Abstract
Background and Aims:
The purpose of this study is to report our 20-year experience with the surgical management of renal cell carcinoma extending into the inferior vena cava using a novel classification system.
Materials and Methods:
We retrospectively reviewed the data of 103 patients (69 males, 34 females, mean age: 52.9 ± 12.6 years) with renal cell carcinoma involving the venous system treated between 1993 and 2014. The inferior vena cava tumor thrombus was classified into five levels: 0 (renal vein, n = 12), 1 (infrahepatic, n = 33), 2a (low retrohepatic, n = 26), 2b (high retrohepatic, n = 19), and 3 (supradiaphragmatic, n = 13). Clinical data were summarized, and overall survival, cancer-specific survival, and disease-free survival were examined by Cox regression analysis.
Results:
All patients underwent radical surgery. Complete resections of the renal tumor and thrombus were achieved in 101 patients (98.1%). Two intraoperative and one postoperative in-hospital deaths (2.9%) occurred. In total, 19 patients (18.8%) had a total of 29 postoperative complications. Mean follow-up time was 46 months (range, 1–239 months). The 5- and 10-year overall survival rates were 62.9% and 56.0%, respectively. Metastasis, rather than thrombus level, was a significant risk factor associated with overall survival (hazard ratio = 4.89, 95% confidence interval: 2.24–10.67, p < 0.001).
Conclusion:
Our novel classification system can be used to select the optimal surgical approach and method for patients with renal cell carcinoma and venous thrombus. Its use is associated with prolonged survival and relatively few complications. Metastasis is an independent risk factor of overall survival.
Introduction
Surgical resection of renal cell carcinoma (RCC) with tumor thrombus in the inferior vena cava (IVC) has become the primary treatment for this disease. The postoperative 5-year survival in patients without metastasis is reported to range from 45% to 69% (1, 2). Even in patients with metastasis, cytoreductive surgery can provide a better quality of life and prolonged survival (3, 4). However, caval thrombectomy is technically challenging and is associated with a major perioperative morbidity rate of up to 70% and a mortality rate of up to 48% (4–7).
In order to determine optimal treatment, a number of classification schemes of caval thrombus and various surgical techniques have been developed in the last decades (5, 8–14). The classification schemes attempt to define the most appropriate surgical method for different levels of tumor thrombus.
The purpose of this study is to report our 20-year experience in the treatment of RCC with tumor thrombosis using a novel five-level thrombus classification to determine the method of surgical management.
Methods
Patients
We retrospectively reviewed the records of patients with RCC involving the venous system treated at the three hospitals in our network from 1993 to 2014. This study was approved by the institutional review boards of the hospitals, and all patients or their relatives received a description of the surgical management and provided written informed consent.
All patients had preoperative imaging examinations such as ultrasound, computed tomography (CT), and magnetic resonance imaging (MRI) with three-dimensional reconstruction. All patients had also had preoperative evaluations of cardiovascular, pulmonary, hepatic, and renal status.
Classification and Definition of Ivc Tumor Thrombus
Based on preoperative imaging studies, we classified and defined the caval tumor thrombus into one of the five levels: renal vein (level 0, thrombus limited to the renal vein), infrahepatic (level 1, thrombus extending ≥2 cm below the liver caudate lobe), low retrohepatic (level 2a, thrombus extending from <2 cm below the liver caudate lobe to ≥2–3 cm close to the main hepatic vein (MHV)), high retrohepatic (level 2b, thrombus extending between <2 cm below the MHV and the diaphragm), and supradiaphragmatic (level 3, thrombus extending above the diaphragm or into the right atrium; Fig. 1).
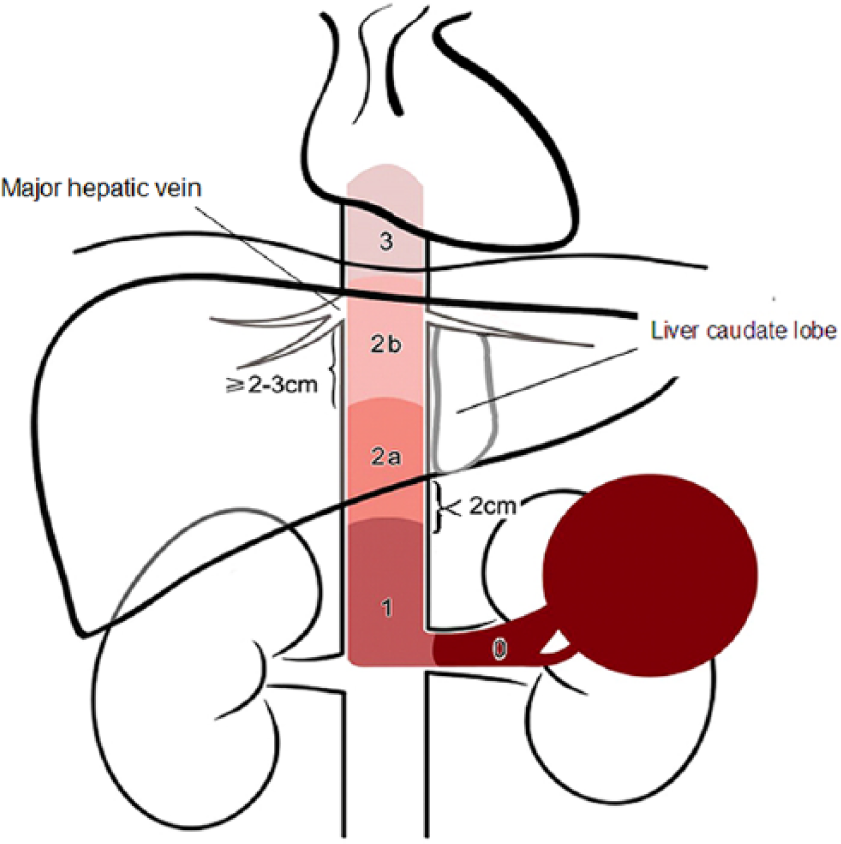
Classifications system consisting of five levels: (1) renal vein (level 0, thrombus limited to the renal vein), (2) infrahepatic (level 1, thrombus extending ≥2 cm below the liver caudate lobe), (3) low retrohepatic (level 2a, thrombus extending from <2 cm below the liver caudate lobe to ≥2–3 cm close to the main hepatic vein (MHV)), (4) high retrohepatic (level 2b, thrombus extending between <2 cm below the MHV and the diaphragm), and (5) supradiaphragmatic (level 3, thrombus extending above the diaphragm or into the right atrium).
Surgical Methods
As a general rule, the IVC was first explored for tumor thrombus, and the renal artery was ligated early. Vessels were controlled before thrombectomy. A modified lymphadenectomy was routinely performed. Bland thrombus attached to tumor thrombus in the distal IVC/iliac veins was resected using a segment-by-segment method after removal of the tumor thrombus.
Renal vein thrombus (level 0)
A transabdominal approach was used for better control of the vessels and performing the lymphadenectomy, especially in patients with a very large tumor. Antegrade radical nephrectomy was performed in a standard manner.
Infrahepatic thrombus (level 1)
A J-shaped abdominal incision was used for right-sided tumors and a Chevron incision for left-sided tumors. After dissecting the IVC and ligating the lumbar veins, Rummel tourniquets (RTs) were preset on the infrahepatic IVC, distal IVC, and contralateral renal vein. The RTs were sequentially tightened following mobilization of the diseased kidney. A cavotomy was made around the ostium of the renal vein. The tumor thrombus was isolated and removed en bloc together with the kidney. The cavotomy was oversewn with running 4-0 polypropylene sutures.
Low retrohepatic thrombus (level 2a)
Percutaneous balloon catheter occlusion (PBCO) was used for resection of thrombus at this level (15). The balloon catheter was first inserted percutaneously via the right internal jugular vein into the retrohepatic IVC, just below the ostium of the MHV, and above the top of the thrombus, in the radiology suite. The patient was then transferred to operating room. During thrombectomy, the catheter balloon was filled with saline to completely occlude the proximal IVC (Supplemental Fig. 1A). After making an infrahepatic cavotomy, the thrombus was completely stripped from the caval wall and then removed gently with a 16–18 Fr Foley catheter. Complete removal was confirmed by inspecting the integrity of the thrombus and checking with an index finger inserted into the IVC. The IVC was flushed with heparinized saline, and the cavotomy was closed with a Satinsky clamp after releasing the distal RT to allow air or clot to escape through the cavotomy. The RT on contralateral renal vein was released and then the catheter balloon was deflated and pulled out.
High retrohepatic thrombus (level 2b)
Caval–atrial bypass (CAB) without an extracorporeal centrifugal pump (16) was used. The incision began abdominally and extended cephalad to make a median sternotomy after the ventral procedures were finished. The supradiaphragmatic IVC was dissociated, and an RT was preset on it. A 28 Fr venous cannula was inserted into the IVC, distal to infrarenal RT, after systemic heparinization. Another cannula was introduced into the right atrium. Both cannulas were directly connected each other to build up the CAB (Supplemental Fig. 1B). Patient was placed in a 20° Trendelenburg position to facilitate venous return. The Pringle maneuver was used to control the hepatic circulation. The thrombectomy procedures were identical to those for level 2a.
In the event that preoperative imaging studies show total IVC obstruction by tumor thrombus in patient with dilated abdominal wall venous collaterals, but without the evidence of lower extremity edema, an attempt to clamp the infradiaphragmatic IVC may be made. If no significant hemodynamic compromise is noted after clamping, the sternotomy and CAB can be omitted.
Supradiaphragmatic thrombus (level 3)
Cardiopulmonary bypass (CPB) with or without circulatory arrest (CA) was used. The operation was coordinated with cardiac surgeons, and CPB was established after all of the abdominal procedures were completed. The tumor thrombus was removed through an infrahepatic cavotomy combined with atriotomy. In the event of a small free-floating atrial thrombus that could be easily pushed back to the IVC below the diaphragm, the CAB technique was considered.
Postoperative Evaluation And Follow-Up
Pathological examination was performed on all specimens, and RCC staging was based on the 2002 UICC/American Joint Committee on Cancer Tumor Node Metastasis (TNM) System. Postoperative complications were recorded up to 90 days after surgery and graded by the modified Clavien system (17). Patients with RCC who had metastasis, lymph node involvement, or positive resection margins were asked for receiving postoperative immunotherapy or targeted therapy with sorafenib or sunitinib beginning in 2008. All patients were followed up at 3, 6, and 12 months postoperatively and yearly thereafter.
Statistical Analysis
Patient overall survival (OS), cancer-specific survival (CSS), and disease-free survival (DFS) were estimated by the Kaplan–Meier method, until the date of last visit on 31 December 2014 or death. The log-rank test was applied to compare OS between patients with and without metastasis. Among all surgical and oncological features of interests, continuous variables were compared using the Kruskal–Wallis test among different thrombus levels, and post hoc Mann–Whitney U tests were performed for those with significant results. Categorical variables were tested using Pearson’s chi-square test. Univariate and multivariate Cox proportional hazard models were used to identify factors associated with OS, including patient age, sex, tumor size, thrombus level, degree of IVC occlusion, histopathological type, tumor stage (T), lymph node involved (N), distant metastasis (M), and targeted therapy. Results were presented as hazard ratio (HR) with corresponding 95% confidence interval (CI) and p-values. All statistical assessments were two-tailed and considered significant at p < 0.05. All statistical analyses were performed with Stata statistical software version 13.0 (Stata Corporation, College Station, TX).
Results
A total of 103 patients (69 males and 34 females) with a mean age of 52.9 ± 12.6 years (range: 21–77 years) were included in the analysis. There were 12 (11.7%) patients with level 0 tumor thrombus, 33 (32.0%) with level 1, 26 (25.2%) with level 2a, 19 (18.5%) with level 2b, and 13 (12.6%) with level 3. Complete IVC occlusion by tumor thrombus and associated bland thrombus attached to the tumor thrombus in the distal IVC/iliac veins were found in 39 (37.9%) and 12 (11.7%) patients, respectively. Preoperative distant metastasis was present in 17 (16.5%) patients.
All patients underwent radical surgery. Complete resections of the renal neoplasm and caval tumor thrombus were achieved in 101 patients (98.1%). Two patients (1.9%) with free-floating infrahepatic level 1 thrombus died of intraoperative pulmonary embolism. Table 1 summarizes the surgical and oncological features by thrombus level. Median blood loss was 2000 mL (range, 50–14,000 mL), and operation time was 330 min (range, 80–835 min). Both were associated with thrombus level (p < 0.01), and occluded vena cava (p < 0.05), but not with tumor size.
Surgical and oncological features by tumor thrombus level.
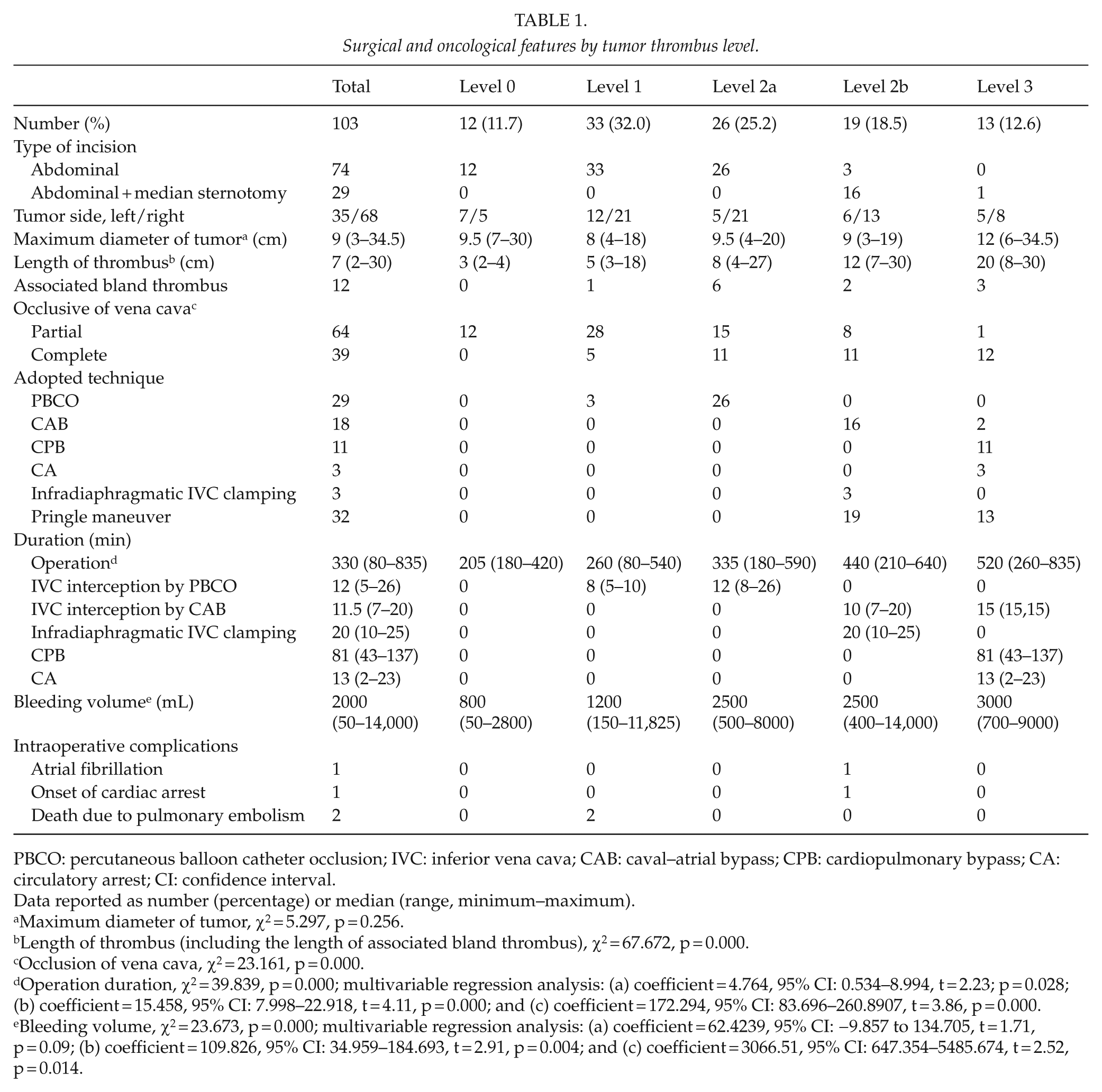
PBCO: percutaneous balloon catheter occlusion; IVC: inferior vena cava; CAB: caval–atrial bypass; CPB: cardiopulmonary bypass; CA: circulatory arrest; CI: confidence interval.
Data reported as number (percentage) or median (range, minimum–maximum).
Maximum diameter of tumor, χ2 = 5.297, p = 0.256.
Length of thrombus (including the length of associated bland thrombus), χ2 = 67.672, p = 0.000.
Occlusion of vena cava, χ2 = 23.161, p = 0.000.
Operation duration, χ2 = 39.839, p = 0.000; multivariable regression analysis: (a) coefficient = 4.764, 95% CI: 0.534–8.994, t = 2.23; p = 0.028; (b) coefficient = 15.458, 95% CI: 7.998–22.918, t = 4.11, p = 0.000; and (c) coefficient = 172.294, 95% CI: 83.696–260.8907, t = 3.86, p = 0.000.
Bleeding volume, χ2 = 23.673, p = 0.000; multivariable regression analysis: (a) coefficient = 62.4239, 95% CI: −9.857 to 134.705, t = 1.71, p = 0.09; (b) coefficient = 109.826, 95% CI: 34.959–184.693, t = 2.91, p = 0.004; and (c) coefficient = 3066.51, 95% CI: 647.354–5485.674, t = 2.52, p = 0.014.
Postoperative complications, pathological findings, and tumor stage are summarized in Table 2. There were 19 (18.8%) patients with 29 complications, including 15 minor complications (Clavien grades 1–2) and 14 major complications (Clavien grades 3–5), of which one patient had sudden cardiac death on the 17th postoperative day. No complications were noted during the 90-day postoperative follow-up period after discharge. Differences of complications between groups categorized by thrombus level were not statistically significance (χ2 = 5.890, p = 0.2075). Pathological investigation and staging of RCC showed that 75 (72.8%) patients had clear cell carcinoma, and 28 (27.2%) patients had positive lymph nodes. Three patients died in the hospital, for an in-hospital mortality rate of 2.9%. The remaining 100 patients recovered and were discharged with a mean postoperative hospital stay of 15.6 days (range, 5–53 days).
Postoperative complications, pathology, and stage by tumor thrombus level.
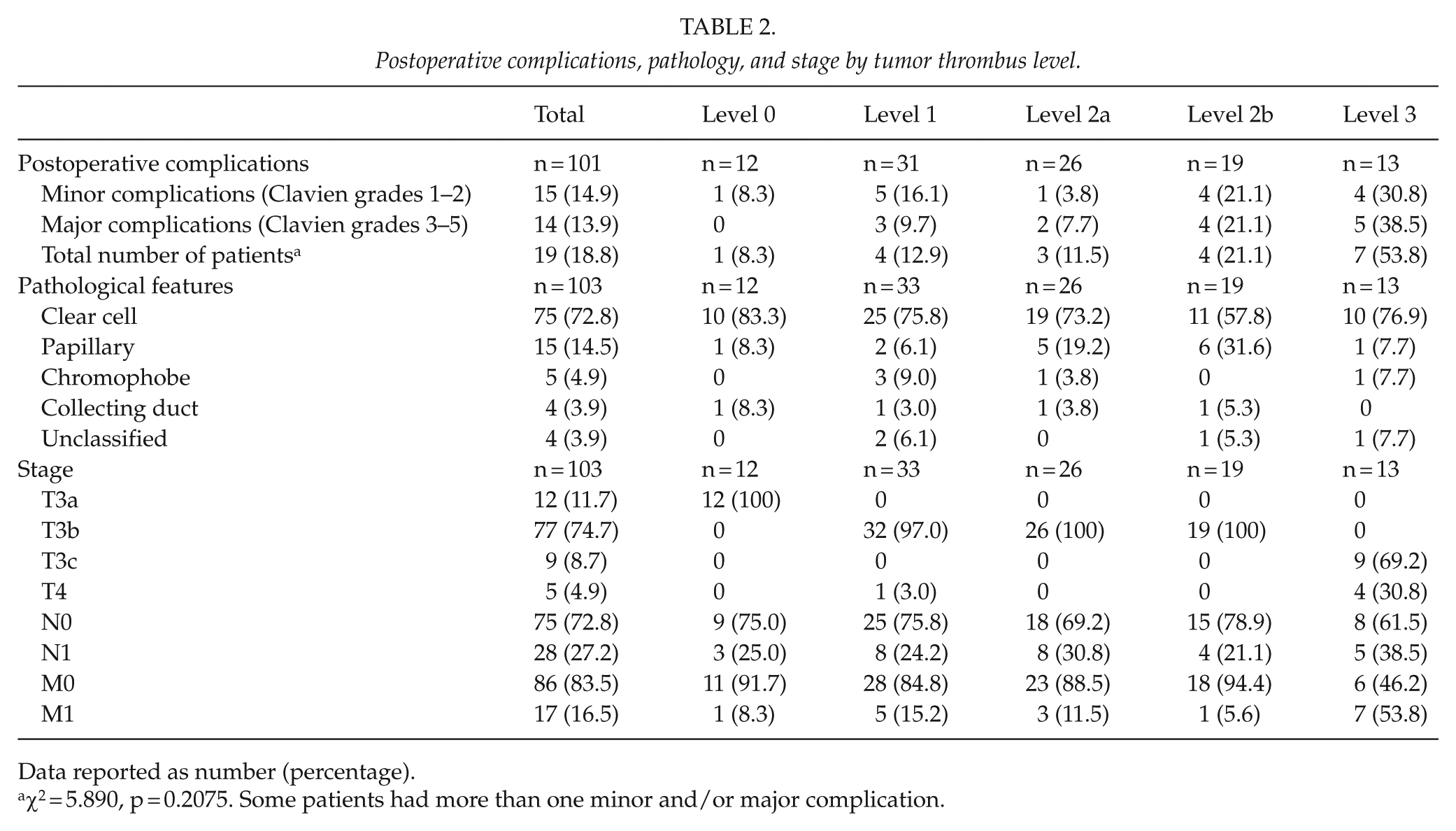
Data reported as number (percentage).
χ2 = 5.890, p = 0.2075. Some patients had more than one minor and/or major complication.
The mean follow-up period was 46 months (range, 1–239 months). In total, 66 patients were alive at a mean of 59 months (range, 1–239 months), 34 died at a mean of 22 months (range, 3–82 months), and 4 patients were lost follow-up at 1 month after discharge. There were 22 (22%) patients who developed distant metastasis postoperatively at a mean of 44 months (range, 3–149 months), of which 75% occurred within 2 years after surgery. The OS rates at 5 and 10 years were 62.9% and 56.0%, respectively; CSS rates were 69.0% and 66.7%, respectively; and DFS rates were 66.0% and 47.6%, respectively (Fig. 2A).
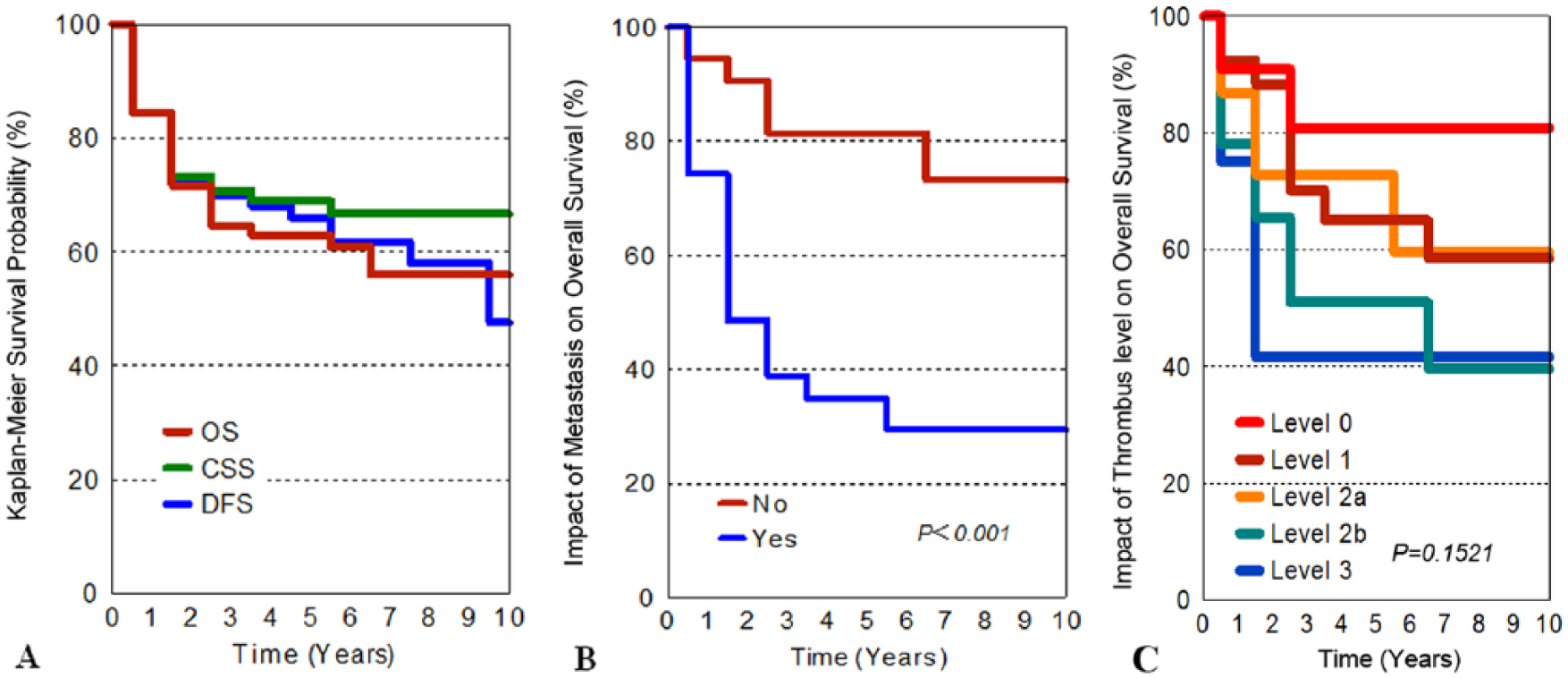
A) Kaplan–Meier survival curves of overall survival (OS), cancer-specific survival (CSS), and disease-free survival (DFS); B) impact of metastasis on OS; and C) impact of thrombus level on OS.
Univariate and multivariate analysis showed that metastasis was a significant risk factor impacting on patient OS (Table 3, HR = 4.894, 95% CI: 2.243–10.675, p < 0.001). Patients without distant metastasis had a 10-year OS of 73.2% (95% CI: 55.12%–84.91%), compared with that of 29.4% (95% CI: 14.03%–46.66%, p < 0.001) in patients with metastasis (Fig. 2B). The relation between level and survival is shown in Fig. 2C. The analysis showed that there was no association between thrombus level and survival.
Univariate and multivariate analysis of factors impacting on OS by Cox proportional hazards model.
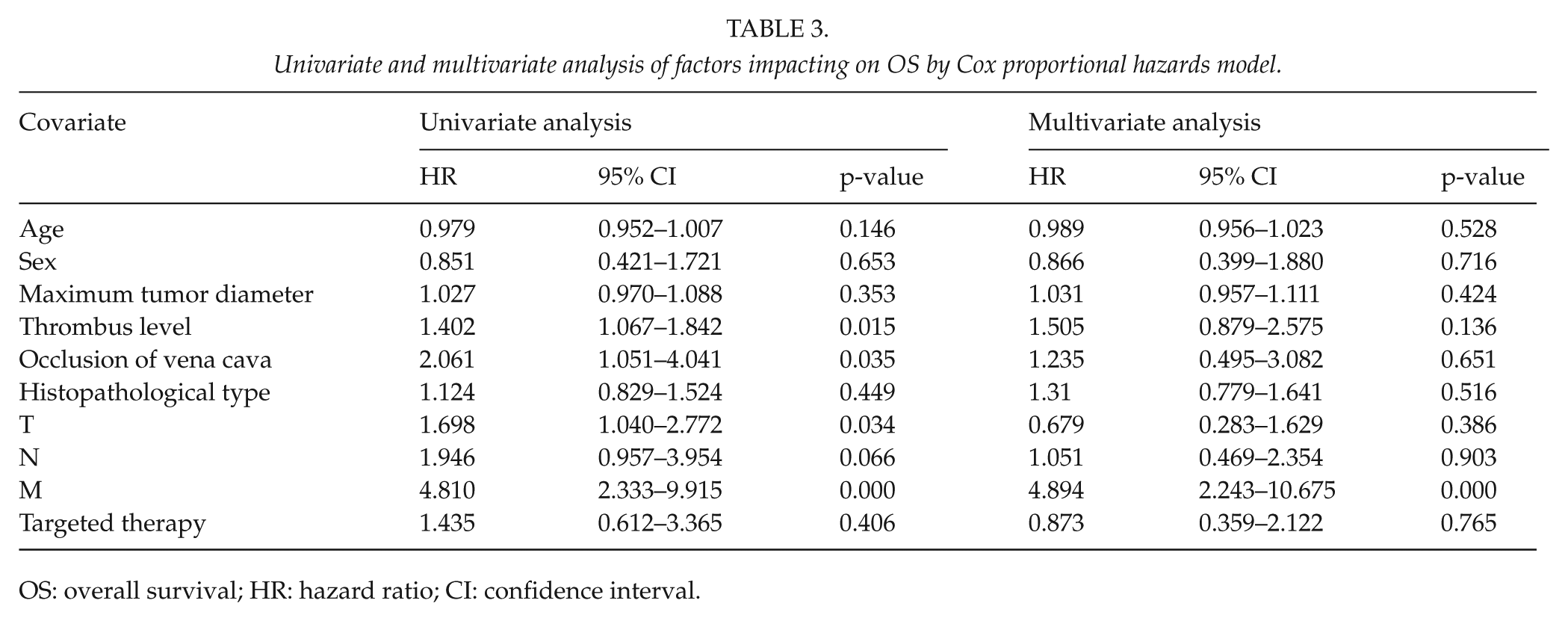
OS: overall survival; HR: hazard ratio; CI: confidence interval.
Discussion
Surgical management offers patients with RCC extending into the IVC the opportunity for prolonging survival. This study indicated that complete resection of renal carcinoma and tumor thrombus has been achieved 62.9% and 56% of OS at 5 and 10 years, respectively, with a postoperative complication rate of 18.8%, which are comparable to rates reported by other authors. Univariate and multivariate analysis of our data showed that metastasis was the most important factors affecting on OS, again consistent with prior reports (4, 18, 19).
The management strategy for caval tumor thrombus depends on the level of the thrombus extension. Since Berg first described nephrectomy and venacavotomy in 1913 (1), many classification schemes of IVC tumor thrombus have been proposed, such as the dichotomized classifications by Libertino (20), Belis et al. (21), and Stief et al. (14); the tri-categories by Wilkinson et al. (22) and Pritchett et al. (13); the four-level classification from the Mayo Clinic by Neves and Zincke (12); and five-level scheme by Blute et al. (5). However, no classification scheme has been commonly accepted, and no uniform operative strategy based on the level of the tumor thrombus has been established (23). Although the Mayo system is currently used in many centers, the distinction between level I and level II is difficult to discern in clinical practice (24). Furthermore, level II is defined broadly (from >2 cm above the renal vein to the level below MHV), so infrahepatic and retrohepatic thrombi may be grouped together. However, the operative management for infrahepatic and retrohepatic thrombi is different. Possibly because of this grouping mode, many diverse operative maneuvers have been reported to resect level II thrombus, such as laparoscopy or robot, liver mobilization, veno-venous bypass, and CPB with CA (5). This has resulted in a number of classification schemes different from the Mayo system being developed in the last two decades and includes those described by Babu et al. (8) in 1998, Staehler and Brkovic (25) in 2000, Tsuji et al. (26) in 2001, Moinzadeh and Libertino (27) in 2004, and Yazici et al. (28) in 2010. Recently, Ciancio et al. (29) from the University of Miami School of Medicine further subdivided level III of the Mayo system into four subtypes (IIIa, b, c, and d), based on the method of thrombus resection (29).
In this study, we classified the caval tumor thrombus based on the method of thrombectomy. Renal vein thrombus of level 0 was separated from level 1 because its operative method is relatively simple and different from that used for infrahepatic caval thrombus. We defined level 1 as infrahepatic thrombus based on the landmark of caudate lobe, rather than the renal vein, because the IVC enters the retrohepatic space at this location. A spatial distance of at least 2 cm between the top of infrahepatic thrombus and the lower edge of liver caudate lobe is safe for isolating and blocking the IVC; a distance <2 cm is considered unsafe and associated with potential complications. We also dichotomized retrohepatic thrombi into low and high subtypes in anatomical relation to the MHV, according to hemodynamic characteristics of blocking the IVC above or below the MHV. This principle of dichotomization should be followed when using any technique or maneuver for resection of retrohepatic thrombus. Based on our results, we believe that the classification scheme we have developed and described in this study is reasonable and feasible for determining the safest and most effective method of caval thrombectomy. However, it should be remembered that any classification scheme is just used to choose an appropriate surgical method to remove thrombus at different levels; overall outcomes are also related to the surgeon’s skills, experience, and pre- and postoperative care.
The best surgical strategy for removal of retrohepatic caval tumor thrombus is a topic of debate. There are many diverse methods used to resect retrohepatic thrombus, such as liver mobilization (29), aortic cross-clamping (30), veno-venous bypass, or even CPB with CA (5, 26). According to the present classification, we used PBCO to temporarily occlude the IVC just below the MHV, so that the outflow of hepatic blood return was not blocked during resection of low-level retrohepatic thrombus (level 2a). This technique was first reported in 2009 by our center (15). For removal of high-level retrohepatic thrombus (level 2b), we used CAB without an additional extracorporeal centrifugal pump to maintain adequate blood return and stable hemodynamics after clamping the supradiaphragmatic IVC during thrombectomy. A Foley catheter was also used to extract the retrohepatic thrombus, thus liver mobilization was omitted. Of course, this maneuver should only be performed after completely stripping the thrombus from the wall of IVC if the thrombus is adherent to the wall. Furthermore, because the maneuver may be accompanied by substantial venous backflow from small veins that drain directly into the retrohepatic IVC (such as short hepatic veins, lumbar veins, inferior phrenic veins, or right adrenal veins), bleeding control by temporary clamping or manual compression of the IVC above the cavotomy immediately after pulling the thrombus out or flushing the lumen of IVC is very important. Other risks associated with this maneuver, such as pulmonary embolization, residual tumor, and IVC disruption, can be avoided so long as the manipulation is performed with care. In the current series, there were no complications associated with the maneuver.
The surgical management of caval tumor thrombus in this study is just one of many strategies used today. Regardless of the technique used to remove a retrohepatic thrombus, such as liver mobilization, classifying the thrombus into low or high level in relation to the MHV is critical for the removal of retrohepatic thrombi. Furthermore, the management should be individualized taking into account for unique characteristics of individual patients. For example, in a patient with a retrohepatic thrombus extending into the MHV and causing Budd–Chiari syndrome, or tumor thrombus invading the retrohepatic IVC wall, with required circumferential segment resection and replacement, the thrombectomy must be performed under CPB with the use of liver mobilization.
The main limitations of this study were the retrospective data collection and the relatively small number of cases, so that the results might be underpowered in statistical analysis. Further study with a larger sample size is necessary to confirm the results and further assess the effect of the classification system and associated surgical approaches and methods on short- and long-term survival. However, given the rarity of this disease, a random comparative study of this nature will be difficult to perform, just as previous reports on this issue (5, 8–14, 20–22, 25–29), and require the cooperation of multiple centers. Beginning in 2008, we began using sorafenib or sunitinib for patients with RCC who had metastasis, lymph node involvement, or positive resection margins. While these treatments are known to prolong survival, probably due to the small sample size, only 19 patients were underwent the targeted therapy, there was no significant impact on OS in our analysis models.
In conclusion, radical resection of the tumor and caval thrombus is the mainstay of treatment in patients with RCC extending into the venous system. We described a novel classification system of caval thrombus corresponding with a specific surgical method and achieved prolonged survival with a relatively small number of postoperative complications. Retrohepatic caval thrombus below or above the MHV should be dichotomized and managed with diverse techniques. Metastasis, rather than thrombus level, is the most important factor affecting patient survival.
Footnotes
Acknowledgements
The authors gratefully acknowledge the assistance of Ms Qing Zhang, Ms Rao Fu, and Ms Xue Mei from The General Hospital of PLA in preparing the figures.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
This study was approved by the institutional review boards of the hospitals, and all patients or their relatives received a description of the surgical management and provided written informed consent.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
