Abstract
Background and Aims:
Although some studies have reported the safety of early oral intake after gastrectomy, it still remains controversial. This study focused on the feasibility of a clinical pathway with early oral intake and discharge setting for exclusively laparoscopic distal gastrectomy.
Materials and Methods:
A clinical pathway was applied to 403 patients until December 2014. In the protocol, patients are allowed to take a sip of water and a soft diet on the first and second days after the operation, respectively, and the discharge day is set as the fifth to seventh day after the operation. Clinicopathological variables were prospectively collected, and risk factors for discharge variances were analyzed.
Results:
The completion rate of the clinical pathway was 76.9%. There were five re-admissions (1.2%). The overall morbidity rate was 18% (n = 72), and major complications (Clavien-Dindo IIIa or greater) occurred in 13 patients (3%). Complications were the causes for discharge variances in 68 cases (73%), while the attending surgeons’ judgment was the cause in 25 cases (27%). On multivariate analysis, age (odds ratio = 2.23, 95% confidence interval = 1.38–3.60, p = 0.001) and operative time (odds ratio = 2.38, 95% confidence interval = 1.45–3.98, p = 0.001) were independent risk factors for discharge variances.
Conclusion:
A high completion rate of a clinical pathway with early oral intake and discharge setting for laparoscopic distal gastrectomy was achievable with an acceptably low re-admission rate. Laparoscopic distal gastrectomy is recommended as a first step for a clinical pathway with an early oral intake and discharge protocol.
Keywords
Introduction
A clinical pathway (CP) is a set of optimal sequencing and timing of interventions by physicians, nurses, and other staff for a particular diagnosis or procedure (1, 2). It is applied to various surgical fields, such as colorectal (2), urological (3), and cardiovascular (4, 5) surgery, and it decreases costs and hospital stays.
Unlike the fields mentioned above, in the gastric field, CPs have not been very frequently applied and reported. The complexity of the procedure and the subsequent hemodynamic changes, as well as the high morbidity rate, are the reasons, and some studies have reported a low CP completion rate in the gastric field (6, 7).
Recently, minimally invasive surgery (MIS), such as laparoscopic surgery and robotic surgery, has been frequently applied for gastric surgery, which results in less postoperative pain, faster recovery, less morbidity, and shorter hospital stays (8–11). Some studies reported open procedure and total gastrectomy as risk factors for variances in CPs (6, 12–14). Therefore, laparoscopic distal gastrectomy may be a good candidate for a CP with a high completion rate, but most of the previous articles regarding CPs in gastric surgery included patients with different approaches (open and MIS) and operation types (total and distal gastrectomy) (6, 7, 12, 13).
Although early enteral feeding has recently been encouraged, and some studies already reported its safety, its feasibility and safety still remain controversial (15–17). The appropriate discharge timing is also an important issue. Early oral intake may lead to early discharge, but it may overlook the latent complications that can be the causes for subsequent re-admission after early discharge.
Thus, in this study, the focus was on the feasibility of a CP with early oral intake and an early discharge protocol for exclusively laparoscopic distal gastrectomy.
Materials and Methods
Patients and Data Collection
From January 2005 to December 2014, 416 patients underwent laparoscopic distal gastrectomy with lymphadenectomy for gastric cancer in the Department of Gastric Surgery of Tokyo Medical and Dental University Hospital. In all, 13 patients with combined colon resection were excluded from the study, and 403 patients were enrolled in this study.
The following information was collected for each patient: age, sex, body mass index, American Society of Anesthesiologists Classification (ASA), clinical stage, extent of lymphadenectomy, operative time, intraoperative estimated blood loss, initial day of water or soft diet intake, postoperative hospital stay, complications, and re-admission rate. T and N stages were classified according to the 7th edition of the Union for International Cancer Control classification system (18). Complications were assessed according to the Clavien-Dindo classification (19). Re-admission was defined as re-hospitalization due to causes related to gastrectomy within 30 days after discharge. This study was conducted in accordance with the Declaration of Helsinki and was approved by the Institutional Review Board of Tokyo Medical and Dental University (M2016-251).
CP
Laparoscopic gastrectomy was introduced and launched in our institute in 1999. The CP for laparoscopic distal gastrectomy was implemented in our institute in 2001. After some modifications, the current CP was finally established in 2005, and it has been used continuously without further modification since then (Fig. 1). On postoperative day (POD) 1, the patient is allowed to have a sip of water and is encouraged to ambulate. On POD 2, a step 1 soft diet (750 kcal and 1130 mL of water) is given to the patient. Epidural and urethral catheters are removed. On POD 3, step 2 soft diet (900 kcal and 1160 mL of water) is started, and the abdominal drain is removed after evaluating the color and appearance of the fluid in the drain. Infusion of lactated Ringer’s solution is also stopped. After POD 5, discharge is recommended if there are no specific complications or complaints. Blood tests and X-rays are performed on PODs 1 and 3. Amylase concentration in the drain is examined on POD 3. Dietary education by a nutritionist is given to patients and their family before discharge. There are no exclusion criteria for the CP. Completion of CP is defined as discharge by POD 7. If a patient was assessed to be eligible for discharge within 7 days, but was not discharged for social reasons, the patient was also regarded as having completed the CP in this study.
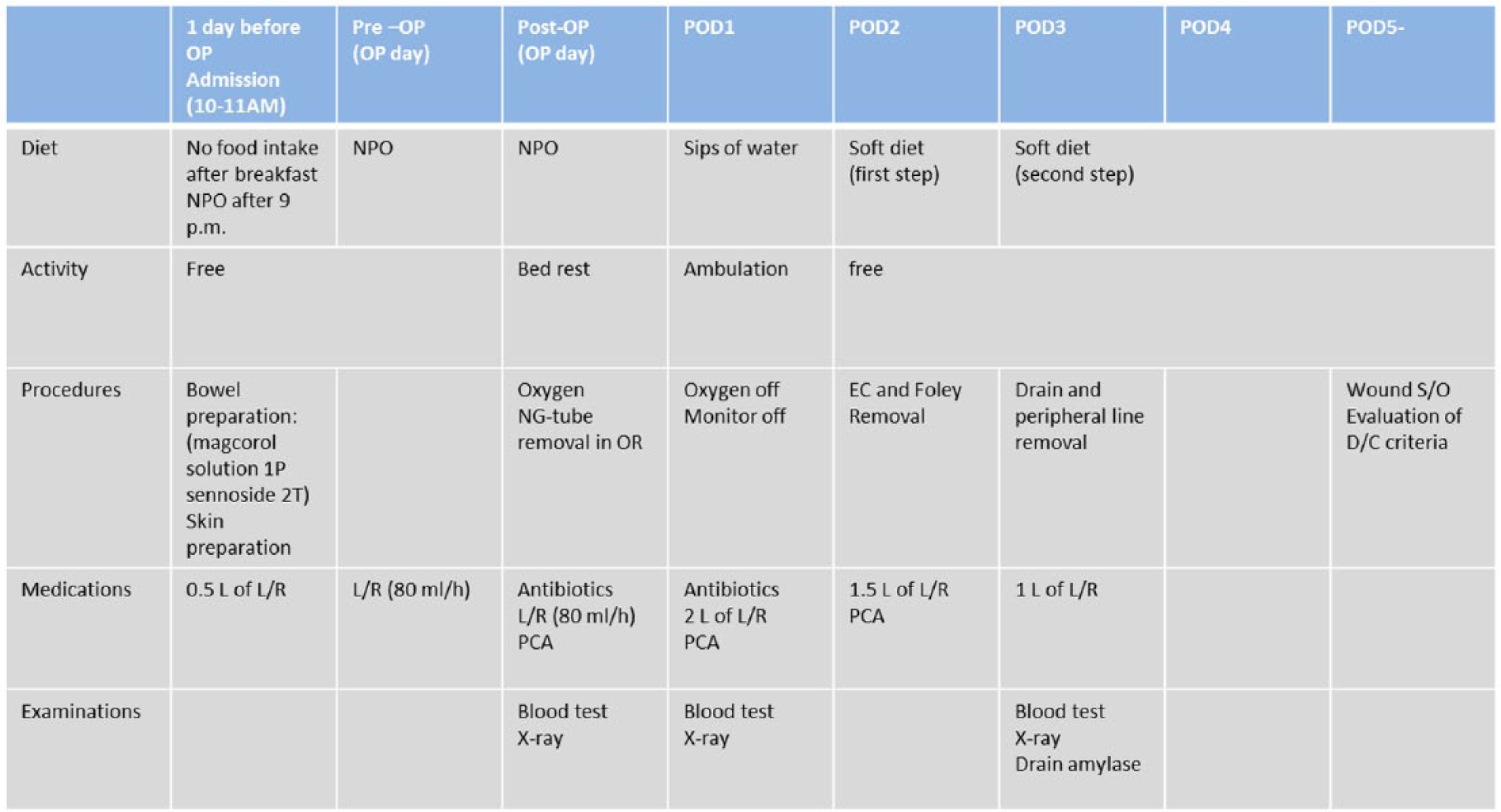
Clinical pathway for laparoscopic distal gastrectomy.
Operative Procedure
Inclusion criteria for laparoscopic distal gastrectomy in our institute are clinical T1-3 and N0-1. The laparoscopic distal gastrectomy was performed as described previously (20, 21). In short, the 5-trocar technique was used. Lymph node dissection was started along the greater curvature of the stomach. After resection of the duodenum, lymph node dissection along the lesser curvature and the suprapancreatic area was performed, followed by resection of the proximal margin of the stomach. Roux-en-Y reconstruction in an isoperistaltic, antecolic manner was performed in all cases. In the early period, a small midline upper abdominal incision 4–5 cm in length was made for reconstruction and removal of the stomach, but after a totally laparoscopic procedure (intracorporeal anastomosis) was adopted, no upper abdominal incision was made, and the stomach was removed via the umbilical incision. A drain was placed through the foramen of Winslow at the end of the operation. A nasogastric tube was inserted at the beginning of the operation and removed before gastric resection by an anesthesiologist.
Statistical methods
Continuous values are shown as means, standard deviations, and ranges. Data were analyzed using independent t-tests when they showed a normal distribution and the Mann–Whitney test when they did not. To identify the risk factors for discharge variances, univariate analyses were performed using logistic regression analyis. Mulitvariate analyses were performed using multiple logistic regression models with the forward likelihood ratio method.
A p value less than 0.05 was regarded as significant for all analyses. Statistical analyses were performed using SPSS version 19.0 software (IBM SPSS, Chicago, IL, USA).
Results
Baseline Characteristics of the Enrolled Population
The baseline characteristics of all (n = 403) patients are shown in Table 1. The mean age was 66 years (range, 29–92 years). A total of 277 patients (69%) were male and 126 patients (31%) were female. Most cases (377 cases; 94%) were diagnosed as clinical stage I. D2 lymph node dissection was performed in 81 patients (20%). No mortality was encountered. The overall morbidity rate was 18% (n = 72), and major complications (Clavien-Dindo IIIa or greater) were seen in 3% (n = 13). There were 262 patients (65%) who were discharged within 7 days, while 141 patients (35%) were discharged after POD 7. Among them, 48 patients (11.9%) were not discharged within 7 days for social reasons (without any complications or complaints). Altogether, CP completion was achieved in 310 patients (76.9%) (Fig. 2). Five patients (1.2%) were re-admitted after discharge, although all of them were discharged by POD 7 following the initial admission. Reasons for re-admissions were as follows: three patients with anastomotic stenosis (one patient needed endoscopic balloon dilatation, and the others recovered with conservative treatment); one with a pancreatic pseudocyst (conservative treatment); and one with duodenal stump leakage (re-operation was performed, and discharged 44 days after second operation).
Baseline characteristics and perioperative results of all patients.
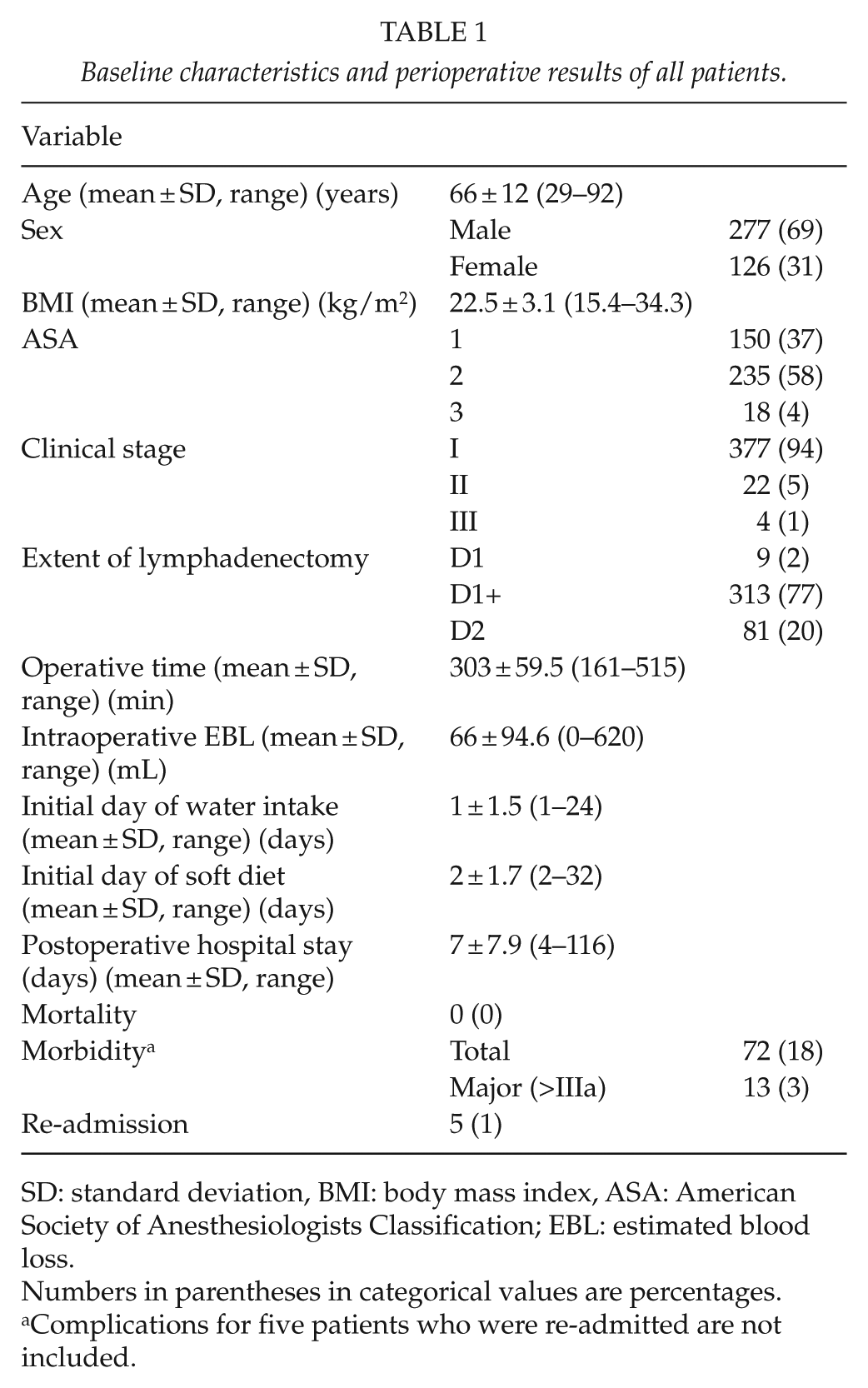
SD: standard deviation, BMI: body mass index, ASA: American Society of Anesthesiologists Classification; EBL: estimated blood loss.
Numbers in parentheses in categorical values are percentages.
Complications for five patients who were re-admitted are not included.
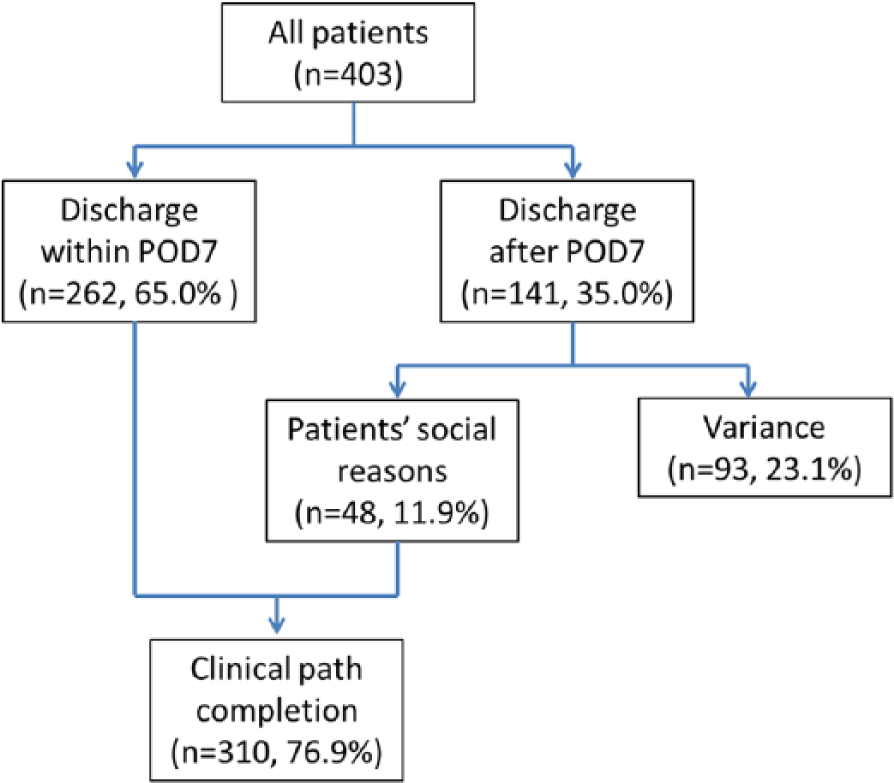
Flowchart of completion of the clinical pathway.
Causes of Discharge Variances
Causes of discharge variances are summarized in Table 2. Complications were the causes of discharge variances in 68 cases (73%) (intra-abdominal 60, systemic 8), while the attending surgeons’ judgment was the cause in 25 cases (27%). The most frequent causes among the complications were abdominal discomfort and pancreatic fistula. Among the 68 cases with complications, the severity of complications was: Grade I, 37 cases (54%); Grade II, 18 cases (26%); Grade IIIa, 6 cases (9%); and Grade IIIb, 7 cases (10%).
Causes of discharge variances and their severity.
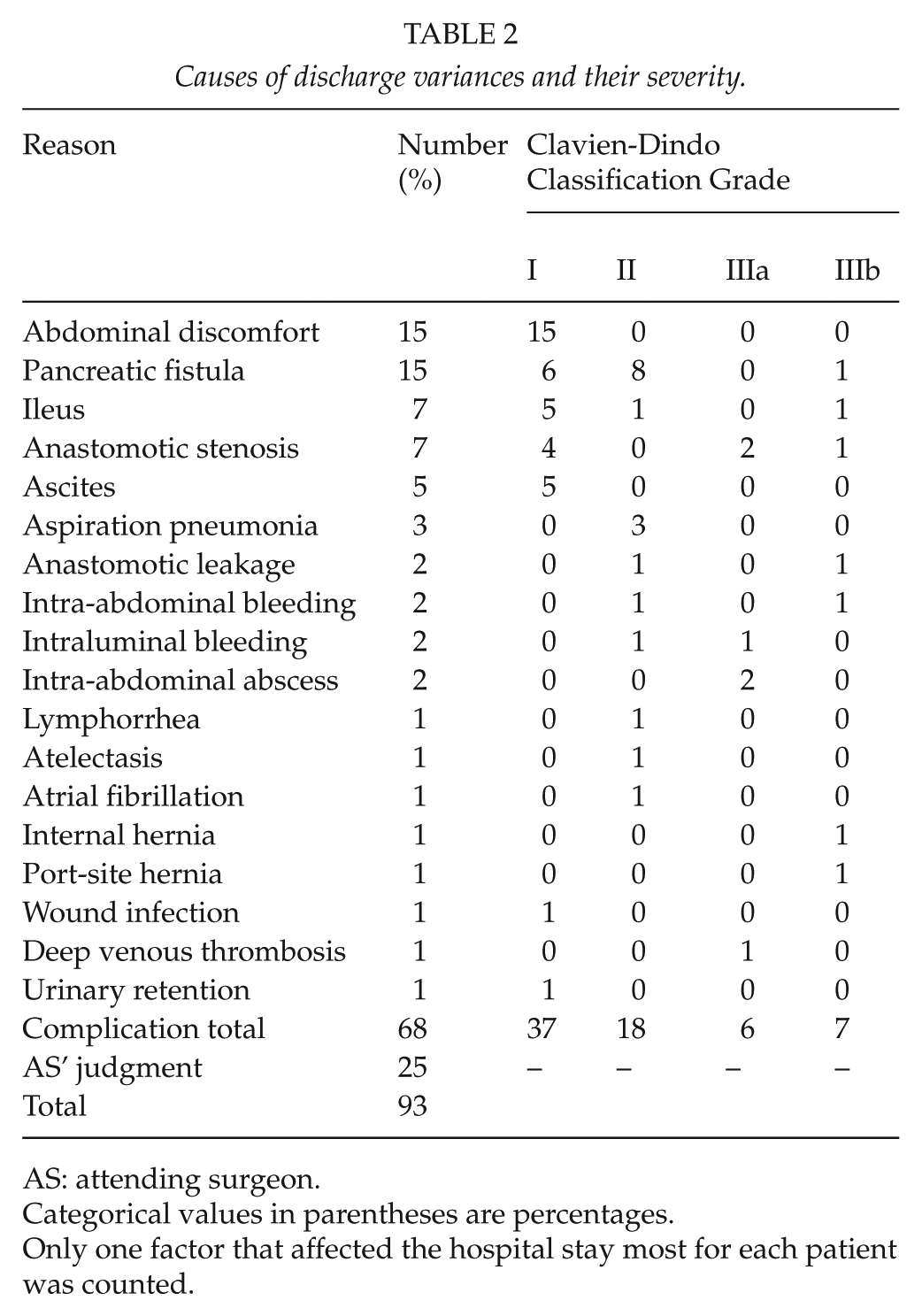
AS: attending surgeon.
Categorical values in parentheses are percentages.
Only one factor that affected the hospital stay most for each patient was counted.
Univariate and Multivariate Analyses for Risk Factors
The results of univariate and multivariate Cox analyses are shown in Table 3. Univariate Cox analyses showed that sex (p = 0.022), age (p = 0.001), ASA (p = 0.031), and operative time (p = 0.001) were significant risk factors for discharge variances. These significant values were included in subsequent multivariate analyses that showed that age (odds ratio (OR) = 2.23, 95% confidence interval (CI) = 1.38–3.60, p = 0.001) and operative time (OR = 2.38, 95% CI = 1.45–3.98, p = 0.001) were the independent risk factors for discharge variances.
Univariate and multivariate analyses of risk factors for discharge variance.
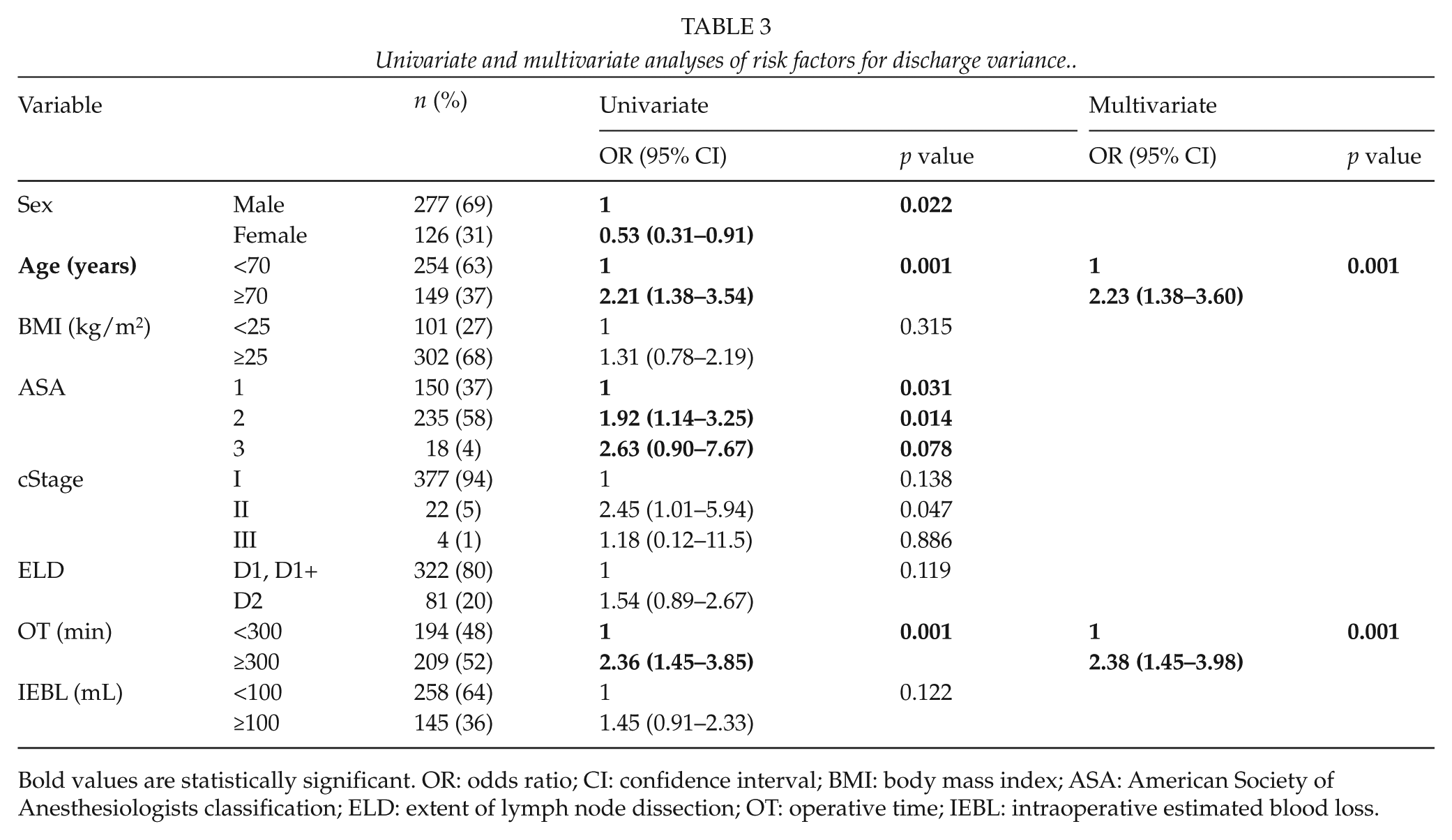
Bold values are statistically significant. OR: odds ratio; CI: confidence interval; BMI: body mass index; ASA: American Society of Anesthesiologists classification; ELD: extent of lymph node dissection; OT: operative time; IEBL: intraoperative estimated blood loss.
Discussion
In this study, the CP completion rate was 76.9%, with a re-admission rate of 1.2%. This completion rate is quite satisfactory with an acceptably low re-admission rate compared to the results reported in previous articles (6, 7, 12, 13).
Table 4 summarizes the previous articles regarding CPs for gastrectomy for gastric cancer, all of which were published from Korean institutes. The risk factors for discharge variances in those studies (not shown in the table) included male sex, older age, open approach, total gastrectomy, combined resection, intraoperative event, comorbidities, and high ASA. In this study, older age and longer operation time were the risk factors for discharge variances. These two factors are relatively common risk factors for complications, which may explain their identification in this study (22–27). Operation time is also a kind of indicator of difficulty and quality of operation and may affect clinical course of patients, even though no complication is identified. In this study, in fact, 29/93 (31%) patients of discharge variances did not experience any complication in the clinical course.
Summary of previously published articles on CPs for gastrectomy.
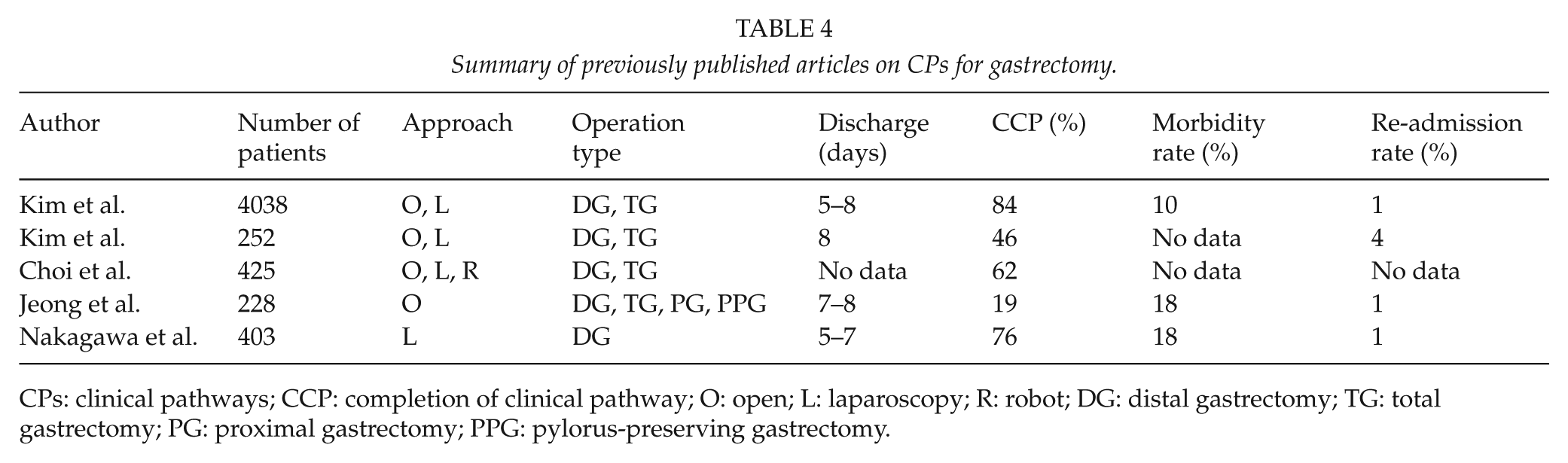
CPs: clinical pathways; CCP: completion of clinical pathway; O: open; L: laparoscopy; R: robot; DG: distal gastrectomy; TG: total gastrectomy; PG: proximal gastrectomy; PPG: pylorus-preserving gastrectomy.
The CP completion rates in the previous articles were quite low, except in the article by Kim et al., in which the CP completion rate was as high as 84% with discharge day-setting of POD 5–8. Unlike this study, the previous studies included various types of approaches and operation types. In this study, only patients who underwent laparoscopic distal gastrectomy were included, and, as we have repeatedly reported, the technique of laparoscopic distal gastrectomy in our institute is well established and standardized (20, 21, 28). That is why the CP completion rate in this study was quite high, although the set discharge day was earlier than in the previous studies.
The morbidity rate is the key for the CP completion rate, because complications are the major factors that affect progress along the CP, since they accounted for 73% of the reasons for discharge variances in this study. The overall morbidity rate in this study was 18%, and major complications over grade IIIa occurred in 3% of the patients. As seen in the results, not only major complications but also minor complications (Clavien-Dindo I and II) can be the reasons for discharge variances.
Recently, enhanced recovery after surgery (ERAS) protocols are becoming popular in the gastric cancer field, and there have been some reports of their safety and feasibility (15, 29, 30). Most of the components of the current CP overlap with those in the ERAS protocol, but there are some components lacking, such as preoperative carbohydrate loading and omission of bowel preparation, which are crucial in the ERAS protocol. The need for epidural catheter and abdominal drain is also controversial and should be reconsidered for the future modification of our clinical path (31, 32). However, one remarkable point about this study is that it was possible to show the safety and feasibility of early oral intake and subsequent early discharge. Thus, these results may encourage more institutes to apply CPs with early oral intake and discharge setting and, consequently, the ERAS protocol in the long run.
This study has several limitations. First, it was a retrospective one-arm study and did not assess the advantages or disadvantages compared to a non-CP group. Second, the cost issue of CP was not investigated and evaluated, and no conclusion about benefits with respect to admission costs can be made based on the results of this study.
In conclusion, a high completion rate of a CP with early oral intake and discharge setting for laparoscopic distal gastrectomy was achievable with an acceptably low re-admission rate. Standardization of surgical technique with a low morbidity rate is the key factor, and laparoscopic distal gastrectomy is recommended as a first step for an early oral intake and discharge protocol.
Footnotes
Author Contribution
All authors contributed to the study concept and design of the study, acquisition and interpretation of data, and final approval. M.N. contributed to data analysis and drafting the article. K.K. contributed to revising the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
