Abstract
Background and Aims:
The impact of body mass index on complications and survival in patients undergoing esophagectomy has been extensively studied with conflicting results. In this study, we assess the impact of body mass index on complications and survival following surgery for esophageal and gastro-esophageal-junction cancer in a Danish population.
Material and Methods:
We identified 285 consecutive patients, who underwent curative-intended treatment for esophageal and gastro-esophageal-junction cancer in the period 2003–2010. We manually reviewed the electronic medical records of all patients included in the study. Body mass index was calculated as weight in kilograms divided by height in meters squared. We grouped patients according to their body mass index, using the World Health Organization definition, as underweight (body mass index < 18.5 kg/m2), normal weight (body mass index: 18.5–24.9 kg/m2), overweight (body mass index: 25–29.9 kg/m2), and obese (body mass index ⩽ 30 kg/m2).
Results:
Median age at surgery was 65 years (range: 27–84 years), of which 207 (72.6%) were males. Patients with the lowest body mass index and the obese patients seemed to have a higher frequency of minor complications. Anastomotic leakage occurred in less than 10% of the patients and was equally distributed across the groups as was the other major complications. There were no differences in the 1-, 2-, or 5-year survival rates between the four body mass index groups after adjustment for possible confounders. Five-year survival rates for the four body mass index groups were 31.8%, 28.7%, 27.9%, and 26.1%, respectively.
Conclusion:
Body mass index over 30 or under 18.5 does not seem to affect survival rates or the presence of serious postoperative complications following esophagectomy in patients with esophageal and gastro-esophageal-junction cancers not receiving neoadjuvant oncological treatment.
Introduction
The incidence of esophageal and gastro-esophageal-junction (GEJ) cancer has increased during the past decades and it is still associated with poor survival rates (1). In Denmark, about 500 new cases are diagnosed each year. Many cases are diagnosed at an advanced stage, limiting the possibility for curative intended treatment (2). Esophageal resection, a complex and challenging procedure, remains the primary curative treatment for non-disseminated carcinomas of the esophagus and GEJ. Today, neoadjuvant oncological treatment is standard in most centers.
Despite the fact that a large proportion of esophageal cancer patients suffer from malnutrition and weight loss, there are still a significant number of patients, who are overweight at the time of surgery. A high body mass index (BMI) is associated with increased perioperative risks, including cardiorespiratory morbidity and impaired wound healing in major intra-abdominal cancer surgery (3, 4). Moreover, a high BMI is reported to be a risk factor for the development of esophageal adenocarcinoma (5). The impact of BMI on complications and survival in patients undergoing esophagectomy has been extensively studied. However, findings remain contradictory and between-study comparisons are hampered due to very heterogeneous study populations. In addition, this potential association has never been examined in a Scandinavian population.
The aim of this study was to evaluate the impact of BMI on complications and survival after esophagectomy in a population of esophageal and GEJ cancer patients without neoadjuvant oncological treatment.
Material and Methods
Setting and Study Population
Using a local procedure registry, we identified all patients, who underwent curative-intended treatment for esophageal and GEJ cancer in the period 2003–2010 at Department of Surgery, Aarhus University Hospital, Denmark. A total of 285 patients were included for final analyses (Fig. 1). Patients were followed up from the day of surgery to the day of death or 30 June 2016, whichever occurred first.
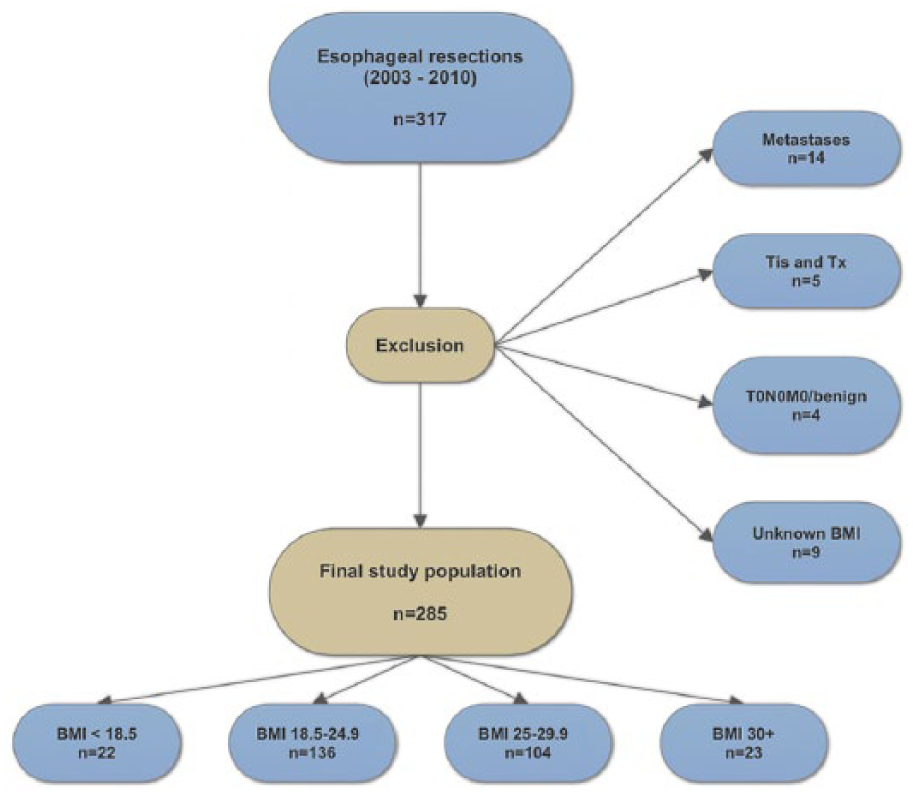
Flowchart of the study population.
Patient Selection
All patients had a standard preoperative work-up including endoscopy (with biopsy), computed tomography (CT) imaging, and a positron emission tomography (PET) scan. After the work-up, patients were evaluated at our multidisciplinary conference. If patients were in performance status 0–1 and the tumor was considered resectable, the patient was referred to surgery with curative intent. Otherwise, patients were referred to palliative oncological treatment.
Information on Patients and Procedures
We manually reviewed the electronic medical records of all patients included in this study. We collected information on age, sex, height, weight, American Society of Anesthesiologists (ASA) score, smoking history (yes/no/former), tumor stage, tumor histology, tumor localization, the use of preoperative stenting, complications, duration of hospital stay, preoperative albumin level, and mortality. BMI was calculated as weight in kilograms divided by height in meters squared. Both weight and height were recorded at the first admission immediately prior to surgery. We grouped patients according to their BMI, using the World Health Organization (WHO) definition, as underweight (BMI < 18.5 kg/m2), normal weight (BMI: 18.5–24.9 kg/m2), overweight (BMI: 25–29.9 kg/m2), and obese (BMI ⩽ 30 kg/m2). Complications were defined according to the Clavien–Dindo classification (6). Minor complications were defined as Clavien–Dindo I–IIIa, and major complications were defined as Clavien–Dindo IIIb–V. Grade IIIa is defined as postoperative intervention without general anesthesia, whereas grade IIIb is defined as postoperative intervention with general anesthesia. We defined anastomotic leakage as any radiologically and/or endoscopically confirmed leakage from the anastomosis regardless of the need for reoperation. Patients were operated ad modum Ivor-Lewis (7). None of the patients received any neoadjuvant chemo-radiotherapy
Information on Death
In our electronic medical records, information on date of death is automatically linked to the Danish Civil Registration System through a unique identification number assigned to all Danes at birth or immigration. The Danish Civil Registration System contains information on vital status for all Danish residents with a 100% coverage (8).
Statistical Analyses
All consecutive variables are listed as medians with range as measures of variation. Categorical variables are listed as numbers with percentages. Multivariate logistic regression models were used to assess the influence of potential confounding variables associated with postoperative complications and survival rates. For each BMI group, survival rates were calculated and presented as Kaplan–Meier plots and compared using the Mantel–Cox test. Statistical analyses were performed using STATA version 13.1 (Stata Corp LP, College Station, TX, USA).
Results
We identified 285 patients eligible for inclusion (Fig. 1). Median age at surgery was 65 years (range: 27–84 years), equally distributed over the four BMI groups, of which 207 (72.6%) were males (Table 1). The range of BMI measurements was 15.6–40.6. Most patients were classified as ASA II. The proportion of males and non-smokers increased with increasing BMI. The majority of tumors were stage 2 or 3. The group with the lowest BMI (<18.5) mostly had squamous cell carcinomas (77.3%) located at the esophagus, whereas the vast majority in the three other BMI groups had adenocarcinomas primarily located at the GEJ. There was a tendency toward a higher use of preoperative stents in patients with a low BMI compared to the patients with higher BMIs. All patients underwent an R0 resection.
Characteristics of 285 patients undergoing esophagectomy for esophageal or gastro-esophageal-junction (GEJ) cancer according to their body mass index (BMI).
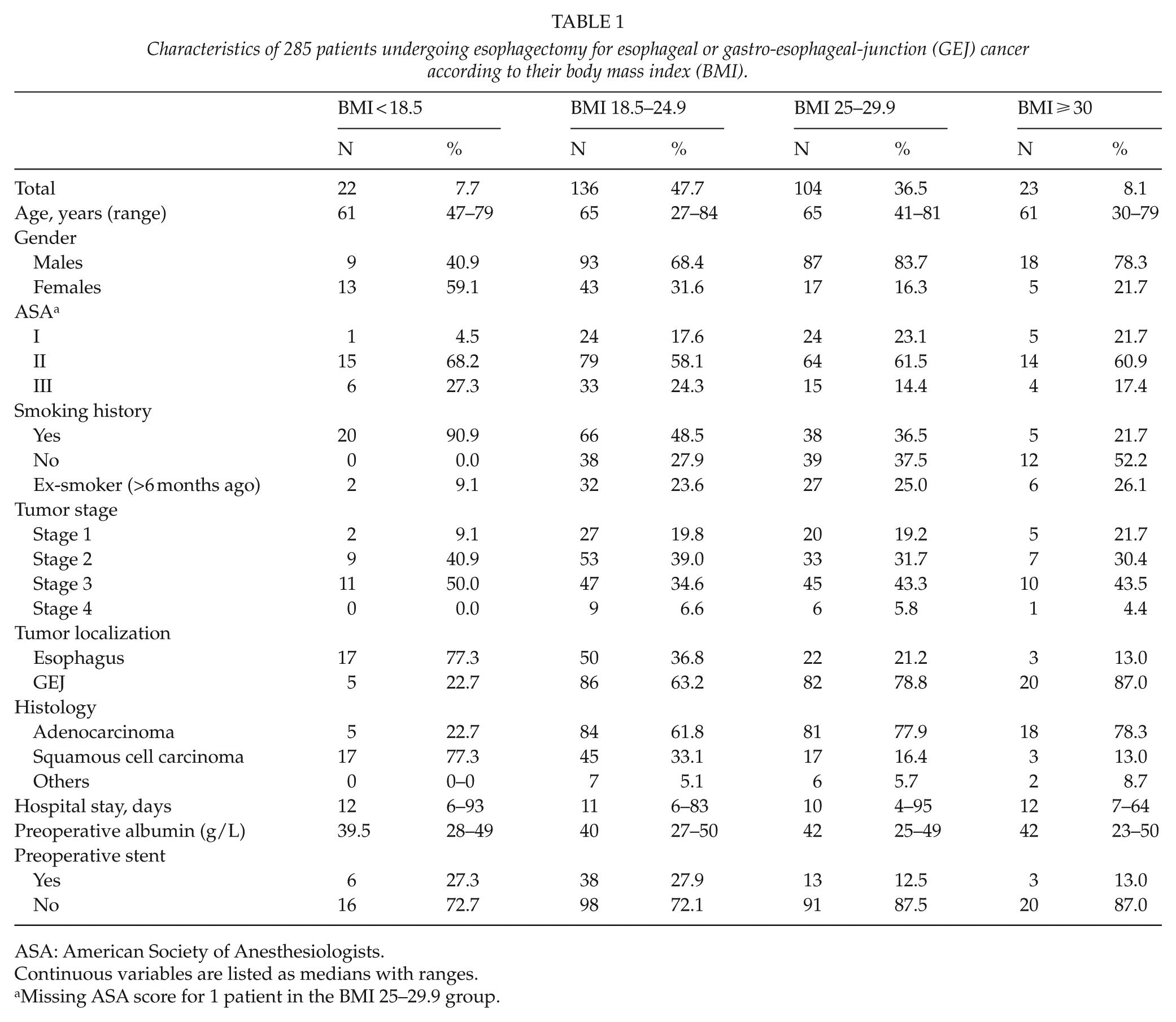
ASA: American Society of Anesthesiologists.
Continuous variables are listed as medians with ranges.
Missing ASA score for 1 patient in the BMI 25–29.9 group.
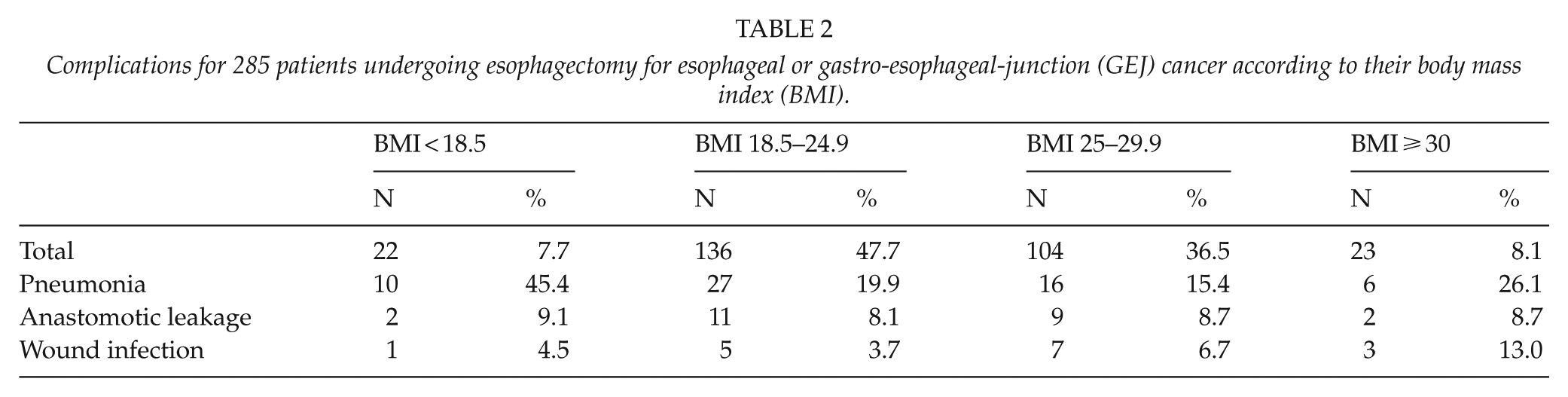
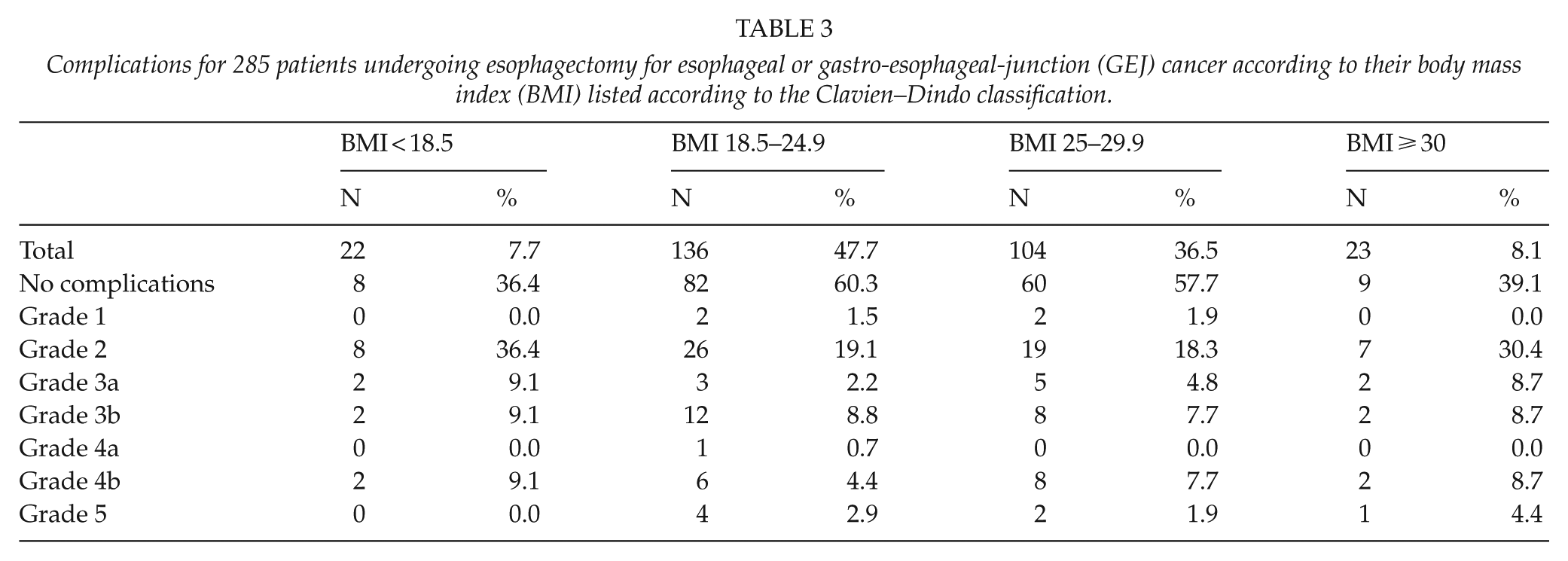
We saw a more pronounced incidence of postoperative pneumonias among patients with the lowest BMI (45.4%) compared to the other groups, which experienced pneumonias in 19.9%, 15.4%, and 26.1% of the cases (Table 2). Anastomotic leakage was equally distributed across the groups occurring in less than 10% of the patients. After adjustment for possible confounders (age, sex, smoking, ASA score, tumor localization, and histology), there were no difference in the risk of anastomotic leak between the four groups. The obese patients tended to have a higher incidence of wound infections (13.0%) compared with the rest of the population (4.5%, 3.7%, and 6.7%). The same pattern was apparent when listing complications according to the Clavien–Dindo classification (Table 3), as patients with the lowest BMI and the obese patients seemed to have a higher frequency of minor but not major complications. However, numbers were small.
Complications for 285 patients undergoing esophagectomy for esophageal or gastro-esophageal-junction (GEJ) cancer according to their body mass index (BMI).
Complications for 285 patients undergoing esophagectomy for esophageal or gastro-esophageal-junction (GEJ) cancer according to their body mass index (BMI) listed according to the Clavien–Dindo classification.
After adjustment for possible confounders (age, sex, smoking, ASA score, tumor localization, and histology), there were no differences in the 1-, 2-, and 5-year survival rates between the four BMI groups. Five-year survival rates for the four BMI groups were 31.8%, 28.7%, 27.9%, and 26.1% respectively (Fig. 2).
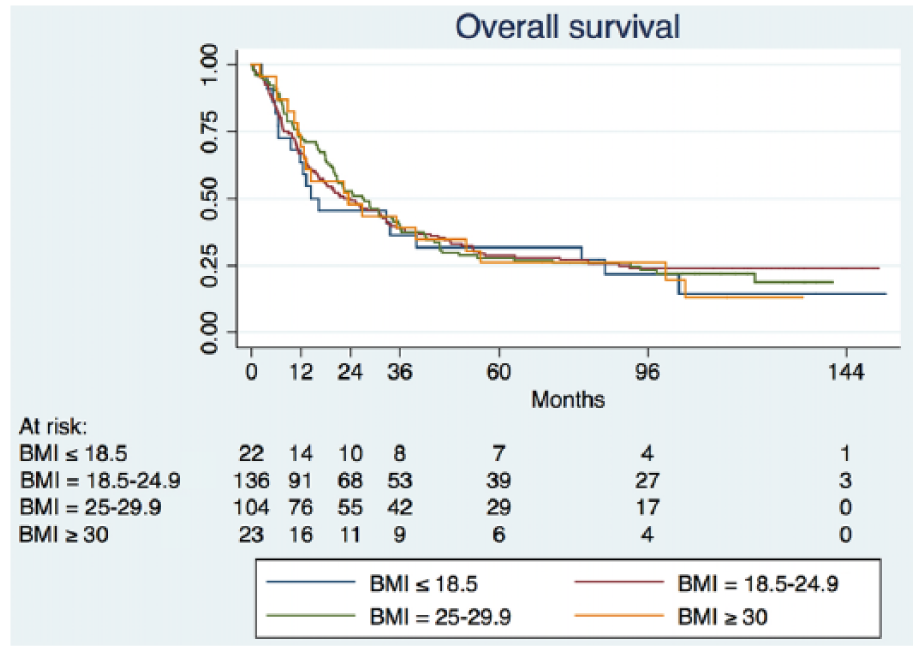
Survival curves according to body mass index.
Discussion
In this study, we examined the impact of BMI on outcome in 285 patients undergoing esophagectomy ad modum Ivor-Lewis for esophageal and GEJ cancers. While there seems to be a higher incidence of minor complications in patients with BMI <18.5 or ⩽30, we did not observe any differences in major postoperative complications or long-term survival between the four BMI groups examined in this study. In this study, none of the included subjects received any kind of neoadjuvant therapy.
This study is, to our knowledge, the first to investigate the impact of BMI on short- and long-term outcome in patients undergoing surgery for esophageal and GEJ cancer in a Scandinavian population. We demonstrate a tendency towards a higher occurrence of minor complications and pneumonias in patients with low BMI (<18.5) and obese patients. Our findings are at a first glance in accordance with a study by Melis et al. (9) of 490 patients undergoing surgery for esophageal cancer. Although their main goal was to investigate the impact of BMI on survival, they demonstrated a slightly higher rate of postoperative complications in patients with a BMI >30. The study populations are, however, not comparable as many of their patients received neoadjuvant chemotherapy, and in addition they did not report complications according to the Clavien–Dindo classification. Our findings are in disagreement with a study by Blom et al. (10), who could not demonstrate a relation between BMI and minor complications in 736 patients, who underwent esophageal surgery for cancer. Their study population was comparable to ours as no patients included in their analysis received neoadjuvant chemotherapy and they too reported complications according to the Clavien–Dindo classification. An explanation for this discrepancy could be that our estimates are associated with a limited precision due to low numbers in each group.
Anastomotic leakage is the most feared complication after esophageal cancer surgery and has been reported to occur in up to 24% of patients (11). In this study, we show a frequency of anastomotic leakage of 9% with no difference between groups according to BMI. Furthermore, we could not demonstrate a difference between the groups in regard to other major complications. Our findings are in disagreement with a study by Grotenhuis et al. (12) of 556 patients, who underwent esophagectomy. They demonstrated a higher frequency of anastomotic leakage and other major complications in obese patients than in non-obese patients. Again, as with minor complications, the discrepancy could be due to imprecision of our estimates. Furthermore, Grotenhuis et al. did not report complications according to the Clavien–Dindo classification and they did not define major complications, which limits comparison.
We found no difference in major complications or survival rates among the four group of patients stratified according to the BMI. Our observed 5-year survival rates for the four BMI groups were 31.8%, 28.7%, 27.9%, and 26.1% ranging from low to high BMI, respectively. These numbers are comparable with some previous studies also examining patients undergoing esophageal surgery without neoadjuvant chemotherapy (12–14), although other studies have demonstrated higher survival rates (10, 15–17). The comparison of our findings with these studies is somewhat limited, as two studies included almost exclusively esophageal squamous cell carcinomas (15, 17), one study provided only disease-free-survival rates (10), and one study included primarily lower stage tumors (16). In addition, most of the studies grouped patients with high BMI and obese into one single category (15–17), whereas two studies grouped low and normal BMI into one category (10, 16). Further studies on survival following esophagectomy for esophageal and GEJ cancer have been published, but they all included patients receiving neoadjuvant chemotherapy, limiting comparison on survival rates.
We demonstrated a higher use of preoperative stents in patients with low BMI. This is probably due to selection by the surgeon, as patients with low BMI ought to have their nutritional status improved prior to surgery. In fact, we discourage the use of stents prior to esophagectomy, as we have recently demonstrated that this may lead to an impaired oncological outcome (18).
When interpreting our findings, some limitations should be kept in mind. We conducted a study based on data collected in a not necessarily uniform way due to the retrospective nature, which may increase the risk of misclassification of some of the variables. However, there were no systematic changes in the classification systems during the study period, which limits this risk. Our multivariate analyses had low precision due to a relatively few number of patients, which limits the interpretation. As in many other studies, we included both esophageal and GEJ cancers. This can be a limitation as the natural course of these diseases may differ.
A major strength of this study is the inclusion of consecutive patients. Thus, all patients operated with esophagectomy for esophageal or GEJ cancer in the period at our institution is included, limiting the possibility for selection bias. Furthermore, our study consisted of a very homogeneous population, as no patients received any neoadjuvant chemotherapy. In addition, due to our individual-level linkage with the Danish Civil Registration System, no patients were lost to follow up, improving the validity of our survival analyses.
In discussing our findings in light of previous reports on patients undergoing esophageal surgery for cancer, we have been very selective. The motivation for this lies in the fact that the existing literature deals with populations that are very inhomogeneous, that is, in many papers some of the patients have received neoadjuvant chemotherapy and others have not. Also, adverse events are being reported very differently, in fact very few studies have reported complications according to the Clavien–Dindo classification. It is important in future studies to focus on homogeneous populations and use appropriate classifications such as the Clavien–Dindo classification for complications and the WHO classification for BMI.
In conclusion, BMI does not seem to affect survival rates or the presence of serious complications following esophagectomy in patients with esophageal and GEJ cancers not receiving neoadjuvant oncological treatment. Studies on patients undergoing neoadjuvant chemotherapy are needed.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
