Abstract
Background and Aims:
There is a risk of decreased physical function, quality of life and persistent pain after open surgery for esophageal cancer. There are currently no studies that evaluate the effect of any postoperative intervention, including physical exercises, after this type of surgery. The aim of the study was therefore to evaluate the effect of a training intervention after Ivor-Lewis resection of the esophagus.
Material and Methods:
Patients scheduled for esophagus resection according to Ivor-Lewis were randomized to an intervention group or a control group. The training intervention started at discharge and lasted three months. Before discharge, patients were given three leaflets with exercises to increase range of motion in the affected area and exercises aiming to restore lung function and physical function. All exercises were described in detail and the patients carried out the ones in the first program under supervision. Before surgery and three months after discharge, the patients estimated their level of physical function, level of physical activity, and quality of life. They also underwent spirometry, measurements of range of motion in the rib cage, spine, and shoulders, and three functional tests. Comparisons of differences within and between the groups were made.
Results:
A total of 43 of 64 randomized patients participated in the follow-up. Postoperatively, the patients in the intervention group had a significantly higher degree of physical function and less deteriorated range of motion in right shoulder flexion and thoracic left lateral flexion. There were no significant differences between the groups in lung function, pain, or quality of life.
Conclusion:
The results of the three-month intervention indicate that specific training can positively affect physical function and range of motion to preoperative values. The intervention was well tolerated, and no side effects were registered.
Introduction
It is well known that open thoracoabdominal esophageal resection for cancer is a procedure with a high risk of complications, and patients’ preoperative status is a crucial factor for outcome. Dysphagia is a common feature of the disease and leads to nutritional difficulties that result in weight loss and muscle weakness. As many are smokers, cardiac co-morbidity and decreased respiratory function are also common (1). Neo-adjuvant oncological treatment increases chances of survival but often further reduces the patient’s preoperative performance level (2). Moreover, the surgical procedure per se is extensive (3). A low preoperative performance status (4) in combination with neo-adjuvant therapy and the extensive procedure significantly increase the risk of postoperative co-morbidity and mortality.
An Ivor-Lewis (3) procedure is common today. This includes a laparotomy for preparation of a gastric tube for a substitute, followed by a lateral–posterior thoracotomy between the fifth and sixth ribs on the right side of the rib cage for the resection and subsequent creation of the esophago-gastric anastomosis. The surgical procedure involves bilateral opening of the pleural cavities, and patients are treated with chest drains bilaterally in order to re-expand the lungs. The procedure carries a high risk of pulmonary complications in the immediate perioperative period. One could therefore assume that the effects of physical training, such as breathing exercises aimed at preventing pulmonary complications in the immediate postoperative period, would be well described. However, to our knowledge, only three clinical studies have evaluated various kinds of breathing exercises aimed at preventing pulmonary complications in the immediate postoperative period by breathing exercises before or after surgery (5–7). Preoperative inspiratory muscle training and especially high-intensity training (5, 6) has been shown to improve respiratory muscle function, but there is no evidence that it reduces postoperative pneumonia (6). The effects of continuous positive airway pressure (CPAP), compared to positive expiratory pressure (PEP), have been evaluated in the postoperative period (7). CPAP was found to significantly decrease the risk of respiratory failure and need for reintubation in comparison to PEP.
As yet, there are no clinical studies that evaluate any other postoperative breathing exercises aimed at improving physical functioning, either in the immediate postoperative phase or in a longer perspective.
The proportion of long-term survival depends mainly on patient’s preoperative tumor stage and performance status at the time of surgery (8, 9). Chances of survival have increased owing to improved surgical techniques, safer clinical staging, and the addition of neo-adjuvant oncological treatment regimens, although only a few reports describe long-term follow-up of physical function. These have shown that respiratory restrictions persist after the operation as well as poorer physical function, range of motion in the rib cage, and limitations in activities in daily living (8–12). Together, this indicates that, while the treatment offers a chance to cure the cancer, it also leads to a reduction in a variety of physical functioning parameters and carries risks of chronic chest pain (13) and poorer quality of life that will persist (8, 9).
Following extensive surgical procedures, a structured rehabilitation may help to reinstate the degree of physical functioning and decrease the risk of physical disability. In surgery for spinal hernia, for example, with trauma to muscles and bone structures in the thorax and thoracic spinal column, there is strong evidence for an active rehabilitation approach in favor of a less active one (14). It is therefore reasonable to assume that early and active physical rehabilitation of patients undergoing esophageal resection for cancer may lead to a faster and better physical recovery in this group of patients as well. There are few, if any, studies that address the value of interventions aimed at improving physical functioning in this patient group after the perioperative period.
The purpose of the trial was therefore to evaluate the effect of a training intervention after open resection of the esophagus according to Ivor-Lewis. The intervention was aimed to decrease the degree of postoperative disability and pain and improve mobility of the chest, respiratory function, and quality of life.
Materials and Methods
Patients scheduled for Ivor-Lewis esophageal resections at Sahlgrenska University Hospital during the semesters between September 2005 and June 2010 or at Karolinska University Hospital between April 2009 and March 2010 were invited to participate. All patients scheduled to undergo surgery perform an exercise electrocardiogram (EECG) and spirometry as part of the preoperative routine work-up. In order to be eligible for surgery, patients must be able to perform 90 W (4), display no serious untreated cardiac disease that may be critical, and have a normal or only moderately reduced lung function so that one-lung ventilation can be undertaken safely during surgery. The inclusion criteria were patients scheduled to undergo Ivor-Lewis esophagus resection with no musculoskeletal or neurological disease or trauma affecting respiration or range of motion in the rib cage and ability to speak Swedish. The regional Ethics Committee for the region of Västra Götaland, Sweden, approved the study protocol, and the patients participated after receiving written and oral information and giving written consent (Registration number: 078-05).
The following tests were undertaken the day before surgery:
Primary end-point
Degree of physical function estimated by the Disability Rating Index (DRI) questionnaire, which includes 12 items covering activities from dressing and going for walks to lifting heavy objects and exercising. The item responses were rated on visual analog scales and higher values indicated a higher degree of disability as compared to lower values (15). The scale was developed in Sweden and has been tested for validity, reliability, and responsiveness (15).
Other end-points
Pain in the neck, rib cage, and shoulders estimated on a 100-mm visual analog scale (16, 17).
Spirometry (forced vital capacity (FVC), forced expiratory volume in 1 s (FEV1), peak expiratory flow (PEF)) performed in a standardized manner (18) using an EasyOne (ndd Medical Technologies, Schweiz).
Range of motion
Chest expansion as measured by the difference in chest circumference at the level of the xiphoid process between maximal inspiration and maximal expiration movements (19).
Thoracic flexion and extension assessed by measuring the distance between skin marks at the seventh cervical spinal process and 30 cm below when the subject was standing erect and after maximal forward and backward bending of the back and the neck. Thoracic lateral flexion was measured at the level of the tip of the index finger on the patient’s thigh in an erect and a maximal lateral bending position (20). Active shoulder flexion and abduction measured with a goniometer (21):
Physical function Stand-ups from a chair were tested in terms of how many seconds the test person needed to perform 10 complete stand-ups without support from a 45-cm-high seat. Heel rising as time needed to perform 10 complete raises without support. The strength of the dominant hand (peak value) was estimated using Grippit (AB Detektor, Göteborg, Sweden) (22).
Level of physical activity by a six-level scale where low figures indicate a sedentary and a high score an active lifestyle (23).
Quality of life was assessed by the EORTC QLQ-C30 (European Organization for Research and Treatment of Cancer Quality of Life Questionnaire Core–30 version 3.0) (24). All responses were converted to a score between 0 and 100 using a linear transformation following EORTC guidelines. High scores indicate good functioning but a high level of symptoms (25). The questionnaire has been validated in Sweden (24).
The patients underwent a standard Ivor-Lewis resection (3). In brief, a gastric tube was prepared via an upper midline abdominal incision. The hiatus was dissected and the left pleural sac opened. The patient then received a left sided chest drain and the wound was closed. The patient was turned on the left side, and a lateral–posterior thoracotomy was conducted on the right side of the chest. The right dorsal latissimus muscle and the right anterior thoracic muscles were transacted, and the posterior mediastinum approached through the fifth or sixth intercostal space. The intercostal nerve and vessels were normally spared. The right lung was deflated and the patients were ventilated with a double-lumen tube and positive end expiratory pressure (PEEP) through the rest of the procedure. The patients with cancer underwent a standard two-field lymphadenectomy. The esophagus remnant was finally connected to the proximal end of the gastric tube with a stapling device or, in some cases, hand sewn. A naso-gastric tube was positioned in the gastric tube. Finally, another chest drain was placed in the right side of the thorax, the lung was expanded, and the wound was closed. The chest drains were held active (15 cm H2O) on average for 2–5 days. In the postoperative period, the patients received epidural anesthesia that was followed some days later by oral analgesics.
Starting on the first day after surgery, the patients underwent standardized physiotherapy including breathing exercises with CPAP while in intensive care (7). After this, deep breathing exercises were practiced with positive expiratory pressure (PEP/RMT set, Wellspect Healthcare, Mölndal, Sweden) every 2 h during the daytime, and there was early and frequent mobilization.
Patients who did not have the scheduled operation and who needed prolonged care at the intensive care unit (>7 days) or additional surgery during the first postoperative week (n = 27) were excluded from the study (Fig. 1). Randomization of the remaining 64 patients to the intervention or control group (1:1) was done a week after surgery, Fig. 1. It was performed using a computer random number generator. The assignment to groups after randomization was performed by a third party independent of the recruitment process for allocation and randomization. The allocation sequence was kept in opaque, identical, and sealed envelopes, locked at the department of surgery at Sahlgrenska University Hospital.
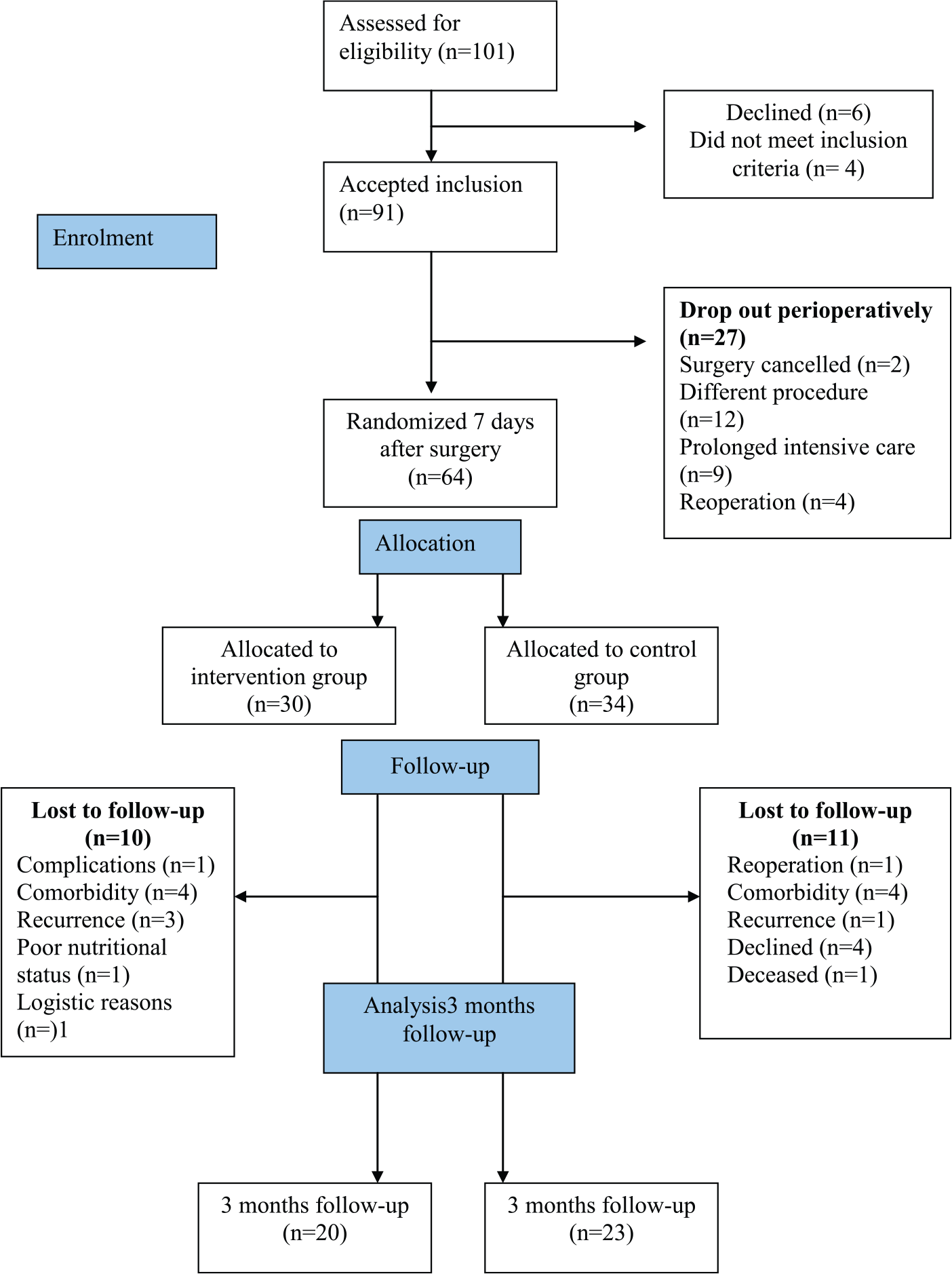
Flowchart.
All patients met a specialized physical therapist before discharge for a discussion about their normal physical activity level and how to regain this function. The patients in the intervention group also received a rehabilitation program that consisted of three leaflets of exercises to restore lung function, range of motion in the thoracic spine and shoulder, and strength exercises for the back extensors, shoulders, and legs (see Appendix 1). The program was gradually increased in intensity through the rehabilitation period. All exercises were described in detail and the patients carried out the exercises in the first program under supervision. The patients were encouraged, unsupervised at home from the day of discharge, to perform the range of motion daily until they felt no restrictions or pain at the end range. The control group was given the information that it is not known whether any training may be beneficial and was told to avoid specific interventions during the first three months postoperatively. Those discharged to convalescence care received no specific additional instructions or exercises to improve rib cage or lung function.
At discharge, the physician in charge prescribed the use of oral analgesics based on individual needs. The patients returned to the hospital three months after discharge and the same tests were undertaken as were done preoperatively.
Statistics
The sample size calculation was based on the difference in the degree of disability assessed by the DRI. To detect a clinically significant difference between groups with 80% power and at the 5% significance level, and a difference in DRI between groups of 15 mm (standard deviation (SD) ± 15 mm) (control group 23 mm (mild disability) (15) and intervention group 8 mm (healthy people with minor ailments)), 17 patients were needed in each group. To be able to detect differences in some of the secondary outcomes and to compensate for dropouts, we planned to randomize 30 patients per group. Because of a high dropout rate and a long period of inclusion, the trial was closed when ⩾20 patients in each group were reached.
SPSS version 15.0 was used for the statistical analyses. The distributions of the variables were given as mean, SD, median, minimum, and maximum for continuous variables and as number and percentages for categorical variables. For comparison within groups, the sign test was used for ordered variables and the Wilcoxon signed-rank test was used for continuous variables. For comparison between the two groups, the Mantel–Haenszel chi-square test was used for ordered categorical variables and the Mann–Whitney U test was used for continuous variables. Adjustment for variable baseline values in tests between the groups was made by analysis of covariance (ANCOVA). All significance tests were two-sided and conducted at the 5% significance level.
Results
There were no significant differences between the groups preoperatively concerning background data and tumor characteristics, Table 1. In addition, there were no significant differences in these variables between the 64 randomized patients and the 27 who dropped out perioperatively and therefore were not randomized. Perioperative data concerning the surgery and postoperative care are given in Table 1. The groups were comparable preoperatively except for a significantly smaller thoracic extension in the control group.
Patient and tumor characteristics for patients who participated in the follow-up or were lost to follow-up; also, perioperative data of patients who completed the study.
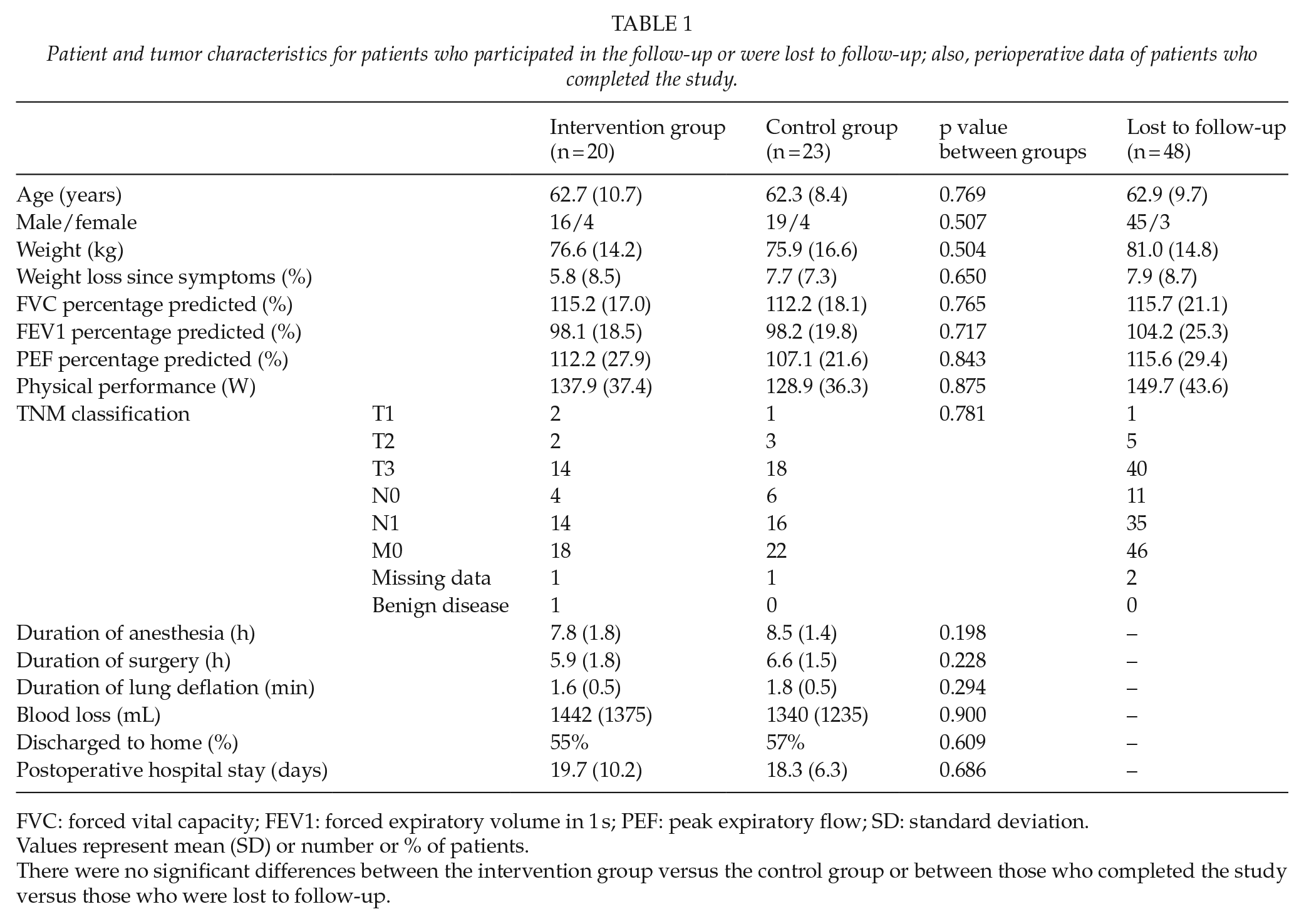
FVC: forced vital capacity; FEV1: forced expiratory volume in 1 s; PEF: peak expiratory flow; SD: standard deviation.
Values represent mean (SD) or number or % of patients.
There were no significant differences between the intervention group versus the control group or between those who completed the study versus those who were lost to follow-up.
The degree of disability measured by DRI is shown in Table 2. There were no significant differences in DRI between preoperative and postoperative values in the intervention group. The patients in the control group scored postoperatively significantly lower in 4 of the 12 activities: outdoor walks, carrying a bag, running, and lifting heavy objects (p < 0.05) and there was a lower sum score (p = 0.013) compared to preoperative values. There were no significant differences between the groups in the separate activity scores, but the patients in the control group had a significantly higher sum score than the intervention group (22.5 vs 6.45 mm, p = 0.041).
Change in physical function (Disability Rating Index (DRI)) between before and three months after surgery in patients included in the intervention study.
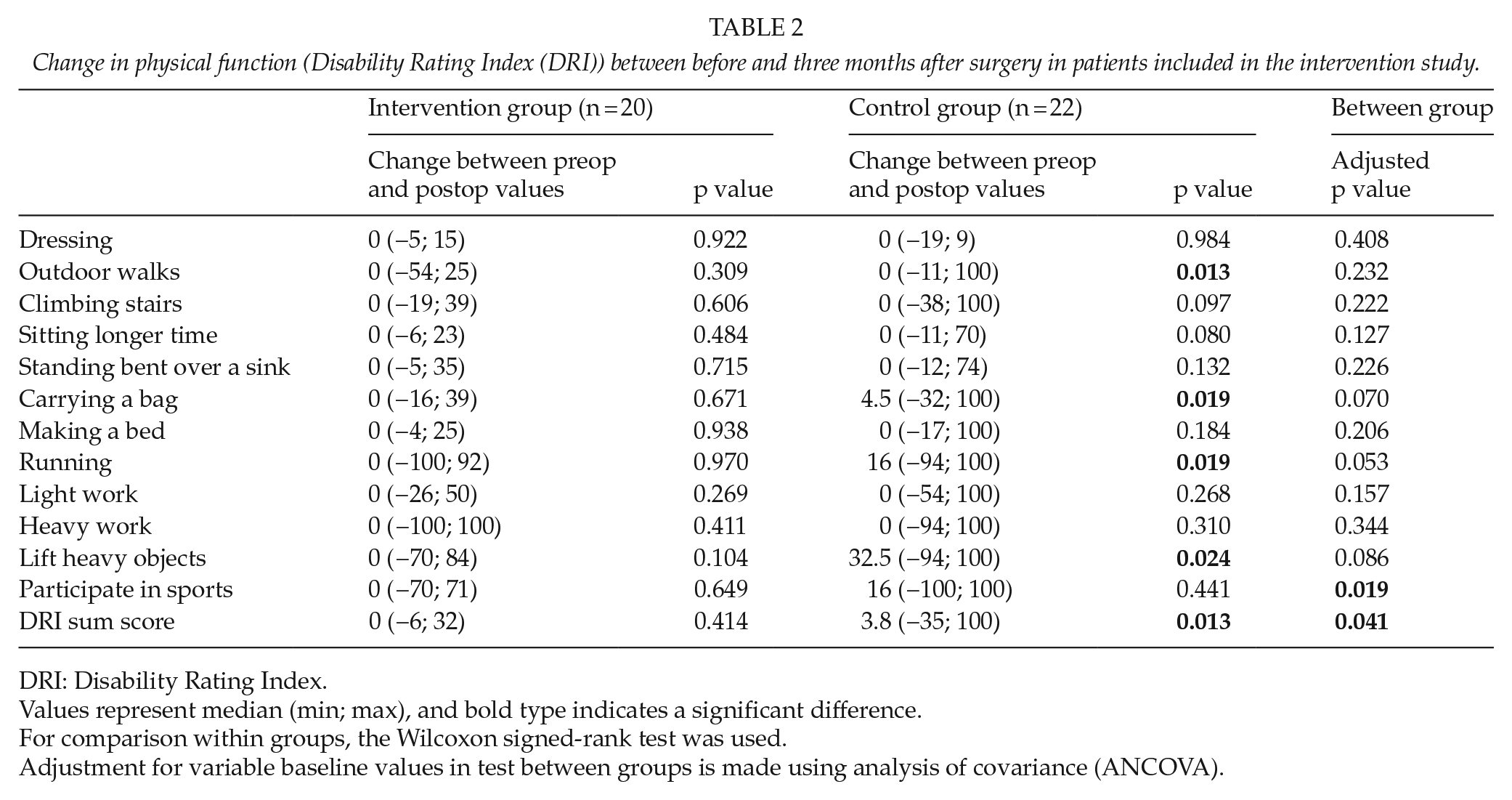
DRI: Disability Rating Index.
Values represent median (min; max), and bold type indicates a significant difference.
For comparison within groups, the Wilcoxon signed-rank test was used.
Adjustment for variable baseline values in test between groups is made using analysis of covariance (ANCOVA).
Pain ratings were low, 0 in median, preoperatively and postoperatively (data not shown). The patients in the control group had significantly more pain in the rib cage postoperatively as compared to before surgery (p = 0.016), which was not seen in the intervention group. There were no significant differences between the groups.
Results of spirometry and the measurements of range of motion both within and between the groups are given in Table 3. Three months postoperatively, FVC was significantly decreased in both groups (p < 0.01), and forced expiratory volume in 1 s and chest expansion were decreased in the control group (p < 0.05). There were no significant differences between the groups. Three months postoperatively, the patients in the control group had a significantly decreased range of motion in left thoracic lateral flexion (p = 0.005) and right shoulder flexion (p = 0.035) compared to preoperatively, while the intervention group had approximately the same levels as preoperatively. This difference was also detected in the comparisons between the groups (right shoulder flexion, a difference of 9.1°, p = 0.031; left thoracic lateral flexion, a difference of 2.5 cm, p = 0.022).
FVC, FEV1, and PEF (percentage of predicted values), range of motion in shoulder and thorax, and functional tests in the intervention and control groups.
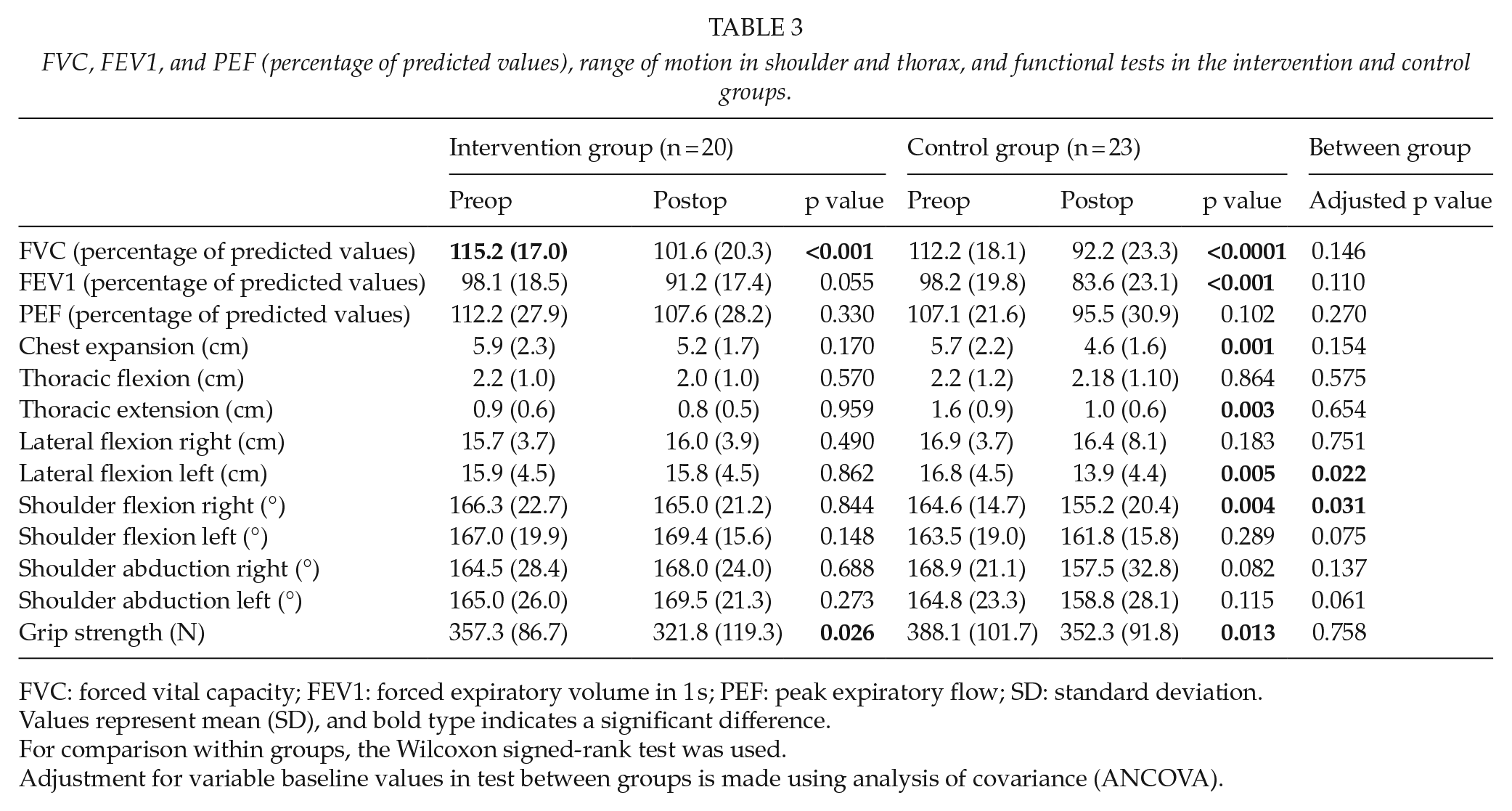
FVC: forced vital capacity; FEV1: forced expiratory volume in 1 s; PEF: peak expiratory flow; SD: standard deviation.
Values represent mean (SD), and bold type indicates a significant difference.
For comparison within groups, the Wilcoxon signed-rank test was used.
Adjustment for variable baseline values in test between groups is made using analysis of covariance (ANCOVA).
The time required to perform the stand-up and heel rising tests was only marginally changed (<1 s) between the test occasions (data not shown) and there were no significant changes either within or between the groups. Both groups had significantly less grip strength postoperatively (p < 0.05), but there were no significant differences between the groups (Table 3). Both groups scored their physical activity level to be, on median, 4 both preoperatively and postoperatively, which corresponds to “moderate exercises 1–2 h a week, e.g. jogging or swimming or light physical activities more than 4 h per week” (23).
The results in the EORTC QLQ-C30 for both groups at diagnosis and at follow-up are shown in Table 4. Both groups reported a poorer quality of life at the follow-up compared to at diagnosis for the majority of scales and single items. A clinically significant deterioration (change of ⩾10 points (25,26)) was found in both groups for problems with fatigue, dyspnea, and diarrhea. In the intervention group, another three significant deteriorations were found for global quality of life, insomnia, and loss of appetite, while the control group reported a significant worsening for physical functioning, role functioning, and pain, and a significant improvement for constipation. No significant differences were found when the two groups’ quality of life was compared at diagnosis. Two differences above 10 points were found at the follow-up, for role functioning in favor of the intervention group and for problems with constipation in favor of the control group. When the changes over time for patients answering the questionnaire at both measurement points were compared between groups, three significant changes in differences were found: the control group’s role functioning deteriorated significantly more compared to the study groups’, while the intervention group had a significantly decreased global quality of life and experienced appetite loss. No side effects of the intervention were recorded.
Results from the EORTC QLQ-C30 at baseline and at the follow-up.
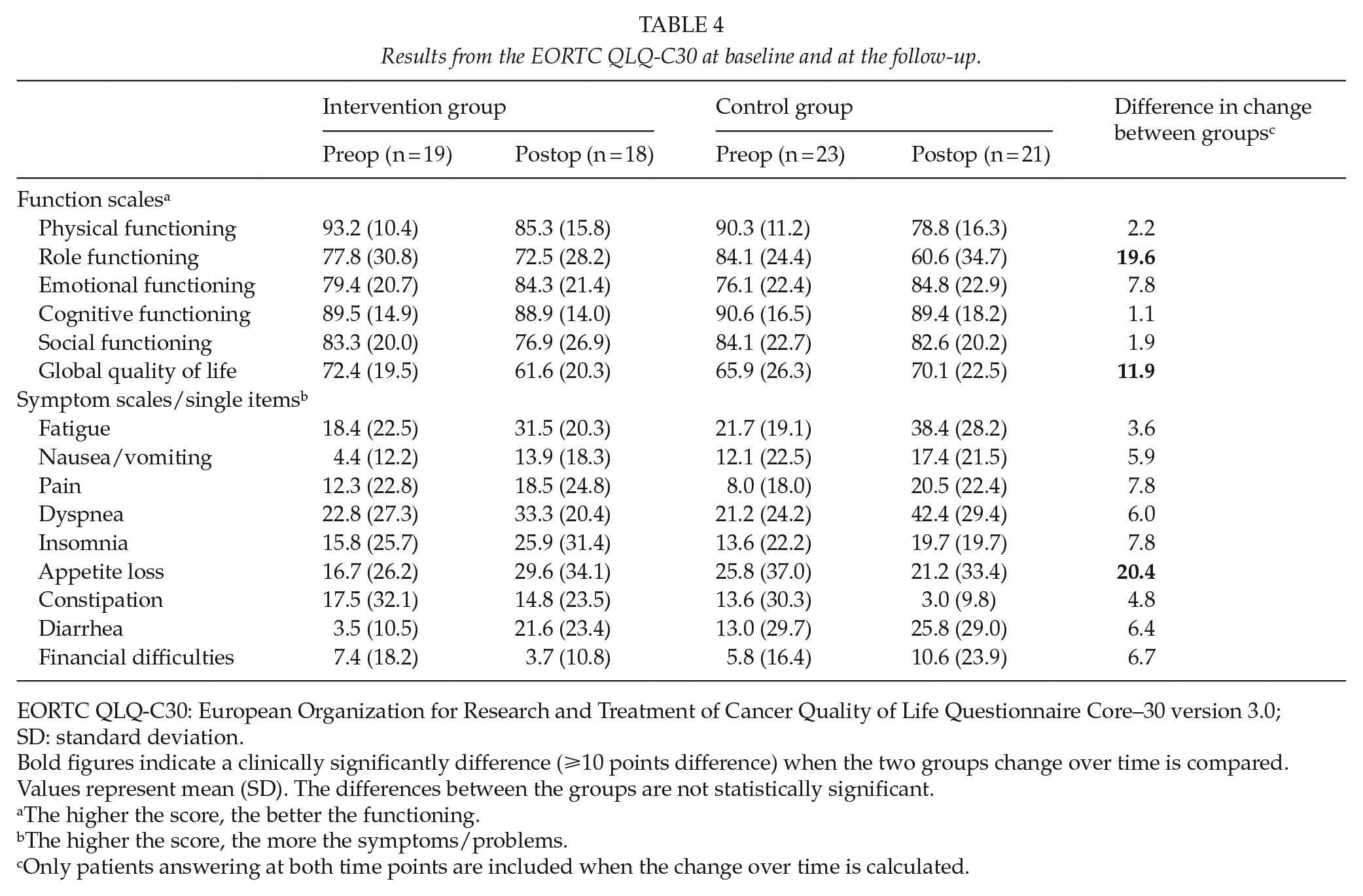
EORTC QLQ-C30: European Organization for Research and Treatment of Cancer Quality of Life Questionnaire Core–30 version 3.0; SD: standard deviation.
Bold figures indicate a clinically significantly difference (⩾10 points difference) when the two groups change over time is compared.
Values represent mean (SD). The differences between the groups are not statistically significant.
The higher the score, the better the functioning.
The higher the score, the more the symptoms/problems.
Only patients answering at both time points are included when the change over time is calculated.
Discussion
Patients undergoing Ivor-Lewis esophagus surgery have an increased risk of developing persistent pain, deceased physical and respiratory function, and decreased range of motion postoperatively (10–13). Since the number of survivors is increasing due to improved surgical techniques, down-staging treatments, modern intensive care, and effective epidural anesthesia, there is an urgent need for further improvement of physical function and quality of life for cured patients. Although some rehabilitation treatments exist, few patients are offered rehabilitation and there is little scientific evidence regarding its possible value in this patient group. This trial is the first to evaluate a three-month-long training intervention aimed at improving thoracic motion, lung function, and physical activity for this category of patients.
The treatment program evaluated in this trial included exercises to facilitate the range of motion in the shoulders and thorax, and the results indicate that the training reduces loss of function caused by surgery. Concerning physical function, the patients in the control group scored significantly higher than the intervention group. This supports the argument that training is beneficial and helps to maintain function after surgery. There were no significant differences between the groups in respiratory function or pain scores, although there were some trends toward a positive effect of the intervention; however, it needs to be emphasized that this study was based on a small number of patients. A sample size of 100 patients per group would have been needed to reach statistical significance for these variables. Therefore, we cannot exclude the possibility that there may be beneficial effects, although less pronounced, on respiratory function and/or pain as well.
The intervention was designed to restore range of motion and physical function and was based on the same model as is used in rehabilitation after spine surgery (27). Earlier follow-ups of the patient group in this study have indicated a reduction in overall strength, as measured by activity scores and functional tests; it therefore seemed essential to evaluate a rehabilitation program that aims to restore function and improve activity. Differences between the groups were found in the sum score in DRI and range of motion in the right shoulder and left lateral flexion, but, since the group of patients is heterogenic (age, sex, preoperative functioning level, etc.), a more individualized intervention may have been preferable. In addition, the intervention evaluated in this trial was started at discharge. It is possible that an earlier start with preoperative training could have increased the effects, and future trials are thus needed.
There is always a risk that some patients develop a passive lifestyle, including avoidance of activities, which can negatively affect them in the already challenged situation caused by their illness. The training protocol aimed to restore physical function and thereby make it possible to achieve a higher degree of physical activity since it is well-established that activity has an impact on wellbeing. It was not possible to show this in our study, probably due to a fragile patient group. However, an advantage of the training protocol evaluated in this study is that it offers a safe opportunity for patients to improve function and thereby an opportunity to have a more active lifestyle. The negative side effects of the program can be a higher degree of fatigue or muscle sourness in addition to the training. This has not been noted in this study. The patients managed to perform the active training protocol without any side effects, and it is important to know that, after this kind of surgery, patients can perform active exercises and start to be active early in the postoperative period.
As expected, a poorer quality of life was found at the follow-up in both groups compared to the time of diagnosis. The patients reported a decline in most functions as well as increases in most problems as compared to preoperative scores. Both groups scored clinically significantly worse on average for fatigue, dyspnea, and diarrhea. Physical functioning had deteriorated in both groups compared to preoperative measures, but according to the results of the analysis within each group, it was only in the control group that the loss of functioning was significant. Few differences were found when the two groups’ quality of life scores were compared at diagnosis and at the follow-up and, owing to the small number of participants, no firm conclusions can therefore be drawn in this regard. Furthermore, these patients have diagnosis of cancer with a small chance of cure and for which the treatment involves extensive surgery with a major impact on wellbeing during a long period of recovery. It may well be that quality of life scores retrieved during the early postoperative period more reflect concerns for survival and/or effects of the physical and mental trauma caused by the treatment, as such, than any possible beneficial effects of a training program.
The risk of malnutrition is high in this group of patients, in whom a large part of the stomach is resected and the vagus nerve is cut. In most cases, patients need nutritional support and dietician assistance for several weeks after the procedure. The main weight loss occurs during the first three months after surgery due to difficulties eating and reduced physical activity resulting in a loss of muscle mass (28, 29). The groups’ body weight or body mass index (BMI) did not differ preoperatively in this study (Table 1). While postoperative weight loss and eventual development of malnutrition were beyond the scope of this study, this may be a confounding factor during the early postoperative period (the first three months after surgery) on quality of life scores. Although all patients were routinely offered nutritional support and assessed by a dietician when needed, no additional nutritional screening or supplementation was offered to the intervention group other than the standard clinical routine. Only one patient in the control group and none in the intervention group had to be excluded owing to poor nutritional status during the study period, but we cannot completely rule out that increased energy intake to compensate for the additional physical exercise could have been beneficial. It is possible that some of the effects of the three-month-long training intervention may become evident at a later stage in the recovery period, when most patients have improved their nutritional intake. It is therefore possible that physical functioning may be further improved over the longer term (>6–24 months).
The trial has some limitations. One is the low sample size and the heterogeneous group of patients, which makes it hard to draw definitive conclusions on effects on respiratory function. Even though the inclusion period was almost five years and we added another study center the last year so that 101 patients were eligible during the study period, 64 patients were randomized 1 week after surgery and only 43 participated in the entire three-month follow-up. It is well known that this type of major intervention carries a high degree of perioperative morbidity (1–3), which was evident in this study as well. The long inclusion period was necessary for logistic reasons and because several patients did not fulfill the inclusion criteria or fulfilled the exclusion criteria. However, there were no major changes in surgery, anesthesia, or care during the study period. In addition, in an attempt to make the groups less heterogenic, patients who needed prolonged care at the intensive care unit or who needed additional surgery during the first postoperative week were excluded from the study. However, patients with complex perioperative courses who spend longer times in hospital and deal with complications during their recovery might actually have greater potential to benefit from postoperative rehabilitation. Another limitation is that even if the groups were comparable preoperatively, perioperative factors could have influenced the results. In addition, the training was unsupervised and we did not ask the patients to fill in a training diary during the intervention, and it is therefore difficult to know the extent to which the patients actually followed the training program. It is possible that the patients may not have exercised enough to make a larger difference between the groups. Supervision of training on some occasions during the study period could have increased compliance. Given the extent of the procedure, three months can be viewed as quite a short period of recovery, and it is possible that beneficial effects of treatment can appear later.
In conclusion, the results of the three-month intervention indicate that a specific training program can positively improve physical function and range of motion to preoperative values. The intervention was well tolerated, and no side effects were registered.
Footnotes
Appendix 1
Acknowledgements
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The regional Ethics Committee for the region of Västra Götaland, Sweden, approved the study protocol, and Registration number was 078-05.
Funding
The trial was undertaken with financial support in the form of research grants from the Research and Development Council for Gothenburg and Southern Bohuslän.
Informed Consent
The patients participated after written and oral information and written consent.
