Abstract
Background and Aim:
The toe skin temperature in vascular patients can be low, making reliable toe pressure measurements difficult to obtain. The aim of this study was to evaluate the effect of heating on the toe pressure measurements.
Materials and Methods:
A total of 86 legs were examined. Brachial pressure and toe pressure were measured at rest in a supine position using a laser Doppler device that also measured skin temperature. After heating the toes for 5 min with a heating pad, we re-measured the toe pressure. Furthermore, after heating the skin to 40° with the probe, toe pressures were measured a third time.
Results:
The mean toe skin temperature at the baseline measurement was 24.0 °C (standard deviation: 2.8). After heating the toes for 5 min with a warm heating pad, the skin temperature rose to a mean 27.8 °C (standard deviation: 2.8; p = 0.000). The mean toe pressure rose from 58.5 (standard deviation: 32) to 62 (standard deviation: 32) mmHg (p = 0.029). Furthermore, after the skin was heated up to 40 °C with the probe, the mean toe pressure in the third measurement was 71 (standard deviation: 34) mmHg (p = 0.000). The response to the heating varied greatly between the patients after the first heating—from −34 mmHg (toe pressure decreased from 74 to 40 mmHg) to +91 mmHg. When the toes were heated to 40 °C, the change in to toe pressure from the baseline varied between −28 and +103 mmHg.
Conclusion:
Our data indicate that there is a different response to the heating in different clinical situations and in patients with a different comorbidity.
Introduction
Toe pressure (TP) measurement is one cornerstone in the assessment of patients with peripheral arterial disease (1, 2). It is less prone to bias caused by mediasclerosis than ankle pressure and thus preferred in diabetic patients. TP is used in the decision-making concerning an intervention for a patient with symptomatic lower limb ischemia, as well as in measuring the success of revascularization and in surveillance after the procedure. The threshold for critical limb ischemia (CLI) is a TP of <30 mmHg in nondiabetic patients and 50 mmHg in diabetic patients (2–4). TPs are, however, sensitive to extrinsic and intrinsic factors, which may lead to false values. Therefore, patients are advised not to smoke or drink coffee prior to measurements. Furthermore, the room temperature should be constant. Coldness of the feet may lead to vasoconstriction in some patients and falsely low TPs. To overcome this, toes can be warmed before the measurement. The measurement protocol is standardized in our institution, and toe temperature is measured from each patient. We have previously studied the repeatability of TPs with three different devices: two photopletysmographic (PPQ) devices and one laser Doppler (LD) device (5). We found that the repeatability was the best with the LD device. Also, LD device was the most sensitive in small TP values. In this study, we evaluate the influence of two different heating methods on TP values.
Materials and Methods
In the Helsinki University Hospital vascular laboratory, approximately 6000 TPs are measured each year by trained vascular nurses. The measurement protocol is standardized, and the Perimed system 5000 (Perimed, Stockholm, Sweden), which is based on LD technology, is used only for a total of seven measurement points. For this prospective study, 46 patients and 86 legs were examined. All patients were examined three times in the supine position, with the feet at heart level, and patients did not leave the bed between the measurements. When a stable signal was obtained, the digital cuff was inflated until the disappearance of the LD signal, usually at 200 mmHg. TP was determined from the point where the LD signal began to rise from the baseline value during the release of the arterial occlusion. Afterward, the signals were checked offline. An LD device also measured the skin temperature. After heating the toes for 5 min with heating pads, we re-measured the TP. A third measurement was performed after using the heating probe to increase the skin temperature locally to 40 °C. The study protocol was accepted in the Institutional Review Board of Helsinki University Hospital.
Statistical Analysis
Statistical analysis was performed using the SPSS statistical software for windows (SPSS version 22.0, SPSS Inc., Chicago, IL, USA). Data are presented as mean values and standard deviation (SD) and median values and interquartile range (IQR). Data on patients with claudication and severe limb ischemia as well as diabetics and nondiabetics are presented separately. To compare the TP values after heating with the baseline value, the paired-sample repeated measure test was used. Median values were compared using nonparametric-related sample test. The proportions were compared using the chi-square test. A p value <0.05 was considered statistically significant.
Results
A total of 46 patients and 86 legs were measured. The basic demographics and risk factors are presented in Table 1. The mean room temperature during the measurements was 22.7 °C. The mean toe skin temperature at the baseline measurement was 24.0 °C (SD: 2.8). After heating the toes for 5 min with a warm heating pad, the skin temperature rose to a mean 27.8 °C (SD: 2.8; p = 0.000). The mean TP rose from 58.5 (SD: 32) mmHg to 62 (SD: 32) mmHg (p = 0.029). Furthermore, after the skin was heated up to 40 °C with the probe, the mean TP in the third measurement was 71 (SD: 34) mmHg (p = 0.000).
Characteristics of the 86 patients.
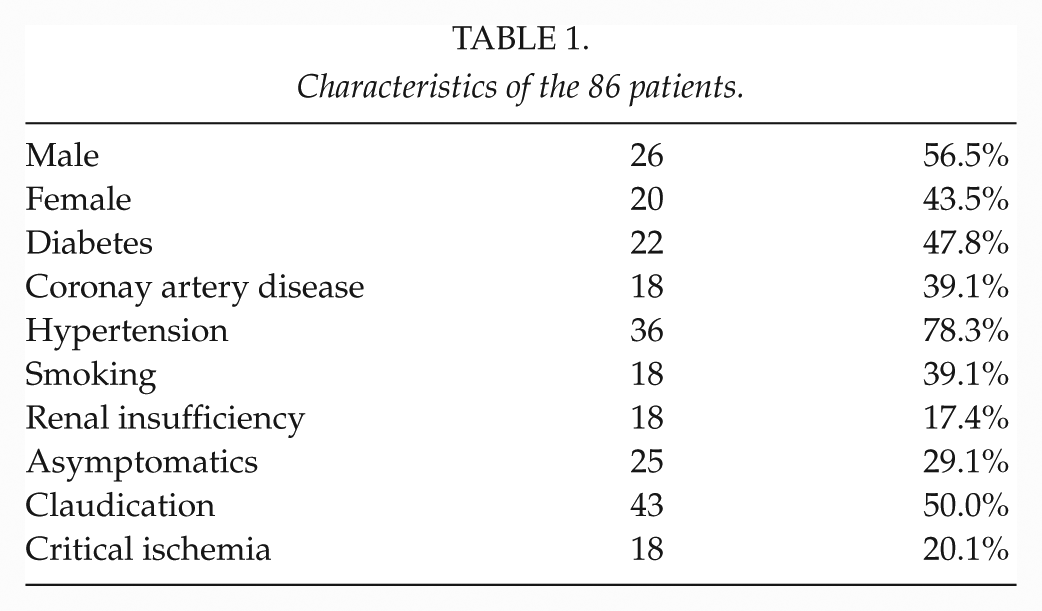
There were 25 legs with no symptoms, 43 with claudication and 18 with CLI. The mean TP at baseline in the asymptomatic legs, claudicants, and CLI legs was 65 (SD:40), 60 (SD: 29), and 47 (SD: 21) mmHg, respectively. In all three groups, the TP increased significantly after the heating, and the increase was the same in all groups (Tables 2 and 3).
The influence of heating to the toe pressure values according to the patient’s symptom and diabetes status.
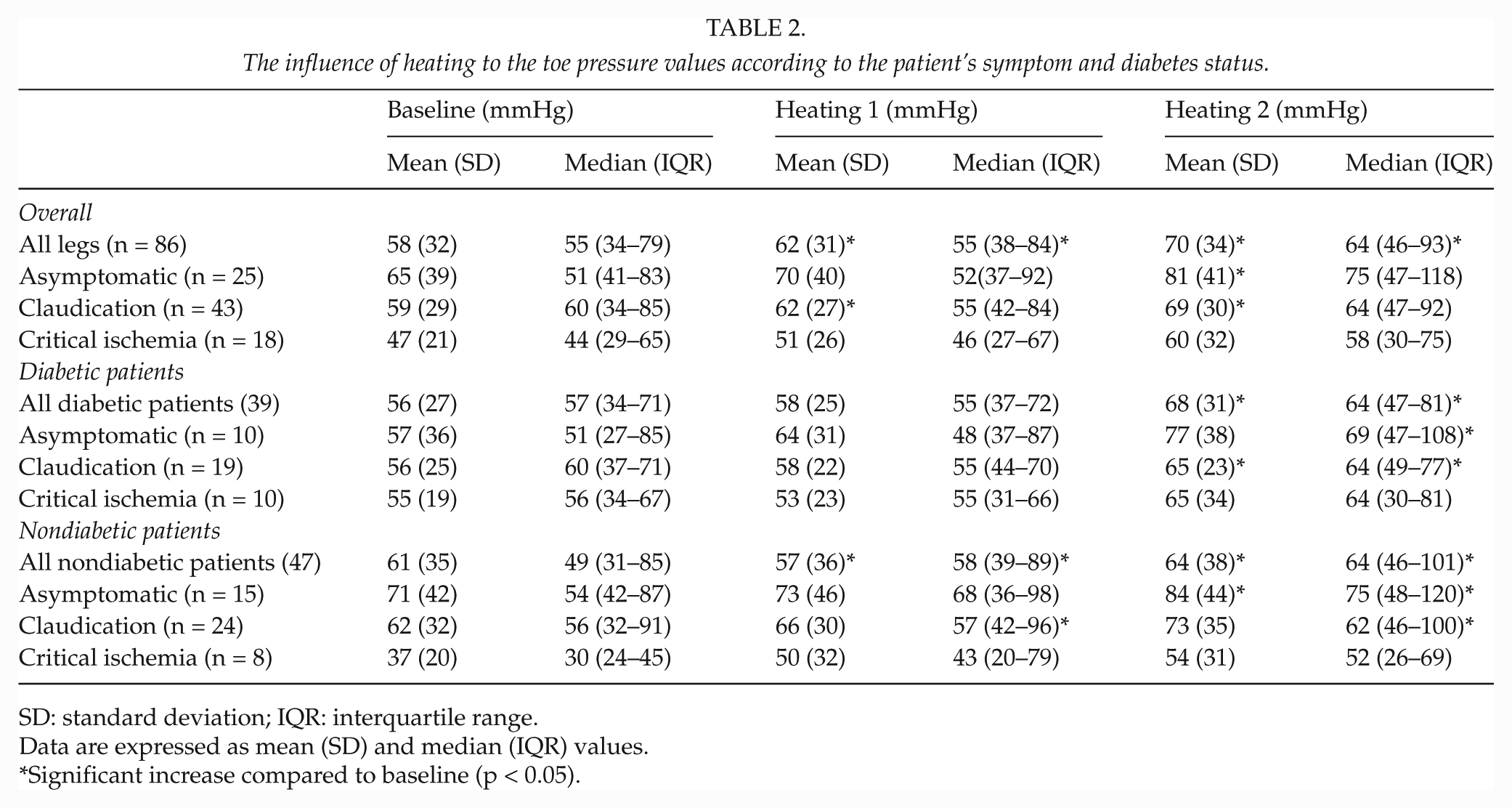
SD: standard deviation; IQR: interquartile range.
Data are expressed as mean (SD) and median (IQR) values.
Significant increase compared to baseline (p < 0.05).
The absolute change in the toe pressure values according to the patient’s symptom and diabetes status.
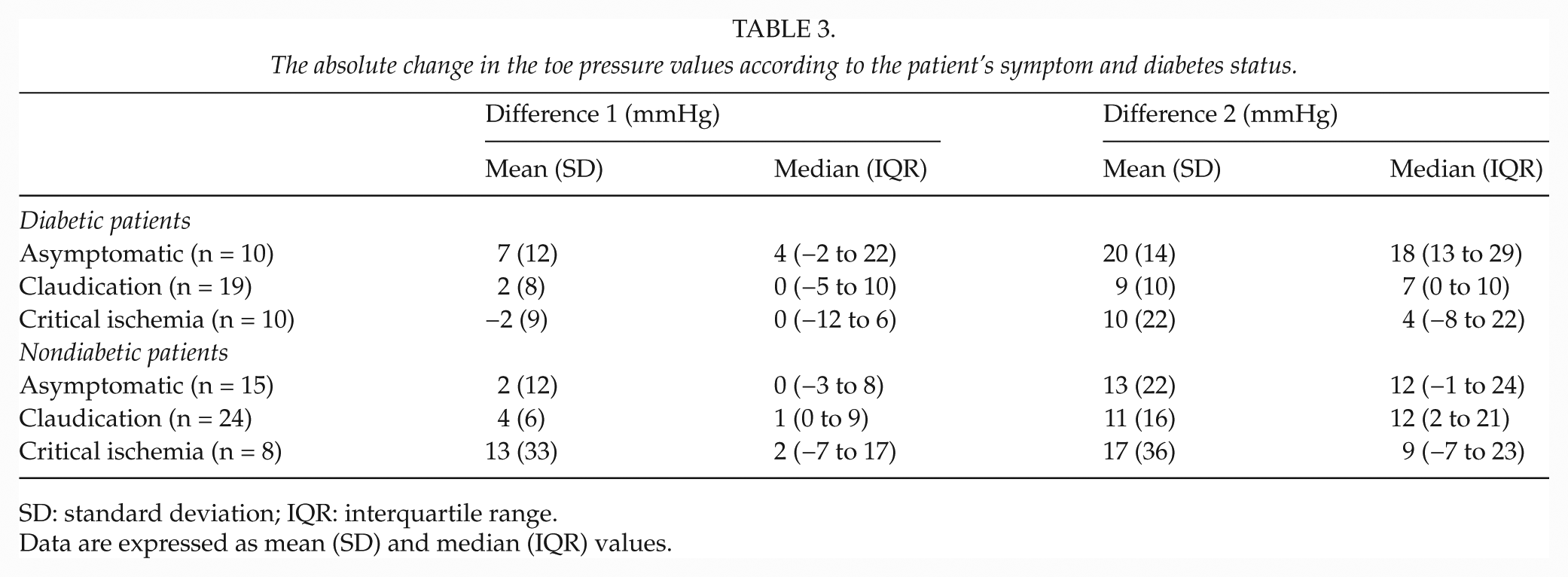
SD: standard deviation; IQR: interquartile range.
Data are expressed as mean (SD) and median (IQR) values.
In 47 limbs of nondiabetic patients, the mean TP was 61 (SD: 36) mmHg, compared to the 56 (SD: 27) mmHg in 39 diabetics. After the first heating, the mean TPs were 66 (SD: 36) mmHg in nondiabetic and 58 (SD: 24) mmHg in diabetic patients. The mean and median values in diabetic and nondiabetic patients in the three groups are presented in Table 2.
Individual Variation
The response to the heating varied greatly between the patients after the first heating—from −34 mmHg (TP decreased from 74 to 40 mmHg) to +91 mmHg (TP increased from 14 to 104 mmHg) (Fig. 1). After the first heating, the TP decreased or remained the same in the second measurement in a total of 40 (46.5%) legs. When the toes were heated to 40 °C, the change in TP from the baseline varied between −28 and +103 mmHg. After the second heating, the TP was unchanged or worse in 22 (25.6%) patients, and the proportion was the same in diabetics versus nondiabetics (25.6% vs 25.5%, respectively) and in symptomatic versus asymptomatic legs (20.0% in asymptomatic legs, 20.9% in claudicants, and 33.3% in CLI legs) (Figs 1 and 2).
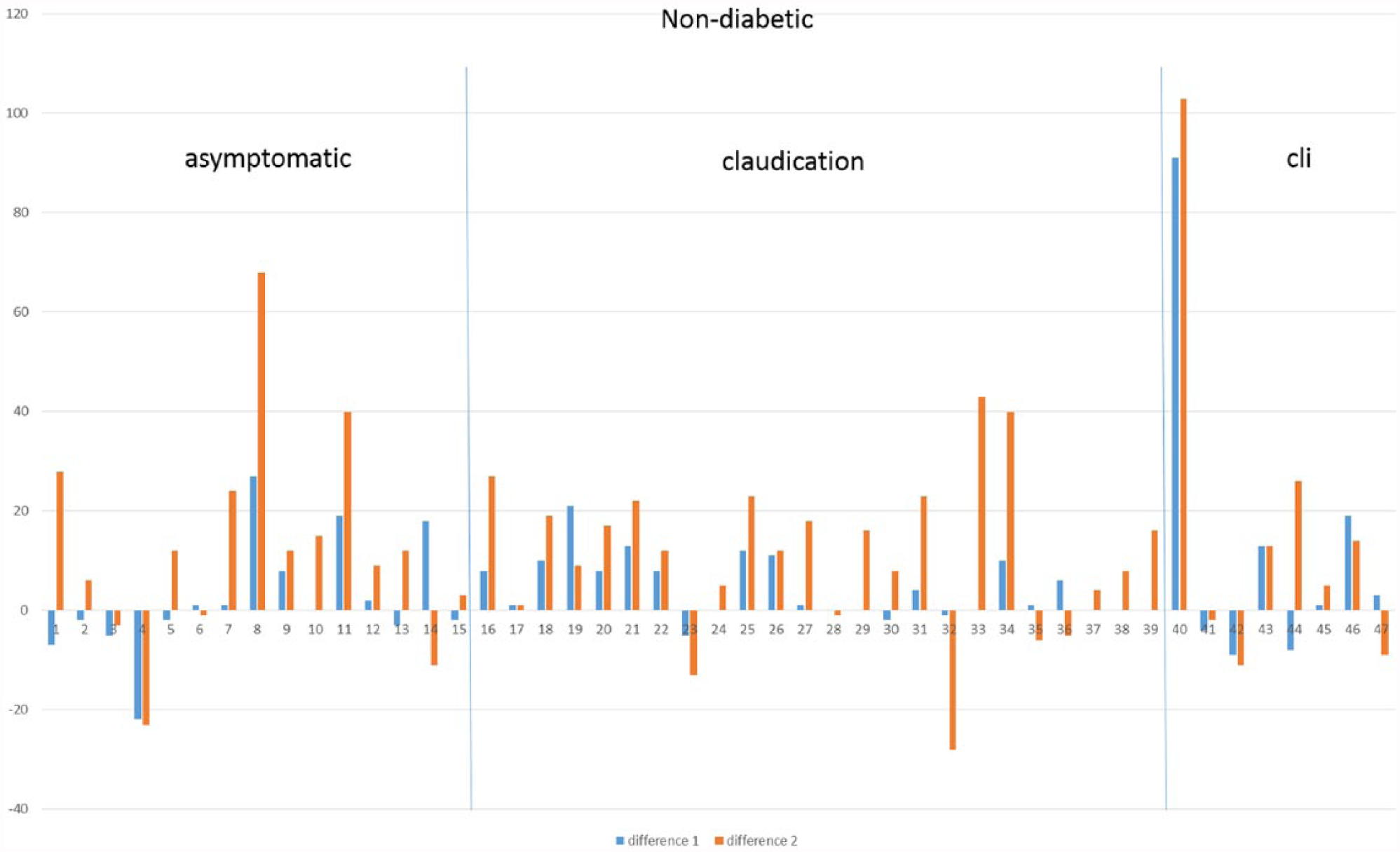
Individual variation in nondiabetic patients.
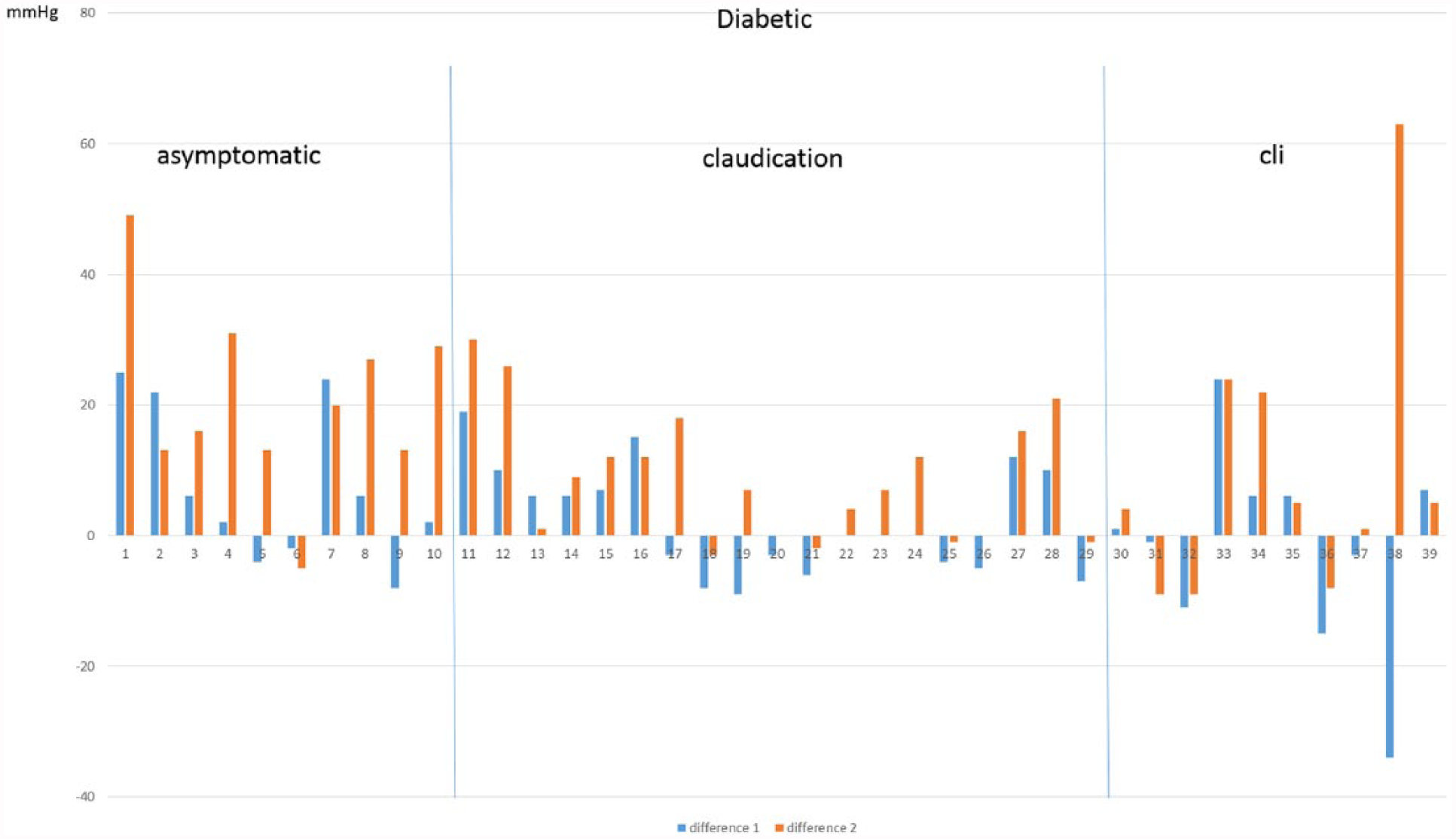
Individual variation in diabetic patients.
Discussion
The Transatlantic Inter-Society Consensus document advocates TP measurements to assess peripheral arterial insufficiency and recommends a cut-off value of 30 mmHg for diagnosing CLI (3, 6, 7). This low value requires an accurate and reproducible measurement technique. TP measurements are useful in the diagnostic workup of patients with a suspicion of peripheral arterial disease, particularly in the presence of mediasclerosis due to diabetes, and especially when ankle blood pressure measurements cannot be assessed reliably (2, 8, 9). However, TP measurements have been shown to be highly susceptible to distal temperature changes, and it has been argued that insufficient heating can lead to disease misclassification (10). Therefore, lower limb heating has been recommended to improve standardization (11, 12), and different heating protocols are used in vascular laboratories.
The LD technique, noninvasively detecting skin microcirculatory perfusion, has been proposed as a useful tool for TP measurements (13–15). In our earlier study, LD device was found to be more repeatable than PPQ TP measurements (4). Although there are studies on the effect of heating, there is no consensus regarding the routine use of heating during TP measurement. Our current study aimed to evaluate the effect of heating on TP. We performed a heating test, measuring the baseline TP, the TP induced by heating for 5 min, and the TP after heating the skin surface locally to 40 °C. We found that heating increased the TPs significantly, especially when the heating was performed with the heating probes to 40°, the mean increase being 12 mmHg compared to the baseline TP. However, there was significant individual variation as in one in five legs, the heating did not increase the TP but even decreased it in many patients.
Local heating reduces the temperature-induced variability in perfusion. The induced superficial vasodilatation allows for a more substantial reperfusion to be detected after the period of arterial occlusion. Furthermore, it facilitates the detection of the baseline signal in case the resting perfusion without local heating is very low, as in the cases in which LD could detect a perfusion signal (16).
The increase in TP after heating has been described earlier. Ubbink (15) studied the effect of heating using two devices, a PPQ and an LD device. They heated the skin with a probe to 37 °C and found a mean increase of 7.7 mmHg in TPs after heating. This is in line with our finding of a mean increase of 12 mmHg after heating the toe locally with a probe to 40 °C. An interesting finding in our study was the relatively high proportion of patients who did not respond to heating or whose TP even decreased after heating. However, some of the patients had a dramatic increase in their TPs. This is a reflection of the fact that every patient has an individual hemodynamic response. Some patients probably had a vasospasm and the influence of heating in these patients was significantly positive. On the contrary, the negative influence of heating can be due to the effect of heating on the sympathetic nervous system, causing vasoconstriction (17). Although the number of patients in the subgroups was too small to make any far-reaching conclusions, there was a trend toward a lower increase in the TPs of diabetic patients. For almost half of the diabetic patients, the first heating had no influence on the TP, and the mean increase was half of that of their nondiabetic counterparts (5 vs 2.5 mmHg). Especially diabetics with CLI had a poorer response. This may be due to the fact that diabetics have significantly decreased metabolic, neurogenic, and myogenic responses to thermal stress (18). One additional mechanism could be the venoarterial reflex that restricts arterial inflow and limits the rise of capillary pressure, protecting the microcirculation from the adverse effects of hypertension (19). A reduction in the precapillary vasoconstriction results in higher capillary pressure in the dependent position (19, 20). This can explain the worse response to heating in diabetic patients affected by CLI.
In our study, we standardized the technique of measurement in order to reduce the bias related to the cold-induced vasospasm. However, there were no significant differences in basal temperatures in the studied patients. The heterogeneous response to the heating in our patients with an increase in TP can be due to the fact that each patient needed a different time of (warm up) adaptation after a possible cold-induced vasospasm. It is difficult to fix the baseline and to avoid all external factors.
The most important question raised by our study is the need for and influence of heating in clinical practice. TP is widely used in the decision-making when the need for as well as the results of revascularization are evaluated. Most probably, many patients have vasoconstriction at baseline without heating, and the values tend to be too low in these patients. However, more hazardous would be the situation where the TP would be too high as a result of heating, presenting a falsely positive picture of the foot circulation or the result of revascularization. In our series, there were eight legs with a TP of <30 mmHg at baseline, and after the first heating, two of these patients demonstrated a clear increase in their TP to >30 mmHg; after the second heating, five of these patients had a TP of >30 mmHg. In the diabetic patients, 13 legs had a TP of <50 mmHg, and after the first heating, the pressure increased to >50 mmHg in 2 of these, while 3 of the 13 legs had a TP of >50 mmHg after the second heating. As the guidelines have been written on the basis of older studies with no information on the heating of the toes during TP measurements, the values after heating should be interpreted with caution. Furthermore, the best solution would probably be to measure TP without and with heating, but this is time-consuming and may be impossible in daily practice. Another solution could be a measurement without heating and, in the cases of low pressure, performing another measurement with heating probes.
The major limitation of our study is the limited number of patients. However, the major findings can be considered reliable, but the analysis in the subgroups is limited due to the fact that there are not enough patients. Especially the number of CLI patients was low, only 18. In addition, the diabetics have been analyzed separately. Even there is a true difference, we are not able to show it in our study as the differences do not reach statistical significance, and the study may suffer from type II error. The strength of the study is that the same person (T.G.) made all the measurements in a standard environment and with the same equipment. Also, the vast experience and long-standing traditions of the vascular laboratory in our hospital make the study results reliable.
Conclusion
Our data indicate that there is a different response to heating in different clinical situations and in patients with different comorbidities. The true meaning of these heterogeneous responses in peripheral arterial disease remains unknown and would be an interesting area of research in the future.
The results suggest that there are many mechanisms that participate in the vasodilatation, sometimes causing high variability in TP results, and that we should be careful in TP measurement to avoid the effects of vasospasm and vasodilatation hiding the real TP.
Further investigations on a higher number of patients are still required to determine whether the pressure measurement after skin heating can be used as a more sensitive strategy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
