Abstract
Background and Aims:
Surgical site infection, in particular superficial incision infection, is a common type of complication following abdominal surgery. Negative-pressure wound therapy has been confirmed to reduce the incidence of surgical site infection in various surgeries, but there are few prospective randomized studies into its application to abdominal surgery.
Material and Methods:
A prospective randomized controlled study was conducted in which patients with abdominal surgery and open surgery were randomly divided into a negative-pressure wound therapy experimental group and a gauze-covering control group. Information about demographic data, type of surgery, surgical sites, incision treatment outcomes, surgical site infection factors, and follow-up was recorded.
Results:
From May 2015 to December 2015, 71 patients were enrolled in this study, including 33 in the experimental group and 38 in the control group. There were 10 cases of incision complications, all superficial infections, with an incidence of 14.1%. The surgical site infection incidence was statistically different between the experimental and control groups (3.0% vs 23.7%, p = 0.031). Multivariate logistic regression analysis showed that incision length ⩾20 cm increased the surgical site infection incidence (odds ratio value of 15.576, p = 0.004) and that the application of negative-pressure wound therapy reduced the surgical site infection incidence (odds ratio value of 0.073, p = 0.029).
Conclusion:
Negative-pressure wound therapy can reduce the incidence of surgical site infection in open abdominal surgery.
Keywords
Introduction
Surgical site infection (SSI) is a common type of complication after surgery. It includes superficial incisional SSI, deep incisional SSI, and organ/space SSI (1). SSI brings patients enormous physical, psychological, and economic burdens. In 2005, scholars in the United States reported that SSI increased treatment costs by more than 1.6 billion dollars and consumed more than 900,000 hospital days (2). Scholars in the United Kingdom report that SSI increases treatment costs for each patient by an average of 814–6626 pounds (3). Many measures have been used to decrease the incidence of SSI, such as these recommended by the US Centers for Disease Control and Prevention (CDC): controlling blood sugar, stopping smoking, lessening preoperative hospital stay, moderate use of antibiotics, strict aseptic intra-operative operation, effective hemostasis, minimizing tissue damage, eliminating dead space, and correct placement of drainage (1). Many scholars have also tried a variety of new preventive measures, for example, laparoscopic technique (4). Nevertheless, the SSI incidence remains high.
Gomoll et al. (5) first reported the application of negative-pressure wound therapy (NPWT) in closed incisions, and their outcomes confirmed that its use for treating closed incisions in orthopedic surgery can reduce the incidence of SSI. A subsequent series of reports also confirmed the effectiveness of NPWT in reducing SSI (6–8). Data from mainland China show that abdominal surgery is accompanied by the highest SSI incidence (8.3%) (9), but few prospective randomized studies have been reported on the application of NPWT in closed incisions after open abdominal surgery. In our study, we employed a prospective randomized controlled trial to evaluate the effectiveness of NPWT in closed incisions after open abdominal surgery. We hypothesized that using NPWT in closed abdominal incision can reduce the incidence of SSI.
Materials and Methods
General Information
The experiment was approved by the Ethics Committee of Daping Hospital of Third Military Medical University and was registered in the China Clinical Trial Registry Center (registration no. ChiCTR-IOR-15006439). All patients signed an informed consent. From May 2015 to December 2015, 71 patients were enrolled in this study. Inclusion criteria consisted of abdominal surgery patients with incision length ⩾10 cm and at least 18 years of age; evidence of at least one of the following high infection risk factors: alcoholism, diabetes, chronic obstructive pulmonary disease (COPD), body mass index (BMI) ⩾24, colorectal surgery, surgical time longer than 3 h, blood loss volume more than 300 mL, preoperative serum hypoalbuminemia (albumin <30 g/L), and preoperative hospital days ⩾5 days. Exclusion criteria included age younger than 18 years, laparoscopic surgery, pre-surgery infection, second surgery with incision not healed, allergy to filling dressing or sealed foil dressings, and patients meeting inclusion criteria but not agreeing to participate. Upon admission, the patient’s general information was recorded, and they were assessed for the high-risk factors mentioned above, according to surgery protocol. Patients with laparoscopic surgery, including laparoscopic which could be converted to open surgery, were excluded. Enrolled patients were randomly grouped by independent researchers. Patients in the experimental group were treated with NPWT, and patients in the control group were treated with traditional gauze dressing.
Surgical Procedures
All patients underwent preoperative mechanical bowel preparation. The prophylactic antibiotic cefuroxime sodium was administrated 30 min before incision. If the operation time exceeded 3 h, an additional dosage was given. Antibiotics were discontinued within 72 h after operation. After cutting the skin layer by layer, surgeons entered the abdominal cavity to perform the operation. A Quill suture (Quill®; Surgical Specialties Corporation, Reading, PA, USA) was used to close the peritoneum, rectus abdominis posterior sheath, and rectus abdominis anterior sheath. No. 3-0 Mersilk® (Johnson & Johnson Medical Company, Shanghai, China) was used to close subcutaneous adipose tissues. No. 2-0 Mersilk was used to suture the skin. The subcutaneous fat thickness and incision length were measured intra-operatively. After the incision suture, patients in the experimental group were administered NPWT black foam (60 cm × 5.0 cm × 2.8 cm; main component: polyurethane; VSD®; Wuhan Medical Science & Technology Co., Ltd, China) as a cover for the incision. The foam was tailored according to the incision length. Biological semipermeable membrane was used to seal the covering. After covering the sucking disk, a vacuum machine (VSD) was externally connected to apply continuous suction (−125 mmHg; Fig. 1). At 3 days after operation, the VSD device was removed and replaced with ordinary dressings. Patients in the control group were given a gauze dressing to cover the incision, which was changed daily.
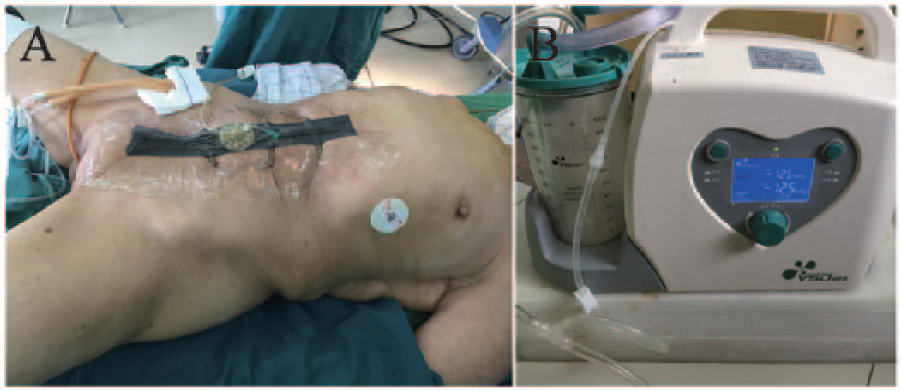
Applying NPWT in abdominal incision. A) A 77-year-old female patient with descending colon and rectal cancer received total colectomy. The incision was covered with polyurethane foam and sealed with biological semipermeable membrane. Vacuum sucker machine was externally connected after covering the sucking disk. B) Vacuum machine. It can display the setting value and the actual measured value.
Randomization, Allocation Concealment, and Blinding
Once the abdominal incision was closed, randomization using random numbers taken preoperatively from a random number table and written in sealed opaque envelopes was done by an independent researcher blinded to the study. Odd numbers were selected for the experimental group and even numbers were used for the control group. The surgical team, clinical staff, and patient were not blinded to the intervention status. The primary outcome of the SSI was assessed by clinical specialist doctors who were blinded to the intervention status. The dressing (NPWT or gauze) was removed by the clinical team prior to the outcome assessors on postoperative day 3 or the day of hospital discharge. The outcome assessors were blinded and assessed the wound daily until discharge after the dressing was removed.
Incision Complications
Incision complications included wound infection, fat liquefaction, wound dehiscence, and wound effusion. Diagnostic criteria for wound infection were based on US CDC criteria, which included one of the following symptoms within 30 days after operation: purulent exudation, red swelling of the skin, local swelling and tenderness, spontaneously dehisces, fever (>38°C), and local abscess formation, all confirmed by histopathologic or radiologic examination (1).
Assessment
The main assessment indicator was the incidence of incision complications. Incisions were observed daily by independent researchers until discharge and outpatient follow-up was performed throughout the 30 days after operation.
Statistical Analysis
A T-test, Kruskal–Wallis test, and chi-square test were used to analyze patients’ general information. The chi-square test (continuity correction) was used for analyzing the main evaluation index, and a logistic regression model was used for the risk factors. SPSS 17.0 software was utilized for statistical analysis and p-value <0.05 was considered statistically significant.
Results
A total of 71 patients were enrolled including 33 in the experimental group and 38 in the control group (Fig. 2). In all, 70 patients underwent selective surgery, while 1 with ulcerative colitis accompanied by massive and difficult controlled bleeding underwent emergency surgery. There was no statistical difference in age, gender, BMI, and American Society of Anesthesiologists (ASA) score between the two groups (p > 0.05) (Table 1).
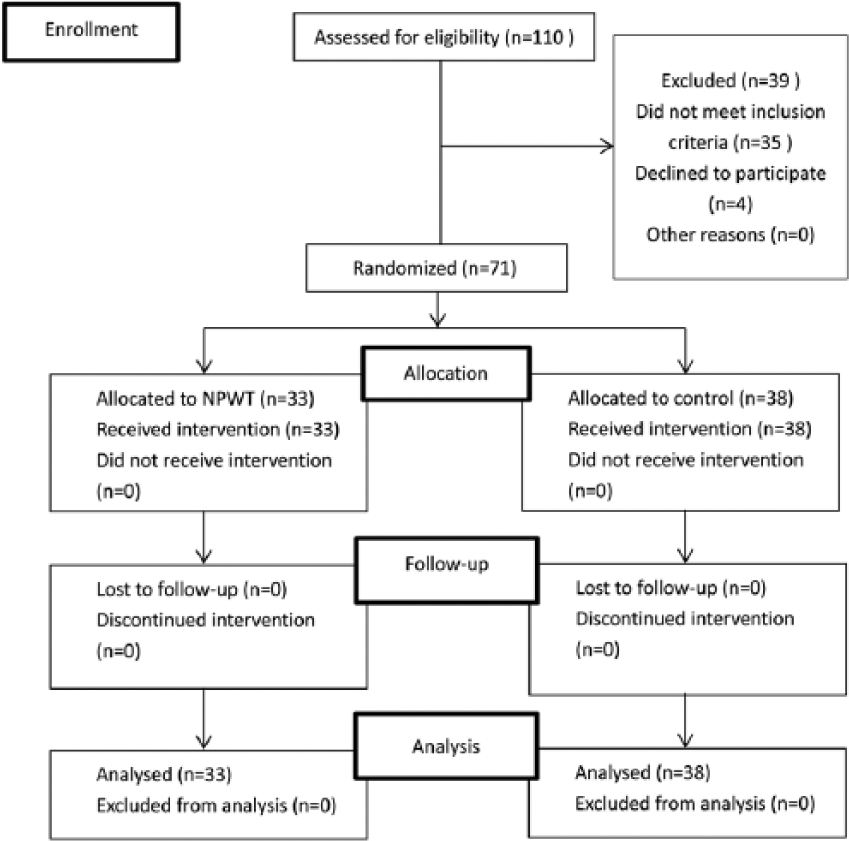
CONSORT flow diagram of the trial.
Comparison of patients’ general information between the experimental group and the control group.
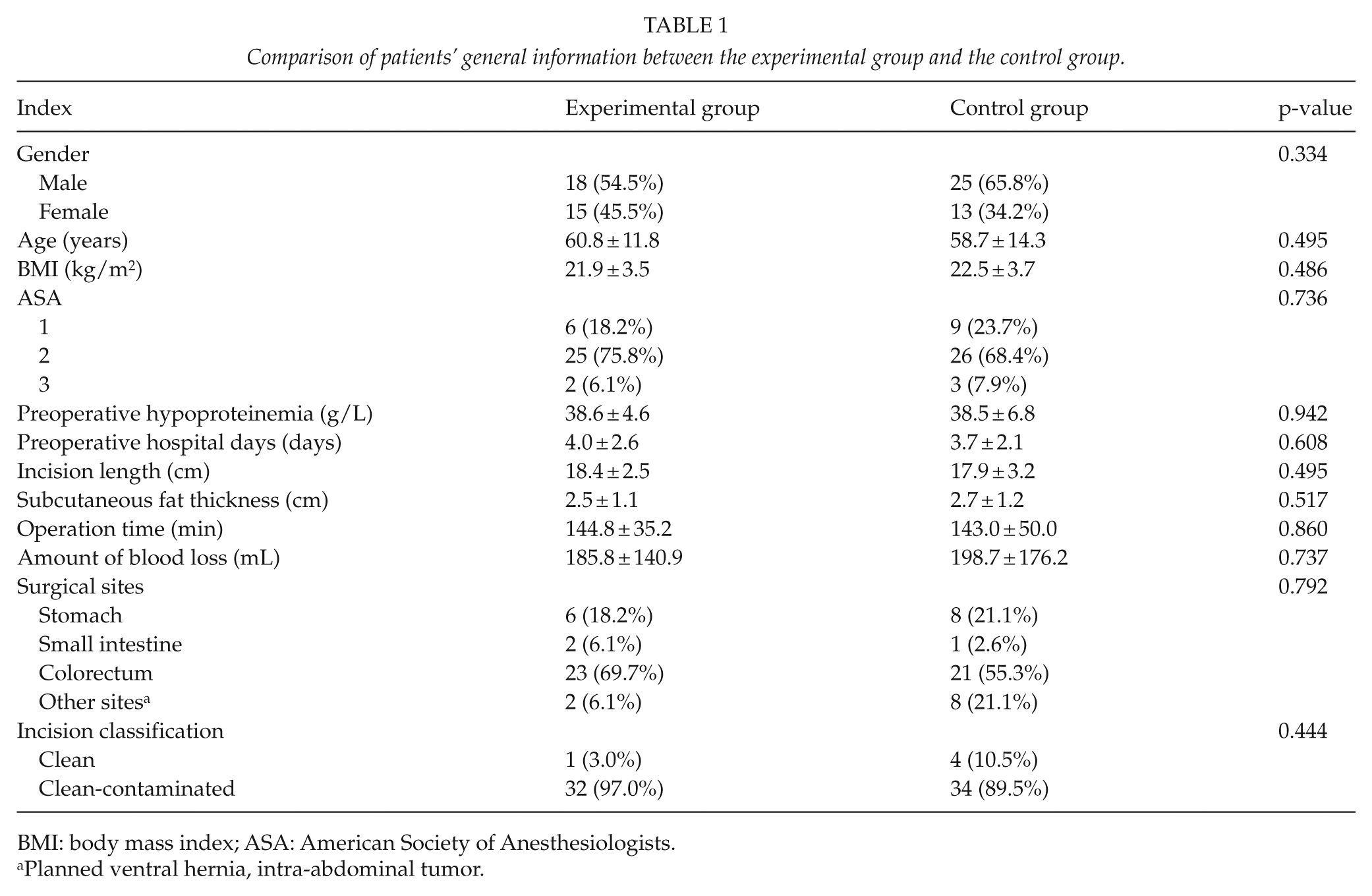
BMI: body mass index; ASA: American Society of Anesthesiologists.
Planned ventral hernia, intra-abdominal tumor.
Incision Complication rates
In all, 10 patients incurred incision complications, with a complication rate of 14.1% (10/71). All were superficial incision infections, without fat liquefaction, wound dehiscence, or wound effusion. There was one case of superficial infection (1/33, 3.0%) in the experimental group and nine cases in the control group (9/38, 23.7%), with the difference being statistically significant (p = 0.031). No site complications were found for stomach and small intestine surgeries. The 10 SSI patients underwent colorectal surgery, planned ventral hernia (PVH), or intra-abdominal tumor resection surgery. The total number of surgical sites (colorectum + other sites) was 54. The incision complication rate was 18.5% (10/54). The infection rate was statistically between the two groups (p = 0.028) (Table 2). Parts of the sutures of the 10 SSI patients were removed and incision secretions were cultured for bacteria. Infrared irradiation was applied to the wound, and hypertonic saline gauze was inserted for drainage. The dressing was changed daily. Antibiotics were added based on the susceptibility results. The incision was sutured after the wound grew fresh granulation tissue. All 10 SSI patients experienced incision healing.
Comparison of the incision infection rate between the two groups.
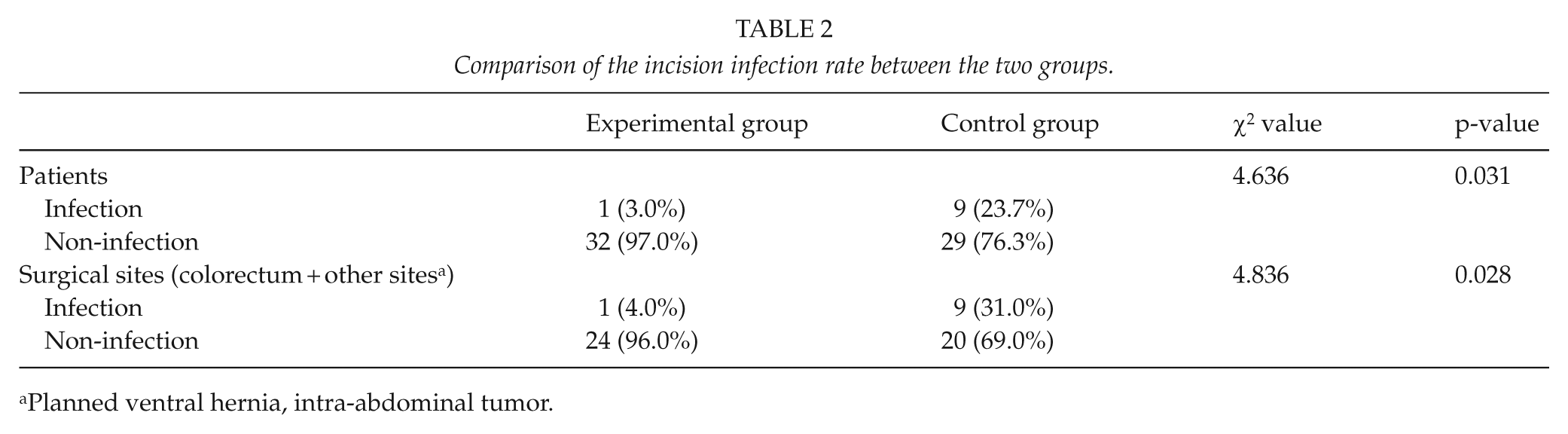
Planned ventral hernia, intra-abdominal tumor.
Risk Factors of Incision Complications
Univariate and multivariate logistic regression analyses revealed two factors relevant to SSI. First, an incision length ⩾20 cm increased the SSI rate (odds ratio (OR) value 15.576, 95% confidence interval (CI) 2.466–98.386, p = 0.004). Second, NPWT usage reduced the SSI rate (OR value 0.073, 95% CI 0.007–0.765, p = 0.029). Factors such as gender, age, BMI, ASA, alcoholism, diabetes, COPD, preoperative hypoalbuminemia, operation time, blood transfusion, fat thickness, number of hospital days before surgery, and amount of blood loss did not affect the incidence of SSI (Table 3).
Univariate and multivariate analyses of incision infection risk factors.
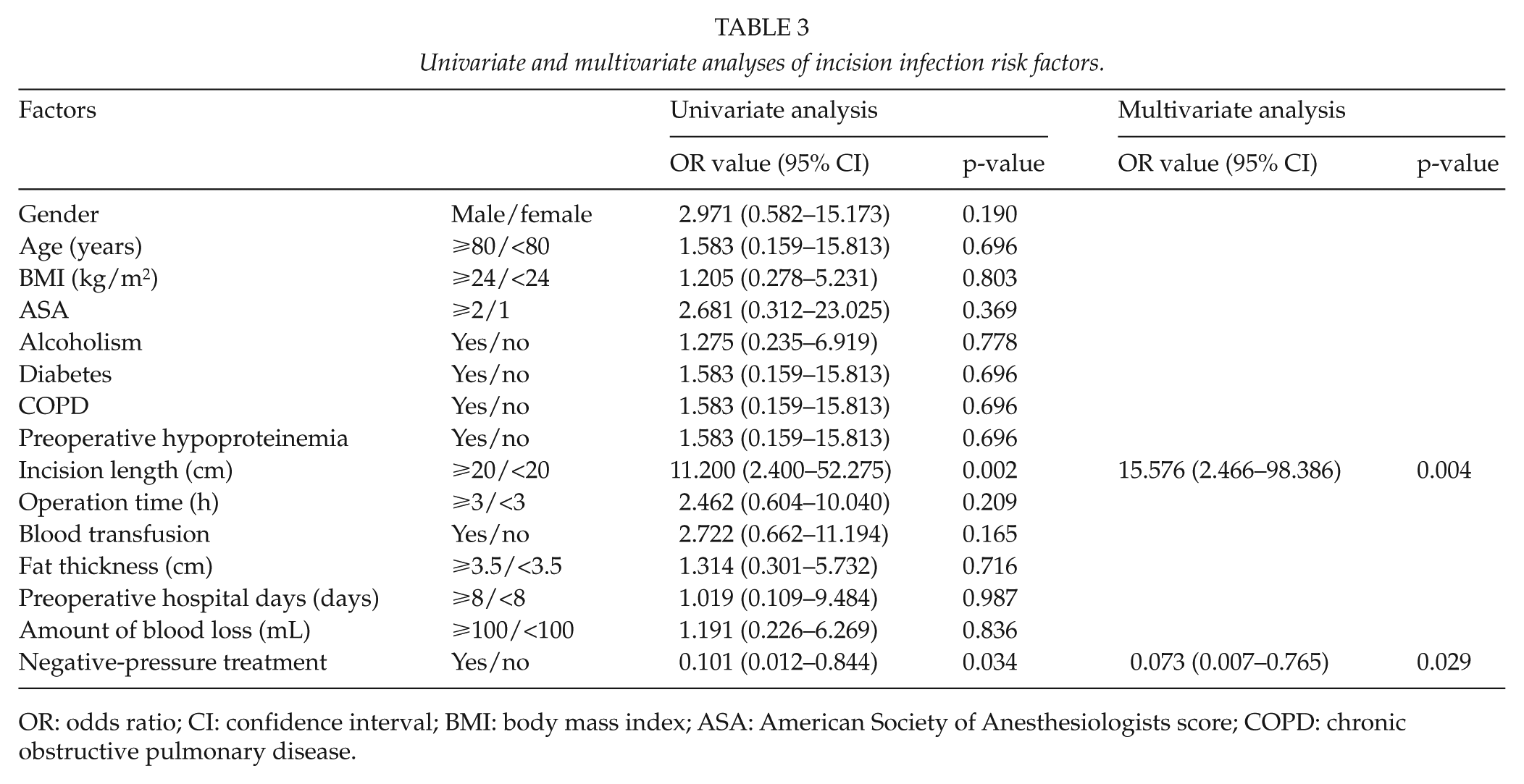
OR: odds ratio; CI: confidence interval; BMI: body mass index; ASA: American Society of Anesthesiologists score; COPD: chronic obstructive pulmonary disease.
Subgroup Analysis
Subgroup analysis showed that if ASA ⩾2 and incision length ⩾20 cm, the SSI rate was statistically different between the experimental and control groups (p < 0.05), registering lower in the experimental group (Table 4).
Subgroup analysis of incision infection.
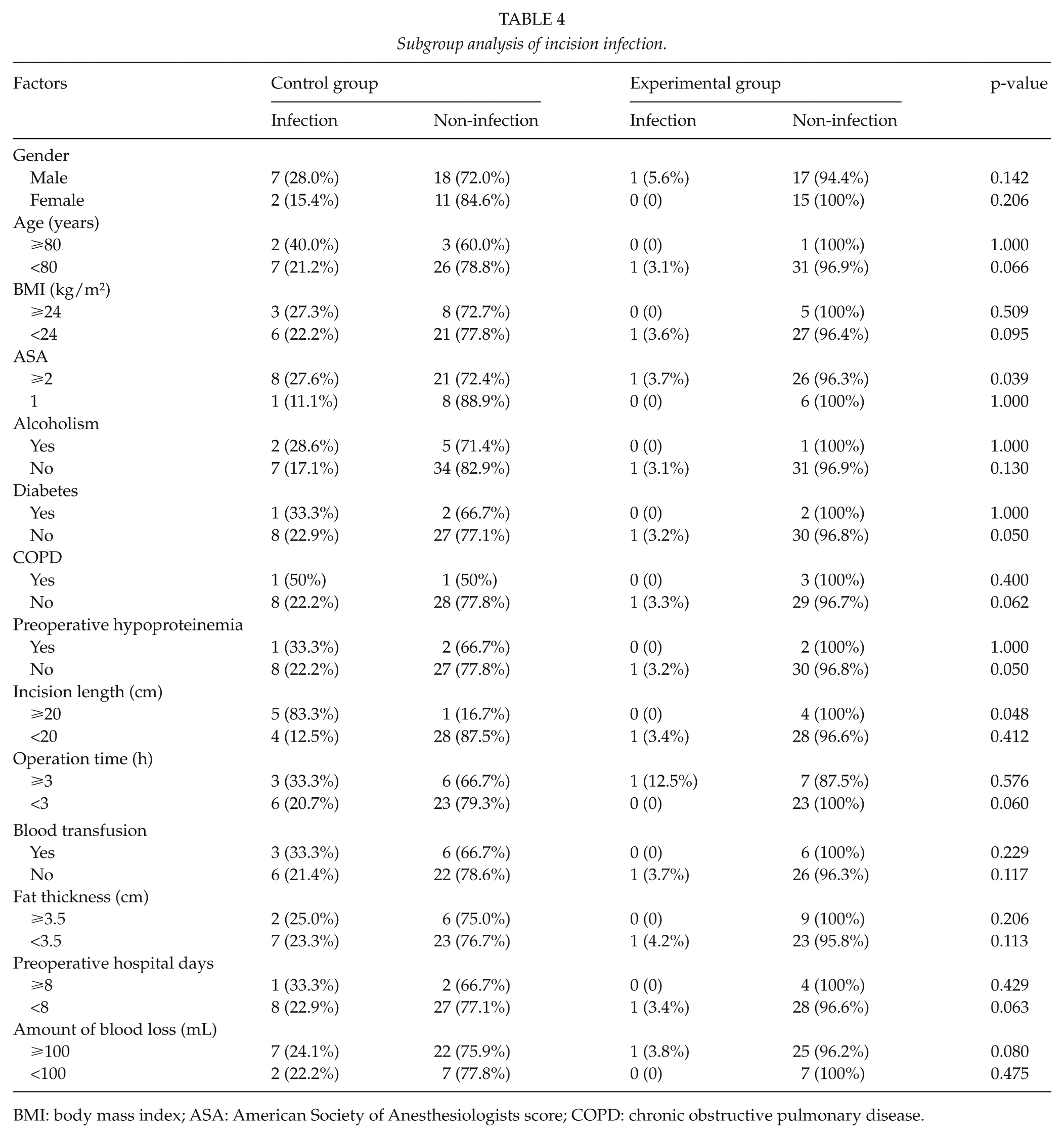
BMI: body mass index; ASA: American Society of Anesthesiologists score; COPD: chronic obstructive pulmonary disease.
Discussion
SSI is the most common surgical complication, depending on the type of incision and the surgical site (10). SSI increases the economic and mental burden of patients, prolongs hospital stay, and increases the rate of intra-abdominal infection, readmission, and mortality (2, 3, 11). The incidence of abdominal SSI rate is 8.3%–32% (9, 12–15), while the infection rate in colorectal surgeries is higher than other surgeries (16, 17). Given the high incidence of abdominal SSI, new prevention measures to reduce SSI rate are needed.
Factors affecting SSI include age, nutritional status, diabetes, smoking, obesity, use of immunosuppressive agents, preoperative hospital stay, operation time, use of antibiotics, surgical techniques, suture material and techniques, surgical drainage, anesthesia, laparoscopic techniques, ASA score, cancer, operation pollution degree, COPD, alcoholism, thickness of subcutaneous fat, blood loss associated with large blood transfusion, and long-term use of hormones (1, 4, 9, 18–20).
NPWT was initially used for acute and chronic wounds (21–23). In recent years, NPWT technology is increasingly being used for closed incisions after surgery to prevent postoperative surgical site complications in high-risk patients. In a multi-center prospective randomized controlled study, Stannard et al. (6) used NPWT for treating lower extremity fractures (calcaneus, pilon, and tibial plateau fractures) with high risk of incision complications and eventually reduced the rate of wound infection and dehiscence. In another prospective study, Grauhan et al. (7) used NPWT among obese patients with cardiothoracic surgery (BMI > 30 kg/m2). Their results showed a reduced SSI rate and also indicated that the Gram-positive bacteria detection rate was lower in NPWT group. Hyldig et al. (24) reported a systematic review and meta-analysis of randomized clinical trials of NPWT compared with standard dressings on closed surgical incisions. Their study included 10 trials, reporting 1311 incisions in 1089 patients. They concluded that NPWT significantly reduced the rate of wound infection and seroma when applied to closed surgical wounds. But few prospective randomized studies have reported on the use of NPWT in the prevention of abdominal incision complications. Mihaljevic et al. (25) cited a study protocol for a randomized controlled trial of NPWT compared with standard dressings on closed incisions following open colorectal surgery. In our prospective randomized clinical study, we aimed to investigate the role of NPWT in the prevention of abdominal incision complications in order to provide better evidence for evidence-based medicine.
In this controlled study, the incidence of abdominal SSI was 14.1%, which was consistent with the above-cited literature. The incidence of SSI in the NPWT experimental group was 3.0% and 23.7% in the control group, indicating significantly statistical differences (p = 0.031) and confirming that NPWT can reduce the incidence of abdominal SSI, as other literature has reported (8). The possible mechanisms include (1) increased skin blood flow and tissue perfusion (26); (2) prompt removal of exudation to avoid fluid on the inter-stitched face and tissue layers; (3) compression of abdominal wall tissues, eliminating gaps, and dislocation movement, thus promoting healing (27); (4) promotion of epithelial cell migration (28); (5) providing a closed environment less susceptible to contamination (27); and (6) reducing local edema (27).
In addition, univariate and multivariate logistic regression analyses showed two factors relevant to SSI: incision length ⩾20 cm increases incidence and is a risk factor (OR value > 1, p < 0.05), and NPWT usage reduces incidence and is a protective factor (OR value < 1, p < 0.05). The subgroup analysis indicated that when patients’ ASA ⩾2 and incision length ⩾20 cm, the incidence of SSI was lower when utilizing NPWT, indicating that NPWT may be useful for such patients. Motie et al. (29) reported that the incision length was closely related to the SSI rate, but they did not give a specific numerical relationship. Harato et al. (30) reported that the incision length was not a wound healing factor for a total knee arthroplasty (TKA) having an incision length of 12.7 ± 1.5 cm (mean ± standard deviation). This may be because the incision classification was clean in TKA surgeries, and the incision classification was clean-contaminated in most abdominal surgeries (e.g. 93.0% incisions were clean-contaminated in this study). Also, it may be associated with the small incision length in TKA surgeries.
Our study has the limitations of few samples and of being a single-center study. In a future study, we will conduct multi-center experiments to confirm our result.
Overall, this prospective randomized controlled study shows that NPWT can reduce the abdominal SSI rate, especially for colorectal, PVH, and intra-abdominal tumor surgeries. Meanwhile, we do not believe that all abdominal surgeries require NPWT to prevent infection. We recommend using NPWT to reduce the incidence of SSI in surgeries with a high risk of infection.
Footnotes
Acknowledgements
China Clinical Trial Registry Center (registration no. ChiCTR-IOR-15006439).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors declare support by grants from “New Techniques of Trauma Care” (2012BAI11B01).
