whereas the hyperplasias showed a range of 0–15 mm, average 8.6, median 10. In the immunohistochemically stratified cohort (meaning, cases with acquired definite diagnoses after the CYP11B2 analyses), the adenomas were found to have a size range between 0 and 24 mm, average size 15.3, median 16, whereas the hyperplasias displayed a size range between 0 and 15 mm, average 8.1, median 9.5. The CT-appreciated lesion sizes did as expected differ significantly between adenoma and hyperplasia in both the “original” and immunohistochemically stratified cohorts (Mann–Whitney U, p = 0.0001 and p = 0.003, respectively).
The AVS lateralization index for adenomas diagnosed histologically without CYP11B2 IHC displayed a range between 2.3 and 50, median 33, and average 32. Hyperplasias showed a range of 3–61, median 14.5, and average 23. There was no statistically significant difference between adenoma and hyperplasia (Mann–Whitney U, p = 0.719). The index for adenomas after specific IHC (CYP11B2) displayed a range of 3–60, median 27, average 29, whereas hyperplasias showed a range of 4–61, median 16, and average 28. There was no statistically significant index difference between adenoma and hyperplasia after reclassification according to CYP11B2 IHC (Mann–Whitney U, p = 0.624).
B2 Ratio As A Clinical Marker
If a B2 ratio cut-off of >8.1 is applied for the proper detection of adenoma (since 8.1 was the highest B2 ratio detected in the hyperplasia group), a sensitivity of 86% and a specificity of 100% would be obtained, and the positive predictive value (PPV) of a B2 ratio of >8.1 would be 100%, whereas the negative predictive value would be 81%. Moreover, if a B2 ratio cut-off of ⩾5.4 was applied (thereby including all adenomas in the cohort), the sensitivity and specificity would be 100% and 69%, respectively, and the PPV and NPV would be 85% and 100%, respectively.
This would mean that a B2 ratio cut-off of >8.1 would have an optimal PPV, while a cut-off at ⩾5.4 would exhibit a perfect NPV. Exemplified, this would mean that a B2 ratio of >8.1 would strongly suggest an adenoma, whereas lesions with a B2 ratio below 5.4 would constitute hyperplasia.
A receiver operating characteristic (ROC) curve for the B2 ratios to discriminate between adenoma and hyperplasia was obtained (Supplemental Figure 2). Area under the curve (AUC) was 0.96 (0.903–1, 95% confidence interval), indicating a well-designed marker for the distinction between these two entities.
Discussion
In this study, patients with PA operated during a 4-year period were evaluated. Of the 39 patients, 22 had an APA, while 13 were judged to have hyperplasia, presumably as part of BAH. IHC contributed to more precise diagnoses of APA and BAH and also made it possible to change the diagnosis from hyperplasia to adenoma in nine patients as only one of several nodules in these adrenals were positive for CYP11B2. We have also determined an IHC-derived B2 ratio which can aid in the final diagnosis of these lesions, as this ratio was significantly greater in adenoma compared to hyperplasia.
In a previous study, we analyzed 120 patients with PA (16), in which functional histopathology including in situ hybridization (8, 10) and polyclonal antibodies was helpful in distinguishing between APA and hyperplasia. Long-term follow-up showed that almost all patients with APA were cured, while patients with hyperplasia had a cure rate of about 62 % (16). With the advent of more specific monoclonal antibodies for the final steps in aldosterone and cortisol synthesis (9), we could now study the role of IHC in the routine histopathological evaluation of adrenals from patients with PA. As it can be difficult to make a safe diagnosis dependent on routine H&E staining only, IHC now makes it possible to identify the enzyme for aldosterone production in each individual lesion.
It seems safe to conclude that the application of these antibodies to the clinical routine will provide a more accurate diagnosis for a large proportion of patients—and probably also discharge them from further surveillance, as adenoma patients are generally cured by surgery alone (16). Indeed, the CYP11B2 IHC appropriately re-allocated a number of hyperplasias to the adenoma group, as shown by the enhanced clinical cure rates in the immunohistochemically corrected adenoma group compared to the routine H&E-diagnosed cohort.
Most diagnostic studies on adrenals without CYP11B2 IHC show that almost all patients display an adenoma and also advocate that operating on hyperplasia is not intentional. When using IHC in a consecutive material as in the work by Volpe et al. (16) and this study, the hyperplasia percentage is in the range of 15%–37%. There is no other consecutive clinical material with IHC to compare with. As further discussed below, it seems beneficial to operate also on patients with hyperplasia.
The equivocal cases (n = 4) constituted a minor subset of cases in our study; nevertheless, we felt the necessity to classify these cases to a specific subgroup with questionable immunohistochemical results, ambiguous B2 ratios, and mixed clinical outcomes. Although discussed internally at our department, no clear consensus regarding their definite diagnosis could be made. When consulting the PASO criteria for clinical cure, only one out of four cases displayed evidence of complete cure, whereas two showed only partial cure and one case showed absence of cure. This group therefore probably contains a mix between adenomas and hyperplasias, and the correct way of handling these cases from a clinical perspective is not known. We therefore advocate that cases with equivocal CYP11B2 staining results should be followed in the same way as bona fide hyperplasia patients.
Since the interpretation of CYP11B1 and B2 IHC was not always straightforward (not least given the equivocal cases), we also included information on the ratio between the largest and the second largest nodules to obtain a numeric difference between adenoma and hyperplasia (B2 ratio). The hypothesis was that a ratio between the different CYP11B2-positive nodules based on size could establish cut-offs for adenomas and hyperplasias. In our material, adenomas displayed significantly higher B2 ratios than hyperplasias—as adenomas usually had a ratio above 9 (median 15.5) and hyperplasia below 5 (median 2.6), but cases in the intermediate interval provided a challenge to establish a final diagnosis (median 6.8). Also, cases with very weak CYP11B2 staining were challenging in this aspect. This was exemplified in four patients (Fig. 4) in which we were not able to establish a definite diagnosis of APA or BAH. This was due to the presence of a small primary CYP11B2-positive nodule or very weak CYP11B2 activity in the largest lesion. The difficulty to safely separate APA and hyperplasia may be due to the fact that APA may possibly develop from hyperplasia (13, 17) as one nodule increases in size and becomes dominant. Genetic studies suggest that nodules in hyperplasia may have a similar spectrum of mutations as APA (18).
Almost all patients had a complete biochemical cure according to the PASO criteria (14). The clinical cure rate was also high, but more patients were completely cured in the adenoma group compared to the hyperplasia group. As these patients were operated during 2013–2016, the follow-up time was short (Table 2) and some patients did not reach the PASO criteria of 6 months. Due to the short follow-up, it was not possible to detect recurrence after initial cure. This constitutes a limitation to this study as well as the small patient cohort.
When the diagnosis of PA had been established, all our patients underwent workup with CT/magnetic resonance imaging (MRI) and AVS. The final decision to select side for the operation was mainly based on the AVS results even if radiology showed no lesion or a lesion on the contralateral side. This may explain why 13 of the 39 adrenals were judged to have hyperplasia, although the traditional opinion has been to treat patients with hyperplasia medically. It seems reasonable to conclude that many patients have an asymmetrical hyperplasia (13) and that these patients have a beneficial effect of adrenalectomy, although potentially for a limited period of time.
The identification of somatic mutations in genes involved in protein formation in ion channels in adrenocortical cells (19) has shed light over the development of adenomas in primary aldosteronism. More than half of APAs seem to harbor a mutation in some of these genes (20). Subsequently, it was postulated that the identification of such driver mutations in adrenals from operated PA patients would prove the lesion to be an adenoma and not hyperplasia. However, the picture has become much more complicated, with mutations found in transitional lesions and APCC (21, 22), in hyperplasia (23), and even in normal adrenal glands (12). Hence, the role for mutational analysis of adrenal lesions in establishing a final diagnosis is yet to be established.
Our results confirm previous findings that almost all patients with APA have a various number of small cell clusters, APCC, positive for CYP11B2. Only in a few cases, CYP11B2-positive cells could be seen in a histologically normal zona glomerulosa (12). Moreover, no diffuse general hyperplasia was found in our material. Interestingly, the zona glomerulosa rearranges with age, and in adults, there is usually not a continuous layer of aldosterone-producing cells (24) but mainly APCC scattered in the outer part of the cortex.
In conclusion, IHC with specific antibodies for the final step in aldosterone and cortisol syntheses could provide a considerable aid in establishing a correct final histopathological diagnosis. Moreover, the establishment of a B2 ratio seems to be helpful when discriminating between adenomas and hyperplasias. We suggest that these procedures should be considered to be used in routine histopathological examination in adrenals from PA patients, as a safe final diagnosis is of great help in the long-term follow-up of PA patients.
Supplemental Material
Supplementary_figure_1 – Supplemental material for Impact of immunohistochemistry on the diagnosis and management of primary aldosteronism: An important tool for improved patient follow-up
Supplemental material, Supplementary_figure_1 for Impact of immunohistochemistry on the diagnosis and management of primary aldosteronism: An important tool for improved patient follow-up by C. Volpe, B. Hamberger, J. Zedenius and C. C. Juhlin in Scandinavian Journal of Surgery
Supplemental Material
Supplementary_Figure_2 – Supplemental material for Impact of immunohistochemistry on the diagnosis and management of primary aldosteronism: An important tool for improved patient follow-up
Supplemental material, Supplementary_Figure_2 for Impact of immunohistochemistry on the diagnosis and management of primary aldosteronism: An important tool for improved patient follow-up by C. Volpe, B. Hamberger, J. Zedenius and C. C. Juhlin in Scandinavian Journal of Surgery
Footnotes
Acknowledgements
The authors thank Dr Anders Höög for aiding in the evaluation of histopathology as well as managing scanning of all slides. Dr Jan Calissendorff has been of great help with endocrinological evaluation, and Ms Lisa Ånfalk has been of invaluable help in allocating all tissue specimens and organizing the histopathology analyses. They also thank Dr Adam Stenman and Dr Ninni Mu for statistical calculations and interpretation.
Declaration Of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study has been approved by the Ethical committee of Karolinska Institutet.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was financially supported by grants generously provided through The Cancer Research Funds of Radiumhemmet and the Swedish Medical Association.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
