Abstract
Background and Aims:
The International Study Group of Rectal Cancer has proposed that a pelvic abscess in the proximity of the anastomosis is considered an anastomotic leak, whether or not its point of origin is detectable. This study describes how the inclusion of pelvic abscesses alters the leakage rate.
Material and Methods:
Risk factors and postoperative complications in patients with visible anastomotic leakage (“direct leakage”), pelvic abscesses alone in the vicinity of a visibly intact anastomosis (“abscess leakage”), and no leakage were retrospectively evaluated.
Results:
In total, 341 patients operated with anterior resections and who received an anastomosis within 15 cm as measured from the anal verge were included. A total of 37 patients (10.9%) had direct leakage, 13 (3.8%) had abscess leakage, and 291 (85.3%) had no leakage. The overall anastomotic leakage rate was 14.7% (50 patients). In accordance with the grading system outlined by International Study Group of Rectal Cancer, 7 patients (2.1%) experienced Grade A leakage, 19 (5.6%) Grade B, and 24 (7.0%) Grade C. Direct leak patients had more often a reoperation due to anastomotic complications (odds ratio = 19.7, p = 0.001), a permanent stoma (odds ratio = 28.5, p = 0.001), and a longer hospital stay than abscess leak patients (29.0 vs 15.5 days, p = 0.030).
Conclusion:
Abscess leakage accounted for over one-fourth of the overall leakage rate, raising the leakage rate. Direct leak patients were at a higher risk of requiring a reoperation, permanent stoma, and longer hospital stay than abscess leak patients. Abscess leak patients were at a greater risk for a urinary tract infection, wound infection, and postoperative intestinal obstruction than non-leak patients.
Introduction
Overview
Anastomotic leakage remains one of the most serious complications following sphincter-saving surgery of the rectum. Postoperative leakage can lead to sepsis, organ failure, and a prolonged hospital stay, thus increasing morbidity and mortality in these patients (1–3). Definitive associations between anastomotic leakage and postoperative functional outcomes, such as low anterior resection syndrome (LARS), are still unclear (4–6).
The etiology of anastomotic leakage is multifactorial. An assessment of the literature on anastomotic leakage yields a range of leakage rates between 3% and 19% in rectal surgery where an anastomosis was created (7, 8). Although this discrepancy in reported leakage rates could be attributed to publication bias and differences in the quality of care among institutions, an important explanatory factor may be variations in the definition of anastomotic leakage (9–11).
The diagnosis and evaluation of anastomotic leakage varies in the literature, but is primarily based on a combination of clinical symptoms, laboratory results, radiological imaging, and findings during endoscopic examination. Clinical symptoms, if present, consist mainly of abdominal/pelvic pain or distention, fever, fecal/purulent vaginal or rectal discharge, and/or fecal/purulent postoperative drain content. Laboratory findings consist primarily of leukocytosis and/or elevated C-reactive protein (9–11). Radiological signs of anastomotic leakage are less systematically studied, but the leakage of rectal contrast medium from the endolumen to the perianastomotic space is considered a reliable sign of anastomotic leakage. A number of radiological signs such as perianastomotic fluid collection, free air, and distant fluid collections are from a radiological standpoint suggestive, but not conclusive, of anastomotic leakage (12, 13).
The Definition Proposed by Isrec
The International Study Group of Rectal Cancer (ISREC) proposed a standardized definition and grading system for anastomotic leakage in 2010, aiming for its universal acceptance in future studies. In their study, the authors proposed that anastomotic leakage after anterior resection of the rectum should be defined as a communication between the intra- and extraluminal compartments due to a defect in the integrity of the intestinal wall at the anastomosis or neorectal reservoir (J-pouch or transverse coloplasty). Furthermore, the authors recommended that a pelvic abscess in the proximity of an anastomosis should be considered leakage, whether or not its point of origin is detectable (14).
The grading system provided by ISREC consists of three leakage grades that are easily applied in clinical practice. Grade A leaks are subclinical (asymptomatic) and require no changes in therapy, Grade B leaks require non-surgical therapeutic intervention, and Grade C leaks require a reoperation (14). Since its introduction, several studies have been published using this grading system (15–18).
Although several authors of previous studies have accepted the inclusion of pelvic abscesses (3, 19–21), the majority of studies on anastomotic leakage do not take abscesses into account when defining leakage. Furthermore, only 51% of British colorectal surgeons participating in a recent survey agreed that Grade B leakage (leakage requiring non-surgical treatment) was in fact leakage. The authors attributed this disagreement to an uncertainty regarding the origin of pelvic abscesses, arguing that a pelvic collection with no fecal fistula to the rectum could be the result of either a small leak or an infected hematoma (22). As comparisons of treatment and complication rates are gaining significance on an international level, agreement on definitions used in the description of anastomotic leakage becomes essential.
The primary purpose of this study is therefore to describe how the inclusion of pelvic abscesses in a definition of anastomotic leakage alters the leakage rate. The secondary purpose is to describe characteristics of patients with anastomotic leakage, with pelvic abscesses alone and patients without anastomotic leakage in terms of risk factors and postoperative complications.
Material and Methods
Study Population
All patients treated surgically for rectal/rectosigmoid adenomas or adenocarcinomas between 1 January 2006 and 31 December 2011 at Haukeland University Hospital were retrospectively studied. This hospital is designated as a regional cancer-treating hospital on the west coast of Norway. Patient data were accessed through the hospital network and stored on a secure hospital server. A database consisting of 22 preoperative and 10 intraoperative parameters related to anastomotic leakage was thereby created. Furthermore, we recorded 21 different postoperative complications related to leakage. A high level of data completeness was obtained (data not shown) that correlated well with a comparable data set from the same institution (23).
Inclusion/Exclusion Criteria
Patients presenting with a primary rectal/rectosigmoid adenoma or adenocarcinoma at the time of diagnosis and who received an anastomosis within 15 cm of the anal verge were included in the study. Measurements were obtained by rigid scope. Anterior resection was performed for both benign and malignant tumors. Measurements were obtained by rigid scope. Patients who did not undergo surgery for their primary rectal/rectosigmoid tumor were excluded. Patients who underwent an abdominoperineal resection, Hartmann’s procedure, transanal endoscopic microsurgery (TEM), or local resection and therefore did not receive a colorectal anastomosis were also excluded.
Recent studies show that complications arise for patients presenting with early (⩽30 days postoperatively) and late leakage/abscesses (>30 days postoperatively) and that a significant number of leaks occur more than 30 days after surgery (11, 17, 24). Both early and late leakages were therefore included in this study. Patients were diagnosed within 1 year of their primary surgery.
Reoperations due to anastomotic complications during the total follow-up time were recorded. These included laparotomies, Hartmann’s procedures, or abdominoperineal resections due to anastomotic leakage. They also included reoperations due to pelvic abscesses, wound infections, fistulas, and anastomotic stenosis. Reoperations due to wound dehiscence, intestinal obstruction, dehydrating stomas, small bowel perforation, and stoma revision were investigated and analyzed separately.
Definition of Anastomotic Leakage
The study population was divided into three groups: patients with visible anastomotic leakage (defined in our study as “direct leakage”), patients with a pelvic abscess in the vicinity of the anastomosis, but no visible failure of the anastomotic integrity (defined in our study as “abscess leakage”), and patients with no leakage. In accordance with national guidelines concerning oncological follow-up, all patients received routine rectoscopy and radiological evaluation every 6 months postoperatively. Patients also underwent rectoscopy before temporary stoma closure or when anastomotic leakage was suspected.
The classification of direct leakage in this study depended on the visual failure of anastomotic integrity in an endoscopy (rectoscopy/sigmoidoscopy) report, by a clinician after digital palpation of an anastomotic defect, in a radiological report (computed tomography (CT)/magnetic resonance imaging (MRI) scan reporting visual leakage of rectal contrast medium from the endolumen to the extraintestinal space, usually perianastomotic), or in a reoperation report after visualization by a surgeon. Although many patients presented with symptoms indicative of leakage (abdominal/pelvic pain and/or distention, fecal/purulent postoperative drain content, purulent discharge from the rectum or vagina, and fever), leukocytosis, and elevated C-reactive protein, the presence or absence of such findings did not influence the classification of direct leakage. In our study, we defined a fistula as a communication between the rectum and a presacral sinus, skin, intestine, vagina, or male genitourinary system.
Abscess leakage was diagnosed by evaluation of CT and/or MRI abdominal and pelvic scan reports when the radiologist described both a lack of rectal contrast medium leakage from the endolumen to the extraintestinal space and additional findings of a presacral, pelvic, perirectal, or perianastomotic abscess, air, and/or pelvic fluid collections in the radiological report. The scans were performed either when the patient presented with clinical symptoms (abdominal/pelvic pain and/or distention, fecal/purulent postoperative drain content, purulent discharge from the rectum or vagina, diarrhea, or fever) or during a routine postoperative oncological checkup. Patients diagnosed with pelvic abscesses did not routinely undergo rectoscopy to inspect the anastomosis. Rather, rectoscopy was performed on an individual basis dependent on the clinical condition of the patient. Pelvic abscesses were drained with radiological guidance, if indicated by the patients’ clinical condition. Patients with both direct leakage and pelvic abscesses were placed in the direct leakage group.
Statistical Analysis
First, the leakage rates for patients with direct leakage and abscess leakage were calculated for the 6-year period. Second, leakage severity was classified according to the A, B, and C grading criteria provided by ISREC. Analysis was thereafter focused on testing for statistical differences between patients with direct leakage, abscess leakage, and no leakage in terms of the frequency of preoperative risk factors, intraoperative variables, and postoperative complications. Comparisons between direct leak and non-leak patients have been made in multiple previous studies and are therefore not performed in this study (1, 25). Statistical analysis of the collected data was performed using SPSS version 21–25 (SPSS, Chicago, IL, USA). For categorical variables, statistical differences were calculated using Fisher’s exact test with risk estimate due to small group sizes. Odds ratios (OR) and 95% confidence intervals (95% CI) were calculated for categorical variables using the statistical software MedCalc version 15.2 (MedCalc Software bvba, Acacialaan 22, 8400 Ostend, Belgium). For continuous variables, one-way analysis of variance (ANOVA) tests with Games-Howell post hoc procedures were used to calculate mean differences and 95% CI due to unequal group sizes. Effect sizes were calculated for continuous variables as specified by Rosenthal, R (26). Effect sizes <0.30 were considered small, 0.30–0.50 moderate, and >0.50 large (26). Two-sided p values <0.05 were considered statistically significant.
The Regional Ethical Committee (REK) in Norway approved this study.
Results
Study Population
Between 1 January 2006 and 31 December 2011, anterior resections of primary rectal/rectosigmoid adenomas or adenocarcinomas were performed in 341 patients. For patients with rectal tumors, total mesorectal excision (TME) was performed in 220 patients (64.5%) while 63 patients (18.5%) with high rectal tumors received a partial mesorectal excision (PME). For four patients (1.4%) with rectal tumors, the mesorectal technique was not described by the surgeon and therefore unknown. For the 54 patients (15.8%) with rectosigmoid tumors, anterior resection with PME was performed. All patients received an anastomosis within 15 cm of the anal verge as measured by a rigid scope. The median follow-up time for the study population was 5.0 years (2.9–8.8 years). The mean age of the study population was 65.55 years and 56% of the patients were males. Of the 120 patients who received preoperative radio- and chemotherapy, 117 (97.5%) also received a temporary stoma before or during their primary surgery. This coincides with department policy that recommends temporary stoma creation, either pre- or intraoperatively, in all patients receiving preoperative radio- and chemotherapy, a known risk factor for leakage (25, 27). In contrast, only 49 of 221 patients (22.2%) without preoperative therapy received a temporary stoma. The rate of preoperative radio- and chemotherapy increased from 27.5% in 2006 to 40.7% in 2011. The total reoperation rate due to postoperative complications was 15.5% (53/341 patients). This rate includes reoperations due to wound dehiscence, intestinal obstruction, Hartmann’s procedures or amputations due to leakage, pelvic abscesses, wound infections, fistulas, and anastomotic stenosis.
Anastomotic Leakage Rate
In our study population, 37 patients (10.9%) were diagnosed with direct leakage, 13 (3.8%) with abscess leakage, and 291 patients (85.3%) experienced no leakage (Table 1). The overall anastomotic leakage rate in this study was therefore 14.7% (50 patients). Of the 37 patients with direct leakage, 29 (78.0%) also experienced pelvic abscesses.
Leakage grade among the three patient groups in accordance with ISREC criteria.
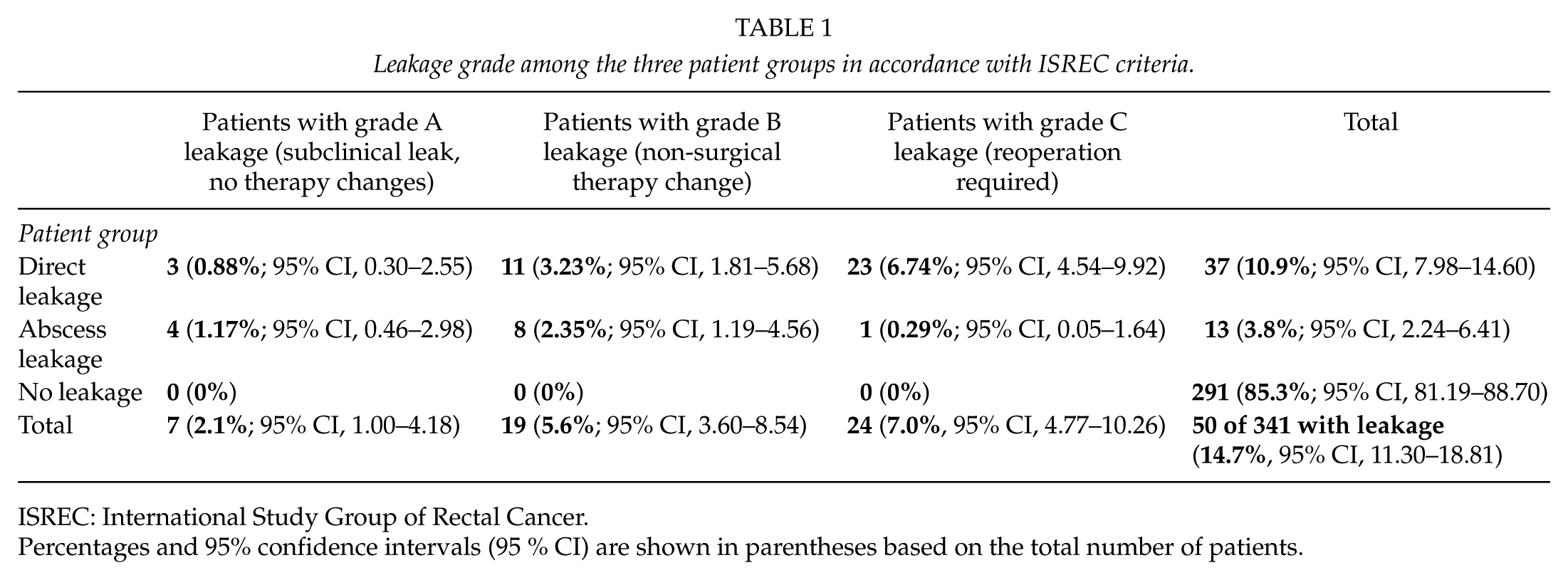
ISREC: International Study Group of Rectal Cancer.
Percentages and 95% confidence intervals (95 % CI) are shown in parentheses based on the total number of patients.
Patients with leakage were graded according to the system outlined by ISREC. Seven patients (2.1%) met the criteria for Grade A leakage (no intervention), 19 patients (5.6%) met the Grade B criteria (non-surgical therapy), and 24 patients (7.0%) met the Grade C criteria (reoperation). Only one of the 24 patients with Grade C leakage was found in the abscess leakage group, while the other 23 patients were found in the direct leakage group (Table 1). One direct leak patient with Grade C leakage died postoperatively due to severe complications and multiple comorbidities.
Preoperative and Intraoperative Risk Factors
The distribution of preoperative risk factors for leakage among each patient group is shown in Table 2. There was no significant difference between direct leak patients and abscess leak patients in regard to the tumor to anal verge distance (p = 0.218), although abscess leak patients had significantly lower tumors compared to non-leak patients (8.39 cm vs 11.60 cm, mean difference = 3.21, 95% CI = 0.48 to 5.94, p = 0.021). The effect size for this difference was found to be 0.16 and therefore small. Comparisons between the other leakage groups, including comparisons between direct leak and non-leak patients, regarding the tumor-anal verge distance and anastomotic level were non-significant. Any differences regarding other preoperative and intraoperative factors were also non-significant.
Distribution of preoperative and intraoperative factors associated with leakage.
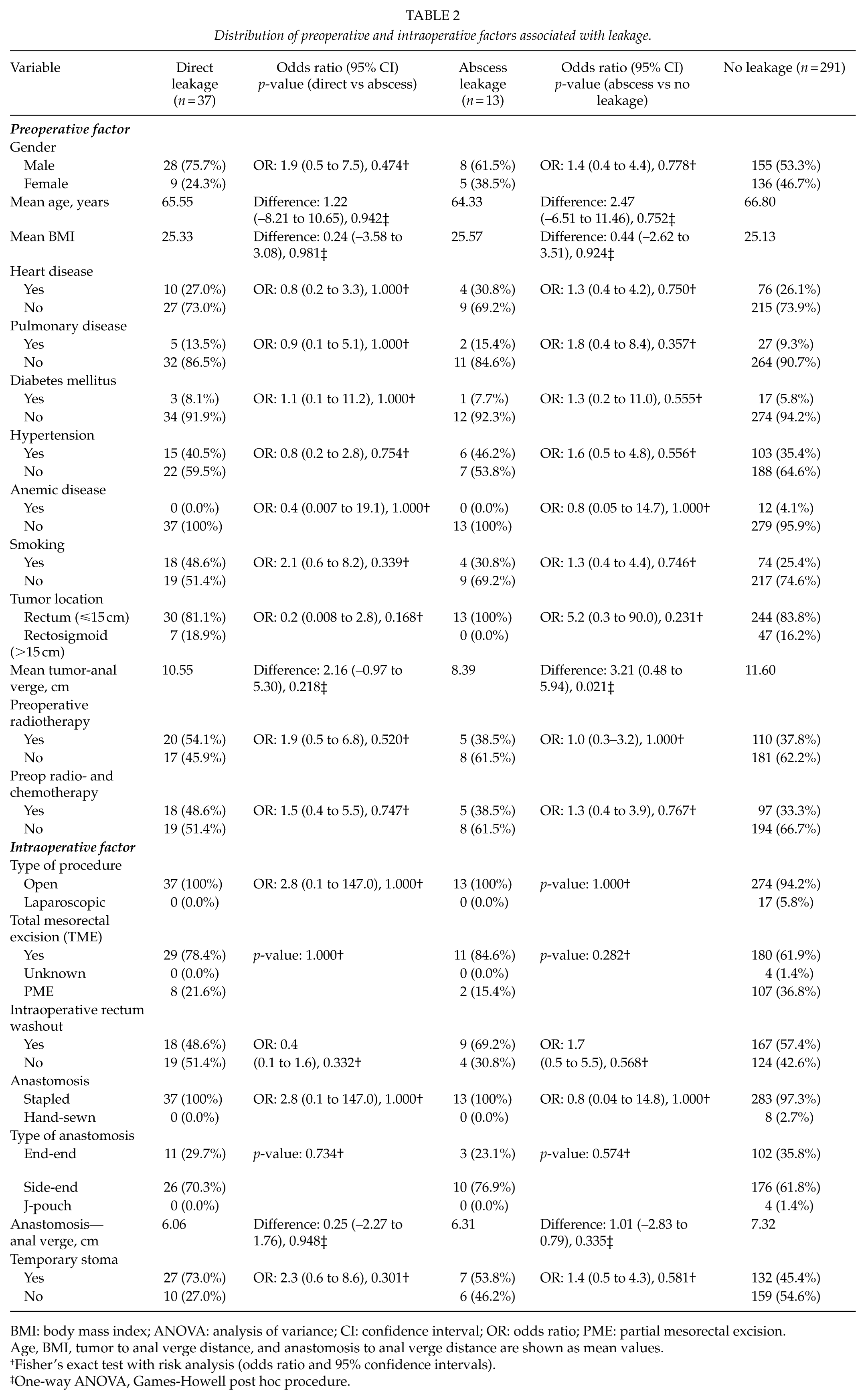
BMI: body mass index; ANOVA: analysis of variance; CI: confidence interval; OR: odds ratio; PME: partial mesorectal excision.
Age, BMI, tumor to anal verge distance, and anastomosis to anal verge distance are shown as mean values.
Fisher’s exact test with risk analysis (odds ratio and 95% confidence intervals).
One-way ANOVA, Games-Howell post hoc procedure.
Postoperative Complications
Time until leakage
Of the 37 patients with direct leakage, 26 (70.3%) experienced early leakage (⩽30 days postoperatively) and 11 (29.7%) experienced late leakage (>30 days postoperatively). Of the 13 patients with abscess leakage, 10 (76.9%) were diagnosed with early leakage and 3 (23.1%) with late leakage. These differences were not significant, p = 0.734. Six direct leak patients were diagnosed with leakage over 100 days after their primary surgery. These patients experienced few or no clinical symptoms of leakage, and they received the diagnosis either after a routine CT/MRI scan or after a scan to rule out suspected local recurrence. Two abscess leak patients were diagnosed with pelvic abscesses 138 and 168 days after primary surgery. Both cases were diagnosed by CT scans due to clinical symptoms shortly following temporary stoma closure.
Direct leakage versus abscess leakage
Postoperative complications among direct leak patients were different in nature than those found among abscess leak patients. Due to the serious nature of direct leakage, 23 direct leak patients (62.2%) underwent a reoperation due to anastomotic complications compared to only one abscess leak patient (7.7%), p = 0.001 (Table 3). The OR for this difference was 19.7 (95% CI, 2.3 to 168.5). Of those reoperated due to direct leakage, 4 patients were reoperated with laparotomy, 15 patients with Hartmann’s procedure, and 3 patients with abdominoperineal resections. One direct leak patient was reoperated due to a deep pelvic infection following leakage. The abscess leak patient was reoperated due to anastomotic stenosis after multiple attempts at dilatation.
Distribution of postoperative complications associated with leakage.
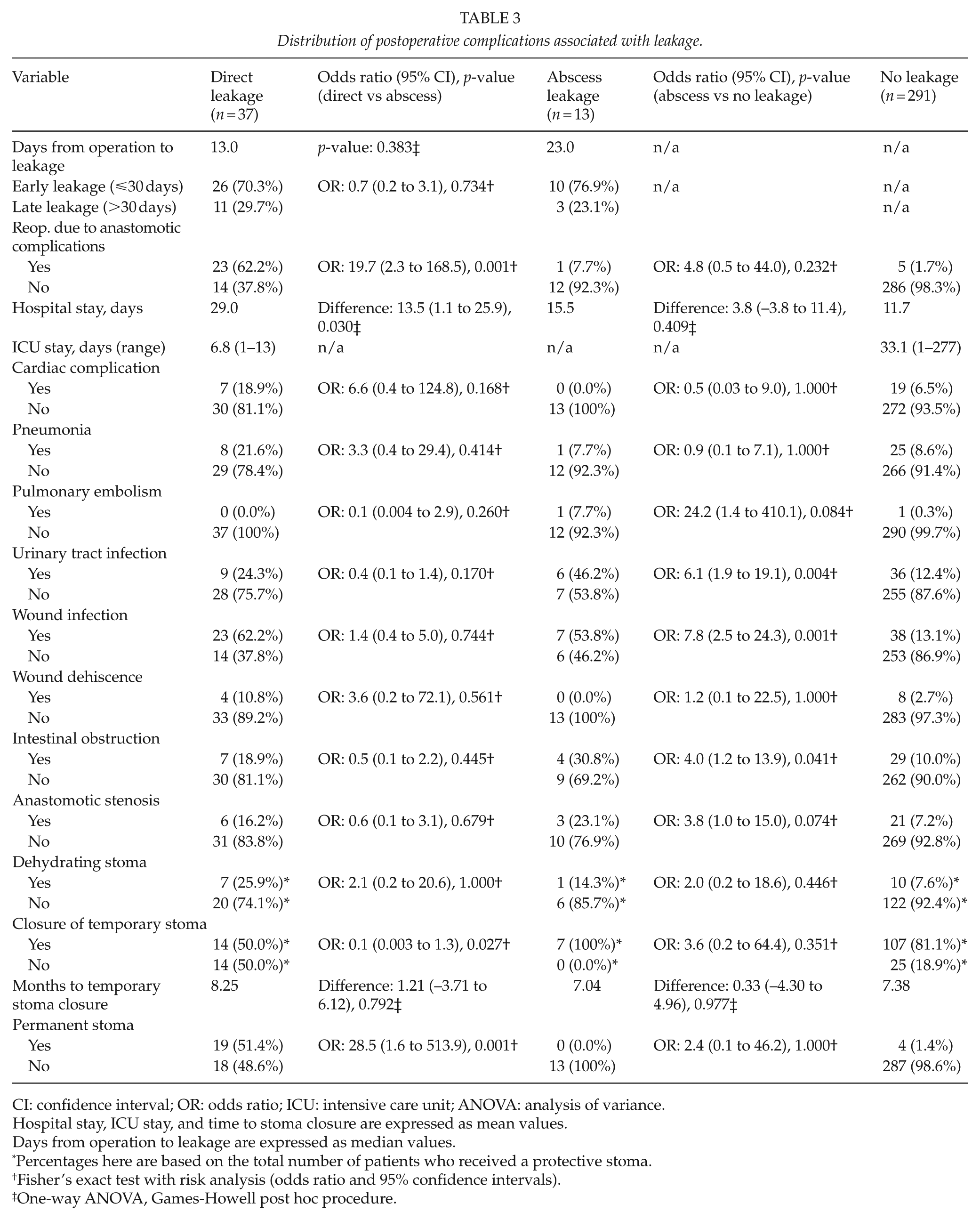
CI: confidence interval; OR: odds ratio; ICU: intensive care unit; ANOVA: analysis of variance.
Hospital stay, ICU stay, and time to stoma closure are expressed as mean values.
Days from operation to leakage are expressed as median values.
Percentages here are based on the total number of patients who received a protective stoma.
Fisher’s exact test with risk analysis (odds ratio and 95% confidence intervals).
One-way ANOVA, Games-Howell post hoc procedure.
Direct leakage had an impact on stoma closure as well. Of the 27 direct leak patients who received a temporary stoma, only 14 (50.0%) were fit for stoma closure postoperatively compared to all seven (100%) of the abscess leak patients, OR = 0.1, 95% CI = 0.003 to 1.3, p = 0.027. Of all the patients with direct leaks, 19 (51.4%) maintained a permanent stoma compared to none of the abscess leakage patients, p = 0.001 (Table 3). The risk of requiring a permanent stoma was therefore 28.5 times higher if a patient developed direct leakage rather than abscess leakage (95% CI, 1.6 to 513.9). Furthermore, it took an average of 8.25 months before the temporary stoma could be closed in direct leak patients, 1.21 months longer than stoma closure for abscess leak patients (95% CI, –3.71 to 6.12), p = 0.792 (Table 3).
Direct leak patients experienced the most prolonged postoperative recovery time of the patient groups, spending on average 29.0 days in the hospital, 13.5 days longer than abscess leak patients who spent on average 15.5 days in hospital (95% CI = 1.1 to 25.9, p = 0.030, Table 3). The effect size of this difference was 0.33 and therefore moderate.
Abscess leakage versus no leakage
Patients in the abscess leak group experienced postoperative complications more frequently than patients without leakage (Table 3). In particular, the risk of wound infections (OR = 7.8, 95% CI = 2.5 to 24.3, p = 0.001), urinary tract infections (OR = 6.1, 95% CI = 1.9 to 19.1, p = 0.004), and postoperative intestinal obstruction (OR = 4.0, 95% CI = 1.2 to 13.9, p = 0.041) was significantly higher among patients with abscess leakage than non-leak patients. Compared to only one abscess leak patient, five patients (1.7%) without leakage were reoperated due to complications associated with their anastomosis. This difference was not significant (p = 0.232). One non-leak patient was reoperated due to peritonitis and deep inflammation, two patients due to recurrent anastomotic stenosis, and two patients due to rectovaginal fistulas. All other differences regarding postoperative complications between non-leak and abscess leak patients were non-significant.
Discussion
Of the 341 patients enrolled in this study, 37 patients (10.9%) were diagnosed with direct leakage (95% CI = 7.98 to 14.60) and 13 (3.8%) with abscess leakage (95% CI = 2.24 to 6.41), giving a total of 50 leakage patients (14.7%, 95% CI = 11.3 to 18.81) as defined by ISREC. Abscess leaks accounted thereby for 26% of all leakage, thus increasing the overall leakage rate. In regard to differences in preoperative risk factors, the tumor-anal verge distance in abscess leak patients was significantly shorter than the distance for non-leak patients (8.39 cm vs 11.60 cm, difference = 3.21 cm, 95% CI = 0.48 to 5.94, p = 0.021), although the effect size of this difference was small (0.13). Surprisingly, no significant differences when comparing the other groups (including comparisons between direct leak and non-leak patients) regarding tumor-anal verge distance and anastomotic level were found. The small number of patients in the abscess leak group (n = 13) can possibly explain the significant difference in tumor-anal verge distance between abscess leak and non-leak patients. One abscess leak patient with a tumor at 10 cm was found to have a significant effect on the average tumor-anal verge distance for the group by lowering the mean from 9.0 to 8.4 cm. Furthermore, the small number of abscess leak patients also made it difficult to run multivariate analysis. The limited number of patients in this study, especially in the abscess leak group, warrants caution in interpreting the results from subgroups, as they are prone to significant type II errors.
Analysis of postoperative complications showed that direct leak patients were at increased risk of requiring a reoperation due to anastomotic complications, requiring a permanent stoma, developing a postoperative fistula, and experiencing a prolonged hospital stay compared to abscess leak patients. These are complications related to a larger contamination of bowel contents into the abdominal cavity, often requiring surgical intervention (23 of 24 grade C leaks were also direct leaks). Abscess leak patients were at significantly greater risk of developing a urinary tract infection, wound infection, and postoperative intestinal obstruction than non-leak patients. Such complications may be related to the detrimental effects of a pelvic abscess and chronic inflammation on bowel function and the urinary tract. It must be emphasized that such changes are also found among patients with visible leakage (28, 29). These conditions can usually be managed with radiological drainage or antibiotics, avoiding surgical intervention (only one patient in the abscess leak group presented with extensive pelvic inflammation and required a reoperation).
In accordance with local department policy, 117 of 120 patients (97.5%) with preoperative radio- and chemotherapy received a temporary stoma compared to only 49 of 221 patients (22.2%) without preoperative therapy. This indicates that patients at higher risk of leakage were provided with a stoma. Furthermore, 9 of 10 (90.0%) direct leak patients without a temporary stoma suffered from Grade C leakage (reoperation) while only 14 of 27 (51.9%) direct leak patients with a temporary stoma experienced Grade C leakage, suggesting that a temporary stoma reduces the severity of leakage and the reoperation rate. These findings are consistent with results from a systematic review and meta-analysis by Huser et al. (30) where a stoma did not influence the occurrence of leakage, but reduced the clinical severity of leakage.
The overall anastomotic leakage rates in our study correspond well with results from studies published after the introduction of the ISREC definition and grading criteria. Testing the validity of the new ISREC grading system, Kulu et al. (15) reported an overall leak rate of 7.5% and found that seven (12.5%) patients with anastomotic leakage also had an abscess in the vicinity of the anastomosis without a visible fecal fistula. Morks et al. (17) reported an overall leakage rate of 20% and data showing an abscess leakage rate of 3.6% (5/141 patients), consistent with our abscess leakage rate of 3.8%. Although other studies describe lower leakage rates than our own (7, 31), this discrepancy can in part be explained by the fact that we, similarly to Morks et al., include both early (⩽30 days postoperatively) and late leakage patients (>30 days postoperatively) in the study population. In our study, for example, three patients were diagnosed with direct leakage over 200 days after the primary operation and another two patients received the diagnosis over 300 days postoperatively. These five patients experienced asymptomatic, subclinical leaks that were discovered radiologically during a routine oncological control or after local recurrence was suspected. All five patients were initially diagnosed with pelvic abscesses. Because patients diagnosed with pelvic abscesses did not routinely undergo rectoscopy, it is possible that these patients experienced leaks that went undetected until radiological examination. Even though all patients in the study were followed with routine rectoscopy and radiological evaluation every 6 months postoperatively, detection of microleaks may be difficult.
Including patients with late anastomotic leakage and chronic pelvic abscesses remains clinically important. Evidence suggests that these patients experience complications even years after surgery (10, 11, 17, 24, 26). Although some previous studies do include abscesses as leakage, many do not. In a systematic review of leakage definitions, Bruce et al. (11) found that only 10 of 29 studies included abscesses as signs of leakage. Snijders et al. (32) found a similar result in a meta-analysis where only 10 of 19 studies mentioned abscesses in their definitions of leakage, let alone considering abscesses as leaks in themselves as ISREC does.
Identifying and treating pelvic abscesses early may help prevent more serious postoperative complications such as chronic presacral sinuses from developing later on (29). Furthermore, distinctions between pelvic abscesses and direct leakage may be relevant in the development of LARS and pelvic dysfunction, thus influencing counseling before temporary stoma closure (33, 34).
In conclusion, we found a direct leak rate of 10.9% (95% CI = 7.98 to 14.60) and an abscess leak rate of 3.8% (95% CI = 2.24 to 6.41), giving a total leak rate of 14.7% (95% CI = 11.30 to 18.81). We found no significant differences between abscess leakage and direct leakage preoperatively or intraoperatively. Postoperatively, direct leak patients were at a higher risk of requiring a reoperation, permanent stoma, and prolonged hospital stay compared to abscess leak patients. Furthermore, abscess leak patients were at a greater risk for a urinary tract infection, wound infection, and postoperative intestinal obstruction than non-leak patients, although the small number of patients in the abscess leak group warrants caution in interpreting these results. The results of this study suggest that patients with pelvic abscesses after anterior resections of rectal/rectosigmoid adenomas or adenocarcinomas could benefit from early intervention such as abscess drainage or close clinical surveillance to prevent long-term complications from chronic abscesses.
Footnotes
Acknowledgements
The authors would like to thank statistician Karl Ove Hufthammer at Haukeland University Hospital for helpful advice during this study.
Declaration Of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by the local ethical committee in accordance with hospital guidelines.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
