Abstract
Background and Aims:
The lungs participate in the modulation of the circulating inflammatory factors induced by coronary artery bypass grafting. We investigated whether aprotinin—which has been suggested to interact with inflammation—influences lung passage of key inflammatory factors after coronary artery bypass grafting.
Material and Methods:
A total of 40 patients undergoing coronary artery bypass grafting were randomized into four groups according to aprotinin dose: (1) high dose, (2) early low dose, (3) late low dose, and (4) without aprotinin. Pulmonary artery and radial artery blood samples were collected for the evaluation of calculated lung passage (pulmonary artery/radial artery) of the pro-inflammatory factors interleukin 6 and interleukin 8, 8-isoprostane, myeloperoxidase and the anti-inflammatory interleukin 10 immediately after induction of anesthesia (T1), 1 min after releasing aortic cross clamp (T2), 15 min after releasing aortic cross clamp (T3), 1 h after releasing aortic cross clamp (T4), and 20 h after releasing aortic cross clamp (T5).
Results:
Pulmonary artery/radial artery 8-isoprostane increased in patients with high aprotinin dose as compared with lower doses (1.1 range 0.97 vs 0.9 range 1.39, p = 0.001). The main effect comparing high aprotinin dose with lower doses was significant (F(1, 38) = 7.338, p = 0.01, partial eta squared = 0.16) further supporting difference in the effectiveness of high aprotinin dose for pulmonary artery/radial artery 8-isoprostane.
Conclusion:
According to the pulmonary artery/radial artery equation, the impact of aprotinin on 8-isoprostane after coronary artery bypass grafting is dose dependent. Aprotinin may aid the lung passage of circulating factors toward a beneficial anti-inflammatory milieu.
Introduction
Coronary artery bypass grafting (CABG) with cardiopulmonary bypass (CPB) induces a systematic inflammation depicted by the release of oxygen-free radicals, circulating cytokines, and other inflammatory factors (1–3). Circulating cytokines such as interleukin (IL) 6, IL8, and IL10 interact with the vasculature during inflammation (1, 3, 4). Surgical trauma, including CPB and aortic cross-clamping, induces ischemia–reperfusion injury. Ischemia–reperfusion injury is associated with increased production of reactive oxygen species that reflect the production of prostaglandins, 8-isoprostane (5), and myeloperoxidase (MPO, (6)).
We have previously demonstrated that CABG impacts on pulmonary vascular resistance upon inflammation (7), which determines the state of the vascular endothelial bed of the lungs. The lungs interact with the circulating cytokines and other inflammatory factors after CABG by filtrating, metabolizing, and producing inflammatory agents (2, 8). The net inflammatory profile of the patient is significantly dependent on the impact of the lungs. Minimal pulmonary artery (PA) blood flow during CPB affects the severity of endothelial reperfusion injury of the lungs (9–11). The net effect of the lungs may be estimated by comparing the concentrations of the inflammatory factors in the peripheral radial artery (RA) blood sample to those in the simultaneous PA blood sample (1, 7). As being in between these two samples, the pulmonary vasculature may reflect the overall effect of the lungs as expressed by the PA versus RA (PA/RA) value of the cytokines (1, 7).
Aprotinin has emerged as a potential anti-inflammatory agent (12, 13). Aprotinin inhibits serine protease, thromboxane A, and the kallikrein–kinin pathway in a dose-dependent manner (12). Aprotinin lowers pulmonary vascular resistance, thus preserving the endothelial lining of the pulmonary vasculature (14).
In the present study, we investigated whether aprotinin administered during CABG impacts lung passage of some key inflammatory factors. With the aid of the PA/RA equation, IL6, IL8, IL10, MPO, and 8-isoprostane were investigated in patients with CABG treated with high aprotinin dose and compared with patients with lower aprotinin regimen.
Materials and Methods
Patient Population
The study was approved by the Tampere University Hospital Ethics Committee (R06173M, 9/16/2006) complying with the principles laid down in the Declaration of Helsinki (recommendations guiding physicians in biomedical research involving human subjects, adopted by the 18th World Medical Assembly, Helsinki, Finland, June 1964), and all patients gave written consent. The National Agency for Medicines was consulted for the approval to use aprotinin (11/15/2007). Forty elective primary CABG patients were enrolled in the study. Randomization was made using sealed blinded envelopes as previously described according to the aprotinin dose (14). Briefly, four groups of patients were scheduled for elective CABG using on pump CPB technique: (1) a high aprotinin dose group receiving 280 mg (2 × 106 kallikrein inactivation unit (KIU) of aprotinin immediately after induction of anesthesia, followed by 280 mg in pump prime, and a constant infusion of 70 mg/h (5 × 105 KIU) until chest closure (high aprotinin dose); (2) an early low aprotinin dose group receiving 280 mg after induction, and 140 mg in pump prime (early low aprotinin dose); (3) a late low aprotinin dose group with 280 mg in pump prime and 70 mg/h infusion until chest closure (late low aprotinin dose); (4) a control group treated with a constant infusion of 500 mg of tranexamic acid just before chest closure (no aprotinin). In all patients, a manual recruitment maneuver was carried out at the end of CPB, keeping airway pressure up to 40 cmH2O for 15 s. Thereafter, a positive end-expiratory pressure (PEEP) level of 5 cmH2O was applied. The exclusion criteria were acute coronary syndrome or myocardial infarction during the previous month, pre-existent pulmonary disease, pulmonary hypertension with systolic pulmonary arterial pressure (PAP) of >40 mmHg, renal insufficiency, previous ischemic cerebral event, smoking, use of corticosteroids or nonsteroidal anti-inflammatory drugs or cyclooxygenase-2 (COX-2) inhibitors, use of angiotensin-converting-enzyme inhibitors in diabetics or known hypersensitivity to aprotinin. Anesthesia was induced with propofol (0.5–1.0 mg/kg) and sufentanil (0.8–1.0 µg/kg) and rocuronium was used for the induction of anesthesia. Halogenated anesthetic gases were avoided due to possible influence on pulmonary hemodynamics. Occasional hypertensive episodes were controlled with nitroglycerin or labetalol.
Hemodynamic Measurements
Technically, surgery was uneventful for all patients. As previously reported, there were no differences between the groups considering preoperative variables (14). Postoperatively, there were no statistically significant differences between the groups observed at any time-point in terms of oxygenation index, alveolar–arterial oxygen gradient, and pulmonary shunt. Pulmonary vascular resistance index (dynes s cm−5) remained low in patients receiving aprotinin as compared without aprotinin despite any correlation with the cytokines.
Sample Collection and Elisa Measurements of IL6, IL8, IL10, 8-Isoprostane, and MPO
For evaluation of IL6, IL8, IL10, 8-isoprostane, and MPO, arterial blood was collected from both RA and PA immediately after induction of anesthesia (T1), 1 min after releasing aortic cross clamp (T2), 15 min after releasing aortic cross clamp (T3), 1 h after releasing aortic cross clamp (T4), and 20 h after releasing aortic cross clamp (T5). The samples were available from all 40 patients at every time-points mentioned. All samples were anticoagulated with ethylenediaminetetraacetic acid, immediately cooled at 4 °C, and centrifuged within 30 min (1200 × g for 10 min); plasma was transferred to polypropylene test tubes and stored at −70 °C until analyzed. IL6, IL8, IL10, and 8-isoprostane levels in plasma samples were determined by enzyme immunoassay (EIA) using commercial reagents (IL6 and IL10: PeliPair ELISA, Sanquin, Amsterdam, the Netherlands; IL8: OptEIA BD Biosciences, Erembodegem, Belgium; 8-isoprostane: Cayman Chemical Company, Ann Arbor, MI, USA; MPO was determined by radioimmunoassay (RIA): Pharmacia AB, Uppsala, Sweden). No adjustment was made for hemodilution. The detection limits were 0.3 pg/mL for IL6 and IL10 and 0.8 pg/mL for IL8. For MPO and 8-isoprostane, the detection limits were 0.8 and 10 pg/mL, respectively.
Statistical Analysis
Data are presented as median (range) and expressed as pg/mL. The ratio of blood cytokine and 8-isoprostane concentration measured in the PA and the RA and adjusted to baseline describes the net production or elimination during passage through the lungs (1). PA/RA > 1 indicates a decrease of the inflammatory factors (e.g. due to metabolism or diffusion) in the lungs, while PA/RA ≤ 1 indicates that the factors are released from the lungs (1). Statistical analysis was performed with SPSS software (SPSS Inc, Chicago, IL, USA). A nonparametric Kruskal–Wallis test was employed to account for comparison of changes in cytokine and 8-isoprostane levels between all groups at different time-points. The observed changes in levels of cytokines and 8-isoprostane in patients with high aprotinin dose were compared with other doses and within all groups at different time-points by the nonparametric Mann–Whitney U test. The predictive value of 8-isoprostane to indicate patients who received high aprotinin dose was assessed by receiver operating characteristic (ROC) curve. Test of normality was assessed by the Shapiro–Wilk test. Repeated measure test was applied to study the interaction of the aprotinin doses and the five time-points on the PA/RA 8-isoprostane levels. A mixed between-within subjects analysis of variance was conducted to assess the impact of the different aprotinin doses including high dose, early low dose, late low dose, and no dose on the PA/RA cytokine levels, across the five time-points. P-values less than 0.05 were considered statistically significant.
Results
Cardiac Index
There was a significant difference in cardiac index at T1 between patients receiving early low aprotinin dose and the other groups (p = 0.043). The mean preoperative left ventricular ejection fraction in every group was normal and the median (range) values of cardiac indexes at T1 in every group were high reflecting normal hemodynamics at baseline (3.5 range 2.1 in high aprotinin dose, 2.7 range 0.8 in early low aprotinin dose, 3.0 range 1.7 in late low aprotinin dose, and 2.8 range 2.2 in no aprotinin).
Levels of Cytokines, MPO, and 8-Isoprostane
An increase of IL6, IL8, IL10, MPO, and 8-isoprostane was observed in all groups in both RA and PA samples (Tables 1 and 2). There were no differences in either RA or PA samples among the groups.
Perioperative and postoperative pulmonary artery cytokines IL6, IL8, IL10, MPO, and isoprostane-8 in patients undergoing coronary artery bypass surgery with different aprotinin doses.
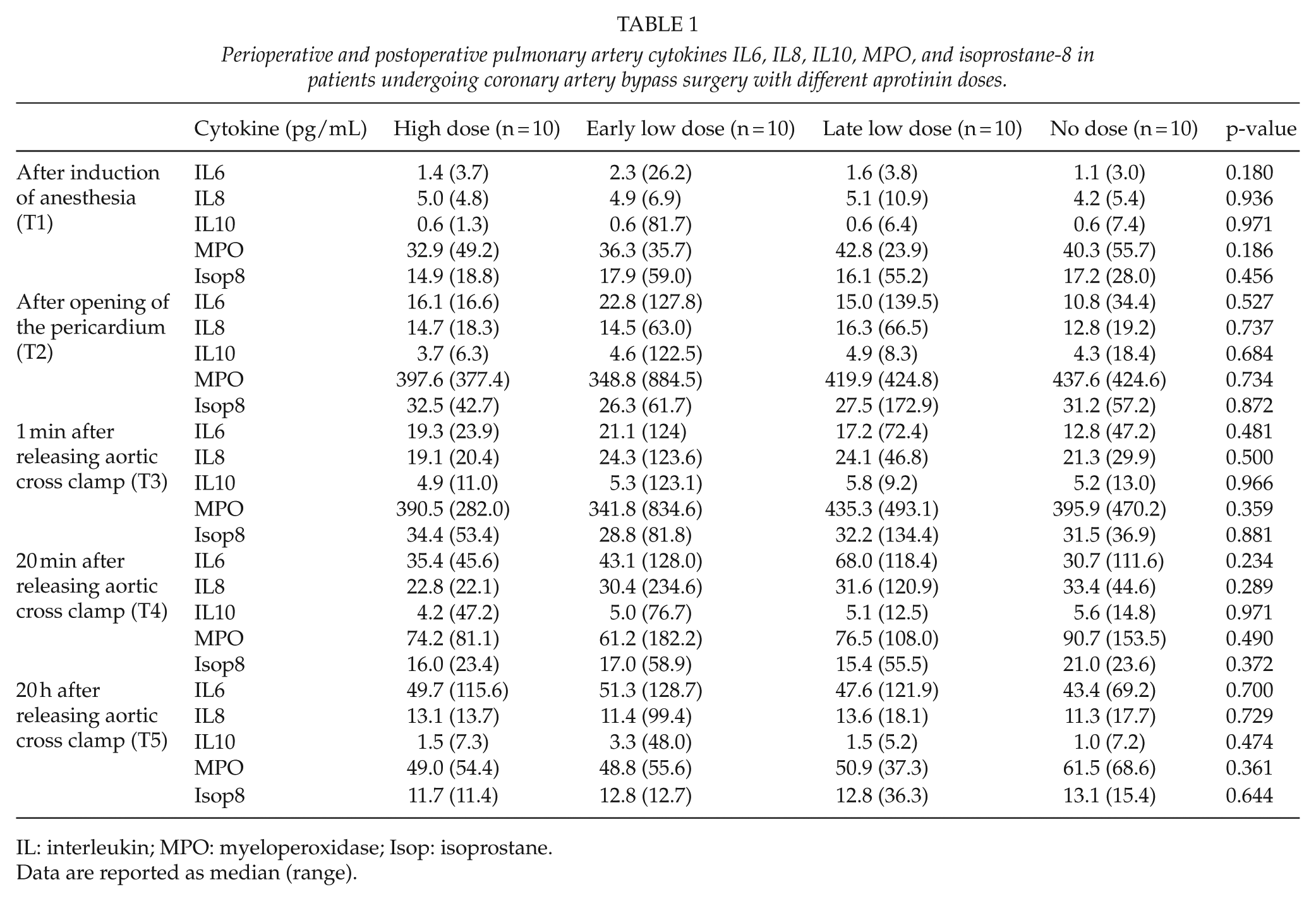
IL: interleukin; MPO: myeloperoxidase; Isop: isoprostane.
Data are reported as median (range).
Perioperative and postoperative radial artery cytokines IL6, IL8, IL10, MPO, and isoprostane-8 in patients undergoing coronary artery bypass surgery.
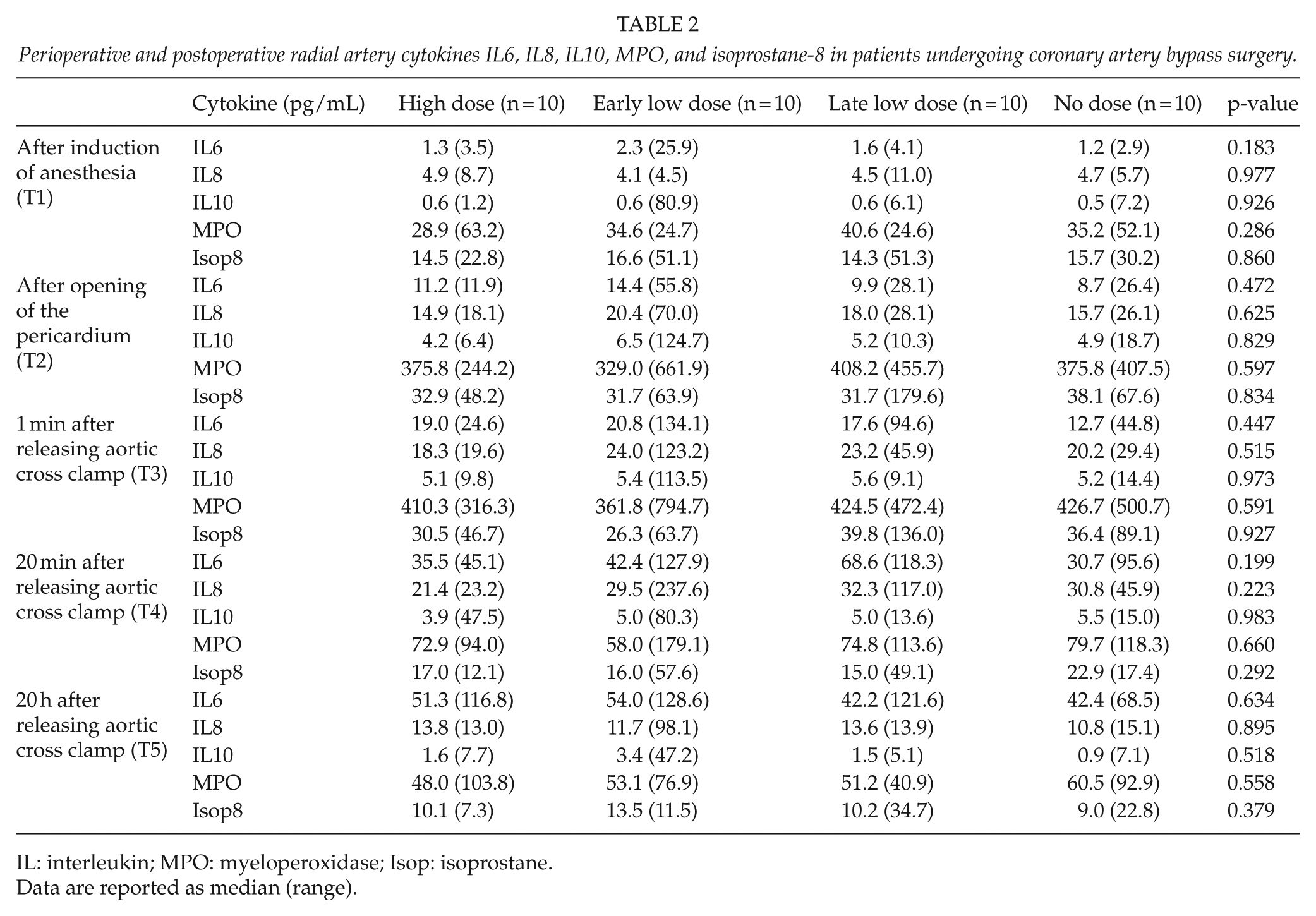
IL: interleukin; MPO: myeloperoxidase; Isop: isoprostane.
Data are reported as median (range).
The Ratio of the Inflammatory Factors in PA Versus RA
To investigate the impact of the lungs on the circulating levels of the measured inflammatory factors, we calculated the ratio of the concentrations measured in simultaneous pulmonary arterial and radial arterial blood samples (PA/RA ratio) during CABG. The PA/RA ratios were adjusted to the baseline values at time-point 1 to eliminate the effect of baseline differences between the patients. There were no differences in PA/RA ratios of IL6, IL8, IL10, or MPO in patients with high aprotinin dose as compared with early low aprotinin dose, late low aprotinin dose, or no dose (1.1 range 1.1 vs 1.0 range 7.4, p = 0.388; 1.1 range 1.1 vs 1.0 range 2.4, p = 0.645; 0.9 range 0.8 vs 0.9 range 0.7, p = 0.308; 0.9 range 1.0 vs 0.9 range 1.0, p = 0.549, respectively). Interestingly, the PA/RA ratio of 8-isoprostane increased in patients with high aprotinin dose as compared with patients with early low aprotinin dose, late low aprotinin dose, or no dose (1.1 range 0.97 vs 0.9 range 1.39, p = 0.001, Fig. 1). The diagnostic performance of PA/RA 8-isoprostane associated with patients who underwent CABG with high aprotinin dose was assessed by ROC analysis. Increased PA/RA 8-isoprostane values were significantly associated with patients with high aprotinin dose (area under the curve (AUC) 0.644; SE 0.044; p = 0.002; 95% CI 0.557–0.730, Fig. 2).
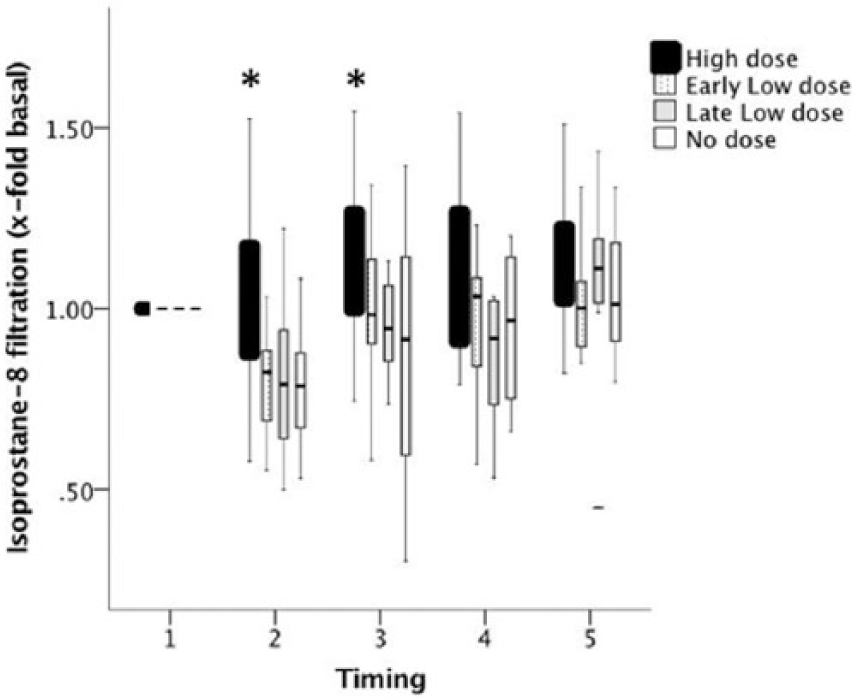
Calculated ratio of the pulmonary artery (PA) 8-isoprostane divided by the radial artery (RA) 8-isoprostane after induction of anesthesia (T1), 1 min after releasing aortic cross clamp (T2), 15 min after releasing aortic cross clamp (T3), 1 h after releasing aortic cross clamp (T4), and 20 h after releasing aortic cross clamp (T5) in patients with high dose, early low dose, late low dose, and no dose (black, dotted, gray, and white columns, respectively). Horizontal lines of the box show the median. Lines above and below the box indicate the 75th and 25th percentiles, respectively. Note increased PA/AR value > 1 in patients with high dose aprotinin, indicating excessive filtration in the lungs of pro-inflammatory 8-isoprostane.
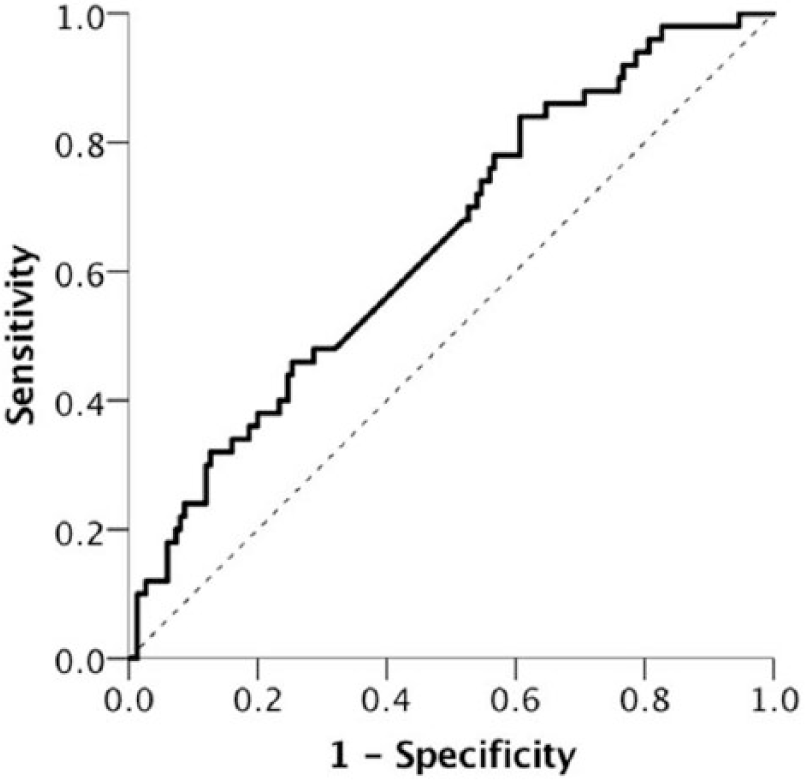
The diagnostic performance of calculated ratio of the pulmonary artery (PA) 8-isoprostane divided by the radial artery (RA) 8-isoprostane associated with patients who underwent coronary artery bypass grafting with high aprotinin dose was assessed by receiver operating characteristic curve (ROC) analysis. Increased PA/RA 8-isoprostane values were significantly associated with patients with high aprotinin dose (AUC 0.644; SE 0.044; p = 0.002; 95% CI 0.557–0.730).
PA/RA 8-isoprostane values were normally distributed. After surgery (T2), PA/RA ratio of the pro-inflammatory 8-isoprostane continued to be increased, namely 1.0 range 0.9, p = 0.031, in patients with high aprotinin dose, while it was 0.8 range 0.7, 0.8 range 1.2, and 0.8 range 0.5 in patients with early low aprotinin dose, late low aprotinin dose, and no dose, respectively. At the end of the study 20 h after restoring ventilation (T3), PA/RA for 8-isoprostane remained increased up to 1.2 range 0.8, p = 0.026, in patients with high aprotinin dose as compared with patients with early low aprotinin dose, late low aprotinin dose, and no dose (0.9 range 0.7, 0.9 range 1.1, and 0.9 range 1.0, respectively).
For PA/RA 8-isoprostane, there was no significant interaction between the dose of aprotinin and time (Wilks’ lambda = 0.12, F(4, 35) = 1.94, p = 0.125, partial eta squared = 0.18). There was no significant effect for time (Wilks’ lambda = 0.84, F(4, 35) = 1.60, p = 0.196, partial eta squared = 0.15) with the groups. The main effect comparing high aprotinin dose with early low aprotinin dose, late low aprotinin dose, and no dose was significant (F(1, 38) = 7.338, p = 0.01, partial eta squared = 0.16), further supporting the connection of aprotinin dose and the PA/RA ratio of 8-isoprostane.
Discussion
This study shows that the major inflammatory response during CABG may be influenced by high aprotinin dose as assessed by the estimation of pulmonary passage of 8-isoprostane. The impact of the lungs on the circulating concentrations of IL6, IL8, IL10, MPO, and 8-isoprostane was estimated by comparing the PA blood sample with the simultaneous peripheral RA blood sample. As being in between these two samples, the pulmonary vasculature may reflect the metabolism/filtration of the inflammatory 8-isoprostane. PA/RA ratio of 8-isoprostane increased in patients receiving high aprotinin dose as compared with the other groups, indicating increased consumption/metabolism/filtration of 8-isoprostane in the lungs.
The lungs play an important role in regulating circulating cytokines and other inflammatory factors during CABG with CPB (2, 8). A previous study demonstrated that the concentration of some cytokines is strongly altered during their passage through the lungs (1). We investigated whether the lung passage of pro-inflammatory IL6, IL8, MPO, and 8-isoprostane or the anti-inflammatory IL10 interacts with high aprotinin dose as compared to lower aprotinin doses. We utilized the PA/RA ratio adjusted to the baseline for all patients. As no differences were observed in IL6, IL8, IL10, or MPO, the mixed between-within subjects analysis of variance was applied to investigate the interaction of aprotinin and PA/RA 8-isoprostane level during CABG. This confirmed that the main effect comparing high aprotinin dose with early low aprotinin dose, late low aprotinin dose, and no dose was significant implicating difference in the effectiveness of aprotinin dose for PA/RA 8-isoprostane.
In contrast to our study, Liebold et al. (2) showed no differences in sinus versus RA blood samples of 8-isoprostane, IL6, IL8, IL10, or MPO. Several other cytokines, such as IL4 and interferon-gamma (IFN-γ), must also be involved in the complex cytokine network (15). The individual plasma cytokine levels reflect poorly the cytokine expression at any organ level, least to say the effect of lung passage (1). Mainly based on the interpretation of individual cytokine changes, a burden of previous studies emphasizes the importance of inflammatory processes due to CABG. A combination of low expression of pro-inflammatory cytokines together with a high expression of anti-inflammatory cytokines may also favor good outcome after surgery (15). The changes of cytokines may be masked by the heterogeneity of the patients recruited in some studies. Importantly, the decrease of vascular reactivity does not exclude the possibility of complete reversible endothelial dysfunction and subsequent recovery. The pulmonary endothelial dysfunction due to CABG (11) may over long-term be reversible despite the effect of lung passage on the concentration of some cytokines. We avoided over-distension of the lungs in our patients including a lung-protective ventilation strategy (8) that may partly explain the statistically non-significant changes in the PA/RA ratio of the other studied inflammatory factors.
Aprotinin is a serine protease inhibitor and a thromboxane A inhibitor (12). 8-Isoprostane interacts with endothelial cells (16). Endothelial cell activation and attraction of circulating monocytes may differ from thromboxane A activation (17). Both MPO and 8-isoprostane are strong markers of oxidative stress (6). Since aprotinin as a kallikrein–kinin inhibitor did not impact on MPO, the inhibition of neither the kallikrein–kinin cascade nor the serine protease may be as important as the thromboxane A inhibition (18). Since aprotinin impacted early before ischemia–reperfusion, the decrease of associated reactive oxygen species may not be the mechanism of action (17). Instead, a direct effect by aprotinin on 8-isoprostane seems plausible (17). The preserved vascular integrity of the endothelium of the lungs may explain these observations (10, 12, 17, 19).
Isoprostanes are prostaglandin-like compounds produced non-enzymatically by free radical-induced peroxidation of arachidonic acid (5). Isoprostanes act as biomarkers and mediators of oxidative stress in various conditions including ischemia–reperfusion injury (5). Low 8-isoprostane levels may predict decreased incidence of acute kidney injury (20), whereas high 8-isoprostane levels may indicate decreased postoperative lung compliance (21). 8-Isoprostane is a vasoconstrictor in a variety of vascular beds and activates monocyte functions (5, 16, 18, 22). 8-Isoprostane is a potentially atherogenic agent as it induces monocyte adhesion to endothelial cells (16) and increases scavenger receptor A activity in macrophages resulting in the formation of long-lived foam cells (23). The interaction of 8-isoprostane with endothelial and other cells via thromboxane receptors seems plausible and may be associated with a specific isoprostane receptor combined to the thromboxane receptor or via a part of the thromboxane receptor (16). The effect of aprotinin in reducing the circulating 8-isoprostane as estimated by lung passage may signify a shift toward an anti-inflammatory milieu (18) and reduced risk for pulmonary vascular resistance (14).
Limitations of this study include the relatively small sample size of our study that precludes assessing any differences in the clinical outcome. Only stable patients with preoperatively impeccable lung function were included. The clinical nature of our study excluded the possibility to perform vasculature histology. The strict inclusion criteria provided comparable patients since smoking, anti-inflammatory medication, and recent acute events were among the exclusion criteria (14). Long-term outcome associated with low intraoperative 8-isoprostane levels clearly needs to be investigated.
In summary, the concentration of 8-isoprostane may pharmacologically be regulated by aprotinin as observed by comparing circulating levels of PA with RA. Estimating lung passage of cytokines and other inflammatory factors with the PA/RA equation provides a practical means for the evaluation of the impact of aprotinin after CABG; it remains to be shown whether increased 8-isoprostane after lung passage may affect long-term clinical outcome after CABG. This study suggests that aprotinin may add to the inflammatory protection after CABG. This message may direct clinical practice on protecting the pulmonary vasculature during CABG using high aprotinin dose.
Footnotes
Acknowledgements
The authors alone are responsible for the content and writing of the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Competitive State Research Financing of the Expert Responsibility area of Tampere University Hospital, the unit of Heart Center Co Grant Z60044, the Finnish Cultural Foundation, and the Tampere Tuberculosis Foundation.
