Abstract
Introduction:
Patients experiencing thermal injuries with an extent of over 20% of total body surface area suffer from systemic catabolic disease. The thermal trauma-induced loss of muscle mass causes a higher incidence for comorbidities and subsequently a higher mortality. In this study, we aimed to investigate the role of myostatin in the interplay with follistatin during muscle cachexia.
Methods:
Patients with burn injuries (>10% total body surface area) between the ages of 18 and 75 were prospectively included within the first 48 h after trauma to determine deviations of parameters connected to muscle catabolism. In the chronic state of burn injury (9–12 months after trauma), we re-evaluated myostatin and follistatin concentrations as well as muscle strength of the non-dominant forearm.
Results:
We were able to show a time-dependent alteration (9–12 months after burn injury) of myostatin with an initial decrease (p < 0.001) and long-term increase (p < 0.001) after thermal injury in blood serum. For follistatin, a reciprocal correlation was observed (r = −0.707, p = 0.001). Accordingly, muscle strength of the non-dominant hand and forearm was significantly decreased 9–12 months after injury in post-burn patients compared with healthy patients with a significant correlation to myostatin levels (r = −0.899, p < 0.001). In addition, initial myostatin serum concentration was predictive for long-term muscle strength impairment.
Conclusion:
With regard to the muscle metabolism after thermal trauma, our data suggest an acute anabolic response, presumably to spare muscle mass, which is converted to catabolic conditions accompanied by muscle strength reduction in the chronic phase. Myostatin plays a crucial role in this orchestration and initial myostatin concentration may predict the long-term muscle strength.
Introduction
Thermal injuries have complex pathological effects influencing body functions, even immediately after trauma. Burn victims are subject to multifactorial damage as a result from injuries affecting more than 20% of the total body surface area (TBSA). The capillary leak syndrome due to a sudden release of vasoactive mediators and the loss of skin integrity are responsible for the vast majority of burn disease damage (1). However, burn injury also results in a chronic inflammatory, hypermetabolic, and hypercatabolic condition affecting muscle mass in burn victims. In animal and human studies, a loss of muscle mass by 10%–20% is reported (2, 3). This loss of lean body mass is harmful both in the acute and the convalescent phase of injury. Even a 10% loss of lean body mass significantly increases infections rates and delays wound healing (4). Furthermore, the associated muscle weakness can prolong artificial ventilation and delay mobilization. Taken together, these physical impairments significantly increase the potential for pulmonary sepsis and mortality in severe burn patients (5).
Although muscle is not directly affected by burn injury, secondary mechanisms leading to muscle atrophy are complex. Insulin-like growth factor 1 (IGF-1) concentration is reported to be reduced in post-burn situations as in most catabolic situations. Immediate systemic substitution of IGF-1 alleviates muscle loss. The underlying pathophysiological pathway, circumstances of injury, and distance from the wound to the muscle determines the proportionate role of different pathways (6).
Certain strategies are elaborated to indirectly modulate muscle wasting after burn injury. For instance, in the acute phase, early wound coverage can reduce metabolic rate impairment and protein loss. An early mobilization can also reduce muscle wasting in burn patients and is one leading strategy against inactivity-induced muscle wasting. There is evidence that early mobilization reduces mortality in patients with burn injuries (7). In contrast, sepsis in acute burn patients elevates the metabolic rate by 1.5×.
In order to counteract these phenomena, pharmacological interventions aim to induce muscle anabolism. One possible strategy is the use of more selective steroids as oxandrolone, which is proven to reduce muscle mass loss in burn injury (8). Administration of recombinant growth hormone is discussed to increase serum concentrations of its mediator IGF-1, improves muscle protein kinetics, maintains muscular growth, and decreases donor site healing time significantly (9). However application of the recombinant growth hormone is accompanied by severe side effects, such as hyperglycemia and increased mortality, in the cohort of critically ill patients who have not sustained burn injuries (10). The secondary mediator of recombinant growth hormone IGF-1 shows similar effects without the adverse side effect of hyperglycemia (11). Insulin, as a substitute in acute thermal injuries, prevents muscle catabolism but bears the risk of a hypoglycaemic crisis. Other strategies to reduce muscle catabolism are anabolic steroids and catecholamine antagonists (3).
One of the most promising targets to alter muscle catabolism toward anabolism may be the activin type II receptor (12). Activin type II receptors modulate signals for proteins belonging to the transforming growth factor beta superfamily. Myostatin is a member of the tumor growth factor (TGF-β) superfamily inhibiting excessive muscle growth by binding to activin type II (13). In muscle cells myostatin promotes protein degradation and acts as a main antagonist of muscle protein synthesis alongside recently discovered growth differentiation factor 11 (GDF 11) (14). Furthermore, myostatin plays a crucial role in cancerogenesis and bone metabolism (15, 16). By inhibiting protein kinase Akt, myostatin negatively interacts with the IGF-1 pathway in muscle cells.
A natural antagonist of myostatin as well as the activin type II receptor is follistatin. It is involved in proliferation, differentiation, and apoptosis, depending on the cell type through blockade of access to both the type I and type II receptor binding sites as well as through blocking the specific activin type II receptor binding protein (e.g. myostatin) directly (17). Follistatin as a natural antagonist of myostatin primarily leads to phenotypic muscle gain. In previous studies, myostatin was closely linked with cachexia in cancer patients, while follistatin was reported not to be dysregulated (18). Nonetheless, activin A as activating binding protein of follistatin is a prognosis factor of survival in cachectic cancer patients (19). To date, data related to myostatin as a regulator of muscle cachexia after thermal injury are rare. In this study, we hypothesize that myostatin plays a central role in muscle cachexia after thermal injury. Moreover, lack of follistatin may contribute to muscle catabolism in post-burn patients. Thus, we aim to investigate dysregulations of myostatin and follistatin in burn patients and subsequent implications for muscle catabolism.
Materials and Methods
Patient Recruitment
Serum harvest and experiments were performed prospectively in accordance and with the ethical committee of the Ruhr University Bochum after approval between September 2015 and August 2017 (approval number: 16-5619-BR). Informed consent was obtained from male patients only to rule out bias due to hormonal variations. Male patients with burn injuries (>10% TBSA) but no direct muscle involvement between the ages of 18 and 75 were included in the patient group within the first 48 h after injury. All patients with severe burn injuries admitted to our institution were asked to be included into the study between 2015 and 2017. In the control group, male volunteers between the ages of 18 and 75 without any hormonal, acute, malignant, infection or autoimmune disease were included. A body mass index between 18 and 30 was set as inclusion criteria for burn and control patients. To measure long-term (9–12 months after burn injury) effects of severe burns on myostatin concentration and muscle strength, serum was collected from identical patients (same patients as in the acute phase testing) between 9 and 12 months after burn injury. Two patients of the acute phase testing passed away and chronic values were only obtained from 13 patients. After acquisition of informed consent, 7.5 mL of full blood was collected into serum tubes. After centrifugation with 3000 r/min for 10 min, serum was obtained.
Enzyme-Linked Immunosorbent Assay
(ELISA) for GDF-8 (myostatin) and follistatin was performed according to the manufacturer’s protocol (PeproTech, Hamburg, Germany). Briefly, after adding 50 µL of sample, control, and standard into the well plate, incubation for 2 h at room temperature on a microplate shaker with 500 r/min was initiated. After a washing procedure, all remaining wash buffer was removed. GDF-8 and follistatin conjugate were added accordingly. After a washing step, substrate solution was added and incubated for another 30 min at room temperature. Stop solution was added and optical density of each well was measured using a microplate reader set to 450 nm wavelength.
Strength Measurements
To facilitate standardized strength measurements, the patient’s grip strength was tested for male patients 9–12 months after burn injury with a Jamar® Hydraulic Hand Dynamometer at level 2 on the non-dominant hand. None of the patients had a burn injury of the non-dominant forearm. Two patients had 2a° burn injury spots on the non-dominant upper arm but did not show differences in strength measurement compared with similar patients. Pinch grip and lateral grip testing was performed accordingly on a Baseline® Pinch Gauge. As a control population, male subjects between the ages of 18 and 75 without any hand injury of the non-dominant hand were selected.
Statistical Analysis
The achieved power (1-β) of the analysis was calculated with the assistance of the G-Power® software (Version 3.1.9.2). An effect size of 0.90 for was calculated by the difference between two independent means and was entered in the analysis with a type I error probability of 0.05. The estimated achieved power was 80%. Statistical significance was assessed by Student’s t-test or by Mann–Whitney test, according to data distribution. Statistical significances were set at a p value < 0.05. Calculation of significant correlation was done with Pearson’s test. All analyses were performed using SPSS Statistics for Windows software (Version 21.0; IBM, Armonk, USA), considering a critical significance value of 5%.
Results
Patients were separated into three groups: control population, acute burn patients (admission as severe burn patients between 48 h after burn injury), and chronic burn patients (9–12 months after severe burn injury; same patients as in the acute phase, two patients passed away in the acute phase). Age, body mass index, and gender of the patients were not significantly different, while creatine kinase, lactate dehydrogenase, myostatin, and follistatin exhibited a highly significant difference between the control and burn patients. The acute burn group consisted of five patients with deflagration and subsequent flame burn, eight patients with flame burns, and two patients with scald burns. None of the patients had a burn injury of the non-dominant forearm. Acute burn patients revealed an increase of myostatin and decrease of follistatin serum levels compared with the control population, while chronic burn patients showed a decrease of myostatin with a simultaneous decrease of follistatin (Table 1, Fig. 1).
Age, total burn surface area (TBSA), and laboratory chemistry of control patients and severe burn patients within the first 48 h after injury.
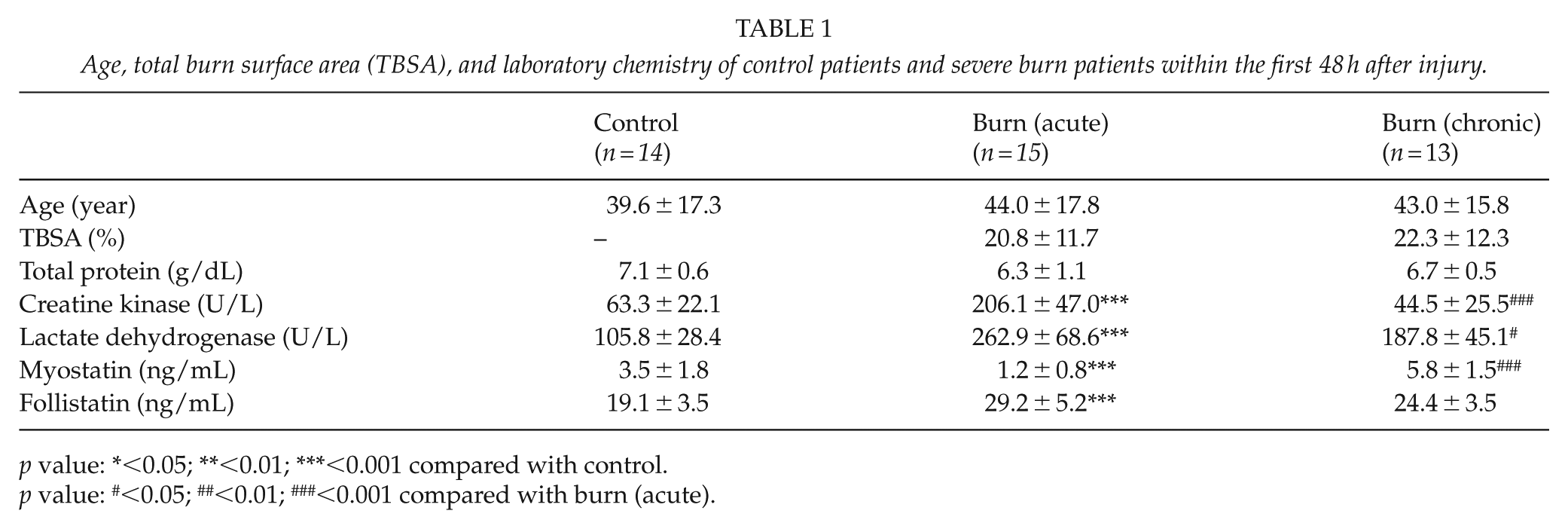
p value: *<0.05; **<0.01; ***<0.001 compared with control.
p value: #<0.05; ##<0.01; ###<0.001 compared with burn (acute).
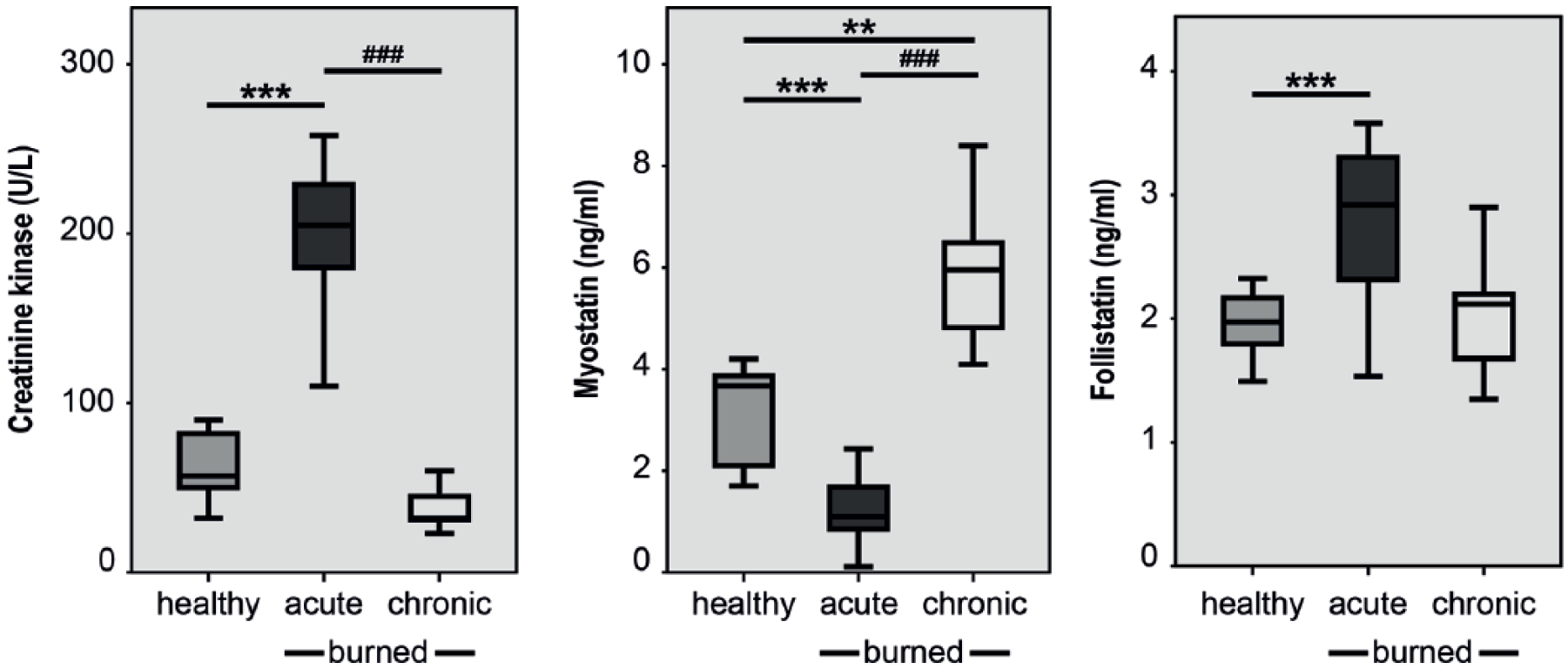
An elevation of serum creatine kinases was observed in acute burn patients. In acute burn patients, a highly significant decrease of myostatin compared with the control population was observed, while chronic burn patients revealed an increase in the control population. Follistatin was increased in acute burn patients compared with the control population. The results are shown as Boxplots (including median, upper, and lower quartiles and upper and lower maximums).
Myostatin and Follistatin Expression are Burn Surface Area Dependent
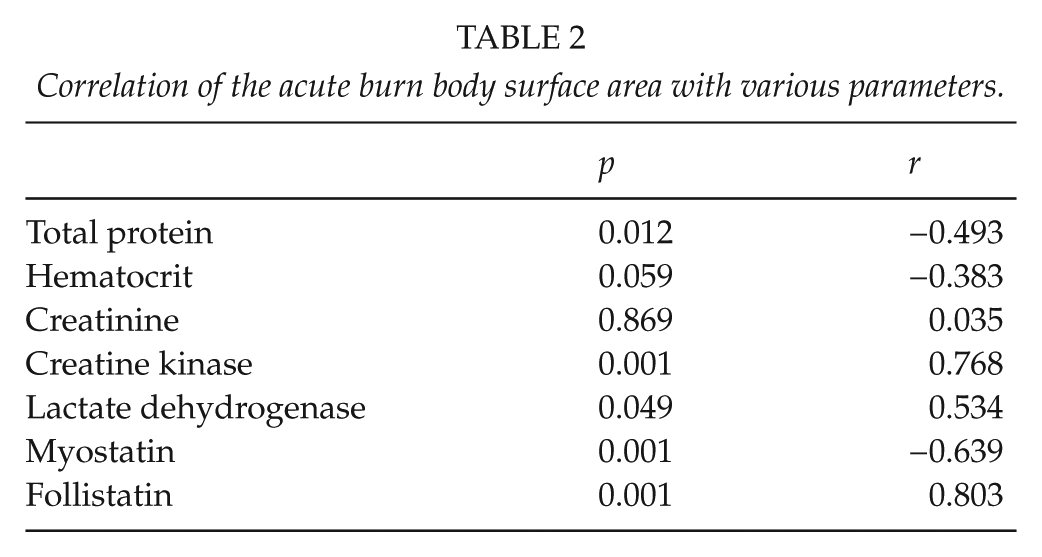
To analyze a correlation of burned body surface area with myostatin and follistatin the Pearson’s correlation coefficient was calculated. Both myostatin (r = −0.639, p < 0.001) and Follistatin (r = 0.778, p < 0.001) showed a highly significant burn surface-dependent decrease and increase, respectively (Table 2, Fig. 2).
Correlation of the acute burn body surface area with various parameters.
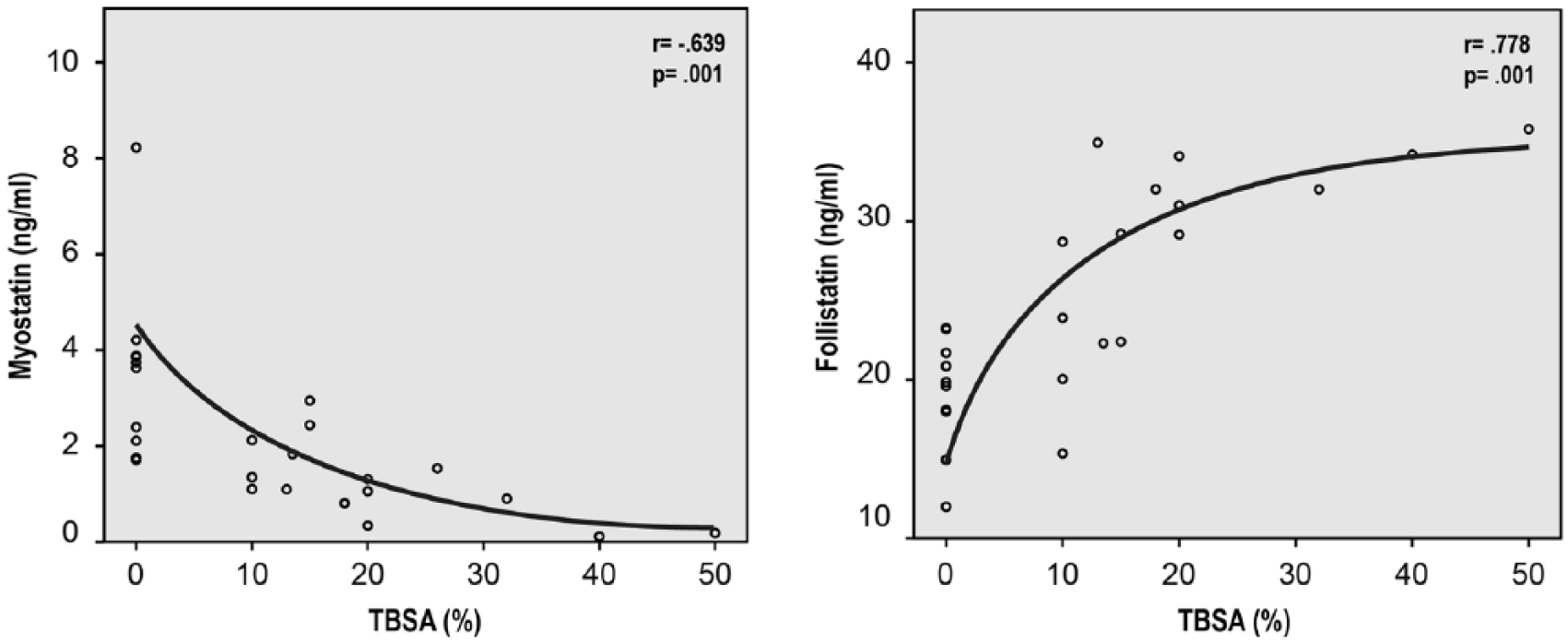
Myostatin and follistatin show dependency to increasing acute burn surface. (Left): ELISA of patients serum shows a highly significant reciprocal correlation between myostatin and burn surface area. (Right): ELISA of patients serum shows a highly significant correlation between follistatin and burn surface area. The results are shown as mean curve (two-sample t-test; Pearson’s regression analysis).
Myostatin and Follistatin Show a Reciprocal Correlation
Data of patient serum show decreasing myostatin levels with increasing amounts of follistatin. To rule out a decreased amount of myostatin level due to reduced total protein concentration, we were able to show that myostatin was not dependent on the total protein concentration comparing healthy and burn patients’ serum. Total protein, hematocrit, and creatinine were not myostatin or follistatin dependent (Table 3, Fig. 3).
Correlation of myostatin and follistatin with various parameters in acute burn.
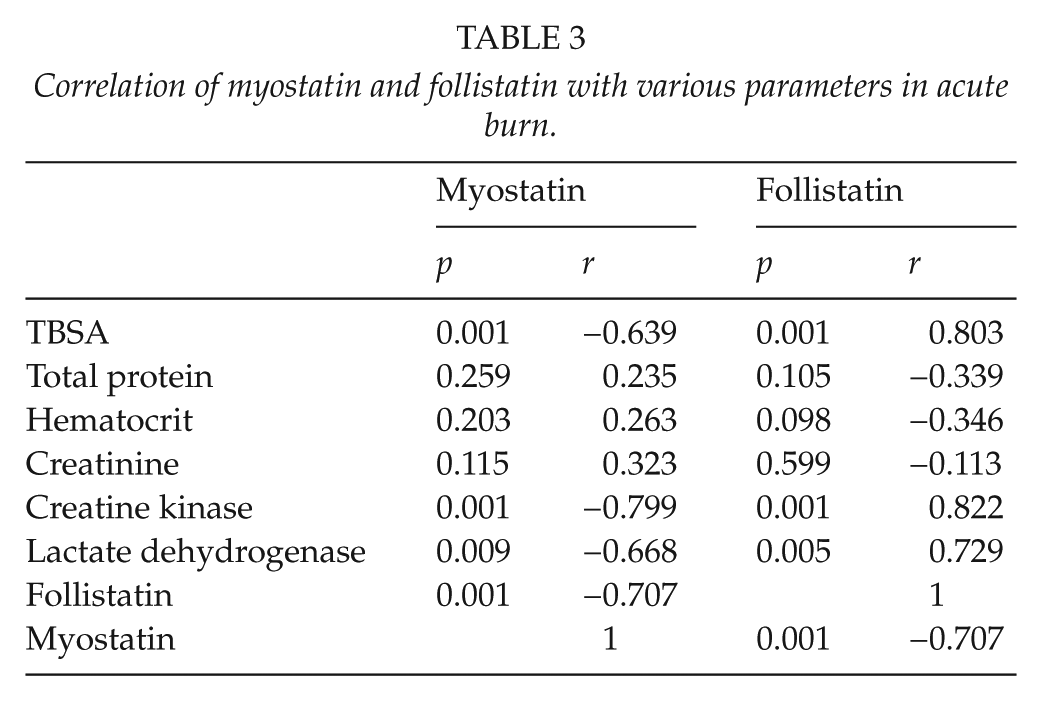
Myostatin decreased with increasing serum follistatin but was not total protein dependent. (Left): Myostatin and follistatin were measured by ELISA in burn patient serum (n = 15). Myostatin concentration showed a follistatin concentration dependency with high significance (r = −0.707; p = 0.001). (Right): Myostatin did not show any protein concentration dependency between burn and healthy patients. The results are shown as mean curve (two-sample t-test; Pearson’s regression analysis).
Strength after Severe Burn Injury is Impaired without Reduction of Muscle Mass
Next, we sought to investigate the long-term (9–12 months after burn injury) outcome on muscle strength in severe burn patients. Patients were evaluated 9–12 months after the severe burn injury. The measurement of the non-dominant upper- and forearm prevents biasing the results by immobilization of affected muscles. There were no significant differences between non-dominant fore- and upper arm circumference (Table 4, Fig. 4A, B). The grip strength, pinch grip strength, and lateral grip strength showed a significant decrease in post-burn patients compared with the control group (Fig. 4).
Baseline data and strength measurements patients 9–12 months after severe burn injury compared with healthy control population.

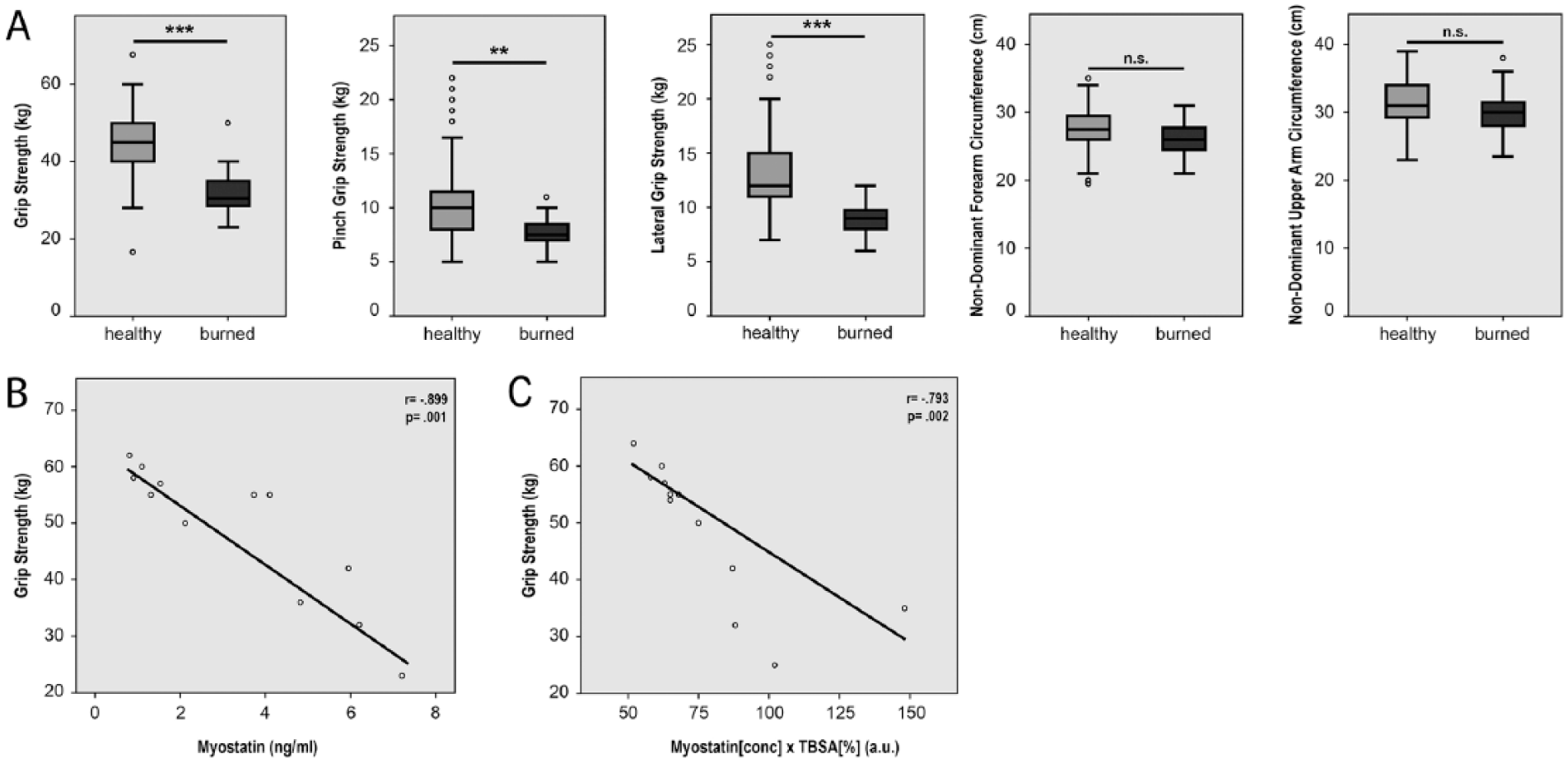
Grip strength, pinch grip, and lateral grip strength were significantly decreased in post-burn patients compared with the control population. (A) Grip strength was measured with a Jamar® Hydraulic Hand Dynamometer at level 2 on the non-dominant hand. Pinch grip and lateral grip strength were tested accordingly with a Baseline® Pinch Gauge. Male control patients (n = 107) showed a significantly higher strength in all three qualities (grip, pinch grip, and lateral grip strength) compared with the male post-burn patients (n = 13). Circumference of the non-dominant forearm and upper arm showed no significant difference between the male control patients (n = 107) and burn patients (n = 13). The results are shown as Boxplots (including median, upper, and lower quartiles and upper and lower maximums). p value: *< 0.05; **< 0.01; ***< 0.001; ANOVA. (B) Grip strength decreases with increasing chronic burn serum myostatin (n = 13; r = −0.899; p = 0.001; two-sample t-test; Pearson’s regression analysis). (C) Grip strength decreases with increasing burn surface area combined with the initial (acute) burn serum myostatin (n = 13; r = −0.793; p = 0.002; two-sample t-test; Pearson’s regression analysis).
Initial Myostatin Concentration Combined with the Burn Surface Area Predicts Long-Term Strength Impairment
Finally, we sought to investigate whether a correlation between initial myostatin concentration and the long-term muscle strength impairment existed. As we demonstrated that decreasing myostatin levels correlate with increasing TBSA (Fig. 2), we wanted to take into account both the factors in correlation to long-term strength. We multiplied TBSA with the myostatin levels in the acute burn serum and showed a highly significant negative correlation between myostatin, TBSA, and long-term muscle strength (Fig. 4C).
Discussion
The aim of the study was to investigate implications of myostatin in muscle catabolism after thermal trauma. We could demonstrate that as an acute reaction to thermal trauma, myostatin levels were downregulated and follistatin as a natural inhibitor upregulated, compared with healthy individuals. These findings were surprising, given the well-known systemic posttraumatic muscle cachexia following thermal injury and suggest a functioning negative feedback loop to prevent excessive muscle wasting in the acute phase. Importantly, downregulation of myostatin and upregulation of follistatin is related to an increased burned body surface area. Interestingly, with a larger burn surface area, myostatin is elevated and linked with decreased grip strength in chronic burn patients.
It is unlikely that protein loss causes an imbalance of follistatin/myostatin in the first days after thermal injury, since there is no statistical significance in the relationship of myostatin levels and protein levels. In this study, no patient with the need for escharotomy or fasciotomy was included. We have no clinical evidence for any muscle injury in our patient collective. One reason for both the CK level rise and the follistatin/myostatin reaction associated with larger burn surface can be a systemic inflammatory reaction involving the muscle. The rise of follistatin levels under systemic stress has rarely been described before: systemic inflammatory processes can cause an elevation of serum Follistatin (20). However, these interactions are not understood yet.
Mechanisms of how myostatin affects different tissues are poorly understood. According to our data, acute thermal trauma and other muscle wasting trauma (e.g. infection, septicaemia) lead to an upregulation of follistatin and concordant to a decrease of myostatin compared with healthy individuals (21). In chronic thermal injuries, the situation is reversed: Follistatin is significantly decreased, while myostatin is increased compared with healthy individuals. Importantly, this observation is in concordance with muscle cachexia, clinically observed in chronic burn patients.
In addition, chronic burn patients showed a highly significant reduction of muscle strength in the non-dominant forearm-muscles, while muscle mass in the forearm and upper arm of the non-dominant upper extremity was not reduced. We were able to show a correlation of muscle strength in chronic thermal injuries and a possible correlation with the elevation of myostatin. Other than expected, we encountered a stronger affect on muscle strength than on muscle mass by elevation of myostatin in chronic burn patients. The literature describes myostatin as the primary factor affecting muscle mass, rather than muscle strength. Accordingly, our results suggest a reduction of muscle mass but not significantly. This could be due to the characteristic limitation of muscle mass gain and loss in the measured anatomical location, given that the forearm circumference with a large proportion of tendons and bones tend to be relatively unaffected by muscle mass gain and loss (22). However, more recent studies have demonstrated a complex interplay between myostatin and muscle strength (23). In our clinical data, we were able to identify myostatin as a factor influencing muscle strength in patients after severe burn injuries.
One major finding of the study was the correlation of the initial myostatin concentration combined with TBSA and long-term muscle strength. As we have shown earlier, myostatin is TBSA dependent, however, the weighting of myostatin (SD: 67%) in this long-term strength correlation is greater than TBSA (SD: 55.2%), because of the higher standard deviation of myostatin (Table 1). This circumstance—that initial myostatin levels play a major role in the long-term muscle strength outcome—may be used to evaluate an anti-catabolic therapy based on the initial myostatin concentration and TBSA.
One main problem in burn patients is muscle loss through cachexia, a hypermetabolic state with wasting of muscle to meet the severely increased nutritional demand. A proposed strategy to overcome this issue could be the pharmacological administration of myostatin antagonists accompanied with adequate nutrition of the patient.
Limitations of the Study
One major limitation of the study is the small sample size. This study was intended to serve as a pilot project to outline the role of myostatin and follistatin in the complex burn-dependent muscle cachexia. In spite of the small sample size we were able to demonstrate a highly significant correlation between the TBSA, myostatin, follistatin, and muscle strength.
Unfortunately the medical circumsatances within the study impede an initial muscle strength measurement. Most of the patients with more than 10% TBSA were intubated and sedated and a measurement within the first 48 h would be not reproducable. Thus, we were not able to identifiy the baseline muscle strength of every individual.
Conclusion
Taken together, our data show that the acute phase of burn injury in humans is accompanied by upregulation of follistatin and myostatin blockade concordantly, presumably in order to prevent muscle catabolism. This effect is reversed in the chronic, catabolic state of burn injury, leading to a net muscle strength reduction. A treatment strategy to keep follistatin levels elevated or to inhibit activin receptor type II receptor as target of GDF 8 might prove to be pivotal to prevent cachexia in burn injury.
Footnotes
Acknowledgements
C.W., M.B., H.J., and B.B. wrote the main manuscript text and prepared all the figures. Experiments were conducted by C.W., H.J., M.B., J.M.W., M.D., and K.H. All the authors reviewed the manuscript.
Declaration Of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Consent To Participate
The authors certify that they comply with the ethical guidelines for authorship and publishing. The study was performed in accordance with the ethical committee of the Ruhr University Bochum after approval (Approval Number: 16-5619-BR).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Grant of FoRUM K108-16 (Research grant by the Ruhr University Bochum, School of Medicine).
