Abstract
Background and Aims:
Treatment of synthetic mesh infections has previously often resulted in mesh explantation. Negative pressure wound therapy has been used in these situations with encouraging results. The aims of this study were to evaluate wound healing, mesh preservation, and patient-reported outcome after negative pressure wound therapy of mesh infections.
Material and Methods:
Medical records of patients treated with negative pressure wound therapy for mesh infection and age-matched mesh-operated controls without postoperative complications were scrutinized in a retrospective study. An abdominal wall complaints questionnaire was used to evaluate patient-reported outcome.
Results:
Of 722 mesh operations performed 2005–2012, negative pressure wound therapy was used for treating postoperative mesh infections in 48 patients. A total of 48 age-matched controls were recruited from patients without wound complications. No differences were found between groups regarding preoperative characteristics. The following peroperative characteristics were significantly more frequent in the negative pressure wound therapy group: emergency operation, dirty/infected surgical field, surgical techniques other than laparoscopic intraperitoneal onlay mesh repair, implantation of more than one mesh, larger mesh size, longer duration of surgery, and mesh not completely covered with anterior rectus fascia. The entire mesh was salvaged in 92%, while four meshes were partly excised. Wounds healed in 88% after a median of 110 (range 3–649) days. In total, 85% in the negative pressure wound therapy group and 75% in the control group answered the questionnaire. There were no significant differences regarding pain, other abdominal wall symptoms, and satisfaction with the final result in favor of the controls.
Conclusion:
No mesh had to be explanted and wound healing was achieved in the majority of patients when negative pressure wound therapy was used for treatment of mesh infections. However, time to healing was long, and numerous procedures were sometimes needed. Positive long-term outcome was more frequently reported among controls.
Keywords
Introduction
Mesh implantation has become gold standard in incisional hernia repair (1). The mesh is used for reinforcement of the re-approximated fascia or as permanent substitute for native fascia when bridging of the hernia opening is necessary. The use of prosthetic mesh has its main advantage in decreased recurrence rates. In a Cochrane review from 2011, the recurrence rate after incisional hernia mesh repair was reported to be 16.4% compared to 33.3% for sutured repairs (2). The disadvantage is mesh-associated complications such as mesh infection, mesh extrusion, visceral adhesions, and enterocutaneous fistula formation (3).
Infection rates of close to 10% have been reported after ventral or incisional hernia mesh repair (4–6). Treating surgical site infections in the presence of a synthetic implant is a challenge, many times ending with explantation of the mesh. Meshes of different materials seem salvageable to a variable extent, for example, expanded polytetrafluoroethylene (ePTFE) implants are reported more likely to be explanted than those of polypropylene (7). If an infected mesh has to be removed, the patient faces the drawbacks of hernia recurrence and morbidity related to additional surgical procedures (8–12). Thus, the problems associated with mesh infection are considerable, and some surgeons have even proposed recommendations against the use of a synthetic mesh in situations where patient comorbidities, previous surgical site infection, or present surgical circumstances indicate an increased risk for infection (13).
Negative pressure has been increasingly used to treat wounds since it was introduced by Argenta and Morykwas (14). Negative pressure wound therapy (NPWT) is considered to stimulate healing by removal of excessive interstitial fluid, increase in tissue blood perfusion and oxygenation, stimulation of angiogenesis, accelerated formation of granulation tissue (14, 15), and reduction in bacterial load, although the decrease in bacterial colonization is debated (16).
NPWT has also been used for treatment of mesh infections, and the results from smaller patient series have been published (8, 9, 12, 17–22). In 2013, a larger study by Berrevoet et al. (4) reported excellent results with only 4.8% of the infected meshes being partly excised or totally explanted, and the rest successfully salvaged.
The aim of this retrospective study was to evaluate wound healing and mesh preservation after NPWT was used for the treatment of postoperative mesh infections. A group of NPWT patients were compared with age-matched mesh-operated controls without wound complications, with regard to patient and operation characteristics, to identify factors associated with later use of NPWT. Furthermore, patient-perceived long-term outcome is reported.
Material and Methods
Patients and Controls
Patients, who had undergone abdominal wall reconstruction using a surgical mesh at Skåne University Hospital in Malmö between January 2005 and June 2012, were identified from the hospital computerized operation registry. Patients with inguinal or femoral hernia mesh repair were not included. Postoperative complications and use of NPWT were searched in the hospital in-patient and out-patient registries 2005–2012. This was done for identification of the study group (NPWT-treated mesh infections), for identification of the cohort from which controls should be recruited (uneventful postoperative recovery) and for identification of patients with a wound complication without NPWT treatment (for description of the total incidence of wound complications). The NPWT patients and the patients with an uneventful postoperative recovery were sorted according to age, and subgroups with age-spans of 5 years were formed. For each NPWT patient, one control was randomly selected from the corresponding age group by drawing lots.
Hernia Repair Techniques
Peroperative 2 g of cloxacillin was routinely administered intravenously (Ekvacillin®; Meda AB, Solna, Sweden). The dominating hernia repair technique during the study period was a standardized open retromuscular repair in which the hernia sac was dissected subcutaneously and the anterior rectus abdominis fascia freed on both sides on a distance of 2–3 cm from the hernia opening. The retrorectus space was entered through an incision of the anterior rectus fascia on both sides and dissected to the lateral border of the rectus sheath to enable 5 cm of mesh overlap from the hernia opening. The total length of the former incision plus 5 cm cranially and caudally was covered with mesh. Different meshes were used with a predominance of heavy-weight, microporous mesh early and low- or mid-weight, macroporous mesh during the last third of the study period. The fascia was closed anteriorly of the mesh, or sutured to the mesh when fascia-to-fascia closure was not possible, whereafter redundant skin was resected and the skin closed intracutaneously. With this technique, and especially in patients with larger hernias, it was often not possible to cover the entire mesh with fascia, leaving part of the mesh exposed to the subcutaneous fat. The laparoscopic intraperitoneal onlay mesh (IPOM) repair during the study period was performed using three or four ports. Sharp adhesiolysis of the anterior abdominal wall and retraction of the hernia contents was followed by placement of a macroporous composite mesh, with an overlap of 5 cm. The mesh was fixated using non-resorbable tacks with a double-crown technique. The hernia opening was not closed, and transfixating sutures were not used.
Definitions
A mesh infection was defined as local signs of a wound infection with positive culture, purulent discharge, or abscess formation at the level of the mesh, that is, either underneath a closed anterior rectus fascia or subcutaneously in patients without anterior fascia covering the mesh.
Wound closure was defined as a wound closed by suturing, skin grafting, or completely healed by secondary intention. The classification of wounds according to the likelihood and degree of contamination applied by the Centers for Disease Control and Prevention (CDC) in the CDC guideline for prevention of surgical wound infections (23) was applied. The wounds were accordingly divided into clean, clean-contaminated, contaminated, and dirty or infected. The Ventral Hernia Working Group (VHWG) grading system for assessment of risk for surgical site occurrences, dividing the hernia/patients in grade 1 (low risk), grade 2 (co-morbid), grade 3 (potentially contaminated), and grade 4 (infected), was also applied (13). Follow-up was defined as time from mesh operation to 31 January 2013 or to date of death for deceased patients.
Chart Review
Patient charts including operative records, laboratory reports, daily notes made by hospital staff, discharge summaries, and clinical out-patient notes were reviewed in February 2013. A protocol was outlined to describe preoperative characteristics including sex, age, body mass index (BMI), comorbidities (diabetes mellitus, asthma, chronic obstructive pulmonary disease, malignancy, and immunosuppressive therapy), presence of a stoma, and index operation (defined as the operation preceding the operation with mesh implantation). The studied characteristics for the mesh implantation operation were as follows: emergency or elective operation, wound classification according to the CDC, hernia grading according to the VHWG system, abdominal wall repair technique, size and type of the implanted mesh, duration of surgery, and whether the mesh was partially or totally covered with anterior rectus fascia. The registered postoperative characteristics were as follows: postoperative complications and treatment, NPWT duration, number of dressing changes, treatment after finishing NPWT (other wound dressing until secondary healing, delayed suturing, or skin grafting), explantation or partial excision of the mesh, bacterial etiology, type of antibiotics, time to wound healing, later complications, and total follow-up time.
Npwt and Follow-Up of Wound Healing
NPWT for a postoperative infection engaging the mesh was commenced once the mesh infection was diagnosed and the wound opened. Surgical revision of the wound was performed in the operation theater during general anesthesia or bedside after systemic administration of morphine, whenever needed. All patients were initially treated with the V.A.C.® system by KCI (San Antonio, TX, USA). With few exceptions, black sponge was used and the negative pressure was set to −125 to 150 mm Hg during the entire NPWT treatment. In the beginning of the NPWT, dressing changes were performed every second or third day. As the healing advanced, the dressing was changed twice a week. After closing the vacuum pump, ropivacaine (Ropivakain®; Kabi Fresenius, Uppsala, Sweden) was locally administered in the sponge 30 min prior to the dressing changes. The principal for finishing NPWT was mesh covered with granulation tissue. After finishing NPWT, the wounds were treated in one of three different ways: with other wound dressings until secondary healed, secondary sutured, or skin grafted. Infected wounds underwent surgical debridement when needed and if the wound was closed by secondary suturing, skin revision was performed at the same occasion. In cases where secondary suturing or skin grafting was performed, systemic antibiotic administration was based on the result of bacterial cultures.
All patients were seen by specialized nurses at the out-patient clinic 7–10 days after discharge and as long as needed, that is, until wound healing in case of wound complications. Diagnoses and treatment codes were registered by the nurses, and wound appearance was recorded in special wound charts.
Abdominal Wall Complaints Questionnaire
A slight modification of the validated Ventral Hernia Pain Questionnaire (VHPQ) (24) was approved by the inventors of the VHPQ. The 21 questions intend to reflect how the patient experiences the wound or scar with regard to pain, cosmetic appearance, social limitations, and satisfaction with the final result. Patients experiencing pain at the time of answering the questionnaire were asked additional questions to characterize the pain in terms of frequency, duration, use of pain medication, and pain-related impairment of physical function. The questionnaire was sent to all patients and followed by one reminder to patients who failed to respond.
Statistics
Data were analyzed using IBM SPSS Statistics version 22. McNemar’s test for related samples was used for analysis of categorical data. p-Values were not calculated for variables with less than six recorded events. Continuous data are presented as median (range), and comparison between matched pairs was analyzed by Wilcoxon signed-rank test. Continuous data for subgroups of NPWT patients were analyzed by Mann–Whitney U-test. In all tests, p <0.05 (two-sided) was regarded as significant.
Results
A total of 722 mesh operations were performed in 699 patients, that is, 23 patients (3.3%) had a second operation due to a recurrent hernia during the study period. The number of open mesh repairs was 588 (81.4%) and laparoscopic IPOM operations 134 (18.6%). Any wound complication was registered after 233 of 722 (30.3%) operations, while NPWT was used for treatment of postoperative mesh infections in 48 of these (6.5% of all operations). In all, 489 (67.7%) operations had an uncomplicated postoperative course. See Fig. 1 for a study flow diagram. The median follow-up time in the NPWT group was 28 months (range 1.3–84 months) and in the control group 31.6 months (range 2.5–96 months).
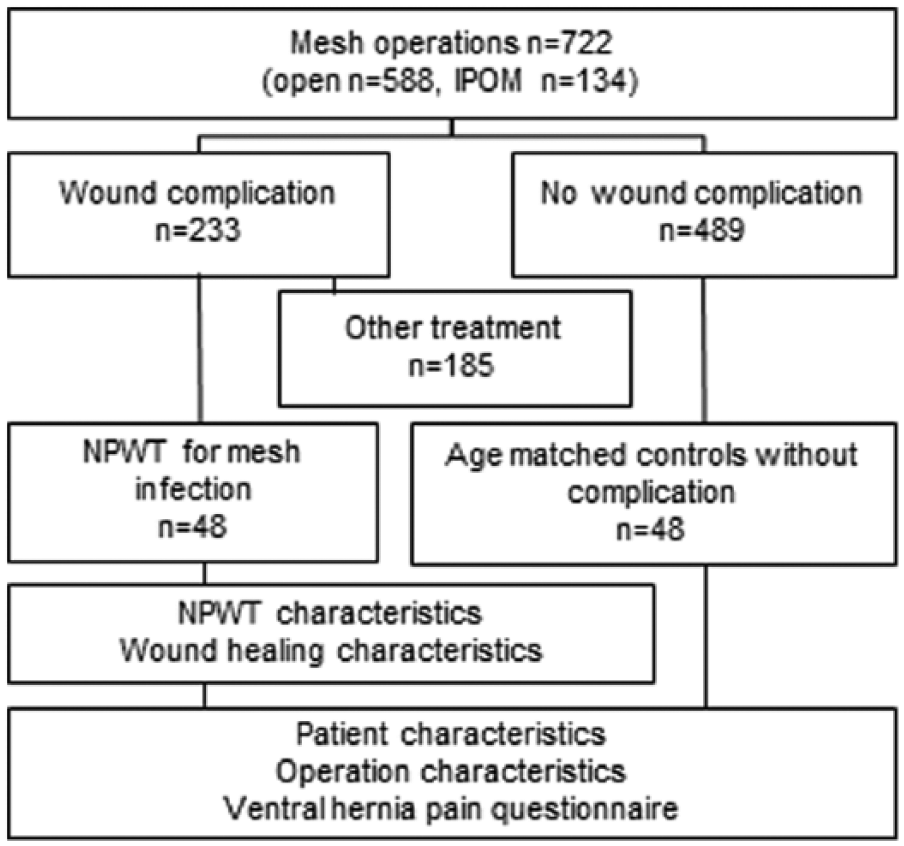
Study flow diagram.
Preoperative Characteristics
Preoperative patient characteristics for the NPWT and control group are listed in Table 1. No differences were detected.
Preoperative characteristics.
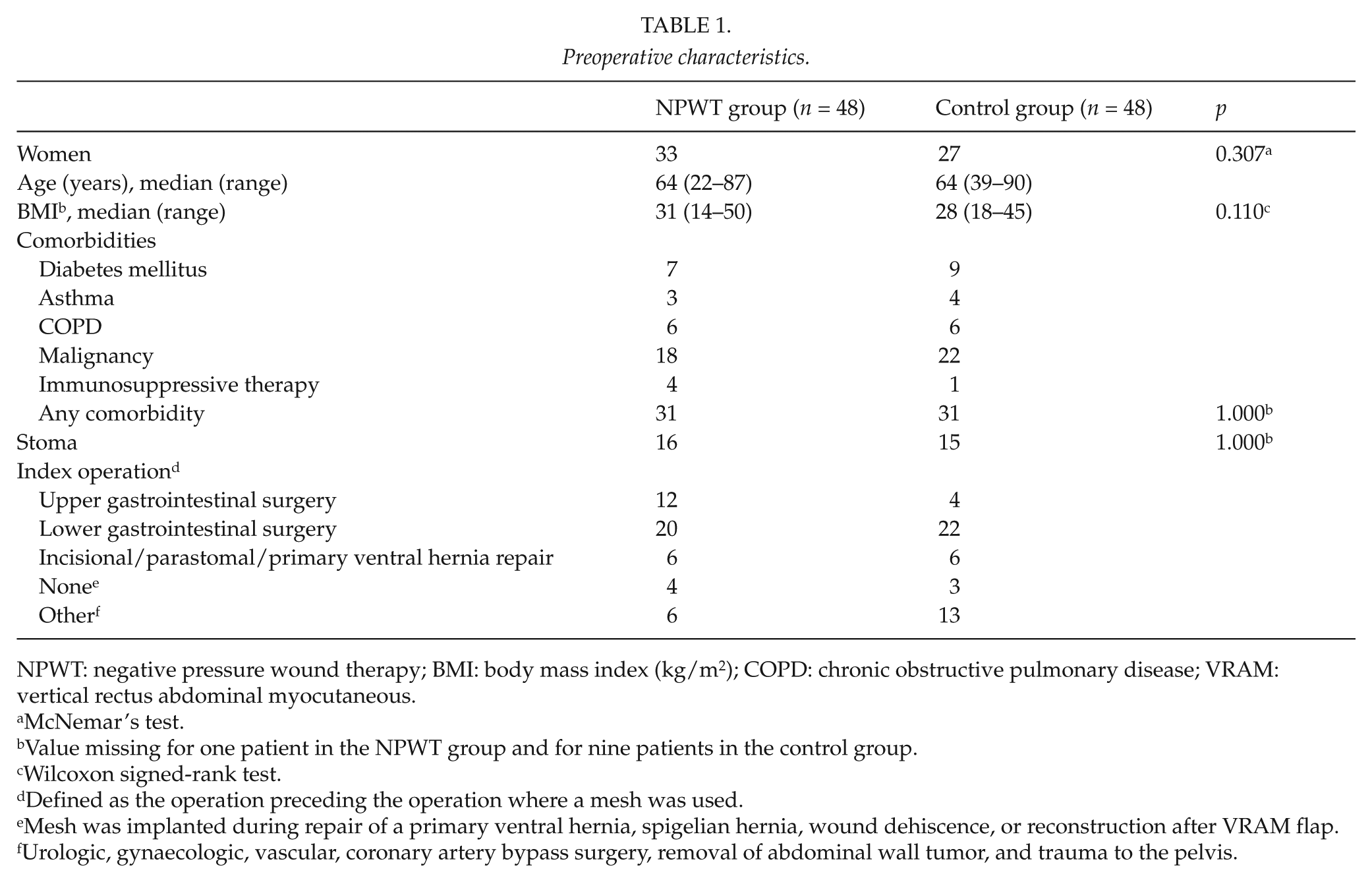
NPWT: negative pressure wound therapy; BMI: body mass index (kg/m2); COPD: chronic obstructive pulmonary disease; VRAM: vertical rectus abdominal myocutaneous.
McNemar’s test.
Value missing for one patient in the NPWT group and for nine patients in the control group.
Wilcoxon signed-rank test.
Defined as the operation preceding the operation where a mesh was used.
Mesh was implanted during repair of a primary ventral hernia, spigelian hernia, wound dehiscence, or reconstruction after VRAM flap.
Urologic, gynaecologic, vascular, coronary artery bypass surgery, removal of abdominal wall tumor, and trauma to the pelvis.
Characteristics for the Mesh Implantation Operation
The mesh operation characteristics for the two groups are shown in Table 2. The following peroperative characteristics were significantly more frequently found in the operations followed by NPWT-treated mesh infections: emergency operation, wound class IV/dirty–infected according to the CDC classification, grade 4/infected according to the VHWG grading system, surgical technique other than laparoscopic IPOM repair, implantation of two meshes, mesh size larger than median, duration of surgery longer than median, and mesh not completely covered with fascia. There was no difference in distribution across VHWG grade 1 versus grades 2–4 when NPWT patients and controls were compared (p = 0.664).
Mesh operation characteristics.
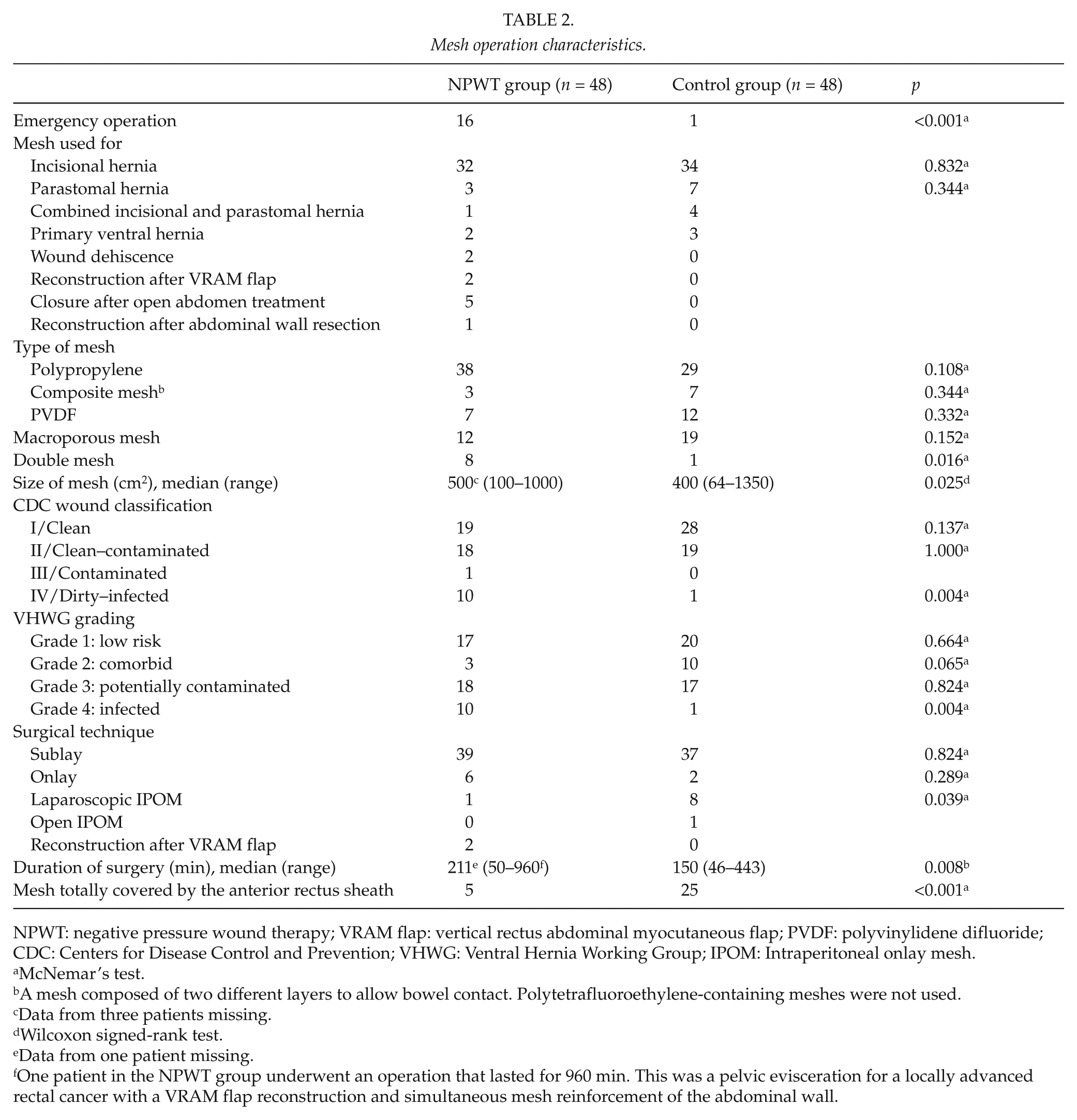
NPWT: negative pressure wound therapy; VRAM flap: vertical rectus abdominal myocutaneous flap; PVDF: polyvinylidene difluoride; CDC: Centers for Disease Control and Prevention; VHWG: Ventral Hernia Working Group; IPOM: Intraperitoneal onlay mesh.
McNemar’s test.
A mesh composed of two different layers to allow bowel contact. Polytetrafluoroethylene-containing meshes were not used.
Data from three patients missing.
Wilcoxon signed-rank test.
Data from one patient missing.
One patient in the NPWT group underwent an operation that lasted for 960 min. This was a pelvic evisceration for a locally advanced rectal cancer with a VRAM flap reconstruction and simultaneous mesh reinforcement of the abdominal wall.
Mesh Infection Treatment Characteristics and Wound Healing
NPWT-treated patients showed the following results of the initial bacterial culture: 12 had Staphylococcus aureus or other skin-derived bacteria; 16 had Escherichia coli or other bowel-derived bacteria; 14 had a mixture of skin and bowel-derived bacteria; 1 had Candida albicans; and 5 had negative cultures, reasonably due to antibiotic treatment instituted before a culture was taken. All but one of the NPWT-treated patients received antibiotic treatment according to the sensitivity analysis. In 17 of the patients, this initially resulted in the use of two antibiotics simultaneously. In total, 13 different antibiotics were used in the 48 patients. The duration of the antibiotic treatment was retrospectively not possible to extract with certainty from the charts.
The characteristics of the NPWT treatment and wound healing are presented in Table 3. One of six patients, not completely healed at follow-up, died from pneumonia and sepsis 39 days after the mesh operation. The present status for the other five patients is as follows: wound relapse after skin grafting in a patient not fit for major surgery, low-output small bowel fistula in a patient not fit for major surgery, recurrent infected subcutaneous sinus planned for extirpation, small wound remaining after secondary suture, and small wound remaining after recent closure of a gastrocutaneous fistula and minor mesh excision.
Characteristics of negative pressure wound therapy and wound healing.
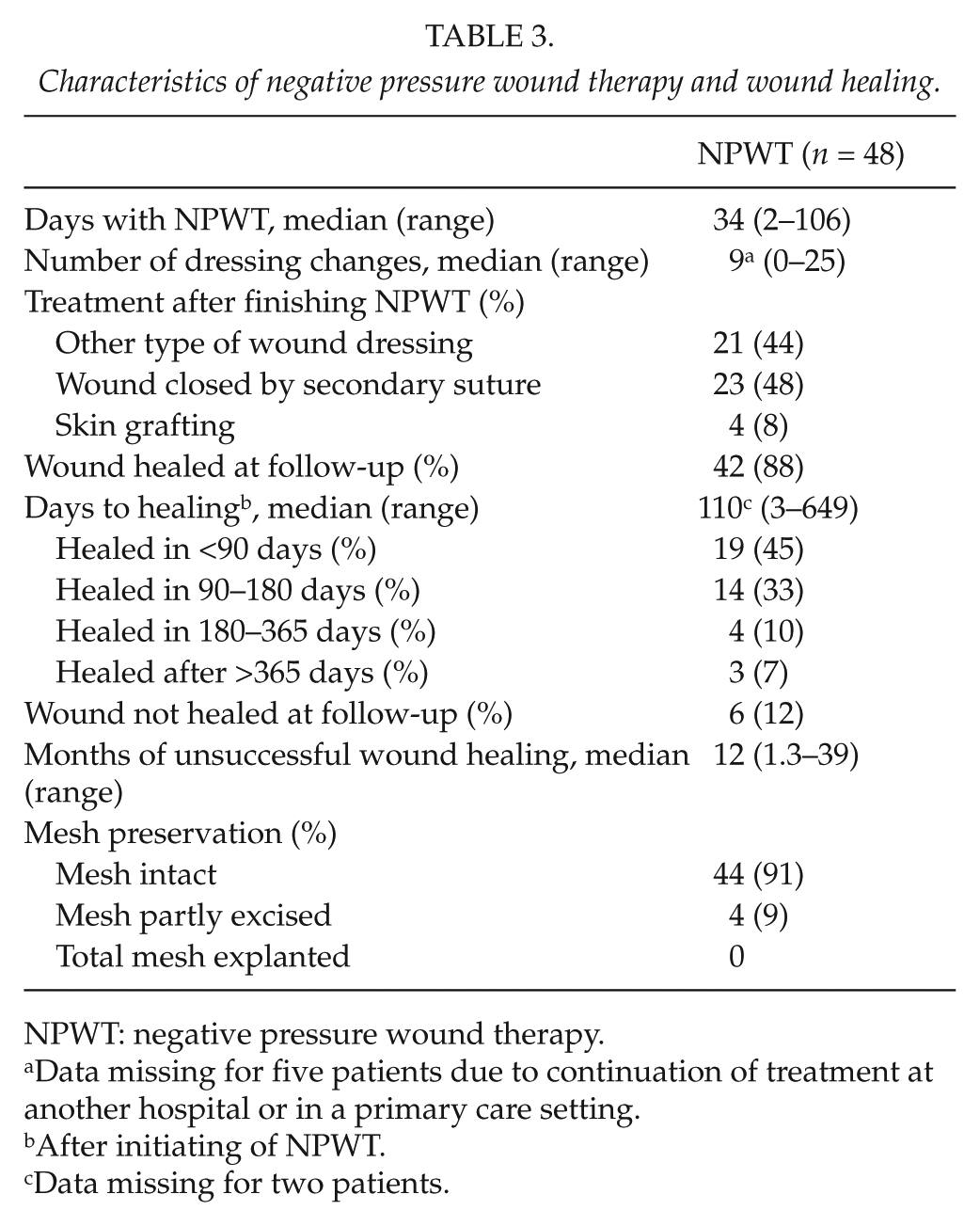
NPWT: negative pressure wound therapy.
Data missing for five patients due to continuation of treatment at another hospital or in a primary care setting.
After initiating of NPWT.
Data missing for two patients.
After finishing NPWT, 21 patients were treated with other wound dressings, that is, without active surgical measures (secondary suture or skin grafting) for closure of the wound. The median healing time after finishing NPWT in the 18 patients healed at follow-up was 107 (13–451) days. Of the 23 secondary sutured patients, 21 healed in a median of 19 (0–642) days after finishing NPWT, which was significantly shorter than for patients not receiving active surgical intervention (p < 0.002). Skin grafting was performed in four cases with successful results in three. The forth patient was not fit for major surgery and was not healed after 35.5 months due to mesh remnants in the wound.
Four patients had their meshes partly excised. All other meshes were salvaged.
Other Complications and Mortality
The only fistula developing during NPWT was a rectocutaneous fistula after a Hartmann’s procedure. Incisional hernia recurrences were found in one NPWT patient and two control patients, respectively. Two additional parastomal hernia recurrences were seen in the control group.
There were two in-hospital deaths and five later deaths in the NPWT group, and the corresponding figures for controls were zero and eight, respectively. The in-hospital deaths in the NPWT group were caused by respiratory insufficiency due to impaired chronic obstructive pulmonary disease (COPD) and pleuritis in one and pneumonia and sepsis in the other patient. All deaths were unrelated to NPWT.
Abdominal Wall Complaints Questionnaire
Forty-one patients in the NPWT group and 40 in the control group were alive and received the abdominal complaints questionnaire. A total of 35 of 41 patients (85%) in the NPWT group and 30 of 40 (75%) patients in the control group responded. A set of selected questions from the questionnaire is presented in Table 4.
Selected questions from the abdominal wall complaints questionnaire (affirmative answers are presented).

NPWT: negative pressure wound therapy.
McNemar’s test.
For six of the eight patients in the NPWT group, who experienced pain that “could not be easily ignored,” the pain adversely affected their physical function and all six patients were on pain medication. No impaired physical function or need for pain medication was reported by the three controls with pain that “could not be easily ignored.” Four of the six NPWT patients with pain had problems with standing up, sitting and standing for more than 30 min, as well as practicing sports. Three patients had problems with climbing stairs and one patient experienced difficulty when driving a car. The wounds of three of these six patients were not healed at the time of answering the questionnaire.
Discussion
This retrospective age-matched case–control study was conducted to evaluate NPWT for treatment of prosthetic mesh infection and patient-reported outcome.
There were no differences between the NPWT and the control group in the preoperative characteristics. The characteristics of a mesh implantation operation associated with mesh infection treated with NPWT in this study were as follows: emergency operation, wound class IV/dirty–infected according to the CDC classification (23), grade 4/infected according to the VHWG grading system (13), surgical technique other than laparoscopic IPOM repair, implantation of two meshes, mesh size larger than median, duration of surgery longer than median, and mesh not completely covered with fascia. This is not to be interpreted as these variables being independent risk factors for mesh infection but the ones that are possible to influence could be of interest to evaluate in a future study. According to previous studies, a prosthetic infection is more likely to occur in some patients than others and proposed risk factors are tobacco use, steroid use, diabetes mellitus, chronic obstructive pulmonary disease, malnutrition, obesity, deep infection after an earlier operation, and the duration of surgery (6, 21, 25–28). A decreased blood perfusion and an immunosuppressive element are two proposed mechanisms linking an increased infection rate with these comorbidities (10). Of these earlier proposed risk factors, only long duration of surgery was more frequently found in patients with mesh infection and NPWT than in controls in our study. One of the weaknesses of this study is the absence of information on tobacco use. This information was either not registered in the charts or vague and incomplete, making it impossible to include tobacco use in the statistical analysis. Furthermore, we were not able to find confident information on malnutrition and deep infection after previous surgery. This might partly explain the discrepancy in our findings compared to previous studies, besides the effect of a relatively small number of patients included in this study.
In 2010, Breuing et al. (13) in the VHWG published recommendations for grading and technique of repair for incisional ventral hernias. They dissuade from using a synthetic mesh in situations where patient comorbidities, surgical site infection after previous surgery, or present surgical circumstances indicate an increased risk for infection, that is, patients with VHWG grades 2–4. In this study, 65% of the NPWT patients with mesh infection and 58% of controls had VHWG grades 2–4. There was no difference between NPWT patients and controls in the prevalence of grade 1 compared to grades 2–4. Only grade 4 patients were more frequently found among patients with mesh infection and NPWT than among controls. These results challenge the VHWG recommendations for not using synthetic meshes in these situations.
There are some previous reports on the use of NPWT for treatment of infected mesh to avoid mesh removal (4, 8, 9, 12, 17–22). In this context, it is important to use a unanimous definition of mesh infection to be able to compare outcome. In this study, we have focused on infections truly engaging the mesh and excluded superficial wound infections in cases where the mesh was covered by anterior fascia.
Baharestani and Gabriel (17) reviewed the medical records of 21 patients with an exposed and infected mesh. Successful wound closure was achieved in 18 patients, but 13 of the patients had their mesh either totally or partially removed. The authors concluded that it is important to initiate NPWT at an early stage and to combine NPWT with numerous debridements, to decrease edema and bacterial burden. Greenberg (12) reported on treatment of infected composite meshes in 11 patients. With NPWT, antibiotics, wound debridement, and partial mesh excision, seven of the infections could be healed while four patients needed mesh removal. The author highlights the importance of individualizing the care these patients receive and concludes that infected mesh can be salvaged as long as the infection is limited and the patient shows no signs of sepsis. Stremitzer et al. (21) used NPWT for wounds larger than 2 cm or if the mesh was exposed. By combining NPWT with other conservative wound therapies, they managed to preserve the mesh in 17 of 31 patients (55%). The results from these studies are encouraging in terms of the overall management of mesh infections but in many patients still at the cost of mesh removal and hernia recurrences in the long run.
Berrevoet et al. (4) published a prospective observational study in 2013, in which 63 patients were treated with NPWT due to an infectious complication following primary ventral or incisional hernia repair, with improved results compared to earlier reports. The infection rate was 8.7% (63/724 patients) compared to 7.5% in this study. In the Berrevoet et al. study, all retromuscular meshes were salvaged while three multifilament polyester meshes were either completely or partially removed after open intraperitoneal mesh placement. Thus, in 95% of their patients, the entire mesh was successfully salvaged compared to 92% in our patients. In both studies, one enterocutaneous fistula developed during NPWT, indicating that NPWT is a safe treatment regimen for mesh infections after abdominal surgery. In the above-mentioned aspects, the Berrevoet et al. study and this study show similar results.
Regarding time to healing, there is, however, a considerable difference between the studies. We found the time to wound healing to be a median of 110 days compared to a mean of 44 days in the Berrevoet et al. study. There are several possible explanations for this difference. Large pore meshes were used in the Berrevoet et al. study, probably advantageous from an infection eradication point of view compared to small pore meshes which were used in a majority of our patients. Only 10% of our patients with mesh infection had their meshes completely covered by the anterior fascia at the mesh implantation operation. Differences in fascia coverage between studies may have implications on healing times once an infection occurs. It is reasonable to assume that it takes longer for a larger area to become covered with granulation tissue and finally healed.
In the Berrevoet et al. study, secondary suturing was part of the protocol and was performed as soon as the wound became granulated, resulting in secondary suture in 78% of their patients. In the present retrospective study, decision on secondary suturing was the responsibility of the surgeon in charge, resulting in a 50% secondary suture rate. Many of the wounds that were not secondary sutured healed well initially, but the time to complete healing was found to be surprisingly long despite relatively small remaining wounds. The majority of the patients with unhealed wounds at follow-up had not received active surgical measures mainly due to poor general condition or impossibility of skin advancement for coverage of the mesh. If secondary suture was used, wound closure without further complications was successful in 56%, while 7% of patients with a complicated course after secondary suturing were not healed at follow-up. In the group without surgical intervention, 18% of the patients were not healed. The time to wound healing for secondary sutured patients was significantly shorter than the group treated with other wound dressing regimens after stopping NPWT. Although other circumstances may contribute to this difference, these findings indicate a potential benefit for earlier surgical intervention for closure of the wounds after NPWT.
In this study, we used an abdominal wall complaints questionnaire to evaluate the patient-reported long-term results. The NPWT patients reported pain, cosmetic dissatisfaction, social limitation, and abdominal wall stiffness twice as often as controls. Satisfaction with the final result was also higher in the control group. Even though the differences did not become statistically significant, it seems as if patients with mesh infection treated with NPWT have a less favorable result.
This study suffers from all the limitations and weaknesses accompanying a retrospective chart review, with missing information due to the absence of a prospective study protocol. A benefit for the study is that patients with complications were seen by specialized nurses at the out-patient clinic until they were healed. At these visits, treatment measures and wound appearance were recorded in special wound charts ensuring good quality data concerning healing times. The use of a questionnaire for patient-reported outcome also supplies valuable, earlier not available, information.
In conclusion, no mesh had to be explanted and wound healing was eventually achieved in the vast majority of patients when NPWT was used for treatment of an infected mesh. Time to healing was long, and numerous procedures were needed in the most complicated cases. The results of this study indicate potential benefits for IPOM compared to open repair, anterior rectus sheath coverage of the mesh for possible prevention of mesh infection, and surgical wound closure interventions for earlier wound healing after complete NPWT. NPWT patients reported abdominal wall discomfort and less satisfaction with the final results more frequently than controls, even though the differences did not reach statistical significance. Based on the study findings, the following changes in treatment strategy have been implemented at our department: complete shift to use of large-pore meshes, surgical technique modification which enables us to cover the mesh subcutaneously with fascia or hernia sac, and compliance with a protocol for treatment of wound and mesh infections including secondary suturing. Further prospective studies need to be conducted to evaluate these changes in treatment strategy.
Footnotes
Acknowledgements
This study was presented at the First World Conference on Abdominal Wall Hernia Surgery in Milan, Italy, 25–29 April 2015.
Declaration of Conflicting Interests
S.N. and P.R. declare no conflict of interest. U.P. declares conflict of interest not directly related to the submitted work (honorarium for lectures arranged by KCI).
Ethical Approval
This study was approved by the Regional Ethics Committee in Lund (2012/726).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Patients and controls were informed in writing and approved participation by answering the questionnaire.
Research Data
Underlying research data related to this article can be accessed from the corresponding author.
