Abstract
Background:
Relatively little is known about the risk factors and treatments for postpancreatectomy hepatic steatosis.
Methods:
The records of patients who underwent pancreaticoduodenectomy or total pancreatectomy between 2005 and 2010 and were followed up by periodic imaging were reviewed retrospectively. Risk factors and treatment for postpancreatectomy hepatic steatosis were analyzed.
Results:
A total of 253 patients were included in the analysis, including 137 males and 116 females, of median (5, 95 percentile) age 67 (47, 81) years. Of these 253 patients, 75 (29.6%) developed postpancreatectomy hepatic steatosis. Multivariable logistic regression analysis showed that female gender (p = 0.005; odds ratio: 2.387; 95% confidence interval: 1.293–4.386), body mass index > 22.5 kg/m2 (p = 0.007; odds ratio: 2.330; 95% confidence interval: 1.261–4.307), operative duration > 540 min (p = 0.018; odds ratio: 2.286; 95% confidence interval: 1.153–4.533), and delayed gastric emptying (p < 0.001; odds ratio: 4.598; 95% confidence interval: 1.979–10.678) were independent risk factors associated with postpancreatectomy hepatic steatosis. Treatment consisted of maintenance- or high-dose digestive enzyme replacement therapy. Of patients without obvious tumor recurrence after 6 months, 12 of 15 treated with high dose and only 6 of 35 treated with maintenance-dose digestive enzyme replacement therapy showed improvements in postpancreatectomy hepatic steatosis (p = 0.006).
Conclusion:
Female gender, obesity, longer operative time, and occurrence of delayed gastric emptying are risk factors for postpancreatectomy hepatic steatosis. High-dose digestive enzyme replacement therapy may improve postpancreatectomy hepatic steatosis.
Keywords
Introduction
Pancreaticoduodenectomy (PD) is the only potential cure for patients with pancreatic head and periampullary tumors. Because of the malignant potential of these tumors, overall survival remains generally unsatisfactory even after PD, although some long-term survivors have been described (1, 2). It is known that some long-term survivors develop pancreatic exocrine insufficiency (PEI) (3–5). One consequence of PEI is postpancreatectomy hepatic steatosis (PPHS), which has been reported to occur in 7.8%–40.0% of the patients after PD (6–14).
Possible risk factors for PPHS include pancreatic fistula (9), absence of insulin use (11), postoperative loss of body mass index (BMI) (11), amount of operative blood loss (12), low remnant pancreatic volume (7, 14), postoperative diarrhea (7), infection (14), female gender (8), pancreatic head cancer (7, 10), and postoperative PEI (13), but none of these has been found responsible for the occurrence of PPHS comprehensively. Furthermore, methods to prevent and treat PPHS have not been established.
This retrospective study was, therefore, designed to identify risk factors for PPHS and to determine effective treatments for this condition.
Materials and Methods
Study Populations
Records of patients who underwent PD or total pancreatectomy (TP) at the Department of Gastroenterological Surgery, Cancer Institute Ariake Hospital, between 2005 and 2010 were reviewed retrospectively. Patients with obvious cancer recurrence, those who did not undergo periodic imaging, and those who deceased within 6 months after pancreatectomy were excluded from this analysis.
Definitions
PPHS was diagnosed noninvasively by unenhanced computed tomography (CT), with mean CT attenuation values of the liver <40 Hounsfield units (HU) defined as diagnostic of PPHS (7, 15, 16). CT attenuation values were obtained by averaging parenchymal attenuation value of four sectors (right lateral, right paramedian, left paramedian, and left lateral) of the liver (Fig. 1). CT was performed 6 months after pancreatectomy and/or at times of certain postoperative events, including elevation in tumor marker concentrations. Once PPHS was detected, steatosis was followed up by performing unenhanced CT every 6 months and at times of postoperative events. An increase of >10 HU on unenhanced CT was regarded as an improvement in PPHS. Delayed gastric emptying and pancreatic fistula were evaluated according to the criteria of the International Study Group on Pancreatic Surgery (17) and Pancreas Fistula (18), respectively.
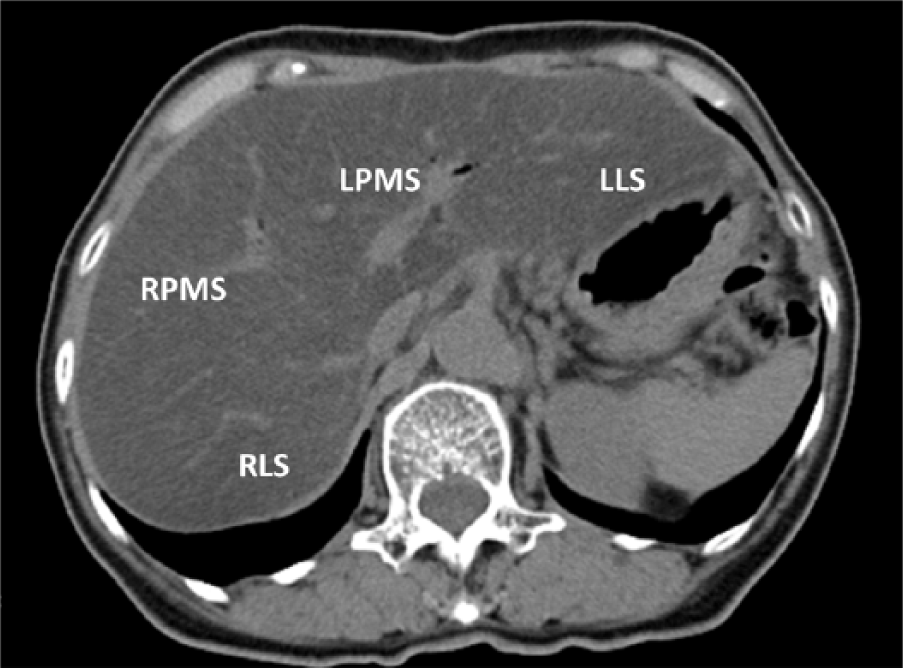
Determination of CT attenuation values in patients following pancreatectomy. Unenhanced CT was used to determine parenchymal attenuation values in four sectors of the liver.
Surgical Procedures
Standardized subtotal stomach preserving PD was performed as described in Hayashibe et al. (19). In cases of adenocarcinoma, concomitant lymphadenopathy and nerve plexus removal were performed. Digestive tract anastomosis was performed using a retrocolic Billroth II type reconstruction with duct-to-mucosal pancreaticojejunostomy in an end-to-side fashion.
Postpancreatectomy Digestive Enzyme Replacement Therapy
All patients received digestive enzyme replacement therapy after PD and TP using Excelase® combination capsules or granules (Meiji Seika, Tokyo, Japan), containing sanactase 125 mg/g, meicelase 125 mg/g, proctase 250 mg/g, olipase 50 mg/g, and the pancreatic digestive enzyme TA 250 mg/g. Although the standard dose of Excelase for general indigestion is 1.2 g/day (three capsules), these patients received 1.2–3.6 g/day Excelase (=3–9 capsules) for postpancreatectomy indigestion. Once PPHS was confirmed, the dose was frequently increased at the outpatient clinic. Patients who received 1.2–3.6 g/day Excelase were defined as the maintenance-dose group and those who received more than 3.6 g/day were defined as the high-dose group. All patients received proton pump inhibitors to prevent anastomotic ulcer.
Study Endpoints
The primary endpoint was the incidence and risk factors for PPHS. The secondary endpoint was to determine an effective treatment for PPHS.
Statistical Analysis
Continuous variables were presented as median (5, 95 percentile) or median (range) as appropriate and compared by Mann–Whitney U tests. Categorical variables were compared using χ 2 tests with Yate’s correction. Parameters shown by univariable analysis to be associated with the occurrence of PPHS (p < 0.10) were included in a multivariable logistic regression analysis to identify independent predictors of PPHS. Possible cutoff values for continuous valuables were determined by receiver–operator characteristic (ROC) curve analysis. Statistical significance was defined as p < 0.05. Among patients diagnosed with PPHS, the improvement rate was compared in those given normal and high doses of Excelase. All statistical analyses were performed using SPSS version 19.0 software (SPSS, Chicago, IL, USA).
Results
From March 2005 to December 2010, 316 patients underwent PD or TP at the Department of Gastroenterological Surgery, Cancer Institute Ariake Hospital. Of these patients, 63 were excluded from analysis because of early recurrences (n = 37), follow-up at another institution (n = 17), death within 6 months (n = 5), or follow-up without regular imaging analysis because of pathologically confirmed benign disease (n = 4). Thus, a total of 253 patients were included in the analysis. These patients consisted of 137 men and 116 women, of median (5, 95 percentile) age 67 (47, 81) years. Median follow-up period was 21 months (range, 6–63 months). PPHS occurred in 75 (29.6%) patients at a median 6 months (range, 3–36 months).
Preoperative indications for pancreatectomies were pancreatic adenocarcinoma (n = 132), duodenal carcinoma including carcinoma of the papilla of Vater (n = 56), lower bile duct carcinoma (n = 34), intraductal papillary mucinous neoplasia (n = 9), gastric cancer (n = 5), neuroendocrine tumor (n = 4), metastatic pancreatic carcinoma (n = 3), pancreatic invasion from retroperitoneal sarcoma (n = 3), colon cancer (n = 2), gastrointestinal stromal tumor (n = 2), solid and pseudo-papillary tumor (n = 2), and mucinous cystic neoplasm (n = 1). Before pancreatectomy, three patients had already developed features of hepatic steatosis, as detected by CT. Of the 253 patients, 248 (98.0%) underwent PD, including 47 who underwent concomitant portal-superior mesenteric vein reconstruction, and 5 (2.0%) underwent TP. Pancreas parenchyma were cut on the portal vein (PV) in 137 patients (54.2%), on the superior mesenteric artery (SMA) in 61 (24.1%), and on the left side of the SMA in 35 (13.8%), including all 5 patients who underwent TP. Postoperative complications, other than PPHS, included postoperative diarrhea in 71 (28.1%) patients, delayed gastric emptying in 34 (13.4%), and pancreatic fistula in 31 (12.3%). Of the 71 patients with postoperative diarrhea, 16 needed opioid antidiarrheal agents for nervous diarrhea possibly caused by nerve plexus dissection around the SMA for pancreatic head adenocarcinoma. A total of 132 patients received adjuvant chemotherapy, with 96 treated with gemcitabine, 17 with gemcitabine plus S-1, 16 with S-1, 2 with imatinib, and 1 with cisplatin plus CPT-11. Patient characteristics are described in Table 1. Univariable analysis showed that female gender, obesity, preoperative hepatic steatosis, operative duration, intraoperative bleeding, and occurrence of delayed gastric emptying were risk factors for PPHS. ROC curve analysis indicated that possible cutoffs for PPHS were 22.5 kg/m2 for BMI, 540 min for operative duration, and 740 mL for intraoperative bleeding with sensitivities of 68.8%, 70.2%, and 72.6%, respectively, and specificities of 52.0%, 54.7%, and 45.0%, respectively. Multivariable logistic regression analysis showed that female gender (p = 0.005; odds ratio (OR): 2.387; 95% confidence interval (CI): 1.293–4.386), BMI > 22.5 kg/m2 (p = 0.007; OR: 2.330; 95% CI: 1.261–4.307), operative duration > 540 min (p = 0.018; OR: 2.286; 95% CI: 1.153–4.533), and delayed gastric emptying (p < 0.001; OR: 4.598; 95% CI: 1.979–10.678) were independent factors for the occurrence of PPHS (Table 2).
Demographic and clinical characteristics of all patients and that of patients with and without PPHS.
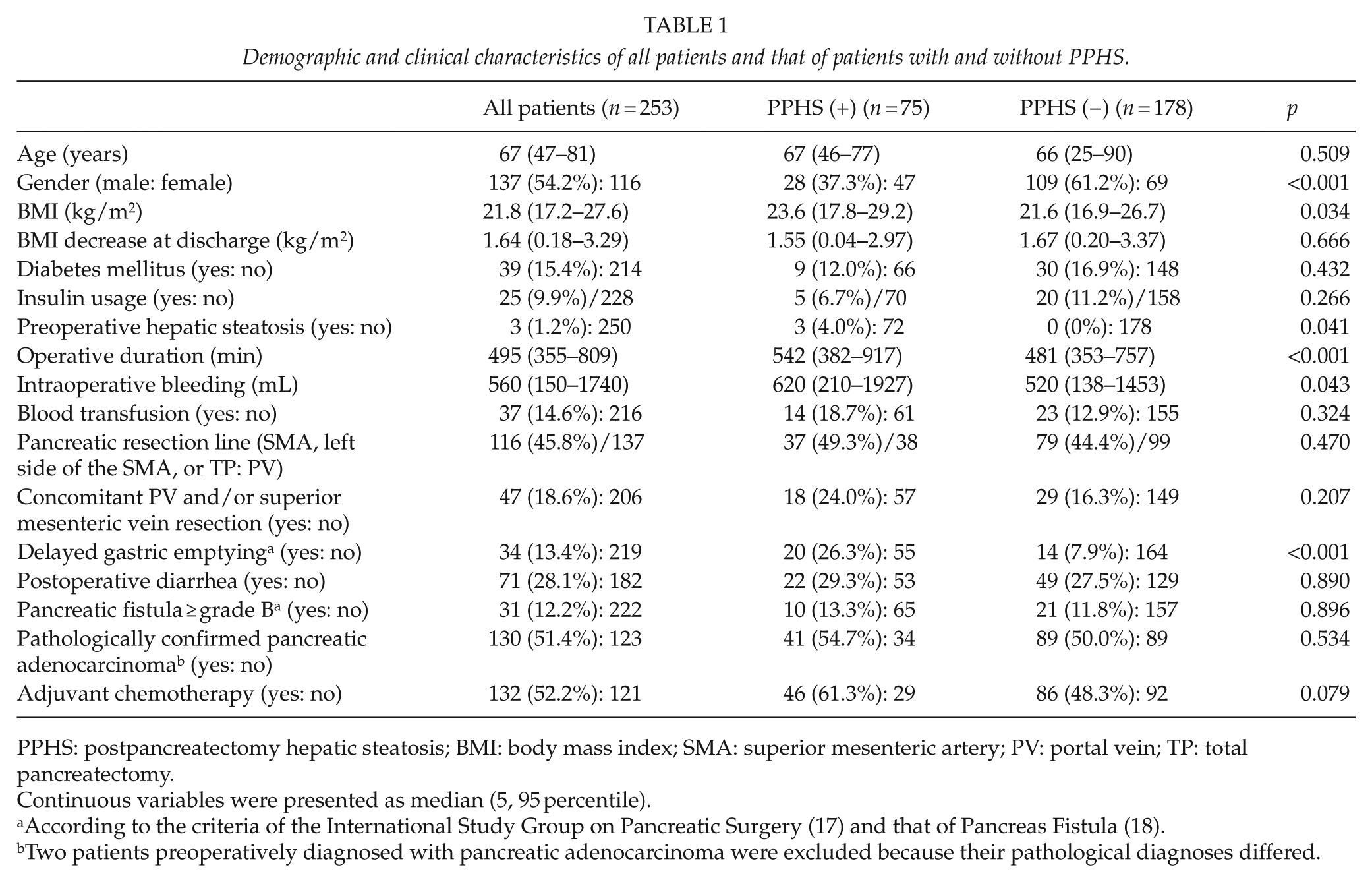
PPHS: postpancreatectomy hepatic steatosis; BMI: body mass index; SMA: superior mesenteric artery; PV: portal vein; TP: total pancreatectomy.
Continuous variables were presented as median (5, 95 percentile).
According to the criteria of the International Study Group on Pancreatic Surgery (17) and that of Pancreas Fistula (18).
Two patients preoperatively diagnosed with pancreatic adenocarcinoma were excluded because their pathological diagnoses differed.
Multivariable logistic regression analysis of the risk factors for PPHS.
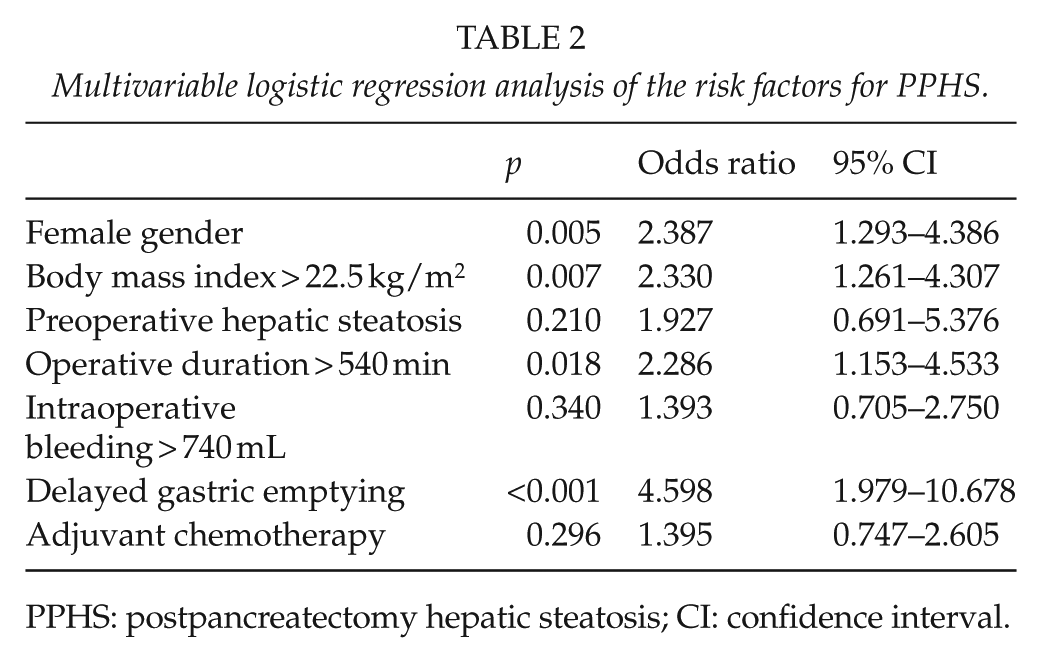
PPHS: postpancreatectomy hepatic steatosis; CI: confidence interval.
Of the 75 patients who developed PPHS, 24 showed cancer recurrence or died of tumor-related causes within 6 months after PPHS was originally diagnosed by CT. The remaining 51 patients were followed up at an outpatient clinic; of these, 15 received high dose (4.8–8.4 g) and 35 received normal (⩾3.6 g) enzyme replacement therapy. A total of 12 of the 15 patients (80.0%) treated with high dose and 6 of the 35 (17.1%) treated with normal dose enzyme replacement therapy showed improvements in PPHS (p = 0.006; Table 3). Preoperative hepatic steatosis, observed in three patients, was not improved by any dosage of enzyme replacement therapy. One patient who developed severe PPHS in the absence of any digestive enzyme died of progressive hepatic failure, which may have been due to the PPHS.
Comparison between normal and high-dose digestive enzyme replacement therapy for PPHS.
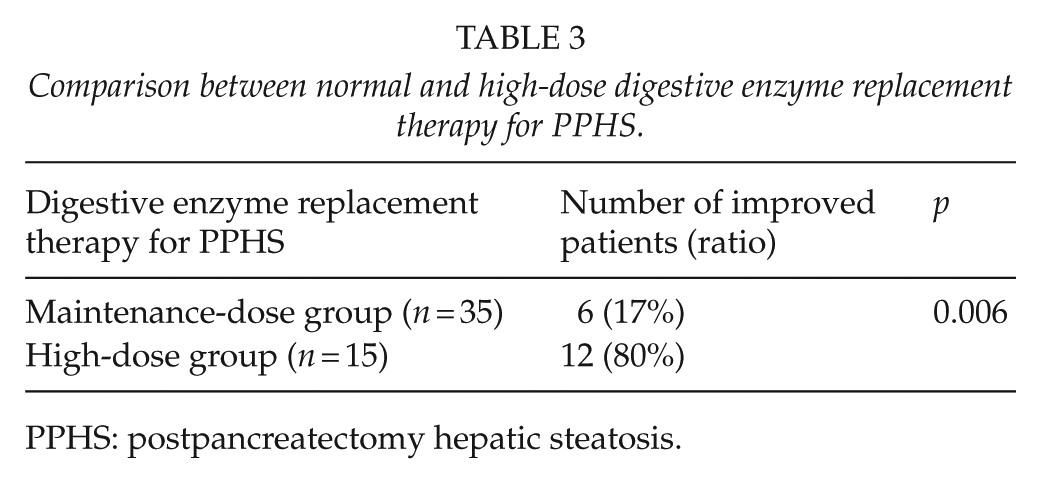
PPHS: postpancreatectomy hepatic steatosis.
Discussion
This study was designed to determine risk factors for PPHS in a large cohort of patients who underwent PD or TP. The study found that female gender, obesity, longer operation time, and delayed gastric emptying were risks factors for PPHS.
Most patients who undergo PD or TP experience a postoperative reduction in body weight caused by insufficient nutritional conditions (3). Reconstruction of the alimentary tract, nervous diarrhea, and delayed gastric emptying can cause postpancreatectomy malnutrition. High BMI and delayed gastric emptying could be associated with perioperative malnutrition, which may induce PPHS. However, BMI decrease was not a risk factor in this study. Obese patients may be naturally more susceptible to hepatic steatosis, and postoperative dietary limitation associated with antrectomy may trigger PPHS. Oral nutritional intake may also be temporarily disturbed by delayed gastric emptying following pancreatectomy.
However, PPHS was not caused only by nutrition-related factors, such as delayed gastric emptying and obesity. PPHS was found to persist in some patients, despite regaining body weight and recovering from postoperative status by sufficient nutritional intake. C-labeled mixed triglyceride breath tests measuring pancreatic exocrine function found that postoperative PEI was a risk factor for PPHS (13). Remnant pancreatic volume (7, 14) and pancreatic adenocarcinoma (7, 10), which often cause obstructive pancreatitis, may also be responsible for insufficient pancreatic exocrine function. PD has also been associated with a deficiency in micronutrients (20), including zinc (21, 22). More than two-thirds of patients who underwent PD showed a lack of zinc associated with PEI (22). This zinc deficiency improved following pancreatic enzyme replacement therapy, further suggesting that zinc deficiency may have an indirect effect on the occurrence of PPHS.
This study also found that female gender was a risk factor for PPHS, in agreement with previous results (8). Estrogen has been reported to prevent nonalcoholic steatohepatitis (NASH) in mice (23). NASH is more frequent in women than in men aged >50 years, perhaps due to menopause (24). This study found that age-related loss of estrogen secretion in females may affect the occurrences of PPHS. Because most of the women in this study were likely post-menopausal, women may be more susceptible than men to the development of PPHS.
In contrast to studies showing that low remnant pancreatic volume was a risk factor for PPHS (7, 14), this study found that low remnant pancreas was not a statistically significant risk factor. It is well known that few patients developed PPHS after distal pancreatectomy (11). Although patients regarded as being at risk of PPHS due to low remnant pancreas or TP might be influenced by occasionally increased doses of digestive enzymes up to 3.6 g/day, thus, taken together, these findings suggest that low remnant pancreatic volume alone is not a risk of PPHS but one of several composite risk factors.
Postoperative diarrhea, pancreatic cancer, intraoperative bleedings, and absence of insulin use were not statistically significant risk factors for PPHS in this study. Increased doses of digestive enzymes may mask the effects of severe diarrhea and of a hard pancreas often caused by pancreatic adenocarcinoma. Intraoperative bleeding may be associated with obesity, making patients susceptive to the effects of nutritional changes after pancreatectomy. Insulin use was reported to reduce the risk of PPHS, suggesting that insulin administration was more important in preventing than in treating PPHS (11). In addition to the 5 patients who underwent TP, the pancreas cut lines were to the left of the SMA in 7 of the 25 patients requiring postoperative insulin. Thus, in agreement with a previous hypothesis (11), there may be some preventable correlation between insulin use and PPHS.
Digestive enzyme replacement therapy has long been considered standard treatment for PEI after pancreatic resection (3, 25, 26). Increased doses of digestive enzymes have been proposed to treat PPHS (6, 7, 10, 13), but dosages and results have remained unclear. This study clearly indicated that higher than standard doses (>3.6 g/day Excelase) may be effective for treating PPHS. Alternatively, PPHS may be treated with pancrelipase, which has been used to effectively treat PEI (27) and PPHS (28). Further research is needed, however, to determine the standard therapy for PPHS.
In this study, one patient died of hepatic failure due to severe PPHS without the recurrence of malignancy while receiving adjuvant chemotherapy. Unfortunately, her enzyme replacement therapy had been discontinued during adjuvant chemotherapy, which may have enhanced her PPHS. Two similar patients who experienced hepatic failure due to severe PPHS have been described (29, 30). Thus, although rare, patients with PPHS may develop hepatic failure if not appropriately treated.
This study had several limitations, including its retrospective design and its inclusion of patients with several underlying conditions, including pancreatic, duodenal, bile duct and gastric carcinoma, intraductal papillary mucinous neoplasia, and neuroendocrine tumor. The study found that female gender was one of the risk factors for PPHS, suggesting that PPHS was susceptible to the effects of menopausal and estrogen. Although most of the women in this study were likely of menopausal age, the number that had undergone menopause was unclear. Furthermore, the optimal dosage of digestive enzyme replacement therapy that could prevent or treat PPHS remains unknown.
As PPHS after PD or TP has several risk factors, care should be taken to monitor its occurrences after pancreatectomy. High doses of digestive enzyme replacement therapy may improve steatosis. Prospective randomized trials are needed to determine the appropriate methods of preventing and treating PPHS.
Footnotes
Acknowledgements
Part of this work was presented at the Meetings of Asian Pacific Association for the Study of the Liver, Digestive Disease Week, 16–19 February 2012, Taipei.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All procedures followed were in accordance with the ethical standards of the Japanese committees responsible for human experimentation and with the Helsinki Declaration of 1975, as revised in 2008. Informed consent was obtained from all individual participants included in the study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
