Abstract
Background and Aims:
Stenosis due to intimal hyperplasia and restenosis after initially successful percutaneous angioplasty are common reasons for failing arteriovenous fistulas. The aim of this study was to evaluate the effect of drug-coated balloons in the treatment of arteriovenous fistula stenosis.
Design:
Single-center, parallel group, randomized controlled trial. Block randomized by sealed envelope 1:1.
Materials and Methods:
A total of 39 patients with primary or recurrent stenosis in a failing native arteriovenous fistulas were randomized to drug-coated balloon (n = 19) or standard balloon angioplasty (n = 20). Follow-up was 1 year. Primary outcome measure was target lesion revascularization.
Results:
In all, 36 stenoses were analyzed; three patients were excluded due to technical failure after randomization. A total of 88.9% (16/18) in the drug-coated balloon group was revascularized or occluded within 1 year, compared to 22.2% (4/18) of the stenoses in the balloon angioplasty group (relative risk for drug-coated balloon 7.09). Mean time-to- target lesion revascularization was 110 and 193 days after the drug-coated balloon and balloon angioplasty, respectively (p = 0.06).
Conclusions:
With 1-year follow-up, the target lesion revascularization-free survival after drug-coated balloon-treatment was clearly worse. The reason for this remains unknown, but it may be due to differences in the biological response to paclitaxel in the venous arteriovenous fistula-wall compared to its antiproliferative effect in the arterial wall after drug-coated balloon treatment of atherosclerotic occlusive lesions. Trial registration: ClinicalTrials.gov NCT03036241
Keywords
Introduction
An important cause of increased morbidity and hospitalization of hemodialysis patients is failing dialysis due to stenotic lesions of the vascular access. These stenotic lesions are traditionally treated with balloon angioplasty (BA), although repeat interventions due to recurrent stenosis are often needed. BA causes intimal injury at the treated site, which in turn is associated with neointimal hyperplasia and eventually restenosis (1). The hemodynamic and biological circumstances in venous bypass grafts resemble those in arteriovenous fistulae in the sense that in both situations, a superficial vein is exposed to arterial circulation. However, in the venous bypass graft the flow is highly pulsatile but with low volume flow due to high peripheral resistance, whereas in the fistula flow velocity is high as the resistance in the venous circulation is low (2). DCBs have been studied in AVFs and vein grafts, but to date, very little data have been published in this field and the results remain inconclusive (3–5).
Intimal hyperplasia causing restenosis after BA is a common problem with AVFs. This occurs usually in the venous segment of the fistula, and leads to prolonged or failed maturation, failed dialysis and potential occlusion, and loss of the access (6). The process of intimal hyperplasia and subsequent stenosis in AVFs seems to be biologically more complex than that in peripheral arterial disease (PAD), and has been linked to a number of variables. These include, among others, age, surgical technique and trauma, shear stress, uremia, hypoxia, smoking, cytomegalovirus infection, diabetes, body-mass index, plasma cholesterol, and other vascular disease (7–10). The molecular pathways of these factors are suspected to be similar (11).
Drug-coated balloons (DCBs) deliver a drug, usually paclitaxel, to the vessel wall during balloon expansion. Paclitaxel inhibits cell mitosis and thus neointimal hyperplasia. It has been associated with better patency rates in coronary arteries and to some degree in manifestations of PAD (12–14).
The purpose of this randomized, prospective controlled trial is to investigate potential benefit from DCBs in failing autologous arteriovenous dialysis fistulae. The study was approved by the Ethical Committee of Helsinki and Uusimaa Hospital District (297/13/03/02/2012). This article reports the results of a registered study, which can be accessed at ClinicalTrials.gov
Methods
Patients were randomized between August 2013 and February 2016. Patients presented by referral from the dialysis unit after suspected stenosis following difficult or failed dialysis or occlusion of the AVF. All patients provided informed written consent before inclusion. Inclusion and exclusion criteria are listed in Table 1. All lesions were treated by certified interventional radiologists. All lesions were in the extra-anastomotic venous segment. No central vein stenoses were included. After ultrasonographic and/or angiographic confirmation of significant venous stenosis the AVF was accessed from an arterial or venous puncture. The patients were routinely administered 5000 IU heparin. The lesion was crossed and predilated for 90 s using an undersized (1 mm below target) plain balloon. A research nurse performed randomization after the predilatation. The stenosis was then treated again with DCB or BA according to randomization (90 s). The DCB used in this trial had a paclitaxel dose of 3.5 µg/mm2 and used urea as excipient (Medtronic IN.PACT, Medtronic, Minneapolis, MN, USA). Technical success was defined as residual stenosis <30%. No high-pressure balloons were used for dilatation. All patients were started on dual antiplatelet therapy (DAPT) for 1 month postoperatively: ASA 100 mg + clopidogrel 75 mg, or warfarin + ASA 50 mg if the patient was already on warfarin. Patients and outcome assessors were blinded to the assigned treatment. Ultrasonographical follow-up was performed by dedicated research nurses immediately after the procedure and at 1, 6 and 12 months. Reinterventions were prompted by recurrent problems with dialysis only. Primary endpoint was any revascularization due to the same lesion (target lesion revascularization, TLR) and any loss of the AV-fistula.
Inclusion and exclusion criteria.
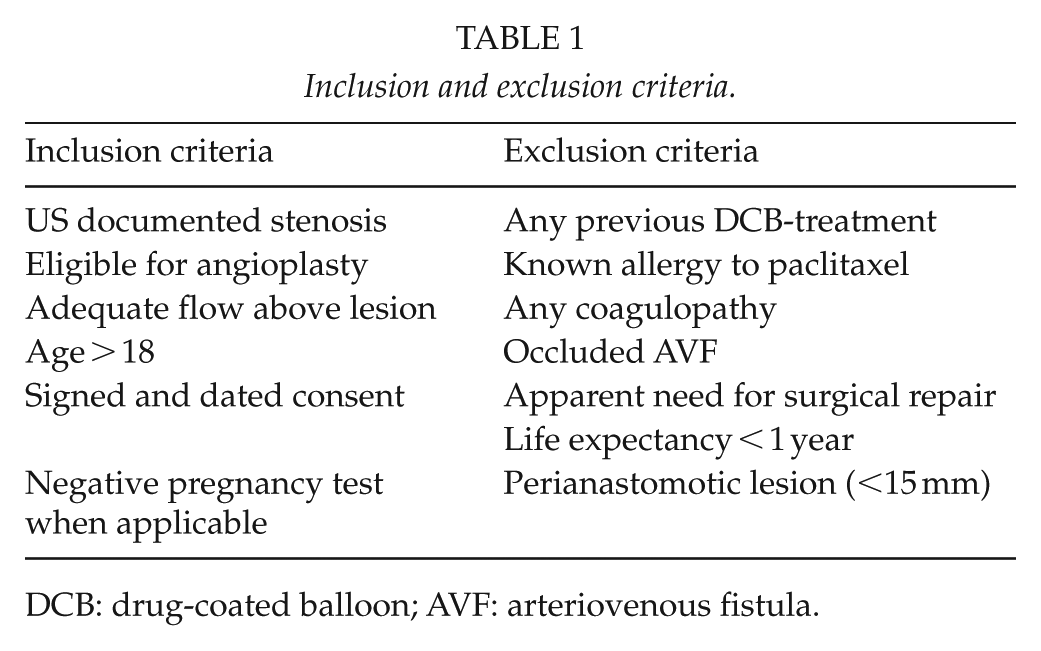
DCB: drug-coated balloon; AVF: arteriovenous fistula.
Based on the published literature on arterial stenosis at the time of trial design, the hypothetical 12-month TLR-rate for the BA and DCB groups is 30% and 10%, respectively (15). With a two-sided 5% significance level and a statistical power of 80%, the necessary sample size was 140 (70 + 70).
Randomization
1:1 block randomization by sequenced concealed envelopes was done after successful crossing of lesion and predilatation. Patient inclusion and allocation was performed by a research nurse.
Statistical Analysis
Statistical analysis was performed with SPSS V.22 (IBM, Armonk, VA, USA). Baseline characteristics were analyzed by Mann–Whitney testing. Log-rank (Mantel-Cox) calculations were used for survival analysis, and as the sample size was small, the TLR-rates were further compared using nonparametric Mann–Whitney testing. Relative risk (RR) was determined by RR = (a/(a + b))/(c/(c + d)). 95% confidence intervals (CI) were calculated using SPSS software. There were no missing TLR data. Missing data for baseline homogeneity was handled by pairwise deletion.
Results
A total of 39 patients were randomized. Baseline patient characteristics are shown in Table 2. The CONSORT flow chart for inclusion can be seen in Fig. 1. The study was discontinued due to slow recruitment. No interim statistical analyses were performed prior to the discontinuation. In cases with duplicate lesions, only the most proximal (with regard to direction of catheterization) was included in the analysis. Three patients were excluded due to aborted intervention after randomization, as the predilated lesion could not be crossed with the stiffer DCB-catheter. Thirty-six patients were eventually analyzed. Mean total time of inflation was 278 s in the DCB group and 274 s in the DCB group (p = 0.84). The respective mean balloon sizes were 4.9 mm (DCB) and 5.7 mm (BA; p = 0.07). 4/18 (22.2%) of the stenoses in the BA group reached an endpoint within 1 year, compared to 16/18 (88.9%) in the DCB group (RR 7.09, 95% CI 0.10–6.73, p = 0.001). Mean time-to-endpoint was 110 (95% CI 71–150) and 193 (95% CI 74–311) days in the DCB and BA groups, respectively (p = 0.06). TLR-free patient numbers can be seen in Fig. 2. In a sensitivity analysis including the three patients excluded due to aborted intervention, the difference remained clearly significant regardless of the allocation of the excluded patients (p < 0.001 for both scenarios). In all, 18 of the primary endpoints were re-PTAs (3 BA, 15 DCB), 2 surgical repairs (1 BA, 1 DCB), and 1 AVF-occlusion (DCB).
Baseline population characteristics.
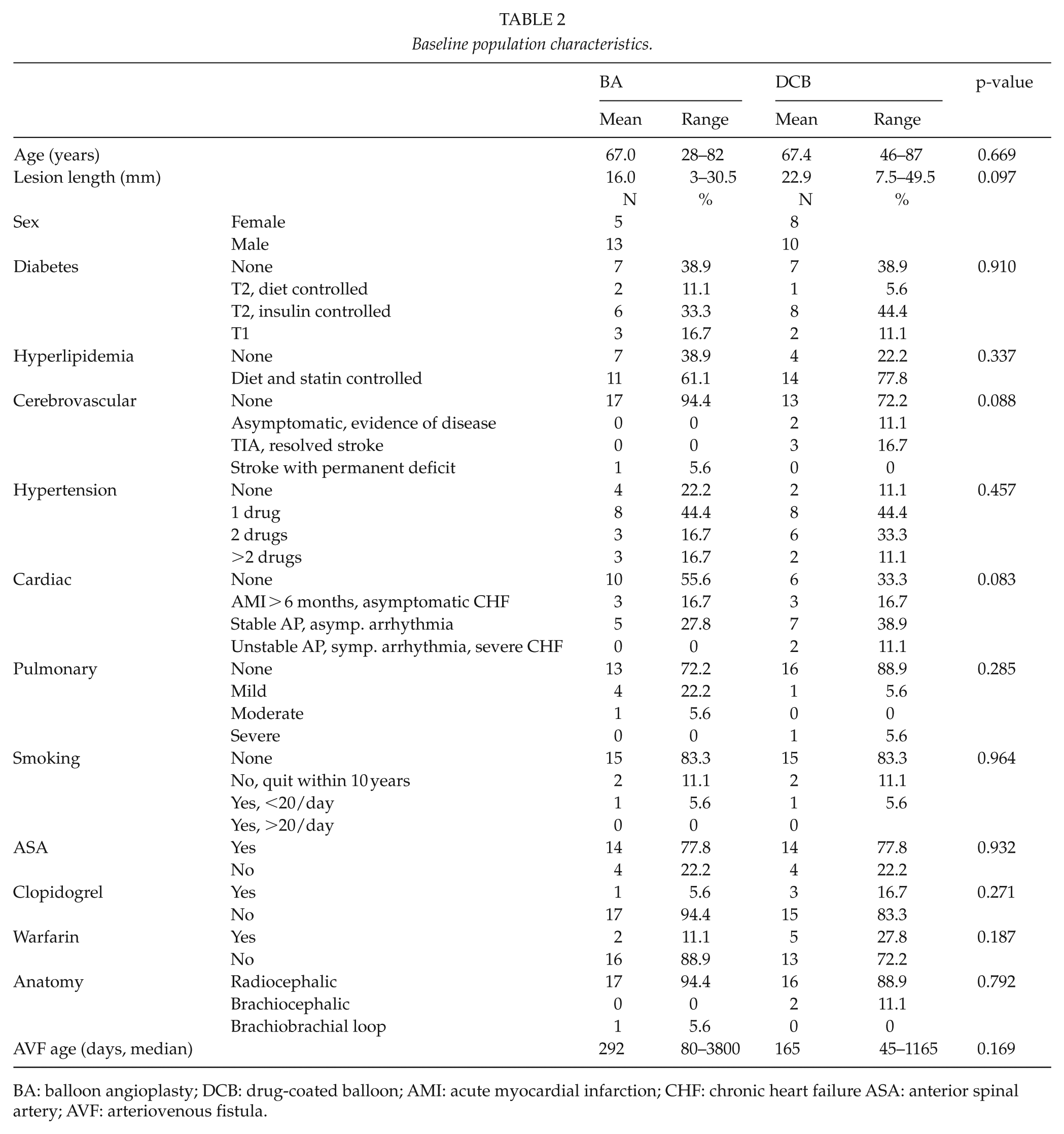
BA: balloon angioplasty; DCB: drug-coated balloon; AMI: acute myocardial infarction; CHF: chronic heart failure ASA: anterior spinal artery; AVF: arteriovenous fistula.
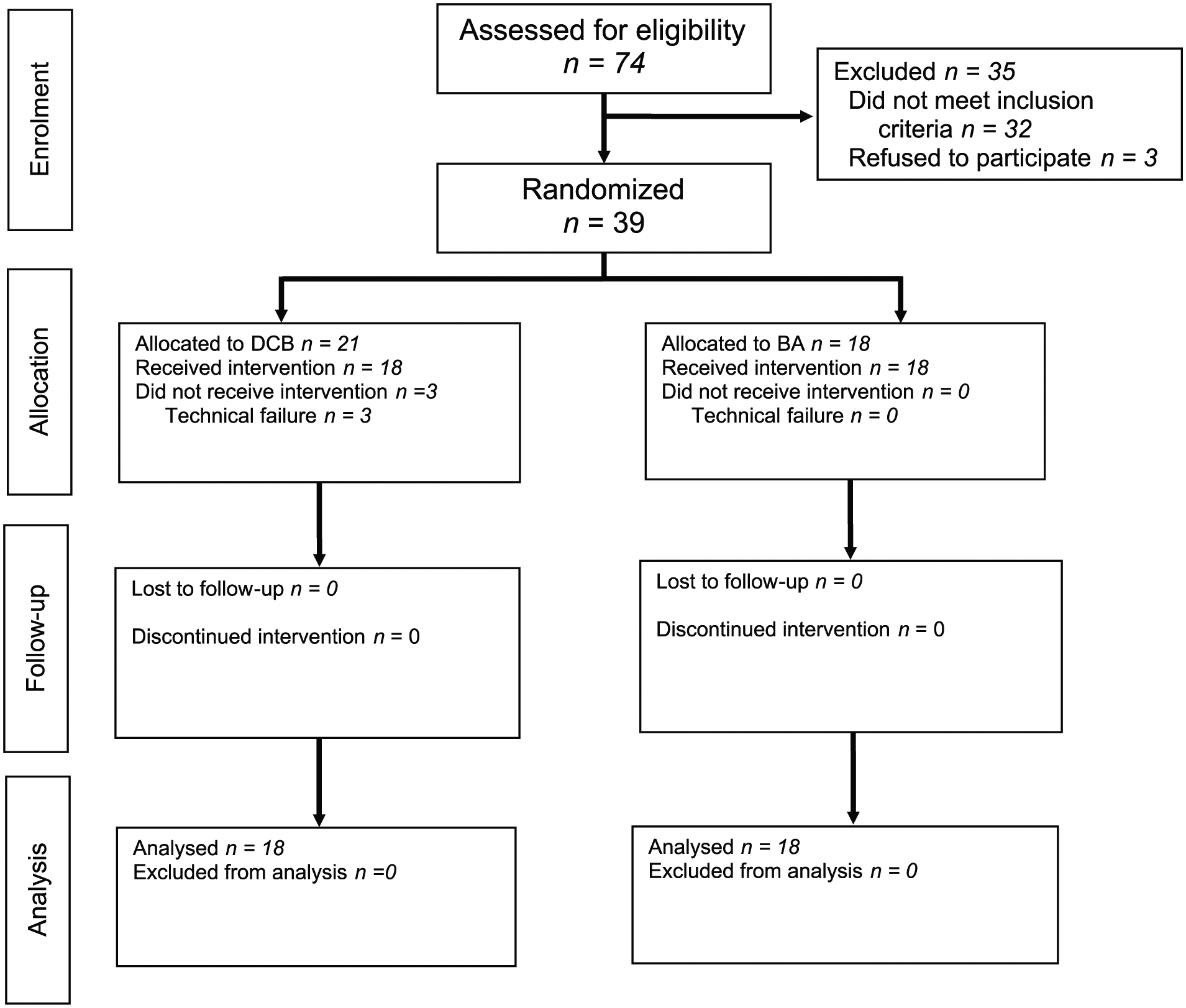
CONSORT flow diagram.
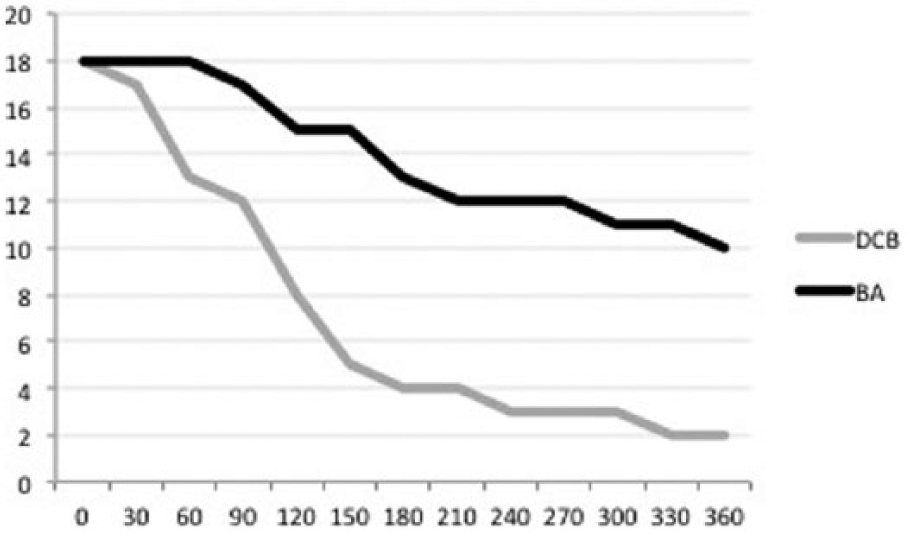
Survival plot for intervention-free survival.
Five patients died during follow-up (four BA, one DCB). The four in the BA group died with an open AVF on average at 145 (100–180) days after the intervention, whereas the one in the DCB group died after re-revascularization at 240 days after the index intervention. Two patients, both from the BA group, were lost to follow-up: one due to withdrawal of consent after randomization and the other for unknown reasons after re-revascularization. TLR-data from these cases could still be included in the analysis, although they did not attend follow-up appointments. Two (one BA, one DCB) patients received a functioning kidney transplant within 1 year of inclusion. As this study addressed patency, the AVFs of these patients were still included in the study, even if dialysis was ceased. In an ad hoc subanalysis, there was no difference between outcomes with regard to previous PTA (p = 0.681).
Discussion
Stenotic AVF-lesions are rarely calcified, and could more often be secondary to neointimal hyperplasia than arterial stenoses. We hypothesized that this study design would demonstrate clinical benefit from hyperplasia suppression by paclitaxel. Our study did, on the contrary, demonstrate harm from DCBs.
The retrospective studies on the use of DCBs in AVFs have generally shown promising results in favor of DCB, although the sample sizes have been limited (16–18). A systematic review of the small randomized and nonrandomized studies published have so far shown increased patency at 6 months after DCB, but this difference vanished at 12 months (19). This review included 254 interventions in 162 patients, and as the material was considered clinically heterogeneous, clinically applicable conclusions could not be drawn.
One prospective randomized trial did show significant benefit from the use of DCBs over plain BA at 6 and 12 months (20, 21). The trial by Kitrou et al. showed 1-year primary lesion patency of 5% (BA) versus 35% (DCB). In our study the corresponding numbers were 75% and 10.5%, respectively. The mean baseline age of the fistulas in our trial was much shorter (6 months vs 2.5 years). This is probably significant and may explain the dramatic differences in outcome. Furthermore, the Kitrou trial included 65% prosthetic AV-grafts, and is thus not directly comparable to our design. In an earlier, small retrospective study we saw possible short-term benefit from DCBs in AV-fistulas (22).
The peripheral arterial trials usually show benefit from the DCB at 6–12 months after intervention, which is consistent with the development of neointimal hyperplasia. In our study, the negative slope of the survival plot for DCBs is quite steep from very early stages of follow-up. This suggests that the difference is not brought on by neointimal hyperplasia, but rather that something damages the AVF immediately after the intervention in the DCB treatment group.
Paclitaxel is an antiproliferative agent that is widely used in cancer treatment. Its potential toxic and inflammatory effect on arterial walls has been studied in animal models, with inconclusive results and unpredictable uptake patterns (23, 24). The results in our study strongly suggest that using DCBs in native AVF can even be harmful, at least in “young,” more immature AVFs created less than 1 year before the intervention. The exact reason for this is unknown, but the complex biological and hemodynamic circumstances probably play a role. Another strong hypothesis is that treating a stenosis in a fairly young and immature AVF practically means essentially treating a venous wall instead of an arterial one, and the thinner venous wall could be more susceptible to the local over dosage and potential toxic effects of paclitaxel. No data have been published on the use of DAPT after endovascular treatment of AVF lesions, and hypothetically the intimal recovery can be slower after DCB, thus benefiting from a more aggressive postoperative antithrombotic regime. This is further complicated by the altered pharmacodynamics of ASA and clopidogrel in patients with chronic kidney disease. One randomized trial showed potential benefit from use of prolonged DAPT after coronary stenting (both drug-eluting and bare metal stents; 25).
This study is limited by its small sample size and the slow rate of randomization. We aimed at a significantly higher number of patients. The fact that a majority of the patients had a perianastomotic stenosis, which was an exclusion criterion in our study, was a major cause for the slow randomization. However, even with a small number of patients the results seem unambiguous. Despite the clear difference in outcome, we cannot exclude the risk for type I error. The power calculation was performed with the assumption that DCBs would lead to clinical benefit, and is rendered pointless by the diametrically opposite results. In fact, using the RR from this study to perform a power calculation, a trial would only need eight events during follow-up to demonstrate the superiority of BA over DCB. Furthermore, if we had done an interim analysis in our study, we would have been obliged to interrupt it due to the significantly poorer outcome in the DCB group.
Conclusions
In conclusion, with 1-year follow-up, the outcome after DCB-treatment compared to conventional BA was clearly worse. The reason for this remains unknown, and probably has to do with the different biological response to paclitaxel in the venous AVF-wall compared to the well-researched arterial wall. Based on this trial, DCB-use in AVF-stenoses, especially in the recently created and/or immature AVF, should be discouraged. Further research should be conducted to investigate the biological and clinical response to paclitaxel in different stages of AVF maturation and age.
Footnotes
Acknowledgements
The authors would like to thank Dr Maria Söderström for help with planning and translation of patient forms, and research nurses Anita Mäkela and Anne Blumen for conducting the inclusion of patients and ultrasonographical follow-up.
Authors’ Note
The abstract has been presented at the Annual Meeting of the European Society for Vascular Surgery in Lyon in September 2017 (both fast-track and qualified rounds). It was also presented at the VEITH Symposium in New York in November 2017.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Committee of Helsinki and Uusimaa Hospital District (297/13/03/02/2012)
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
All patients included in this trial provided written informed consent.
