Abstract
Purpose:
Clinical and endoscopic assessment of the outcome after fundoplication for pediatric gastroesophageal reflux.
Basic procedures:
Hospital records of 279 consecutive patients who underwent fundoplication for gastroesophageal reflux from 1991 to 2014 were reviewed. Underlying disorders, clinical and endoscopic findings, imaging studies, pH monitoring, and surgical technique were assessed. Main outcome measures were patency of fundoplication, control of symptoms and esophagitis, complications, redo operations, and predictive factors of failures.
Main results:
A total of 279 patients underwent 300 fundoplications (277 primaries and 23 redos). Underlying disorders in 217 (72%) patients included neurological impairment (28%) and esophageal atresia (22%). Indications for fundoplication included recalcitrant gastroesophageal reflux symptoms (44%), failure to thrive (22%), respiratory symptoms (15%), esophageal anastomotic stricture (4%), apneic spells (2%), and regurgitation (2%). Preoperative endoscopy was performed in 92% and pH monitoring in 49% of patients. Median age at primary fundoplication was 2.2 ((IQR = 0.5–7.5)) years. Fundoplication was open in 205 (74%; Nissen n = 63, Boix-Ochoa n = 97, Toupet n = 39, and other n = 6), laparoscopic in 72 (24%; Nissen n = 67 and Toupet n = 5), and included hiatoplasty in 73%. Clinical follow-up was a median of 3.9 (IQR = 1.2–9.9) years. Mortality related to surgery was 0.3%. Symptom control was achieved in 87% of patients, and esophagitis rate decreased from 65% to 29% (p < 0.001). Fundoplication failed in 41 (15%) patients. Failure was predicted by esophageal atresia risk ratio = 3.9 (95% confidence interval = 1.3–11, p = 0.01), any underlying disorder risk ratio = 3.1 (95% confidence interval = 1.1–9.1, p = 0.04), and hiatoplasty risk ratio = 2.6 (95% confidence interval = 1.1–6.6, p = 0.03). Of the 23 redo-fundoplications, 32% failed.
Conclusion:
The majority of patients who underwent fundoplication had an underlying disorder. Primary fundoplication provided control of symptoms in almost 90% of patients and also reduced the rate of esophagitis. Failure of primary fundoplication occurred in 15% of patients, and an underlying disorder, esophageal atresia, and hiatoplasty increased the risk of failure.
Introduction
In children, gastroesophageal reflux disease (GERD) may cause severe problems of nutrition, growth, and respiratory function (1). In the management of congenital malformations such as esophageal atresia (EA), laryngotracheal anomalies, and congenital diaphragmatic hernia (CDH), control of GERD is of paramount importance(2). Abnormal esophageal anatomy and function, such as short dysmotile esophagus in EA, lack of diaphragmatic muscular support of the esophagogastric region in CDH, and abnormal neural regulation and impaired esophageal motility in neurological diseases, predisposes to severe GERD, which cannot be controlled by conservative measures, and surgical management is required (2). Although surgery effectively controls GERD, long-term complications such as recurrence of GERD, retching, or dysphagia are not uncommon (2–4).
In this retrospective study, we investigated the outcome of 300 consecutive fundoplications for pediatric GERD in a national tertiary pediatric surgical center from 1991 to 2014. We analyzed operative complications, surgical failures, redo operations, and their predictive factors.
Methods
The hospital review board approved this study. Hospital records of 279 consecutive patients who underwent fundoplication for GERD in our institution during the 24-year period from 1991 to 2014 were completely reviewed. Surgical technique, preoperative and postoperative endoscopic findings, imaging studies, results of esophageal pH monitoring, clinical symptoms, and medication were carefully assessed.
The main indication for fundoplication was complicated GERD unmanageable with conservative treatment. The complications included failure to thrive, chronic respiratory problems, aspiration, apneic spells, esophageal stricture, persisting reflux esophagitis, and uncontrollable gastroesophageal reflux (GER) symptoms. Whenever clinically possible, conservative treatment was first attempted. Conservative treatment of GERD included prone positioning, thickening of feeds and continuous tube feeding via nasogastic tube, gastrostomy or (transgastric) jejunostomy with or without oral proton pump inhibitors (1–2 mg/kg daily), ranitidine (5 mg/kg in two daily doses), or cisapride (1 mg/kg in three daily doses).
Preoperative upper gastrointestinal endoscopy and 24-h esophageal pH monitoring or pH-impedance monitoring were performed when clinically applicable to provide supporting data for the decision to perform surgery. Endoscopies were performed with flexible videogastroscope (Pentax EG 2985, Japan). Esophagitis was graded histologically as none, mild, moderate, or severe. Of the metaplastic changes, only intestinal metaplasia (columnar epithelium and goblet cells) was recorded (5, 6). Esophageal pH monitoring showing acidic reflux over 10% of the total measurement time or 5% of total time minus 2 h after meals or reflux periods exceeding 5 min (7) or moderate or severe histological esophagitis was considered indicative of significant GERD. Preoperative upper gastrointestinal tract contrast study was routinely performed to exclude a small intestinal obstruction, whereas reflux of contrast into the esophagus was not considered diagnostic for GERD. Esophageal manometry or gastric emptying studies were performed selectively. In otherwise healthy children, socially restricting regurgitation unresponsive to anti-reflux medication was considered an indication for fundoplication, although endoscopy and pH monitoring in these patients were generally normal.
The most common surgical techniques for abdominal fundoplication were Nissen, Toupet (2), and Boix-Ochoa (8) and for thoracic fundoplication was Belsey Mark IV (9). Laparoscopic fundoplications were started in 1998. In patients without endoscopic or radiologic evidence of hiatal hernia, routine hiatoplasty in transabdominal fundoplications was abandoned in 2007.
Postoperative weaning from anti-reflux medication was individualized. Patients with EA underwent a programmed endoscopic surveillance from 1 to 15 years of age. Preoperative esophageal pathology, such as moderate or severe esophagitis, stricture, or hiatal hernia, warranted routine postoperative endoscopic surveillance also in other patients. Other indications for postoperative endoscopy, contrast study, or pH monitoring included dysphagia and recurrence of any symptoms or complications of GERD after surgery.
In follow-up endoscopy, patency of the fundoplication was assessed by antegrade viewing of the gastroesophageal junction and retrograde viewing of the fundoplication wrap at esophagogastric junction by inverting the gastroscope in air-filled stomach. Signs of failed fundoplication included loose or absent wrap, wrap retraction into thorax, or hiatal and paraesophageal hernias (10). Esophagitis was assessed histologically. Persisting moderate or severe postoperative esophagitis—after fundoplication with anti-reflux medication—was considered as a treatment failure. Mild esophagitis, a very common finding in GERD, was not considered significant.
The main outcome measure was the patency of fundoplication classified according to the last recorded clinical or endoscopic assessment as “fundoplication intact” or “fundoplication failed.” Clinical outcome was classified as good (full resolution of symptoms), moderate (attenuation of symptoms but some complaints), or poor (inadequate response).
Statistical Analyses
Statistical analyses were performed with StatView® 512 program (Calabasas, CA, USA). Unless otherwise stated, the data are presented as medians and interquartile range (IQR) or as frequencies. The effects of various factors on outcome were assessed by a logistic regression analysis. Frequencies were compared with Fisher’s exact test. Values of p < 0.05 were considered statistically significant.
Results
A total of 279 consecutive patients (155 males) were included. We performed primary fundoplication in 277 patients, 2 patients were referred for a re-fundoplication. Underlying conditions are classified in Table 1. Syndromes and associated disorders included arthrogryposis (n = 1), Asperger (n = 2), CATCH 22 (n = 2), Charcot–Marie Tooth (n = 1), CHARGE (coloboma, heart defects, atresia choanae, growth retardation, genital abnormalities, and ear abnormalities) (n = 9), Cornelia de Lange (n = 3), Cri du Chat (n = 1), Crouzon (n = 1), Down (n = 6), Ehlers–Danlos (n = 1), Goldenhar (n = 1), Kabuki (n = 1), Langer-Giedion (n = 1), Leigh (n = 1), Moebius (n = 2), Nager (n = 1), Noonan (n = 1), Pierre Robin (n = 2), Tourette (n = 1), Townes–Brock (n = 1), Turner (n = 2), VACTERL (vertebral defects, anal atresia, cardiac defects, tracheoesophageal fistula, renal anomalies, and limb abnormalities) (n = 20), and Williams (n = 1).
Underlying diagnosis in 279 pediatric patients who underwent fundoplication for GERD.
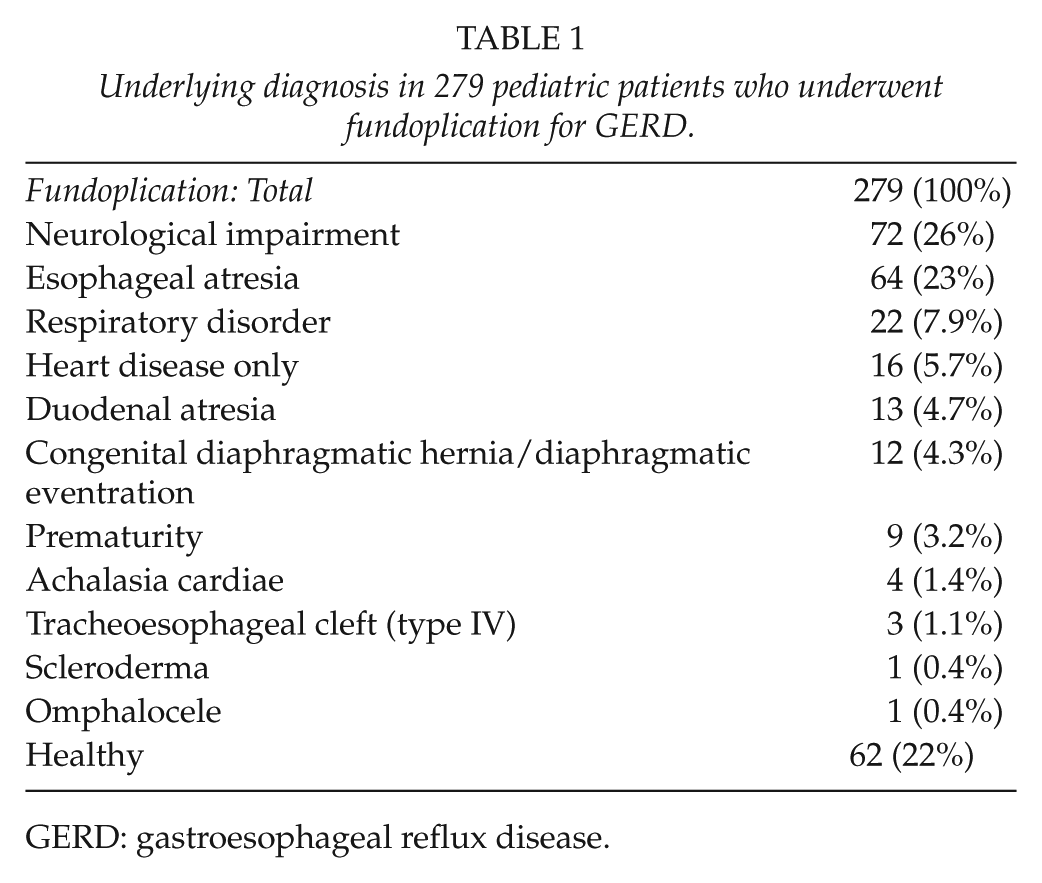
GERD: gastroesophageal reflux disease.
Fundoplications
A total of 277 patients underwent primary fundoplication. Of the 277 patients, 19 (7%) underwent redo-fundoplication—18 in our center and 1 after transition in an adult center. In addition, two patients who underwent the primary operation elsewhere were referred for redo-fundoplication (eventually, one patient underwent one and the other four redo-fundoplications). Total number of fundoplications was 300 (277 primaries and 23 redo-fundoplications). Median age at primary fundoplication was 2.2 (IQR = 0.5–7.5) years.
Only 22% of the patients were otherwise healthy, whereas the most common underlying conditions included neurological impairment (NI; 28%) and EA (23%). The main indications for fundoplication are given in Table 2. Patients with EA underwent fundoplication earlier, at median age of 5.0 (IQR = 3.0–28) months, compared with other children (36 (IQR = 9.3–103) months, p < 0.001). Healthy children underwent fundoplication at a later age of 5.9 (IQR = 2.5–10) years compared to the rest, 1.4 (IQR = 0.4–5.9) years, p < 0.001). Of the 111 primary fundoplications performed before the age of 12 months, only 10 (9%) were performed in healthy children for the following indications: failure to thrive n = 2, reflux symptoms n = 3, and apneic episodes n = 5.
Indications for primary fundoplication in 277 pediatric patients.

GER: gastroesophageal reflux.
Of the 279 patients who underwent fundoplication, 254 (91%) underwent preoperative endoscopy. Mild (n = 66, 29%), moderate (n = 65, 29%), or severe (n = 15, 7%) histological esophagitis was found in 200 (72%) patients, and intestinal metaplasia in one. Other significant findings included anastomotic stricture after repair of EA (n = 24), peptic stricture (n = 1), hiatal hernia (n = 6), paraesophageal hernia (n = 2), and failed primary fundoplication (n = 2, in two referred patients). Esophageal pH monitoring or pH-impedance monitoring showed pathological acid reflux in 137 patients; in addition, impedance monitoring showed impaired esophageal clearance in six patients with EA. Six patients underwent biphasic isotope test for gastric emptying with normal results; no preoperative esophageal manometries were done.
Types of primary fundoplications (n = 277) were Nissen (n = 63), laparoscopic Nissen (n = 67), Boix-Ochoa (n = 97), Toupet (n = 39), laparoscopic Toupet (n = 5), transthoracic Belsey Mark IV (n = 3), or other (Thal, Collis n = 3). Open technique was used in 205 (74%) operations (including 3 operations converted from laparoscopic to open) and laparoscopic in 72 (26%). Hiatoplasty was performed in 170 (83%) of the 205 open operations and in 31 (43%) of the 72 laparoscopic operations (p < 0.001).
Feeding gastrostomy was inserted to 94 (34%) patients before (n = 40), at the time (n = 45), or after (n = 9) fundoplication. Temporary detachment of a previously constructed gastrostomy was required in five operations. Gastrostomy was most common (53%) in patients with NI or cardio-respiratory disorders.
From the start of laparoscopic fundoplications in 1998, we performed a total of 172 fundoplications, including 72 (42%) laparoscopic and 100 (58%) open procedures. Laparoscopic fundoplication was favored in healthy patients (25/41) in relation to patients with an underlying condition (47/131), p = 0.01.
Follow-Up after Fundoplications
Of the 279 patients, 31 (11%) died after a median follow-up of 3.2 (IQR = 0.5–9.5) years. Fundoplication-related death occurred in one case due to sequelae after post-fundoplication small bowel strangulation. Other non-survivors included patients with EA (n = 6), tracheoesophageal cleft (n = 1), duodenal atresia with Down’s syndrome (n = 2), CDH (n = 2), cerebral palsy (n = 14), and heart disease (n = 5). Of the 279 patients, 10 (2%) were lost to follow-up. Clinical follow-up data of the remaining 269 patients were available, and in 75% of these, follow-up exceeded 12 months. Endoscopic follow-up data included 240 of 269 (85%) patients and exceeded 12 months in 180 of 240 (75%). Because of anastomotic complications, two patients with EA underwent esophageal replacement, and a patent fundoplication was surgically removed.
Median clinical follow-up after primary fundoplication was 3.9 (IQR = 1.2–9.9) years. Control of GERD symptoms was good in 219 (81%), moderate in 26 (10%), and poor in 24 (9%) patients. In 20 patients with good (n = 19) or moderate (n = 1) control of GERD symptoms, functional problems (frequent vomiting n = 6, dysphagia n = 8, inability to burp and vomit n = 5, slow gastric emptying n = 1) had mild (n = 15) or moderate (n = 5) negative effect on general outcome. At the time of latest follow-up visit, 77 (29%) of the 269 patients used protein pump inhibitors for anti-reflux medication. In five patients, the preoperative diagnosis of GERD changed to cyclic vomiting (n = 1), esophageal dysmotility (n = 3), and eosinophilic esophagitis (n = 2).
Endoscopic follow-up was performed in 240 (96%) of 269 patients. Median follow-up after primary fundoplication was 2.7 (IQR = 0.9–7.5) years with a median of 2 (IQR = 1–3) endoscopies per patient. Comparison between preoperative and postoperative biopsies was possible in 193 patients. The overall occurrence of esophagitis (mild, moderate, and severe) decreased from 65% to 29% (p < 0.001) and of moderate or severe esophagitis from 36% to 9% (p < 0.001). Postoperative endoscopy displayed no or mild histological esophagitis in 171 (90%) patients. Unhealed or recurred (moderate or severe) esophagitis was observed in 20 (10%) patients. Preoperatively, three patients had pathologic pH monitoring and histologically verified moderate esophagitis with some eosinophil leucocytes consistent with reflux esophagitis but not responding to proton pump inhibitors. Postoperative biopsies at 6 months still showed moderate esophagitis but with high amount of eosinophils (>50 eosinophils/microscopic field) consistent with eosinophilic esophagitis. In two patients, eosinophilic esophagitis was associated with dysphagia, and with oral fluticasone, dysphagia and eosinophilic esophagitis disappeared, whereas in the third non-symptomatic patient, eosinophilic inflammation disappeared without treatment. Five patients (2%), two with EA, one with NI, and two healthy patients—one with pre-fundoplication peptic esophageal stricture and the other with failed fundoplication and recalcitrant moderate esophagitis—developed intestinal metaplasia after 14 (IQR 12–18) postoperative years; all five were programmed for further endoscopic surveillance.
Endoscopic diagnosis of failed fundoplication was made in 38 (16%) patients. Findings included totally loosened wrap (n = 28), wrap retraction into thorax (n = 7), hiatal hernia (n = 3), and paraesophageal hernia (n = 1).
Complications after Fundoplications
Three patients were reoperated for small bowel strangulation (n = 1) and adhesive obstruction (n = 2). Two patients had gastric retention exceeding 1 week after fundoplication. An attempt to relieve pyloric spasm by endoscopic balloon dilatation resulted in gastric perforation, and both patients underwent emergency laparotomy and suture closure of perforations.
Postoperative endoscopic balloon dilatations of the fundoplication wrap were performed in 11 (5%) of 205 patients of whom 8 had persistent dysphagia and 3 a disturbing inability to burp and vomit after Nissen-type fundoplication. Three patients with dysphagia benefited from dilatations. In two patients with unsuccessful dilatations, esophageal biopsies indicated eosinophilic esophagitis and dysphagia attenuated after a course of oral fluticasone 250 mg daily. In three remaining patients with recalcitrant postoperative dysphagia, esophageal manometry showed impaired motility of the esophageal body, and in them, dysphagia attenuated with oral cisapride (5–10 mg three times daily).
Failed Primary Fundoplications
Of the 269 patients with follow-up data, primary fundoplication eventually failed in 41 (15%). Median time to failure was 3.0 (IQR = 0.7–11) years. Cumulative failure rate at 5 and 10 years were 86% and 83% (Fig. 1). Failures were diagnosed by recurrent symptoms combined with endoscopy in 38 or contrast imaging in 2 patients. The failure rate was highest in patients with EA (16/64, 25%) compared with other patients (25/205, 12%; p = 0.02), whereas the respective median lag times of 3.9 (IQR = 0.8–13) and 3.0 (IQR = 0.7–8.0) years from fundoplication to failure were similar (p = 0.61). Long-term failure rate in patients with any underlying disease (n = 209) or with EA or NI (n = 128) was higher than in otherwise healthy patients (n = 60), but the difference was not statistically significant (Figs 2 and 3). Of the 41 patients with failed primary fundoplication, 18 (44%) eventually underwent redo-fundoplication. In 23 patients, redo-fundoplication was not performed for the following reasons: mild GER symptoms (n = 7), GER symptoms managed with medication (n = 10), poor general condition (n = 2), and deceased (n = 4).
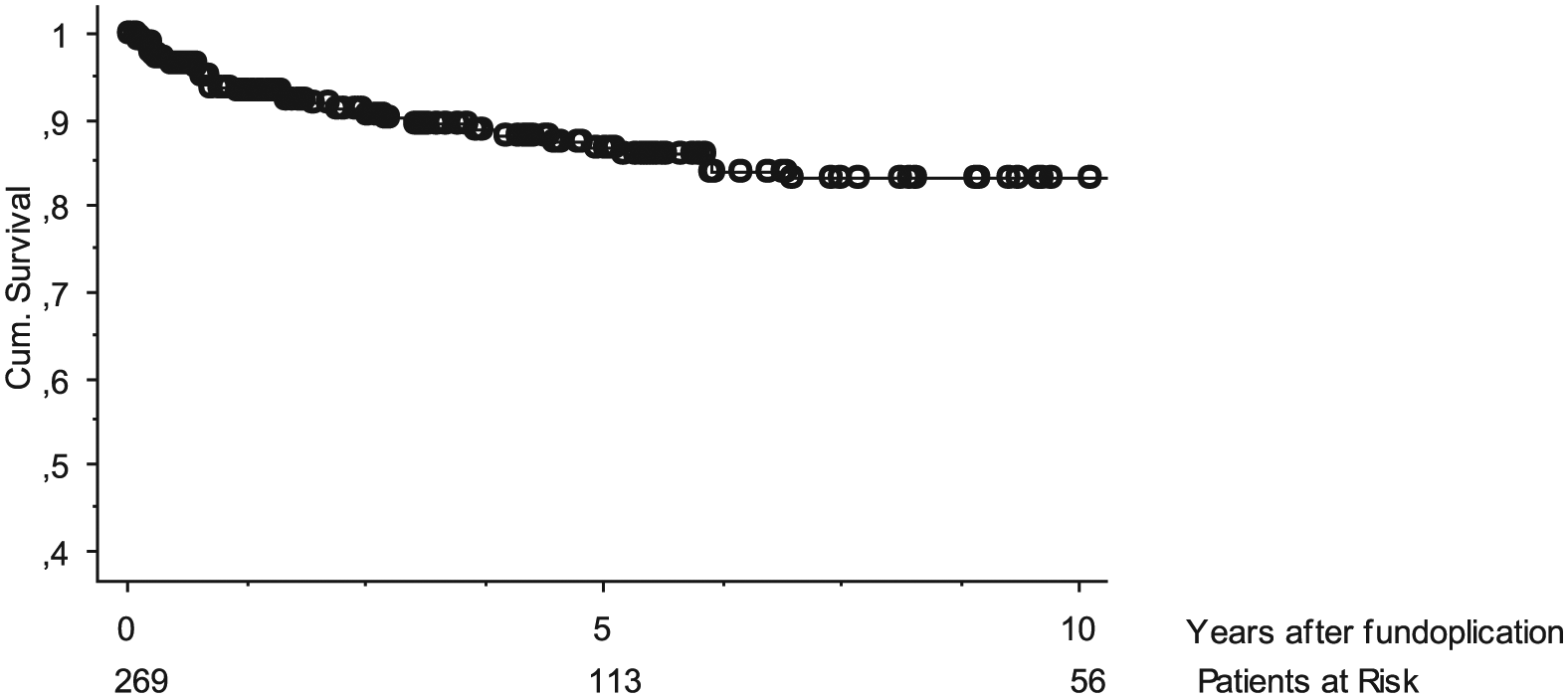
Patency rate of primary fundoplication in 269 patients; 5- and 10-year rate was 86% and 83%, respectively.
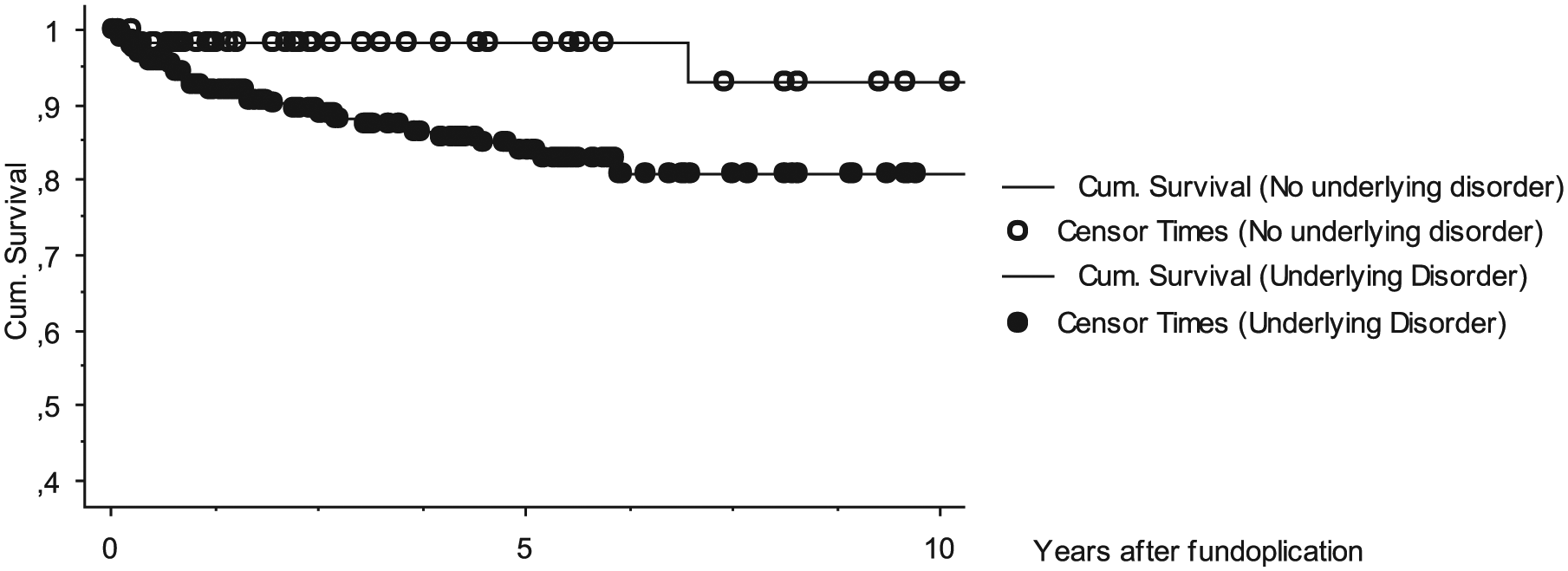
Cumulative patency rate primary fundoplication in patients without any underlying disorder (n = 60; 5- and 10-year survival was 98% and 93%, respectively) and in patients with an underlying disorder (n = 200; 83% and 81%, respectively) (Logrank Mantel–Cox, healthy vs underlying disease, p = 0.08).
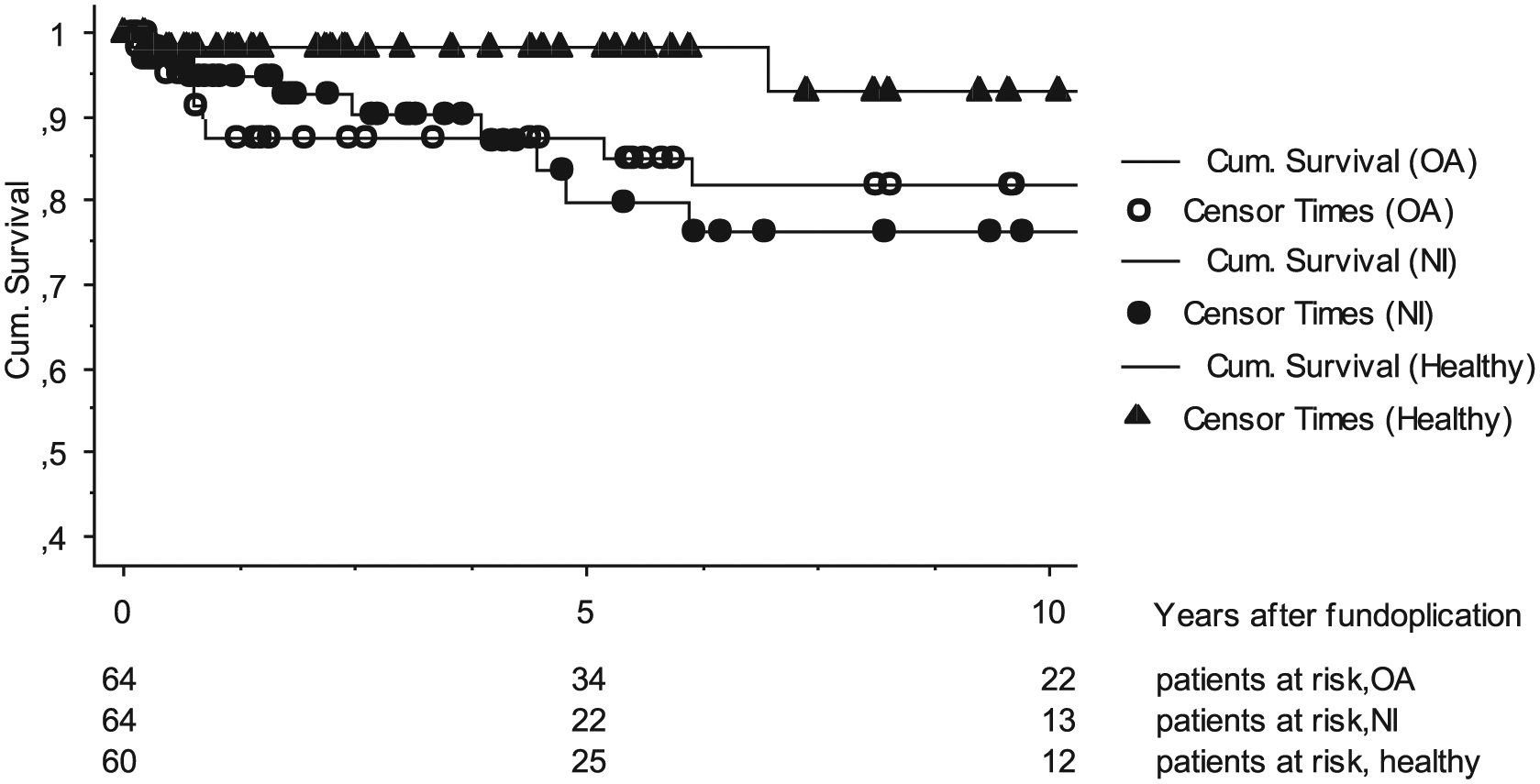
Cumulative patency rate of functioning primary fundoplication in healthy patients, in patients with esophageal atresia (EA), and in patients with neurological impairment (NI). The 5- and 10-year patency rate in healthy patients was 98% and 93%, respectively, in EA patients was 85% and 78%, respectively, and in NI patients was 80% and 76%, respectively (Logrank Mantel–Cox EA and NI vs healthy, p = 0.06).
Redo-Fundoplications
We performed a total of 23 redo-fundoplications in 20 patients. One patient underwent four redo-fundoplications. Preoperative diagnostics included endoscopy (n = 19), pH monitoring (n = 11), and contrast study (n = 7). Indications for redo-fundoplications were failed primary fundoplication in 20 patients, the primary fundoplication of whom was performed in our center in 18 and 2 referred patients; 1 patient required 3 further redo-fundoplications. Median age at redo-fundoplication was 2.2 (IQR = 1.1–5.1) years, and median delay after the primary fundoplication was 11 (IQR = 7–33) months. All the 20 patients had an underlying disorder, including EA (n = 7) with redo rate of 7/64 (11%), NI (n = 6) with redo rate of 6/64 (9%), isolated heart disease (n = 2), respiratory disorder (n = 2), and tracheoesophageal cleft type IV, impaired esophageal motility, and prematurity one each.
Redo-fundoplication techniques were open Nissen (n = 11), laparoscopic Nissen (n = 1), Thal (n = 1), and Belsey Mark (n = 7). One patient underwent three further re-operations with open Nissen (n = 2) and Collis (n = 1) fundoplications.
Nineteen (95%) redo patients had clinical (n = 3) or clinical and endoscopic (n = 16) follow-up data, covering 3.4 (IQR = 1.1–5.6) years. Endoscopically verified failure occurred in 6 (32%) after Nissen (n = 4), Thal (n = 1), and thoracic Belsey Mark (n = 1) fundoplication. One patient with tracheoesophageal cleft died of tracheal bleeding 1 month after redo-fundoplication. In patients with failed redo, underlying conditions included EA (n = 2) and heart disease, tracheoesophageal cleft, NI, and impaired esophageal motility one each. Median time from redo-fundoplication to failure was 5.2 (IQR = 2.7–33) months.
Of the five survivors with failed redo, one patient with impaired esophageal motility underwent three further redo-fundoplications and her symptoms are currently manageable with medication, one patient with EA and anastomotic stricture underwent esophageal resection, one with NI and one with EA have feeding jejunostomy, and one patient is managed medically. Overall, in 14 (74%) patients with redo-fundoplication, the clinical outcome was good.
Overall Control of Gerd after Fundoplication
Of the 269 patients with available follow-up data after primary or redo-fundoplication, effective control of GER was eventually achieved, with or without anti-reflux medication, in 233(87%) patients (primary fundoplication n = 219, redo-fundoplication n = 14). Of the remaining 36 patients, moderate control of GER was achieved by medical treatment in 20, esophageal replacement was done in 3 with EA, 2 had feeding jejunostomy, and GER symptoms remain poorly controlled in 10 (3%) patients (NI n = 7, RA n = 1, respiratory disorder n = 1, and heart disease n = 1).
Predictors of Failed Fundoplication and Unfavorable Clinical Outcome
Tested risk factors for failure of fundoplication, poor or moderate outcome, continued use of anti-reflux medication after fundoplication and non-healing of esophagitis included underlying disorder (EA, NI, heart disease or respiratory disorder, any disorder), age at surgery, fundoplication wrap technique, laparoscopic technique, hiatoplasty, gastrostomy, failed fundoplication, postoperative complications such as dysphagia, inability to burp or vomit, changed diagnosis, and non-healing of esophagitis. Factors were first assessed in univariate logistic regression analysis, and factors with risk ratios (RRs) of statistical significance were further assessed in multivariate logistic analysis.
Statistically significant risk factors for fundoplication failure were EA risk ratio (RR) = 3.9 (95% confidence interval (CI) = 1.3–11), p = 0.01; underlying disorder RR = 3.1(95% CI = 1.1–9.1), p = 0.04; and hiatoplasty RR = 2.6 (95% CI = 1.1–6.6) in univariate analysis and hiatoplasty RR = 2.7 (95% CI 1.1–6.8), p = 0.03, in multivariate analysis.
Risk factors for unfavorable (poor or moderate) clinical control of GER were fundoplication failure RR = 18(95% CI = 8.0–39), p < 0.001; EA RR = 2.6 (95% CI = 1.3–5.0), p = 0.01; and non-healing of esophagitis RR = 2.7 (95% CI = 1.1–6.8), p = 0.03, in univariate analysis and fundoplication failure RR = 27(95% CI = 7.6–93), p < 0.001, in multivariate analysis.
Independent predictors for continued use of anti-reflux medication after fundoplication were failed fundoplication, EA, and non-healing of esophagitis RR = 2.4–9.0 (95% CI = 1.1–19), p = 0.03–0.001. If fundoplication was intact, only EA correlated with continued use of anti-reflux medication after fundoplication RR = 2.8 (95% CI = 1.4–5.8), p = 0.04. Risk factors for non-healed esophagitis, both in univariate and in multivariate analysis, were failed fundoplication and EA—RR = 4.3–9.4 (95% CI = 1.2–47), p = 0.01–0.03, and RR = 5.1–11(95% CI = 1.2–61), p = 0.01–0.03, respectively.
Discussion
The main findings of this study were that (1) 80% of anti-reflux procedures were performed in order to control complications of GERD in children with associated disorders; (2) primary fundoplication provided good long-term control of GERD symptoms in over 80% of patients; (3) during long-term follow-up, 15% of primary fundoplications and 32% of redo-fundoplications failed; (4) underlying disorder and EA increased the risk of fundoplication failure.
The main shortcoming of the study was its retrospective nature and extended study period, leading to large variations in the follow-up times. However, both clinical and endoscopic follow-up covered over 90% of the patients, and in 75% of the patients, follow-up exceeded 12 months.
These results reflect our approach to surgical treatment of GERD in children. The great majority of the fundoplications and all esophagogastric disconnections were performed in children with significant associated disorders. Based on their cohort of over 11,000 patients, McAteer et al. (11) recently criticized the excessive use of surgical treatment of GER in healthy infants aged below 6 months in whom GER is physiological. Our management of GER in healthy infants has been very conservative. During the 24-year study, only 10 healthy children aged below 12 months have undergone fundoplication.
Primary fundoplication controlled GERD symptoms effectively in more than 80% of our patients. These results concur with Fonkalsrud et al. (12) who in a large cohort of 7467 children reported good to excellent results in 85%–95% of patients after 7.3 years. Here, fundoplication also promoted healing of esophagitis. Among the reasons for eventual poor outcome and unhealed esophagitis, fundoplication failure was, not surprisingly, a central risk factor. We found that after fundoplication, a substantial portion (29%) of patients, with or without failed fundoplication or persisting esophagitis, still used anti-reflux medication. This finding concurs with Lee et al. (13) who reported that 4.5 years after Nissen fundoplication, 58% of children used anti-reflux medication. Although fundoplication fails to wean all patients from anti-reflux medication, we think that control of symptoms and complications of GER with combination of fundoplication and medication is still of great value to a patient.
Postoperative dysphagia and inability to vomit are not related to the anti-reflux function of the fundoplication but nevertheless produce significant discomfort to a patient. Post-fundoplication dysphagia has been reported in 10%–24% of patients, and endoscopic wrap dilatations have been required in 4%–12% of cases (4, 14–16). Although we could not assess the exact incidence of dysphagia, incidence of wrap dilatations among our patients was similar, that is, 4%. Post-fundoplication dysphagia may occur in 45% of patients with EA (17), but because dysphagia in EA is associated with impaired esophageal motility and anastomotic stenosis, we did not consider it as a fundoplication-related complication.
During the assessment of postoperative dysphagia, we discovered patients with impaired esophageal motility and eosinophilic esophagitis. Symptomatic esophageal dysmotility can be diagnosed before fundoplication but only in children who are old enough to cooperate in esophageal manometry. Eosinophilic esophagitis and reflux esophagitis may have similar symptoms, and histology and symptom in both types of esophagitis may respond well to anti-reflux medication. A typical feature of eosinophilic esophagitis is dysphagia (18, 19). In two of our patients, eosinophilic esophagitis was found in association with post-fundoplication dysphagia. It is possible that these two patients actually had eosinophilic esophagitis from the beginning, but they underwent fundoplication because preoperative biopsies and pathological acid reflux in pH monitoring pointed to reflux esophagitis. Dysphagia and eosinophilic esophagitis may have manifested after weaning from proton pump inhibitors. We can speculate that with correct preoperative diagnosis of eosinophilic esophagitis, these patients could have been treated with medication without performing unnecessary fundoplication. If esophagitis in a child responds poorly to proton pump inhibitors, a clinician should keep in mind eosinophilic esophagitis and look for typical endoscopic appearance and histology. If the ascertained diagnose is eosinophilic esophagitis, the treatment of choice is medical treatment with oral cortisone with dietary measures, whereas fundoplication may significantly aggravate dysphagia and is contraindicated.
Our overall long-term failure rate of primary fundoplications (15%) fell within the range of 12%–43% reported in the contemporary literature (4, 14–16, 20–22). In this study and in a recent study by Lopez-Fernandez et al. (20), EA was a clear risk factor of fundoplication failure. Patients with EA often underwent open fundoplication at an early age because of GER-induced anastomotic stenosis or apneic spells. In these situations, fundoplication is usually required without delay and the risk of later failure is of little significance. Control of acute EA-related GER symptoms with fundoplication usually sustains the patient over the first years of life. If fundoplication eventually fails, a redo operation is not always necessary because GER symptoms at later age may subside or they can be managed conservatively.
In concordance with Baerg et al. (22), we found that hiatoplasty was also a risk factor for fundoplication failure. Hiatoplasty may be considered an essential part of a classic fundoplication (2), but since the majority of children have no hiatal hernia, routine dissection of crura followed by their suture apposition may not be necessary and minimal esophageal dissection may lead to less frequent fundoplication failures (23).
High failure rate with hiatoplasty may be associated with the fact that the patients in whom hiatoplasty is performed have an underlying disease which aggravates the risk of fundoplication failure. This may be true in patients with EA, but in our series in which only 22% of patients had EA, hiatoplasty remained the strongest overall predictor of failure.
Laparoscopic and open fundoplication were compared in two recent randomized studies. Pacilli et al. (24) found no difference in failure rates during 4-years of follow-up (laparoscopic 20%, open 12.5%), whereas in a study by Fyhn et al. (21), the difference during similar follow-up was significant (laparoscopic 37%, open 7%). In our series, laparoscopic technique was clearly preferred among children without underlying disorders, which may explain the low failure rate.
NI has been considered a risk for failed fundoplication (1, 12), but this could not be confirmed in this study.
Overall, we performed redo-fundoplications in less than half of patients with failed fundoplication. Failure rate of redo-fundoplications was relatively high (33%) and some patients ended up with permanent feeding jejunostomy. Horwood et al (25) reported that in neurologically impaired children, fundoplication with vagotomy and pyloroplasty resulted in lower need for revisional surgery (3%) than a standard fundoplication (17%) after 19.5 months of median follow-up. In NI patients with exceptionally severe complicated GERD and very limited ability to eat, we have used esophagogastric dissociation as a primary procedure in seven patients with satisfactory outcome. With a relatively low 9% redo-fundoplication rate in neurologically impaired patients during the 24-year follow-up, we, however, consider a standard fundoplication as the primary procedure in NI patients.
Anti-reflux surgery in pediatric patients focuses more often on prevention of life-threatening or disabling complications of GERD in children with significant associated disorders rather than to attenuate uncomfortable symptoms. Although anti-reflux surgery in children includes failures and complications, the generally favorable outcome warrants its use in carefully selected patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
