Abstract
Purpose:
Sentinel node is routinely localized with the intraoperative use of a radioactive tracer, involving challenging logistics. Super paramagnetic iron oxide nanoparticle is a non-radioactive tracer with comparable performance that could allow for preoperative localization, would simplify the procedure, and possibly be of value in axillary mapping before neoadjuvant treatment. The current trial aimed to determine the a priori hypothesis that the injection of super paramagnetic iron oxide nanoparticles in the preoperative period for the localization of the sentinel node is feasible.
Methods:
This is a prospective feasibility trial, conducted from 9 September 2014 to 22 October 2014 at Uppsala University Hospital. In all, 12 consecutive patients with primary breast cancer planned for resection of the primary and sentinel node biopsy were recruited. Super paramagnetic iron oxide nanoparticles were injected in the preoperative visit in the outpatient clinic. The radioactive tracer (99mTc) and the blue dye were injected perioperatively in standard fashion. A volunteer was injected with super paramagnetic iron oxide nanoparticles to follow the decline in the magnetic signal in the sentinel node over time. The primary outcome was successful sentinel node detection.
Results:
Super paramagnetic iron oxide nanoparticles’ detection after preoperative injection (3–15 days) was successful in all cases (100%). In the volunteer, axillary signal was presented for 4 weeks. No adverse effects were noted.
Conclusion and relevance:
Preoperative super paramagnetic iron oxide nanoparticles’ injection is feasible and leads to successful detection of the sentinel node. That may lead to simplified logistics as well as the identification, sampling, and marking of the sentinel node in patients planned for neoadjuvant treatment.
Introduction
Since its introduction in the middle of the 1990s, sentinel node biopsy (SNB) has been established as the gold standard for the nodal staging of patients with breast cancer with a clinically negative axilla (1, 2). Therefore, axillary sampling and blind axillary dissections have been practically removed from clinical practice, resulting in substantial decrease in associated surgical complications such as vascular and neural injuries, lymphedema, chronic pain, and wound infections (3). Apart from invasive breast cancer (IBC), SNB is also used in certain subgroups of patients with a preoperative diagnosis of ductal cancer in situ (DCIS) with the rationale that approximately 25%–30% will postoperatively be upgraded to IBC. In spite of controversy and variance in clinical practice, prognostic factors for upgrading invasive ductal cancer (IDC) such as the presence of a mass (clinical or radiological), nuclear grade, the presence of comedo necrosis, size larger than 50 mm, or when a mastectomy is planned (thought to preclude the possibility for a SNB at a second operation) are considered as criteria to perform SNB (4).
However, SNB is not an indolent procedure. A recent meta-analysis has demonstrated early- and late-postoperative morbidities such as restricted shoulder range of motion (up to 37.8% 2 years after surgery), pain (up to 56.6% 1 month after surgery), paresthesia (up to 15.8%), axillary web syndrome (20%), and lymphedema (8.2% 2 years after surgery) among others (5)
The established method of SN tracing with radioisotope colloid (Tc99) and blue dye is highly effective, with detection rates as high as 99% (6), but it is chronically bound to the procedure itself due to the 6-h half-life of the isotope, which mandates access to nuclear medicine facilities on the day of the operation or the day before. However, the blue dye may be allergenic up to 2% of cases and is not administrated before the induction of anesthesia.
Our group recently conducted the Nordic SentiMag trial (7) where super paramagnetic iron oxide (SPIO) nanoparticles were compared to the standard dual technique. In this article, a meta-analysis of all published studies on the same subject was also included. Not only was SPIO found to be comparable with the standard technique in terms of detection rate but also it was demonstrated that techniques were highly concordant in detecting the same nodes with a pooled concordance of 98.3% both in the absence and, most importantly, in the presence of malignancy. The meta-analysis demonstrated that in several other studies, slightly more nodes were detected with SPIO; this, however, might be explained by the decision of respective authors to consider palpable nodes as sentinels and to dissect until no magnetic background was present. In the Nordic trial, a detailed long-term follow-up of the SPIO-induced skin staining was performed for the first time, where it was shown that it was 100% prognostic for ferromagnetic signal and therefore demonstrated prolonged SPIO residual in the tissue. Possible clinical implementations were sought with the first being the feasibility of injecting SPIO to the breast preoperatively.
Study Aim and Design
The fact that SPIO resides in the tissue for a prolonged period of time prompted the idea to investigate whether the injection of SPIO in the preoperative visit to the outpatient clinic, several days before the operation, was feasible. This was a novel idea, as all comparative studies performed until now investigated the perioperative or intraoperative injection of SPIO, including a 5-min massage in the injection site as well as waiting at least 20 min from injection to operation, as this is the estimated time required for SPIO to migrate in the axilla. The perceived advantages would be that this time could be spared from the operating time, as well as that logistics would be simplified. Additionally, this could be a possible method to mark the sentinel node but remove it only in need, helping to avoid unnecessary SNBs nowadays performed, such as in several cases with DCIS.
To assess the feasibility of the preoperative injection of SPIO, a prospective comparative pilot study was conducted in the Breast Department of the Uppsala University Hospital. The study concept was that patients would undergo triple mapping with radioisotope, blue dye, and SPIO, but SPIO would be injected in the preoperative visit to the outpatient clinic. Additionally, a healthy volunteer was injected with SPIO, and the magnetic signal in the breast and axilla was recorded for 6 weeks. The study was approved by the ethics committee in Uppsala University in Sweden and performed according to the 1975 Helsinki Declaration and the Swedish Act on Patient Insurance. This article was prepared according to the CONSORT statement for pilot studies. The flow is illustrated in Fig. 1.
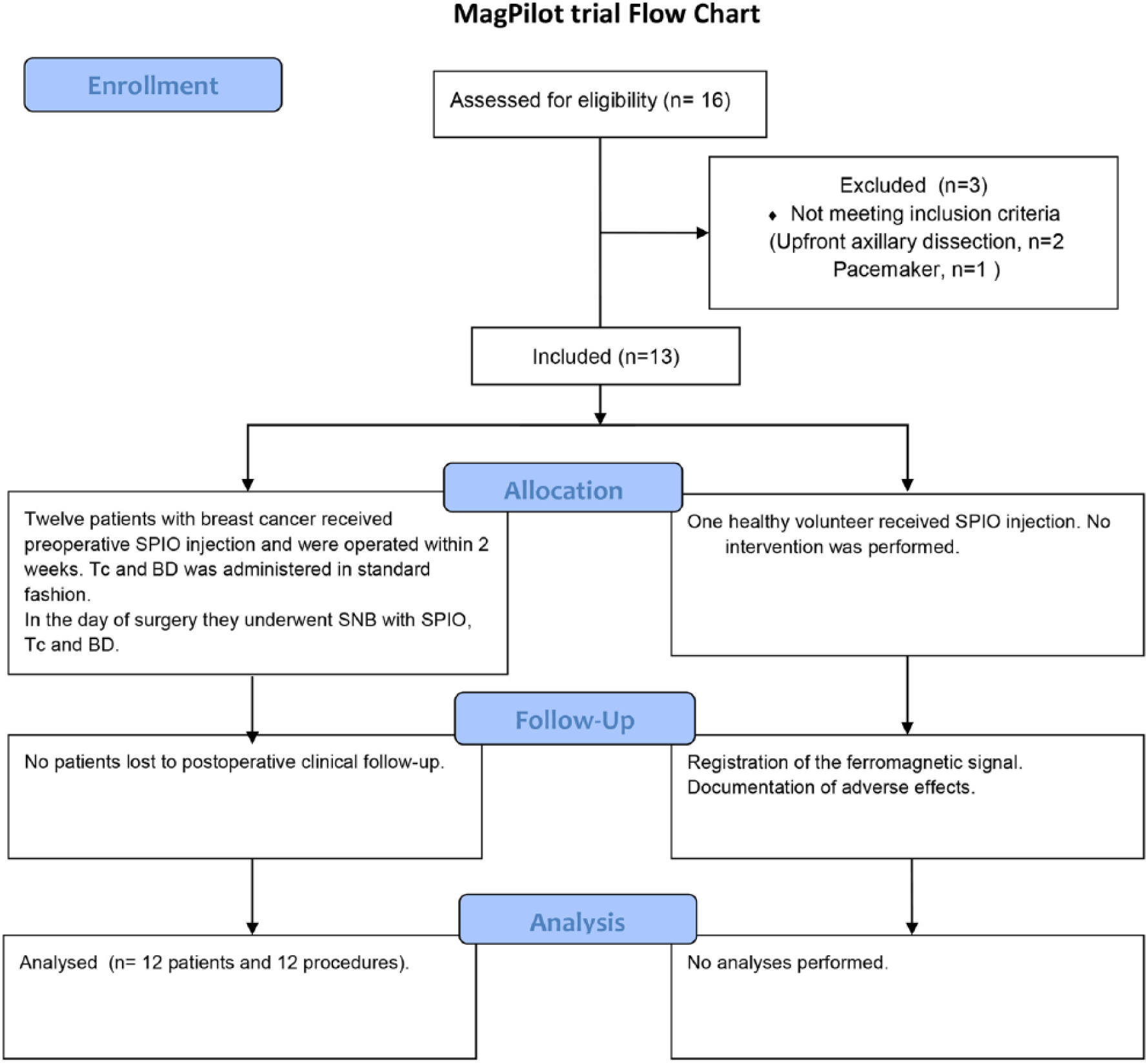
CONSORT flowchart for the pilot study.
Patient Selection
Patients were eligible for recruitment if they were older than 18 years and were diagnosed with IBC and DCIS, with negative axilla in clinical examination and ultrasound (US). Exclusion criteria were hypersensitivity to dextran compounds, iron, or Sienna+; iron overload disease; pregnancy; pacemaker or other implantable metallic devices in the chest wall; or inability to provide written informed consent. All patients had to be available for postoperative follow-up.
Tracer Injection–Operative Protocol
Two milliliters of Sienna+® (Sysmex Europe GmBH, Hamburg, Germany) diluted with 3 mL local anesthetic (Xylocain®, 10 mg/mL) were injected subareolarly during the preoperative visit in the outpatient clinic. The radioactive tracer (99mTc), usually 40–60 mBq, was injected subareolarly either on the day of surgery or the day before. Lymphoscintigraphy was not performed. A vital blue dye (1–2 mL of Patent Blue V®-Laboratoire Guerbet, Aulnay-Sous-Bois, France) was injected in standard fashion after the onset of anesthesia.
During operation, a handheld probe (SentiMag®; Endomagnetics Ltd, Cambridge, UK) was first used to detect magnetic uptake. A short incision was made in the axilla over the area with the greatest uptake, and the sentinel node was sought for primarily using the SentiMag probe. Metal retractors and instruments were removed and plastic ones were used at that point. Thereafter, the finding was confirmed with the gamma probe and the sentinel node(s) removed. All sentinel nodes were excised until the counts were lower than 10% of the highest count or a maximum of four nodes per patient were removed. Blue or brown nodes were also regarded as sentinel nodes. Magnetic counts in the axilla were documented before skin incision as well as ex vivo on the excised nodes.
Pathology
All sentinel nodes were examined with hematoxylin–eosin staining. Frozen section was performed in cases planned for mastectomy, as well as post neoadjuvant treatment. Additional techniques were used according to the pathologist’s discretion.
Statistical Analysis
Descriptive values are presented as medians with range and interquartile range, due to the small number of participants. Comparisons are performed with the Wilcoxon signed rank test. Correlation is performed with the Spearman ρ. Rates are given as percentages (%), and comparison between detection rates is performed with Mc Nemar’s test. Detection rates and comparisons are performed for tracer per tracer separately and also between SPIO and the dual technique. Successful localization with the dual technique was considered as every case with SN was detected by the isotope, the blue dye, or both. To document whether the preoperative injection of SPIO results in the detection of the “correct” node, concordance between methods was assessed. Concordance is defined as the number of both radioisotope and SPIO-positive patients or nodes, divided by the number of patients or nodes marked by the isotope (Tc+ and SPIO+⁄Tc+). All analyses were performed with SPSS V 23.0 (IBM Corp, Armonk, NY, USA). All tests were two-sided, and a p-value of <0.05 was considered significant.
Results
In all, 12 consecutive patients were included, operated from September 2014 to October 2014. Clinicopathological characteristics are presented in Table 1.
Clinicopathological characteristics and study outcomes.
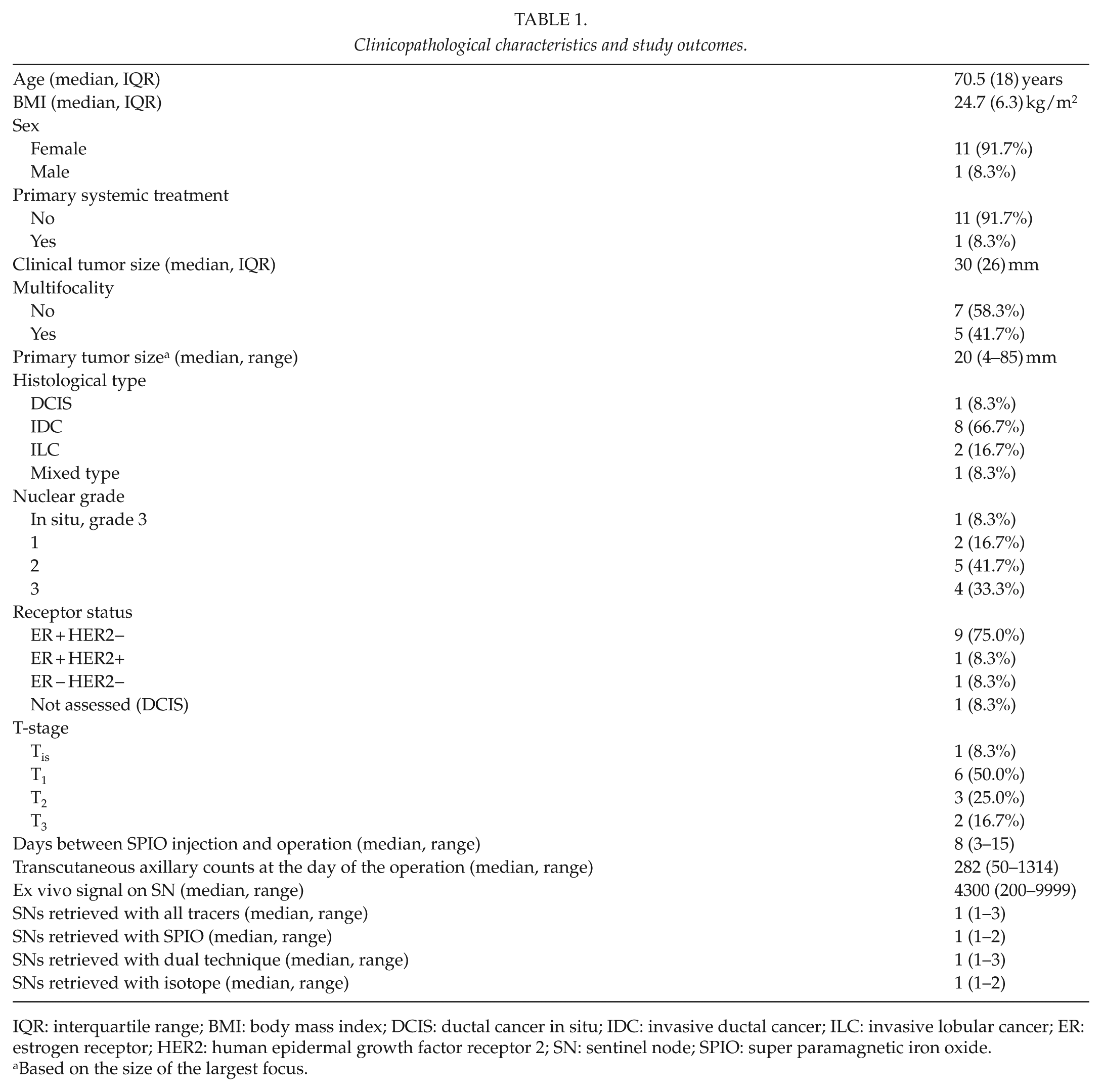
IQR: interquartile range; BMI: body mass index; DCIS: ductal cancer in situ; IDC: invasive ductal cancer; ILC: invasive lobular cancer; ER: estrogen receptor; HER2: human epidermal growth factor receptor 2; SN: sentinel node; SPIO: super paramagnetic iron oxide.
Based on the size of the largest focus.
SPIO was injected 3–15 days before the operation (median 8 days). Transcutaneous magnetic signal was present in all patients at operation, and radioactivity was present in 10. In nine patients (75%), the SN was identified with all three methods. Blue dye was successful in 9 cases (75%) and Tc99 in 10 (83.3%). The dual technique was subsequently successful in 10 cases (83.3%). SPIO was successful in all cases (100%). Therefore, no difference was noticed in detection rates between SPIO and isotope (p = 0.500) or between SPIO and dye (p = 0.250) and SPIO and the combined dual technique (p = 0.500). Concordance between isotope (and the dual technique) and SPIO per case was 100%. In these 10 patients, there was complete nodal concordance for retrieved nodes (13/13, 100%), demonstrating that the exact same nodes were identified as sentinels by SPIO and Tc99. Nodal concordance between SPIO and the dual standard was 81.25% (13/16), because of three nodes that were colored but neither radioactive nor magnetic. A median of one sentinel (Table 1) was retrieved totally as well as per tracer. No differences were found in the number of nodes retrieved with either SPIO or the dual technique (p = 1.000). Metastases were found in three patients (25%). None of the three nodes detected only by the dye harbored metastasis.
The cases where blue dye and isotope failed were examined separately. One of the patients detected only with SPIO had undergone neoadjuvant therapy for an 85-mm grade 2 lobular cancer. The axillary sentinel node contained a macrometastasis, and in the subsequent axillary clearance, 9 of 20 nodes contained macrometastases. The other patient where isotope and dye failed did not present any specific features. Finally, the patient in which only blue dye failed had previously been operated with a wide local resection because of DCIS.
In the present series, transcutaneous magnetic signal was a predictor of successful detection (positive prognostic value 100%). However, no correlation could be found between the transcutaneous counts and the maximum ex vivo signal (p = 0.244), the body mass index (BMI; p = 0.564), or the interval between SPIO injection and operation (p = 0.347).
In the first three patients with palpable lesions, the surgical specimen including the injection site of SPIO was sent for postoperative mammography. SPIO artifacts could not be detected on mammograms, and no disturbance in the visualization of the breast lesions was noted. Then, in patient number 4 with a non-palpable lesion, SPIO was injected and preoperative guidewire localization was performed. The SPIO was not detectable on mammograms before or after surgery and the lesion was clearly seen both preoperatively and in the surgical specimen (Fig. 2A to C). A magnetic resonance imaging (MRI) was also performed after SPIO injection. SPIO-induced artifacts were evident in imaging (Fig. 2D).
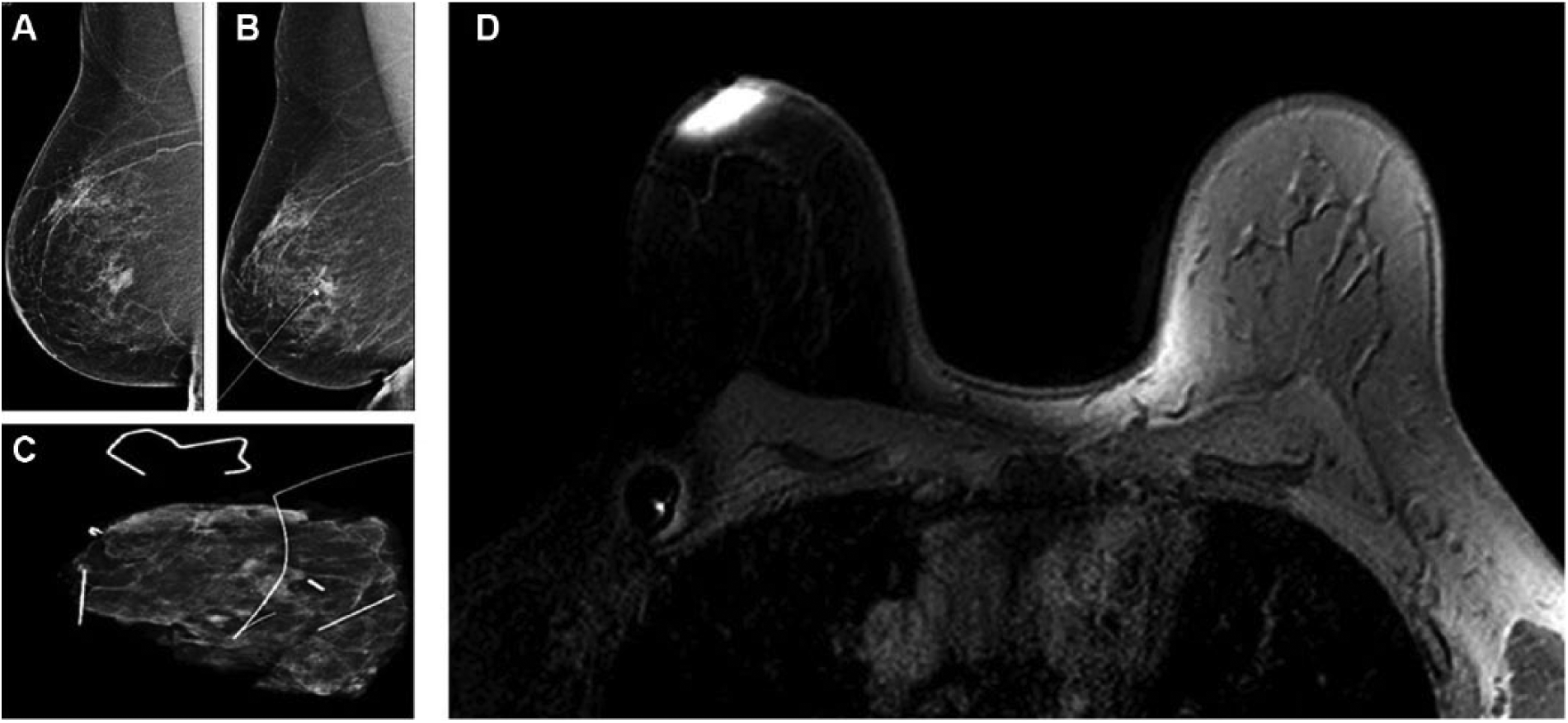
A) Preoperative mammography after SPIO injection. B) Placement of a localization guidewire without problems. C) Specimen mammogram. D) MRI after SPIO injection.
At histopathology, SPIO deposits were examined in relation to the breast lesion and in the SN. There was an uptake of SPIO in macrophages subcutaneously at the injections’ site and in histiocytes in the SN, but there was no uptake in the primary tumors or in lymph node metastases (Fig. 3A, B). On the contrary, the examination of frozen sections of SNs was easier as the SPIO was not accumulated in metastatic cells. We could not see any disturbance of the following cytokeratin MNF (CKMNF) staining. The yellowish/brownish SPIO granules in the cytoplasm of the histiocytes were easily separated from the distinct CKMNF staining of the membranes and cytoplasm of tumor cells (Fig. 3C).
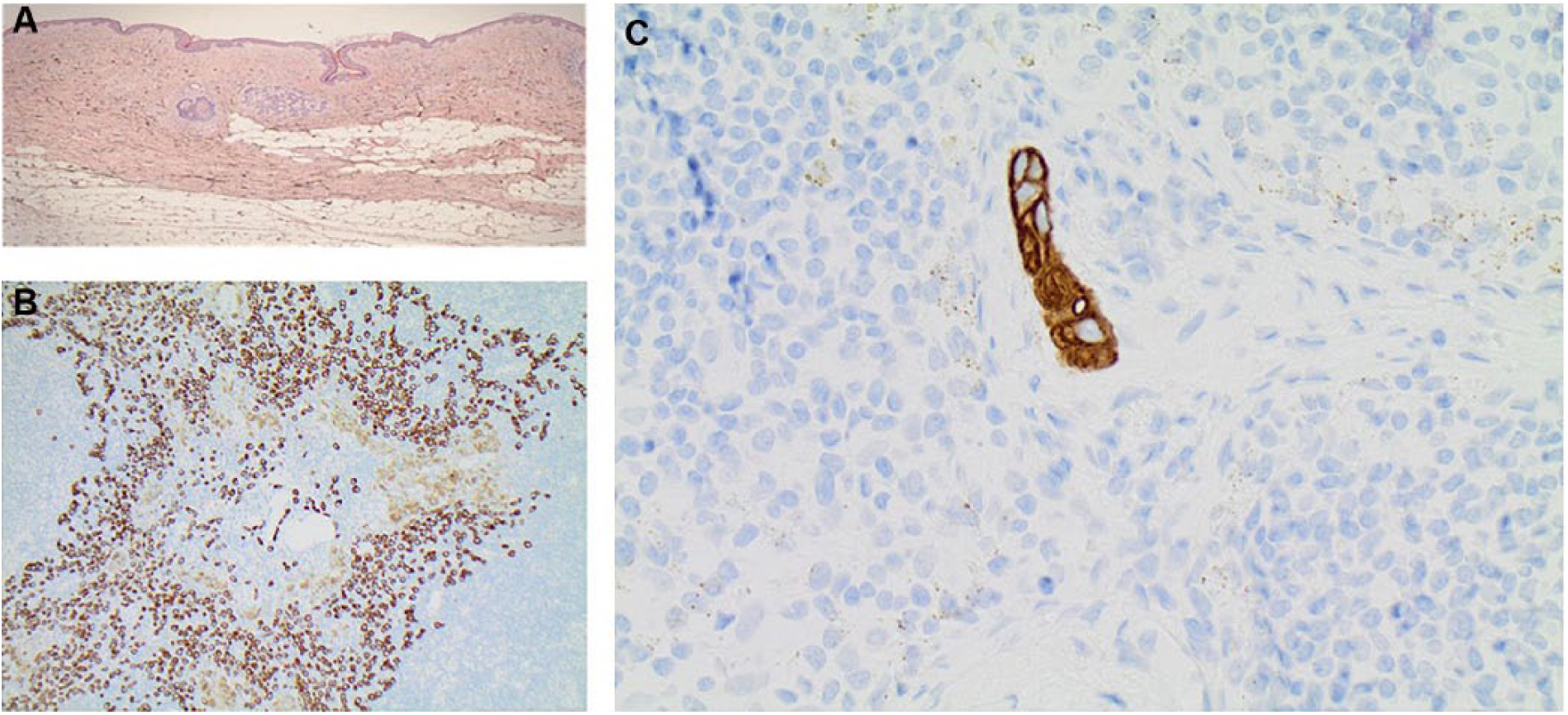
Histopathology in A) the tumor and B) SN without any SPIO artifacts present. C) Cytokeratin MNF (CKMNF) staining of the membranes and cytoplasm is not distorted by the easily identified yellowish/brownish SPIO granules in the cytoplasm of the histiocytes.
The decline in the magnetic signal was followed in the volunteer injected with SPIO. The counts in the axilla increased with a peak 3 weeks after the injection and remained high for more than 4 weeks (Fig. 4); 15 days after the injection, the volunteer passed a metal detector (Ceia02PN20) at Arlanda International Airport without the detection of ferromagnetic signal.
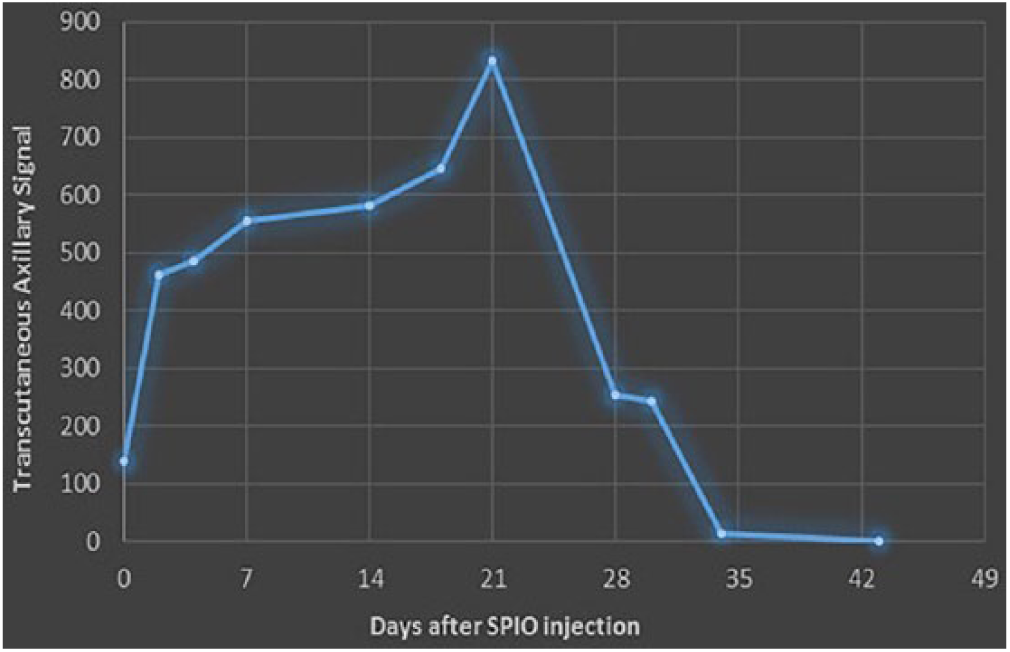
Transcutaneous axillary ferromagnetic signal curve in a volunteer.
No side effects were reported by the patients or the volunteer. Data on the long-term follow-up of SPIO-induced skin staining will be reported elsewhere.
Discussion
The combination of radioisotope and blue dye was highly effective in SNB. The development of new tracers has been motivated mostly by the known drawbacks of this combination, the predominant being the difficulty of regular access to the isotope, the need for nuclear medicine facilities, and the complicated logistics of maintenance, manipulation, and disposal, as well as the discomfort of patients and personnel in the idea of exposure to radiation as well as the allergenic properties of blue dye. Alternative methods have been used in the detection of SN such as indocyanine green fluorescence (ICG) and SPIO injected directly pre- or intraoperatively, with comparable results (8). Interestingly, no direct comparison has ever been performed between the two methods. ICG is known to retrieve higher number of sentinel nodes, leading to a successful SNB with comparable results to the dual technique, but alleged drawbacks of the method include a migrating time of 30 min, meaning that the time frame for the SNB is limited, problems with intraoperative spillage that may limit the success of the method, as well as sporadic reports of skin staining together with the fact that higher numbers of sentinels retrieved do not comply with the minimally invasive character that SNB is supposed to have (9 –11).
With documented detection rates as high as 97.6% (7), SPIO has been verified to be a comparable tracer to the dual standard. None of the studies has demonstrated superiority and there probably is little point—if any at all—in seeking a “better” technique in terms of detection grade. A potential drawback of novel methods could be the lack of studies of SNB in a background of axillary lymph node dissection, but a question of scientific ethics is raised, since the oncologic safety of SNB has been demonstrated (12); this, however, is addressed by the fact that SPIO is at least 96% concordant to the dual standard. Therefore, possible advantages of new technologies have to be sought in novel clinical implementations, as well as simplification of routine practice.
In this pilot study, the feasibility of preoperative SPIO injection is demonstrated in a variety of patients. An obvious advantage is that the intervention is simple and does not require specialized personnel or the presence of a physician, since the injection can be administered by the breast nurse in the outpatient clinic. In this study, successful SNB was performed up to 15 days after SPIO injection. However, considering the positive prognostic value of the transcutaneous signal for a successful SNB, since there is documented magnetic activity in the axilla up to 42 days, it has to be assumed that an SNB should also be feasible within this interval. Apart from the obvious simplification of logistics, this suggests flexibility in the timing of the operation.
Additionally, preoperative injection does not seem to affect diagnostic radiologic routine, localization of non-palpable lesions, or specimen radiology. However, the expected exception of breast MRI may be dealt by the wide time frame between injection and successful SNB. A patient indicated for a preoperative MRI could undergo imaging first and be injected with SPIO afterward. Another important finding in this study is the compatibility of preoperative injection with pathological examination of the SN; on the contrary, it seemed to simplify the SN examination on frozen section. This is a finding in the direction of similar results after perioperative subcutaneous injection (13).
The feasibility of preoperative SPIO injection is of particular interest in patients with a preoperative diagnosis of DCIS and in particular that approximately 25% that will upgrade to IBC after the microscopic analysis of the entire specimen (14), but only 7.5% will be diagnosed with metastatic cells in the SN (15). This questions the necessity of most of the SNBs performed according to current practice. The lack of a predictive model successful in identifying which cases with a preoperative DCIS diagnosis will upgrade to IDC postoperatively is reflected in the largely variant practice of performing SNB in the setting of a preoperative DCIS diagnosis, which ranges from 33% of cases in the United States (16) to 84.5% in Japan (17). At the same time, while SNB may be accompanied by less morbidity than axillary clearance, it is considerably more morbid compared to a non-invasive procedure (18). A way to address unnecessary biopsies resulting in complications is to “load” the SN with SPIO by injecting at the first operation, but remove the SN only if the postoperative diagnosis upgrades to IBC, in a second operation. This does not contradict patient safety, since no exposure risks have been reported.
Finally, the preoperative injection of SPIO could serve for the transcutaneous identification of the SN. That could, in turn, allow for a targeted minimally invasive biopsy, either MRI-guided or even US-guided in the hotspot of the magnetic probe. This could prove to be of particular value in patients with a clinically negative axilla that are planned for primary systemic treatment, since the SN could be localized with the probe on axillary US, biopsied, and marked in a similar way that the index node is marked in cases of a positive axilla before neoadjuvant treatment (19). In that way, the “true” SN could be retrieved easily, minimizing the high false-negative rate, which is a consideration, especially in the presence of metastases (20).
There are limitations in this pilot study. Despite the fact that the feasibility of preoperative SPIO injection was demonstrated, the feasibility of SPIO as a sole tracer for SN detection has also got to be depicted. Additionally, larger numbers are required to document that SPIO performance is not compromised by factors such as axillary metastases, previous surgery, or primary systemic treatment. As far as cost issues are concerned, compared to the intraoperative injection of SPIO, preoperative injection is advantageous as one could spare the 5-min massage at the injection site as well as the 20 min required as minimum for SPIO to migrate to the axilla, thus sparing operative time. However, a small pilot study is not suitable to address cost issues, which is why the authors refrained from presenting data which may not lead to safe conclusions. Currently, trials to assess the efficacy of SPIO as a sole tracer, the cost, and the logistics, as well as the aforementioned implementations, are accruing data in our institution.
Conclusion
Preoperative SPIO injection for SNB is feasible. It does not seem to be affected by the presence of metastases, previous ipsilateral breast surgery, or systemic treatment. It simplifies the logistics and has interesting implementations, especially in patients with DCIS. Further studies are needed to elucidate this interesting new method.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The study was approved by the Ethics Committee in Uppsala University and funded by Uppsala University.
Supplementary Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
