Abstract
Background and Aims:
In mid-rectal cancer, the low colorectal anastomosis is, although feasible, sometimes avoided. The aim was to compare low Hartmann’s procedure with intersphincteric abdomino-perineal excision of the rectum, in patients operated with total mesorectal excision for mid-rectal cancer in whom the low anastomosis was technically feasible but for patient-related reasons undesired.
Material and Methods:
A total of 64 consecutive patients with mid-rectal cancer who underwent low Hartmann’s procedure (n = 34) or intersphincteric abdomino-perineal excision (n = 30) at one colorectal unit were compared regarding patient demography, short-term oncology, surgical outcome at 3 and 24 months, and long-term overall survival.
Results:
There were no significant differences between intersphincteric abdomino-perineal excision and Hartmann’s procedure regarding age, gender distribution, body mass index, preoperative radiotherapy, tumor level, or cancer stages. Operation time was shorter in Hartmann’s procedure as compared with intersphincteric abdomino-perineal excision, median 174 and 256 min, (P < 0.001), and intraoperative blood loss was increased, 600 and 500 mL, respectively (P = 0.045). Number of lymph nodes and circumferential resection margin were comparable. In Hartmann’s procedure compared with intersphincteric abdomino-perineal excision, the need for reoperation was 24% and 3%, (P = 0.020), complications classified as Clavien–Dindo 3–4 occurred in 32% and 10%, (P = 0.031), pelvic abscess in 21% and 10%, (P = 0.313), and mortality within 90 days was 3% and 0%, respectively, (P = 0.938). In intersphincteric abdomino-perineal excision, the perineal wound was not healed at 3 months in 13%, and in Hartmann’s procedure 15% had chronic secretion from the anorectal remnant at 2 years postoperatively.
Conclusion:
The results from this study suggest that intersphincteric abdomino-perineal excision might be an alternative to Hartmann’s procedure in patients with mid-rectal cancer, in whom a low colorectal anastomosis is undesired.
Keywords
Introduction
Hartmann’s operation (HA) for rectal cancer was first described in 1921 and soon developed a reputation of being a safe procedure due to the low mortality reported in early series by Soupault and Hartmann (1, 2). HA has, for many years, been considered an appropriate surgical option in patients with rectal cancer, in whom a low anastomosis was feasible, but for patient-related reasons deemed undesirable (3–5). The reputation of being an operation with a low complication rate was questioned during the early 2000s by some authors, suggesting that dehiscence of the stapler line of the closed rectal remnant with the risk of subsequent pelvic abscess, pelvic sepsis, and possible fistula formation, was underestimated (6–8). On the contrary, in recent publications, lower rates of pelvic abscess have been reported (9, 10). In HA, long-term complications related to the persisting anorectal remnant, such as proctitis, bleeding, and excess secretion, have seldom been described in the surgical literature.
An alternative procedure to HA for rectal cancer is to excise the rectum as well as the anal canal. This operation represents one type of abdomino-perineal excision (APE) of the rectum, and the anal canal can be resected by an intersphincteric excision together with the rectum, or separately. This procedure represents one of three principally different ways to perform an APE, and the term intersphincteric abdomino-perineal excision (IS APE) was recently proposed (11), the two other APE options being the so called extralevator abdomino-perineal excision (ELAPE) (12, 13) and the ischiorectal APE (11).
At this institution, the APE with intersphincteric excision of the anal canal (IS APE) was deemed an alternative to HA and introduced in 2007. The aim of this investigation was to compare surgical outcomes in patients with mid-rectal cancer operated with IS APE or HA with total mesorectal excision (TME), in whom low anterior resection with colorectal or coloanal anastomosis would have been technically feasible, but for patient-related reasons was undesired.
Material and Methods
Included Patients
All patients consecutively operated for adenocarcinoma or adenoma with severe dysplasia of the mid rectum, with either a low HA or an IS APE including a complete TME, between January 2007 and December 2013 by the colorectal unit at the Department of Surgery, Örebro University Hospital, serving a geographically defined catchment area of 290,000 inhabitants, were included in this study Fig. 1. The decision to perform TME surgery without an anastomosis was taken by the colorectal team at the multidisciplinary conference (MDT), while the decision to perform a low HA or an IS APE was at the discretion of the colorectal surgeon in charge of the operation. Cases in which LAR was the planned procedure but where the surgeon in charge intraoperatively decided to avoid the low anastomosis and thereby converting LAR to HA or IS APE were also included. Patients with low rectal cancers in whom the anastomosis was not safe from an oncological point of view underwent ELAPE and were not included, nor were patients with rectal cancer undergoing any type of en bloc rectal resection or pelvic excenteration. Data were retrieved from the local patient files which were scrutinized. Ethical approval was obtained from regional ethical committee of the Uppsala-Örebro Healthcare region.
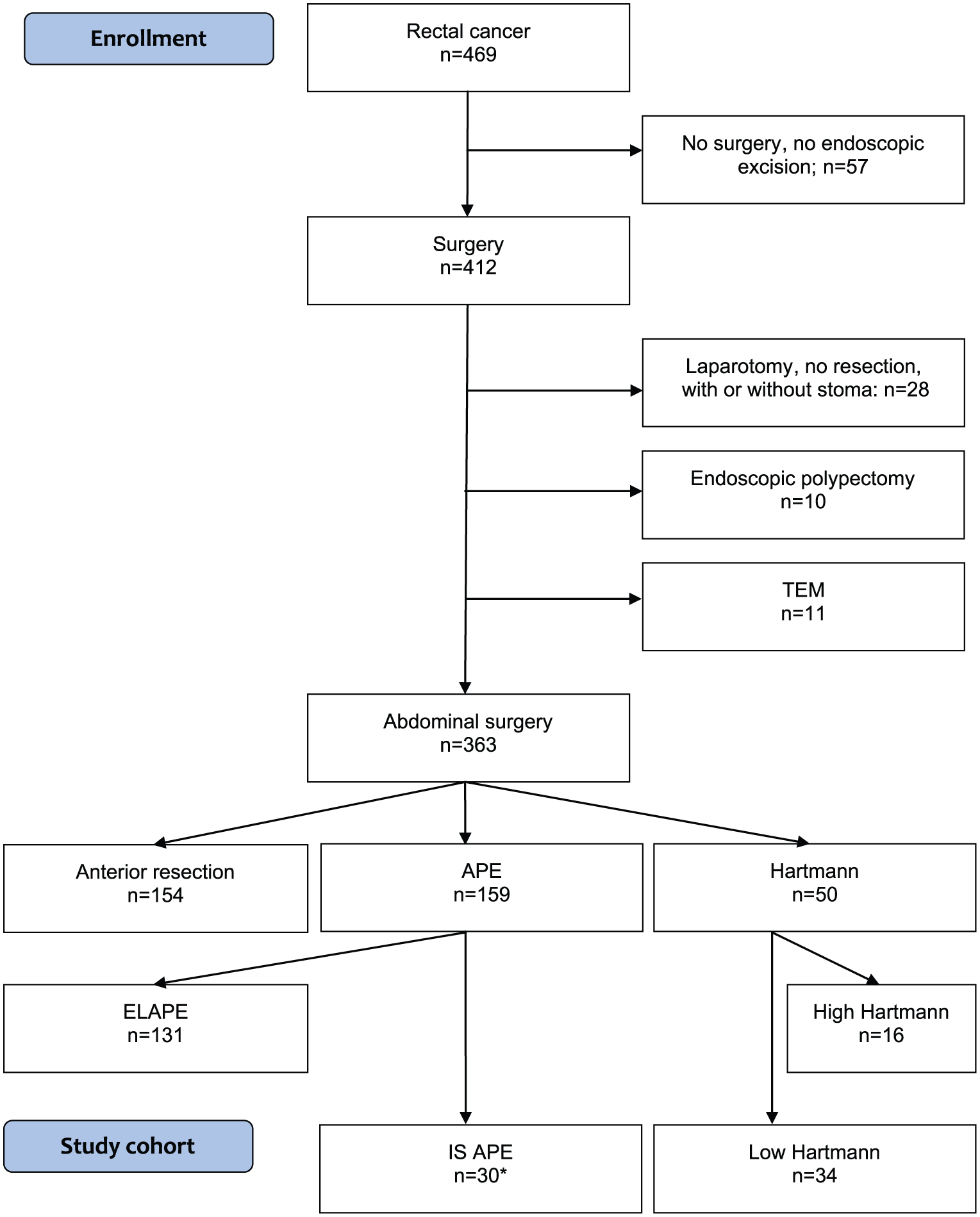
Flow chart for patients diagnosed with and operated because of rectal cancer between 2007 and 2013 at the Colorectal Unit, Örebro University Hospital, Sweden.
Surgical Technique
All operations were performed in accordance with the principles of TME (14), by open or laparoscopic technique. In HA, a TME was performed, and the rectum divided at the pelvic floor at ⩽6 cm above the anal verge. Partial mesorectal excision (PME), defined as transection of the mesorectum at ⩾7 cm above the anal verge, was not included (15, 16). The standard IS APE procedure at our institution was to perform a TME to the pelvic floor where the rectum was divided with a surgical stapler device and the specimen removed trans-abdominally, and the anal canal hereafter excised separately by the perineal approach in the supine lithotomy position. This operation can also be performed without dividing the rectum at the pelvic floor but instead performing the perineal part of the operation with an intact specimen extracted either trans-abdominally or through the perineal incision.
Included Variables
Patient-related variables were age at surgery, gender, body mass index (BMI), Union Internationale Contre le Cancer (UICC) stage, American Society of Anesthesiologists (ASA) score, and preoperative radiotherapy (RT). Perioperative variables were intraoperative bleeding, duration of surgery, pelvic drainage, and time in hospital. Histopathological variables were number of lymph nodes, distal resection margin, circumferential resection margin (CRM), and proportion of patients with positive CRM, defined as <1.0 mm.
Postoperative variables were recorded for the first 90 postoperative days and included complications classified as Clavien–Dindo 3–4 (17), reoperations, unplanned hospital readmission, and mortality for any reason. Adverse events with regard to disturbed healing and inflammation in the pelvis occurring beyond 3 months, and up until 24 months, postoperatively included pelvic abscess, perineal wound healing, chronic secretion, and fistula formation were assessed. Reasons for avoiding an anastomosis, and whether this was a preoperative decision or not, were retrieved. Overall survival at 3 years was also assessed.
Statistical Analysis
For comparison of continuous variables, Mann–Whitney U test was employed.
For comparison of proportions, chi-square test or Fischer’s exact test were used, as appropriate. Kaplan–Meier curves for 3-year overall survival were generated for HA and IS APE patients and compared using the log-rank test. Because treatment assignment was not random, we used inverse probability weighted (IPW) analyses (18) based on propensity scores to adjust the survival curves for observable differences between the two treatment groups (i.e. patients’ age, ASA score, cancer stage and whether the decision to avoid an anastomosis was made preoperative or introperatively). For the propensity score, weighted analyses, a multivariable logistic regression model was fitted with receipt of HA versus IS APE as the dependent variable and age, ASA score, cancer stage and preoperative or introperative decision to avoid anastomosis as independent variables, and predicted probabilities of receiving HA versus IS APE were calculated for each patient. Kaplan–Meier survival analyses were then inversely weighted on these propensity scores (i.e. predicted probabilities) to make the two groups more comparable. Statistical analyses were performed using Stata version 15 (Stata Corporation, College Station, Texas, USA). All statistical tests were two-sided, and P values less than 0.05 were considered statistically significant.
Results
Patient Demography
During the study period 2007–2013, a total of 363 patients underwent abdominal procedures for rectal cancer at the Örebro University Hospital, Sweden, of which 62 patients were consecutively operated with either a complete TME HA (n = 34) or an IS APE (n = 28). Another two patients with adenoma of the mid rectum with severe dysplasia were identified and included in the IS APE group (n = 30; Fig. 1). HA patients undergoing PME, defined as rectal transection at ⩾7 cm above the anal verge, were excluded (n = 16). The procedure employed for IS APE by this colorectal unit was to excise the anal canal separately, which was done in a majority of patients (28/30). Patients with low rectal cancers selected for APE because of oncological reasons were excluded (n = 131) (12, 13). With regard to patient demography, there were no statistically significant differences between the HA and IS APE groups, although some numerical differences were noted. In HA and IS APE, respectively, median age was 78 and 76.5 years, female gender in 18% and 27%, median level of tumor was 8 and 9 cm, ASA grade ⩾3 in 41% and 30%, stage III cancer in 41% and 13%, and stage IV cancer in 26% and 20%, respectively (Table 1). The decision was to avoid the low colorectal anastomosis, and thereby, LAR was taken preoperatively in 65% (22/34) of HA and in 83% (25/30) of IS APE (P = 0.092), and the reasons for avoiding LAR are presented in Table 2.
Patient demography, pathology report details, and cancer stage in patients undergoing intersphincteric abdomino-perineal excision (IS APE) or low Hartmann’s operation (HA) for mid-rectal cancer.
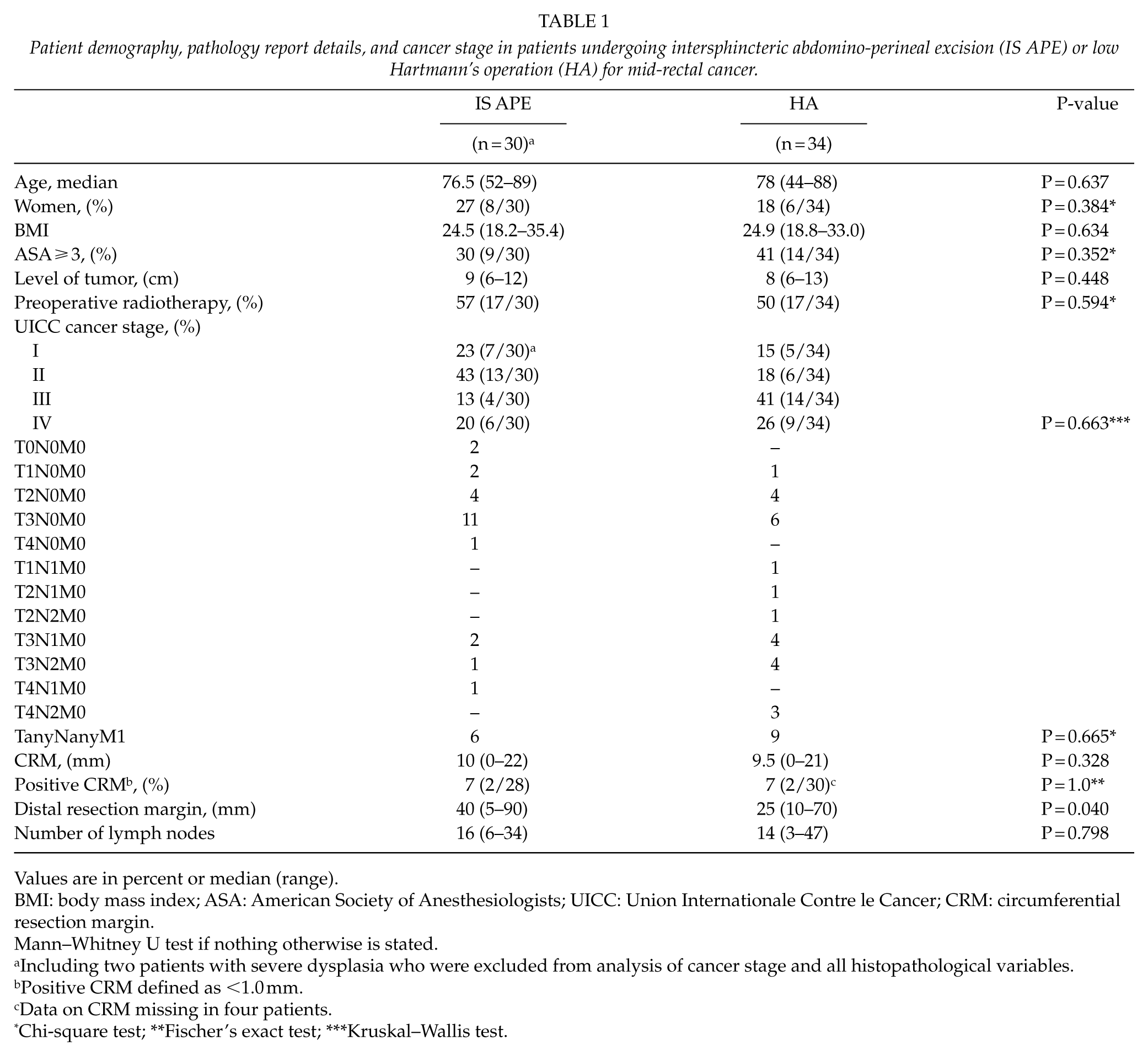
Values are in percent or median (range).
BMI: body mass index; ASA: American Society of Anesthesiologists; UICC: Union Internationale Contre le Cancer; CRM: circumferential resection margin.
Mann–Whitney U test if nothing otherwise is stated.
Including two patients with severe dysplasia who were excluded from analysis of cancer stage and all histopathological variables.
Positive CRM defined as <1.0 mm.
Data on CRM missing in four patients.
Chi-square test; **Fischer’s exact test; ***Kruskal–Wallis test.
Reasons for the decision not to perform LAR in patients undergoing TME surgery for cancer in the mid rectum operated with Hartmann’s procedure (HA) or intersphincteric abdomino-perineal excision (IS APE).
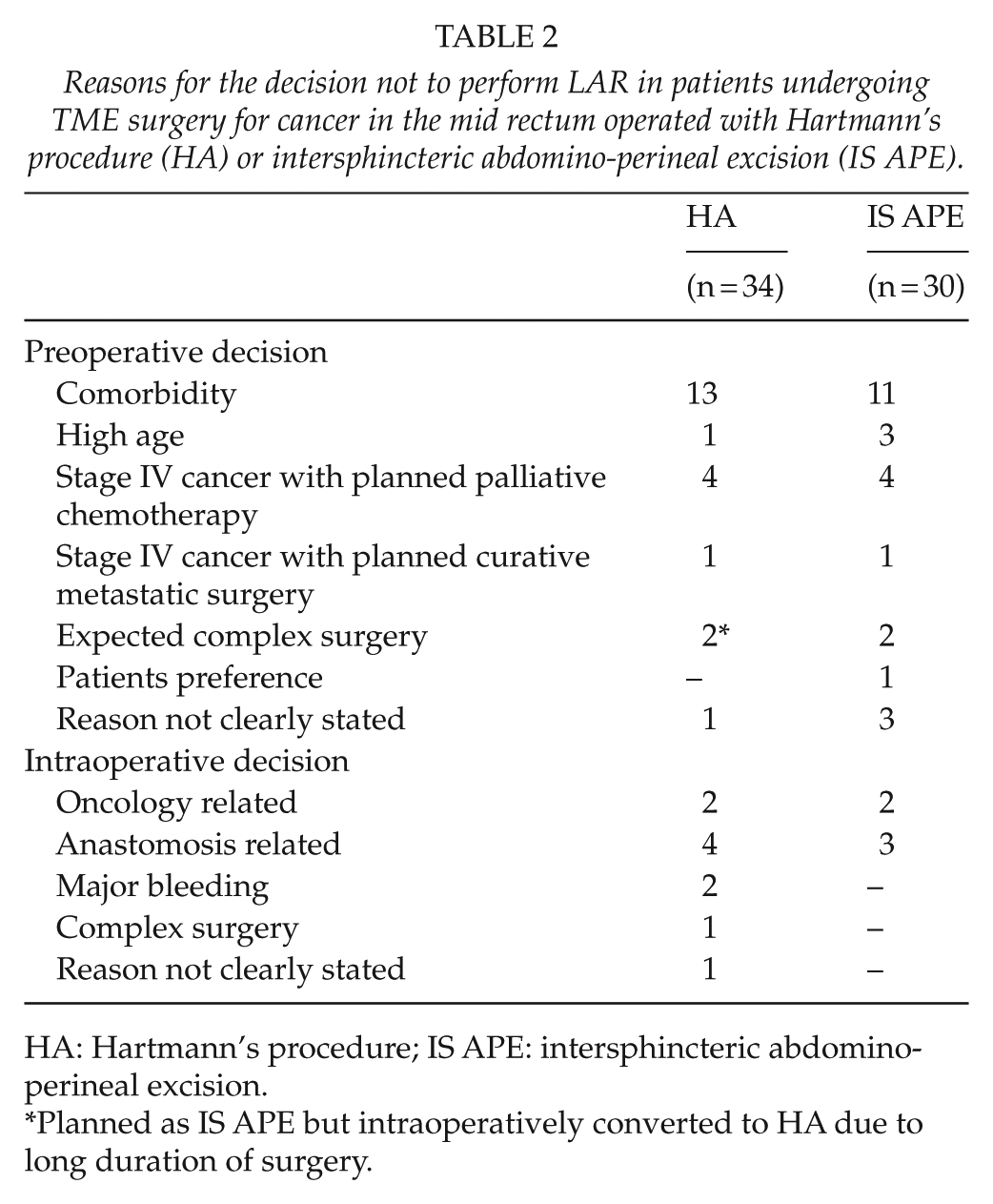
HA: Hartmann’s procedure; IS APE: intersphincteric abdomino-perineal excision.
Planned as IS APE but intraoperatively converted to HA due to long duration of surgery.
Both procedures were performed during each year of the study period with a per annum median (range) of 3 (1–10) for HA, and 4 (1–9) for IS APE. All six colorectal staff surgeons performed both procedures during the study period, HA in median (range) 6 (1–11) and IS APE in 5 (4–6), yearly.
Perioperative Details and Short-term Oncological Outcome
Duration of surgery was shorter in HA compared with IS APE, 174 and 256 min, respectively, (P < 0.001), while intraoperative bleeding was increased, 600 and 500 ml, respectively, (P = 0.045). The short-term oncological outcome was comparable regarding the number of lymph nodes, CRM, proportion of positive CRM, while the median distal margin was shorter in HA compared with IS APE, 25 and 40 mm, respectively (P = 0.040; Table 2).
Complications within 90 Days
In comparison with IS APE, HA had an increased proportion of reoperations, 24% and 3%, respectively (P = 0.020), and patients with complications classified as Clavien–Dindo 3–4, 32% and 10%, respectively (P = 0.031). There was no difference in the proportion of readmission, 15% and 10%, respectively (P = 0.713), or pelvic abscess, 21% and 10%, respectively (P = 0.313). There was no 30-day mortality in either group but mortality within 90 days was 3% in HA compared with none in IS APE (P = 0.928; Table 3).
Perioperative details and postoperative events within 90 postoperative days in patients undergoing intersphincteric abdomino-perineal excision (IS APE) or low Hartmann’s operation (HA) for mid-rectal cancer.
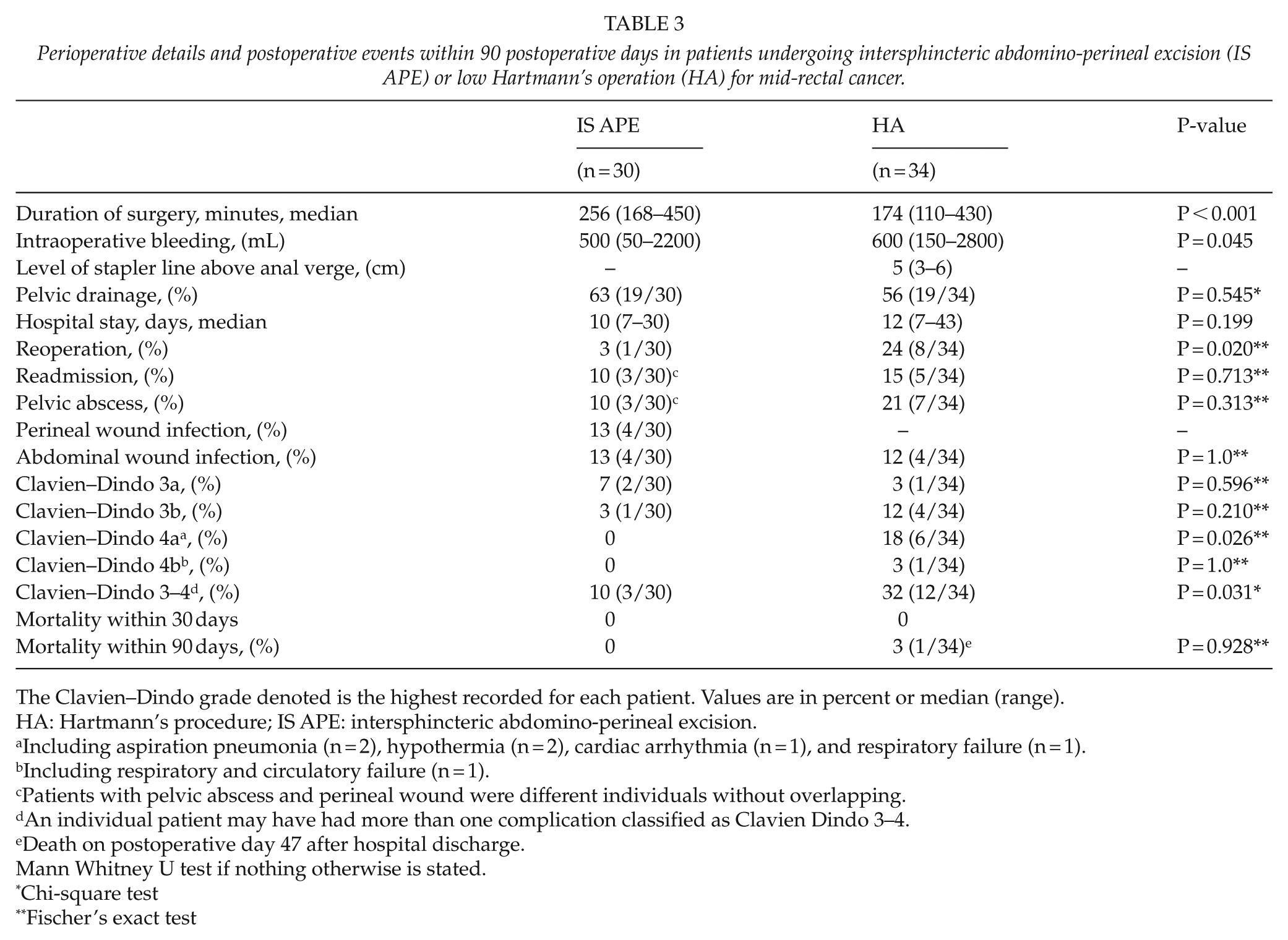
The Clavien–Dindo grade denoted is the highest recorded for each patient. Values are in percent or median (range).
HA: Hartmann’s procedure; IS APE: intersphincteric abdomino-perineal excision.
Including aspiration pneumonia (n = 2), hypothermia (n = 2), cardiac arrhythmia (n = 1), and respiratory failure (n = 1).
Including respiratory and circulatory failure (n = 1).
Patients with pelvic abscess and perineal wound were different individuals without overlapping.
An individual patient may have had more than one complication classified as Clavien Dindo 3–4.
Death on postoperative day 47 after hospital discharge.
Mann Whitney U test if nothing otherwise is stated.
Chi-square test
Fischer’s exact test
Long-term Adverse Events and Survival
In HA, a pelvic abscess not healed at 90 days was present in 9% compared with 3% in IS APE (P = 0.612). In IS APE, the perineal wound had not healed at 90 days in 13%. In HA, 15% had chronic secretion from the anorectal remnant still present at 2 years after surgery, while chronic proctitis and entero-rectal fistula was present in one patient each (Table 4).
Adverse events with regard to disturbed healing and inflammation in the pelvis occurring beyond 3 months and up until 24 months postoperatively (pelvic abscess, perineal wound healing, chronic secretion and fistula formation) in patients undergoing intersphincteric abdomino-perineal excision (IS APE) or Hartmann’s procedure (HA) for mid-rectal cancer.

Total number of individual patients in IS APE (n = 5) and in HA (n = 10). Fischer’s exact test.
Healed within 120 days (n = 2) and 150 days (n = 2).
Crude Kaplan–Meier survival curves were calculated for both treatment groups. Compared to HA, IS APE was associated with improved 3-year overall survival (log-rank test; P < 0.001; Fig. 2). This difference in overall survival was somewhat attenuated in IPW analyses with propensity score adjustment but remained statistically significant (log-rank test; P < 0.001; Fig. 3).
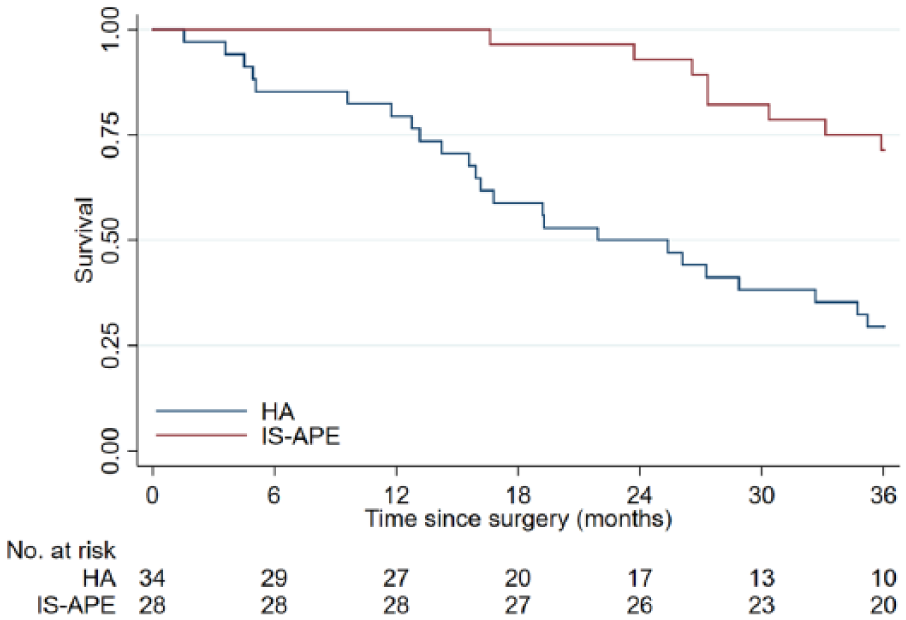
Kaplan–Meier survival curve for patients undergoing intersphincteric abdomino-perineal excision (IS APE) or Hartmann’s procedure (HA) for mid-rectal cancer. Unadjusted 3-year overall survival by surgical procedure. Patients with severe dysplasia not included (n = 2).
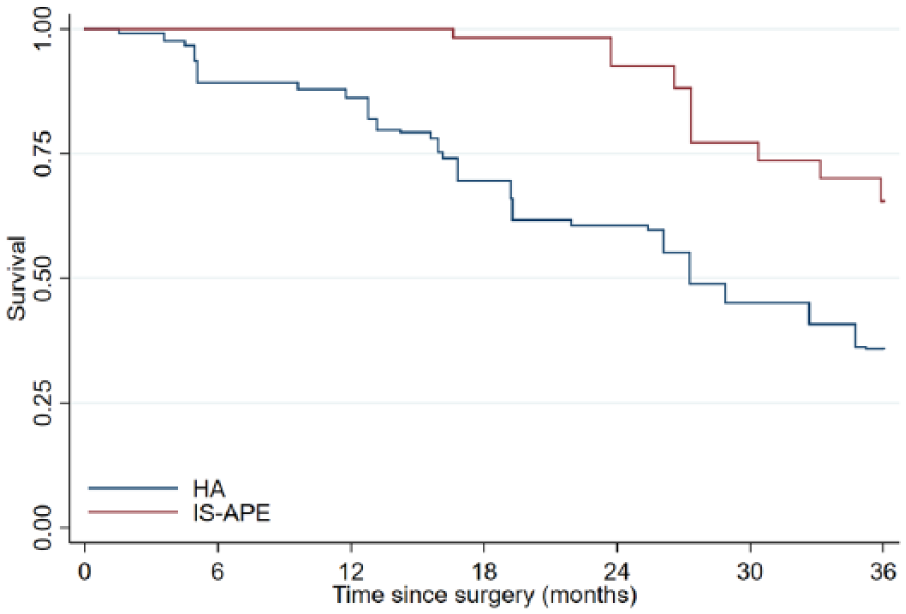
Kaplan–Meier curves for patients undergoing intersphincteric abdomino-perineal excision (IS APE) or Hartmann’s procedure (HA) for mid-rectal cancer. Adjusted* 3-year overall survival by surgical procedure in inversely weighted propensity score analyses. Patients with severe dysplasia not included (n = 2). Log-rank test; P < 0.001.
Discussion
We report a consecutive series of patients with mid-rectal cancer undergoing complete TME surgery with either a Hartmann’s procedure (HA) or an IS APE. In these patients, a low anastomosis would have been technically feasible but was deemed undesired for patient-related reasons. In this study, we found that reoperations and complications classified as Clavien–Dindo 3–4 were more common in HA. Pelvic abscess with clinical symptoms occurred in 21% of HA and in 10% of IS APE, while mortality within 90 days was low and not different between the groups. These findings should be balanced against the fact that patients undergoing IS APE tended to be younger were less often classified as ASA ⩾3, and had less advanced cancer disease, and the notion that we were unable to adjust for these between group differences due to the limited number of events in this specific analysis. However, we were able to adjust the 3-year overall survival curves for between group differences in patient characteristics by using inverse probability weighting based on propensity scores. Compared to HA, IS APE was associated with improved 3-year overall survival, and this difference was somewhat attenuated but remained statistically significant after accounting for certain differences in patient characteristics between the two groups.
We also found evidence of procedure-specific adverse events. In IS APE, the perineal wound was not healed within 3 months in 13% of the patients, although all patients were healed within 5 months after surgery. In HA, one adverse event was chronic secretion from the anorectal remnant still present 2 years after surgery in 15%.
IS APE performed for rectal cancer has previously only rarely been described in the surgical literature (19, 20). Surgical outcomes of HA for rectal cancer have been described, and when reported, adverse events are generally limited to a postoperative follow-up period of 30 days (3–10).
Töttrup et al. (7) analyzed a large series of patients undergoing HA and found pelvic abscess in 19%, although in a subgroup of low HA, defined as transection of the rectum at <2 cm above the pelvic floor, pelvic abscess was diagnosed in 33%. Frye et al. (6) compared HA with standard APE in a smaller study and found pelvic abscess in 17% of HA. Molina Rodriguez et al. (8) also compared HA with standard APE performed for oncologic reasons and found 12% pelvic abscess in HA. These three studies report rates of pelvic abscess lower but still comparable to the 21% found in this study. In contrast to these publications, Sverrisson et al. (9) recently presented a study comparing HA with standard APE for low rectal cancer and noted a rate of pelvic abscess of only 3% in the HA group. Our study is not directly comparable to the publications by Frye et al., Molina Rodriguez et al., and Sverrisson et al., since we included IS APE and exclusively low HA, while they compared standard APE performed for oncologic reasons with HA comprising both TME and PME surgery. Eriksen et al. (19) published a study with 50 rectal cancer patients undergoing IS APE and reported a rate of perineal wound infection of 20%. Recently, Westerduin et al. (20) presented a study similar to ours, comparing 40 low HA with 12 IS APE and found that pelvic abscess occurred in 10% and 17%, respectively, and that the need for reintervention was 28% and 42%, respectively. Compared with these two publications and the abovementioned variables, outcome in our study was better for IS APE and worse for HA.
Several weaknesses of this study need to be addressed. One main weakness was the limited study population size and thereby a subsequent increased risk of statistical type II errors. Another weakness was that the decision to perform HA or IS APE was at the discretion of the surgeon responsible for the operation. One weakness as compared with a randomized setting was that patients undergoing IS APE tended to be more fit and have less advanced cancer stage. Moreover, this series includes the learning curve for IS APE, which was in contrast to HA, a procedure of which the colorectal team had a long experience. This learning curve may have influenced the duration of surgery in IS APE which was increased compared with HA. Strengths of this study were that the patients originated from one defined geographical area, and were consecutively operated by the same colorectal team.
Our results suggest that IS APE might be an alternative to, or at least not inferior to, HA for patients with mid-rectal cancer in whom the low colorectal anastomosis is undesired. Disadvantages of the IS APE was the longer duration of surgery as well as the risk of healing disturbances of the perineal wound, the latter obviously not being a risk factor in HA. On the other hand, the patients operated with an IS APE are spared the relatively frequent and potentially chronic morbidity in HA induced by the persisting anorectal remnant.
This study cannot give a definite answer with regard to whether IS APE or HA is the method of choice for this category of patients with mid-rectal cancer. To approach the question of whether IS APE or HA is the preferable surgical method, a randomized controlled multicenter trial is needed. At present, there is such an ongoing trial, HAPIrect, which hopefully will be able to answer this question (21).
Footnotes
Acknowledgements
The authors thank Judith S. Brand, Clinical Epidemiology and Biostatistics, School of Medical Sciences, Örebro University, Örebro, Sweden, for statistical advice. This work was presented in parts at the ESCP, Berlin, Germany, 20–22 September 2017.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by the Ethical committee of the Uppsala-Örebro Healthcare region.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the Örebro County Council, Örebro, Sweden (Grant No. OLL-615851).
