Abstract
Aims:
We compared laparoscopic and robotic gastrectomies with open gastrectomies and with each other that were held for gastric cancer in Europe.
Methods:
We searched for studies conducted in Europe and published up to 20 February 2015 in the PubMed database that compared laparoscopic or robotic with open gastrectomies for gastric cancer and with each other.
Results:
We found 18 original studies (laparoscopic vs open: 13; robotic vs open: 3; laparoscopic vs robotic: 2). Of these, 17 were non-randomized trials and only 1 was a randomized controlled trial. Only four studies had more than 50 patients in each arm. No significant differences were detected between minimally invasive and open approaches regarding the number of retrieved lymph nodes, anastomotic leakage, duodenal stump leakage, anastomotic stenosis, postoperative bleeding, reoperation rates, and intraoperative/postoperative mortality. Nevertheless, laparoscopic procedures provided higher overall morbidity rates when compared with open ones, but robotic approaches did not differ from open ones. On the contrary, blood loss was less and hospital stay was shorter in minimally invasive than in open approaches. However, the results were controversial concerning the duration of operations when comparing minimally invasive with open gastrectomies. Additionally, laparoscopic and robotic procedures provided equivalent results regarding resection margins, duodenal stump leakage, postoperative bleeding, intraoperative/postoperative mortality, and length of hospital stay. On the contrary, robotic operations had less blood loss, but lasted longer than laparoscopic ones. Finally, there were relatively low conversion rates in laparoscopic (0%–6.7%) and robotic gastrectomies (0%–5.6%) in most studies.
Conclusion:
Laparoscopic and robotic gastrectomies may be considered alternative approaches to open gastrectomies for treating gastric cancer. Minimally invasive operations are characterized by less blood loss and shorter hospital stay than open ones. In addition, robotic procedures have less blood loss, but last longer than laparoscopic ones.
Introduction
Various surgical operations are conducted through minimally invasive approaches, either laparoscopic or robotic (1). In many fields of surgery, minimally invasive procedures are feasible and safe alternative options to the open procedures (1). They are characterized by earlier mobilization, decreased postoperative pain, faster recovery, and shorter hospital stay, along with equivalent oncological results (1–5).
The first laparoscopic gastrectomy was a laparoscopy-assisted distal gastrectomy performed by Kitano et al. (6) in 1994 in Japan. Since then, many attempts have been made in order to perform distal, subtotal, and total gastrectomies laparoscopically and more recently robotically (1–5, 7–11). Many studies have compared laparoscopic and robotic gastrectomies with open ones in terms of feasibility, safety, oncological results, and postoperative outcomes (2–5, 7–11). The majority of studies have been conducted in Asia (1, 3, 5, 7, 10, 11). The fact that most studies come from Asian countries can be explained by the higher incidence of gastric cancer in Asia and especially in East Asia (12–14). This has led to an increased number of both open and minimally invasive gastrectomies, and therefore to an increased experience in gastric surgery in East Asian countries (5, 15, 16). Moreover, many surgeons in East Asian countries favor more extensive lymphadenectomies, which involve para-aortic and other lymph node stations, than the standard D2 lymphadenectomy (16). In addition, screening programs involving endoscopy as a method of pre-symptomatic detection of gastric cancer have resulted in the increased detection and treatment of early gastric cancer in Japan and South Korea (13, 16).
On the contrary, gastric cancer is less frequent in European countries (14). This makes operations for gastric cancer, both open and minimally invasive, rarer and European surgeons less experienced in performing gastrectomies, and even more so minimally invasive ones. Nevertheless, several studies on minimally invasive gastrectomy for gastric cancer have been conducted in Europe. Some of them have compared laparoscopic or robotic operations with open ones. Our aim was to provide an overview of the European experience in performing minimally invasive gastrectomies for gastric cancer in comparison with open gastrectomies for the same reason.
Methods
Search Strategy
We searched for articles published up to 20 February 2015 in the PubMed database. We used the following key words for this search: “laparoscopic,” “robotic,” and “minimally invasive” in combination with “gastrectomy” and “gastric cancer” or “stomach cancer.”
Inclusion and Exclusion Criteria
In our review, we included only articles coming from European countries, written in English, and referred to patients with gastric cancer that compared minimally invasive (laparoscopic or robotic) gastrectomy with open gastrectomy or laparoscopic with robotic gastrectomy. We excluded reviews, case reports, animal studies, studies referring to patients with gastrointestinal stromal tumors, original articles about patients who had undergone one type of gastrectomy only (minimally invasive or open), articles written in languages other than English, and articles coming from countries outside Europe.
Review and Analysis
We extracted data regarding the type of minimally invasive approach (laparoscopic or robotic), the type of operation (hand-assisted or totally minimally invasive), patients’ characteristics (number, gender, age, body mass index), tumor characteristics (location, stage, and diameter), operative characteristics (type of anastomosis, number of retrieved lymph nodes, surgical margins, duration of operation, amount of blood loss, and conversion to open), complications (anastomotic leakage, duodenal stump leakage, stenosis, bleeding, reoperation, and intraoperative and postoperative mortality), the length of hospital stay, and the progression-free and the overall survival. In cases when an article included additional types of gastric pathology apart from gastric cancer, we considered data that had to do with patients with gastric cancer only.
Statistical Analysis
A meta-analysis was performed according to the recommendations specified in the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, using Review Manager Version 5.3 (RevMan 5.3). Survival analysis was carried out using the hazard ratio (HR) for the evaluation of overall mortality, and the risk ratio (RR) was used for the assessment of dichotomous variables, for example, postoperative morbidity and mortality as the summary statistics. Random-effects model was used in all cases due to the expected heterogeneity between the studies and, thus, heterogeneity of the effect across different studies which cannot be excluded a priori. The inverse of variance method was selected to combine results and calculate the heterogeneity among the different groups. Statistical heterogeneity was investigated using Higgins’ I2 statistic (17, 18). The pooled HRs and RRs are reported with 95% confidence intervals (CIs). Assessment for potential publication bias was performed through drawing of funnel plots which were inspected for asymmetry (19).
Results
Search Results
Our search yielded 123 articles coming from European countries. We excluded 105 articles: 18 reviews, 16 case reports, 21 articles referring to patients with gastrointestinal stromal tumors, 19 articles written in languages other than English, and 31 original articles with patients having undergone only one type of gastrectomy (minimally invasive or open). This allowed us to take into account 18 original articles about patients with gastric cancer that compared minimally invasive (laparoscopic or robotic) gastrectomy with open gastrectomy or laparoscopic with robotic gastrectomy. Of the 18 studies, 13 compared laparoscopic with open gastrectomy (20 –32), 3 compared robotic with open gastrectomy (33–35), and 2 compared laparoscopic with robotic gastrectomy (36, 37). Concerning studies with laparoscopic gastrectomies, 6 included totally laparoscopic operations (22, 25, 26, 28, 29, 31), 3 included hand-assisted laparoscopic operations (27, 30, 32), and 6 included both types (20, 21, 23, 24, 36, 37). Concerning studies with robotic gastrectomies, one referred to hand-assisted robotic procedures (33), whereas four referred to both hand-assisted and totally robotic procedures (34–37). A D2 lymphadenectomy was performed in 13 studies (21–23, 25–29, 32–36), a D1 lymphadenectomy was performed in 1 study (31), and either a D1 or a D2 lymphadenectomy was conducted in 3 studies (24, 30, 37). The exact extent of lymphadenectomy was not mentioned in one article (20). Finally, of the 18 studies that were included, 17 were non-randomized trials (20–31, 33–37) and only 1 was randomized controlled trial (32). However, none of the 18 studies included data for all the parameters tested. Of all, 14 articles included data for almost all the variables (21, 22, 24–32, 34, 36, 37) and 4 articles contained information for only a number of the variables (20, 23, 33, 35). Fig 1 depicts the study flowchart, Table 1 shows the types of operations and characteristics of studies, and Table 2 shows patients’ characteristics.
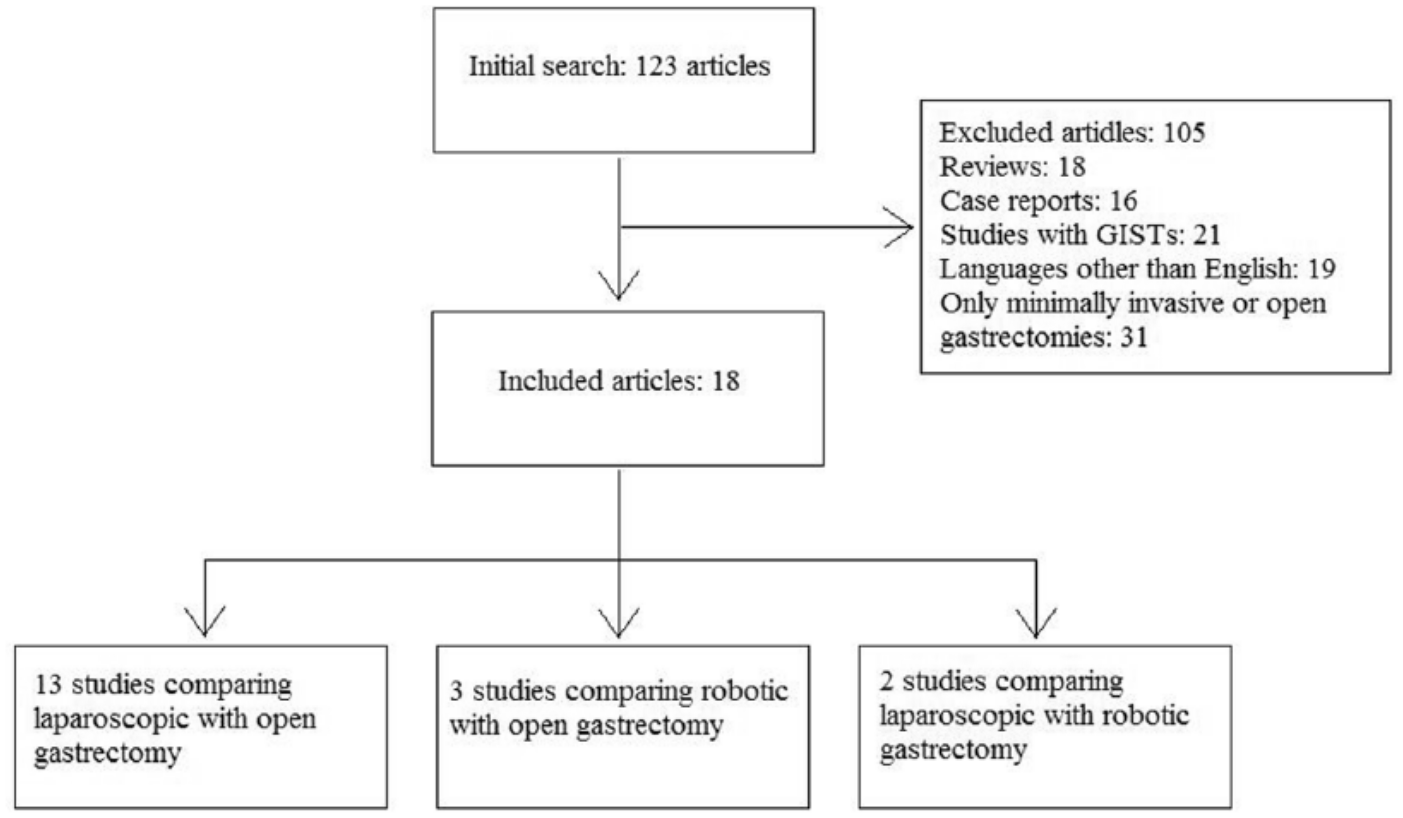
Study flowchart.
Study characteristics.
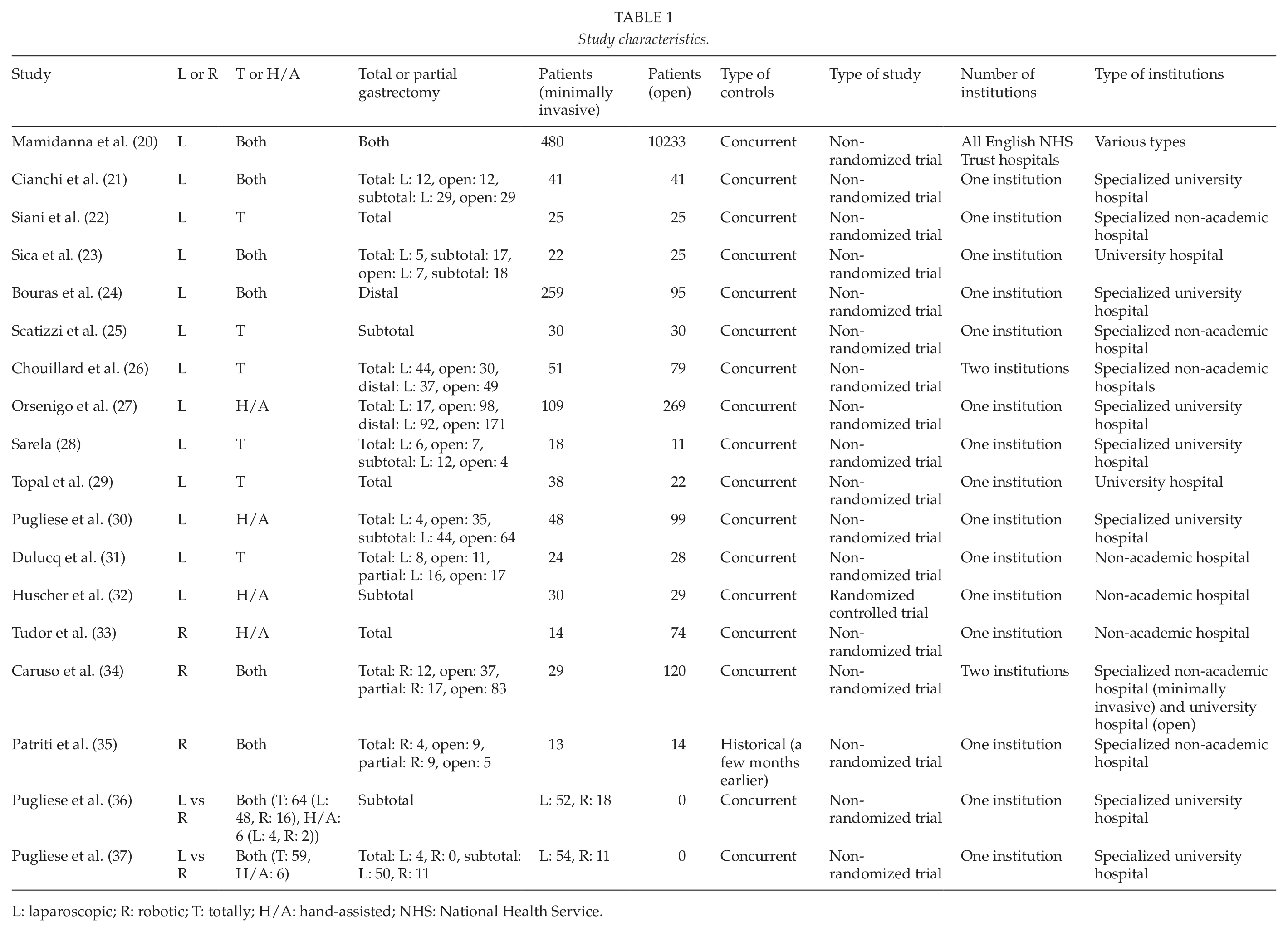
L: laparoscopic; R: robotic; T: totally; H/A: hand-assisted; NHS: National Health Service.
Patients’ characteristics.
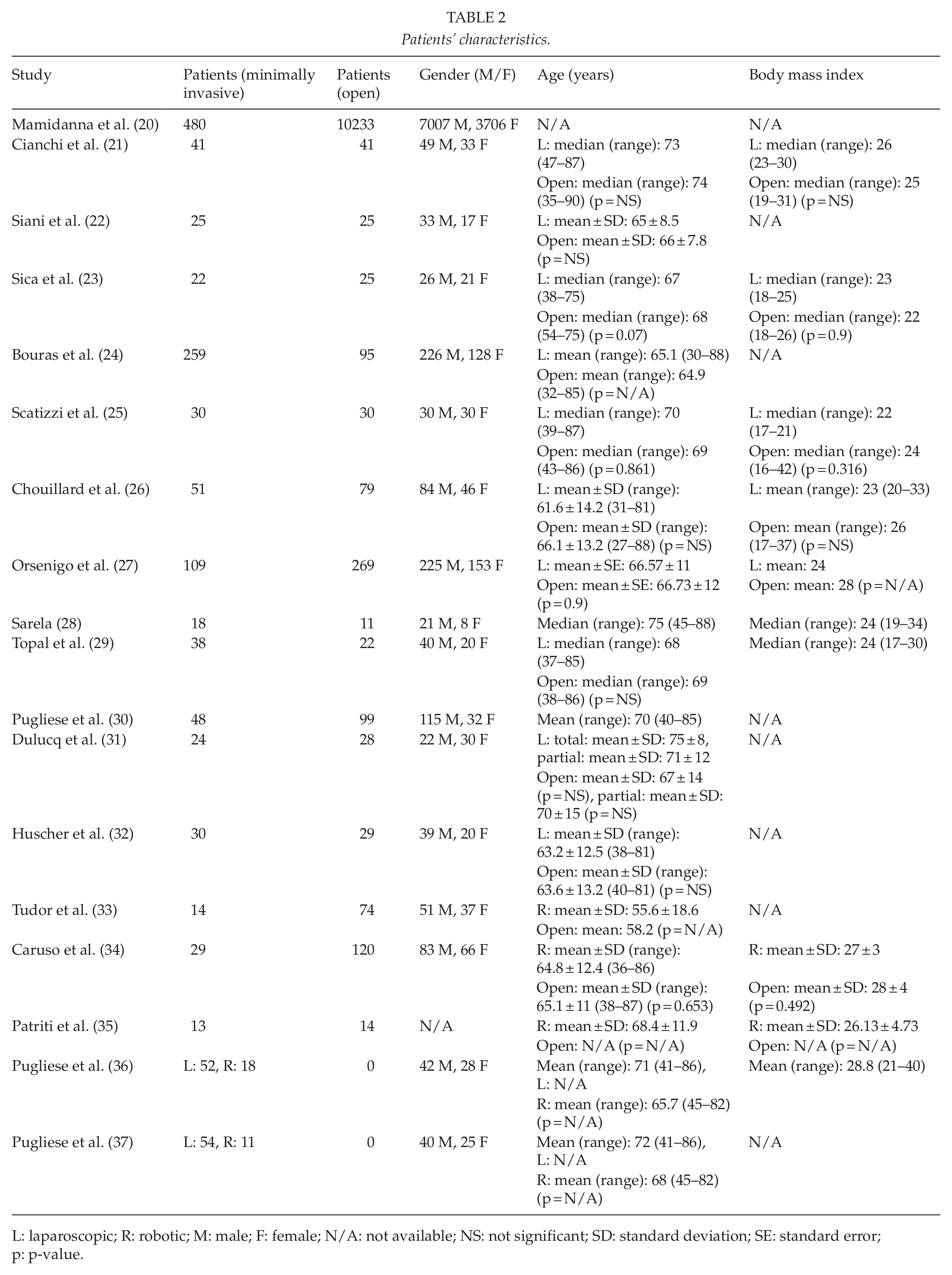
L: laparoscopic; R: robotic; M: male; F: female; N/A: not available; NS: not significant; SD: standard deviation; SE: standard error;p: p-value.
Tumor Characteristics
There were no major differences between minimally invasive and open gastrectomies regarding tumor characteristics. The distribution of tumor location among the upper, middle, and lower parts of the stomach was not significantly different in laparoscopic operations from open ones (21–23, 25, 29, 32). In studies comparing robotic and open procedures, one reported a greater proportion of tumors in the middle and the distal parts of the stomach in robotic gastrectomies and a greater proportion of tumors in the upper and the middle parts of the stomach in open gastrectomies (34), whereas another reported no significant difference between the two approaches (33). Tumor stages were evenly distributed between minimally invasive and open procedures (21 –23, 25, 26, 28, 29, 31, 32, 34). In one study, though, stages tended to be more advanced in open than in laparoscopic gastrectomies (27). Finally, tumor size did not differ significantly in laparoscopic operations from open ones (21, 25, 29, 31), with the exception of one study in which tumors treated by open approach were larger than those treated by laparoscopic approach (27). Data about the tumor characteristics are listed in Table 3.
Tumor characteristics (location, stage, and diameter).
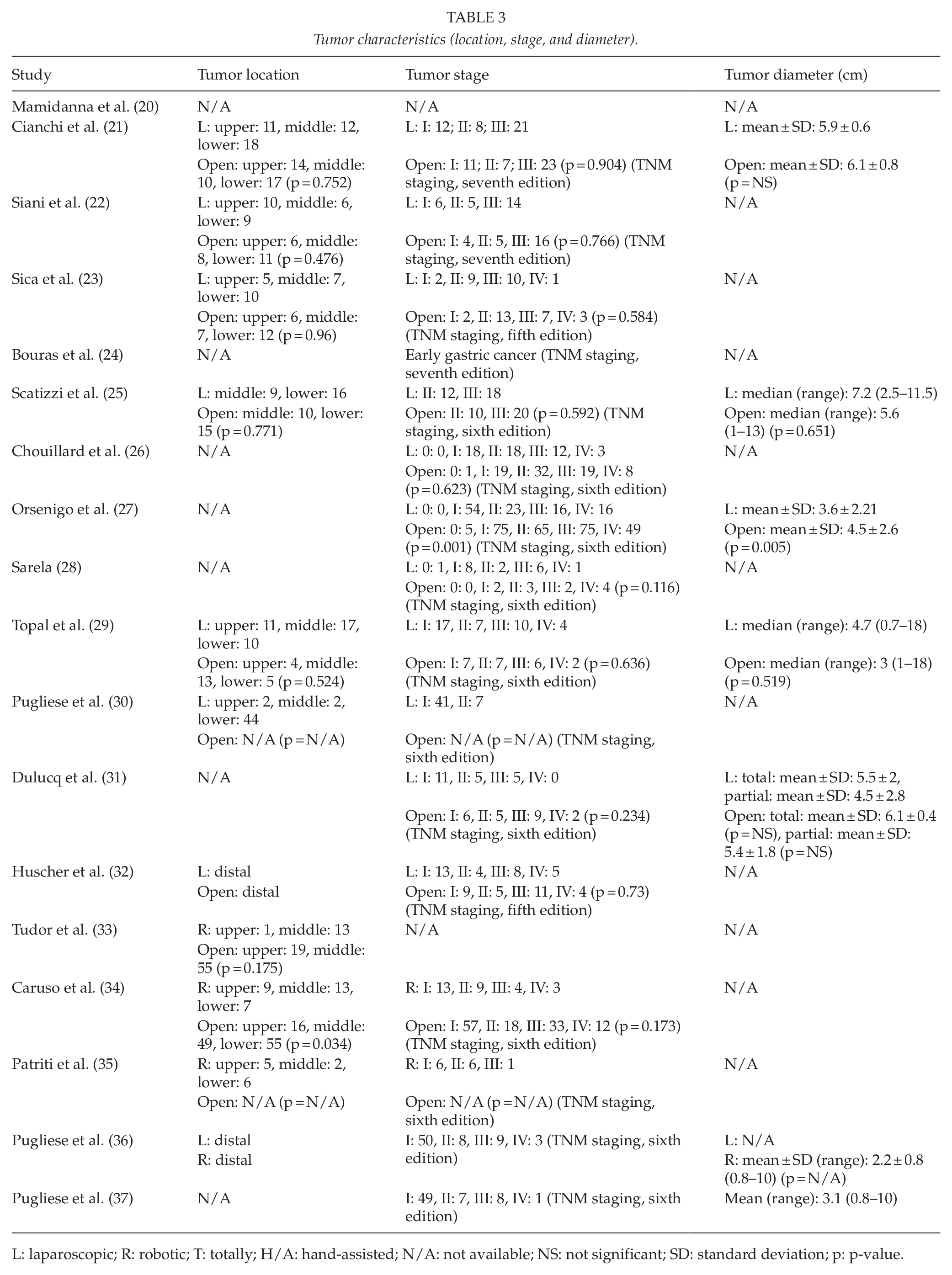
L: laparoscopic; R: robotic; T: totally; H/A: hand-assisted; N/A: not available; NS: not significant; SD: standard deviation; p: p-value.
Operative Characteristics
First, the most frequent type of anastomosis was the Roux-en-Y anastomosis in both minimally invasive and open approaches (21–37). The Roux-en-Y type of anastomosis was used in all total gastrectomies (21–23, 26–31, 33–37) and in most subtotal and distal gastrectomies (23, 25–28, 30, 32, 34–37). The Billroth II type of anastomosis was used in some subtotal and distal gastrectomies (21, 23, 24, 31, 32). No differences were detected between minimally invasive and open procedures concerning the type of anastomosis (21–23, 25–35).
The number of retrieved lymph nodes and the rates of negative resection margins were studied due to their oncological significance. The number of retrieved lymph nodes did not differ significantly in laparoscopic gastrectomies from open ones (21–26, 28, 31, 32). The same was found when comparing only total (22) or only partial operations (24, 25, 32) and when comparing only hand-assisted (32) or only totally laparoscopic operations (22, 25, 26, 28, 31). Sole exception to this was a study about hand-assisted laparoscopic gastrectomies, according to which there was a small advantage of laparoscopic over open procedures (27). The results about robotic operations and the number of retrieved lymph nodes were controversial, as one study reported a small advantage of open over robotic approaches (34), while a second one did not indicate any significant difference between them (35).
When we performed meta-analysis, the number of retrieved lymph nodes did not differ between minimally invasive surgeries and open gastrectomy (21–24, 26, 27, 30–32, 34–37). The observed mean difference was −1.75 (95% CI: −3.73 to 0.24, p = 0.08). However, the evaluated studies showed significant heterogeneity (I2 = 73%, p < 0.00001). The exclusion of the studies of Pugliese et al., which were published in 2009 (37) and 2010 (36) and compared the robotic with the laparoscopic approach, did not alter the observed results, while the heterogeneity remained significant.
No actual difference was found between laparoscopic and open gastrectomies about the rates of negative resection margins (21, 24–31), regardless of the type of gastrectomy (total or partial) (24, 25, 28, 29) or the type of procedure (hand-assisted or totally laparoscopic) (25–31). The same goes for the comparison between robotic gastrectomies and open (34, 35) or laparoscopic ones (36). In most studies, the rates of negative resection margins ranged between 94.5% and 100% for laparoscopic (21, 24–27, 29–31) and between 88.8% and 100% for open operations (21, 24–27, 29–31, 34, 35). The rates for robotic procedures were 100% in three studies (34–36).
Our meta-analysis showed increased rates of negative resection margins in minimally invasive gastrectomies in comparison with open gastrectomies with an odds ratio (OR) of 2.19 (95% CI: 1.16 to 4.13, p = 0.02; I2 = 0%, p = 1.00) (Fig. 2) (21, 26–29, 31, 34, 36). However, when we excluded the study by Orsenigo et al. (27), which had the greatest number of patients among the eight included studies for this particular analysis, the results were not statistically significant (OR = 2.23; 95% CI: 0.92 to 5.40, p = 0.08; I2 = 0%, p = 1.00). The heterogeneity of studies was not significant in both analyses, but the funnel plots depicted a mild asymmetry.
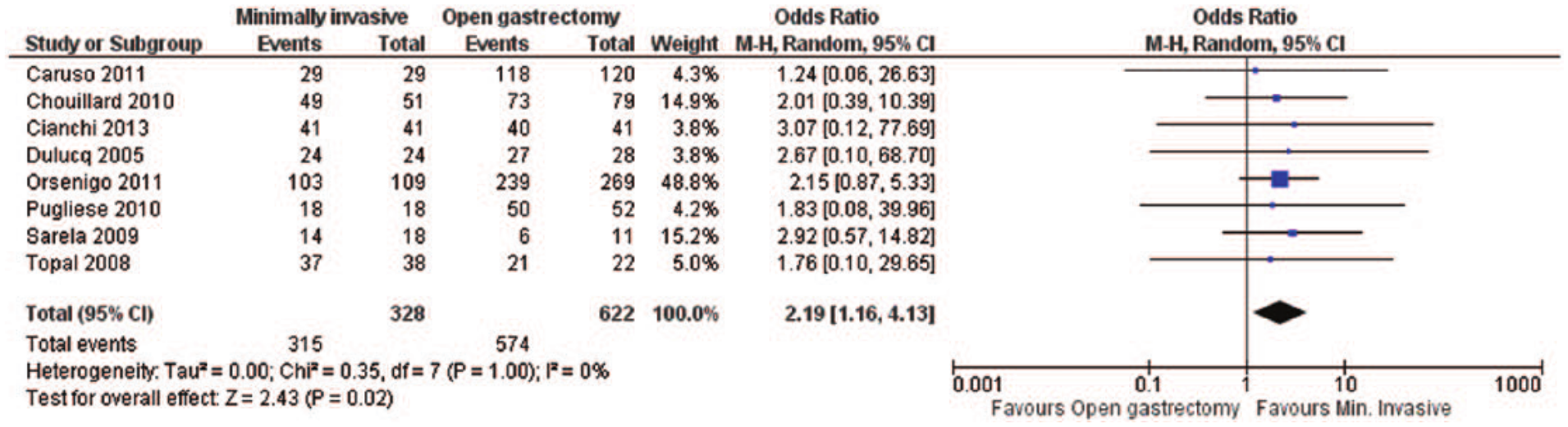
Negative resection margins: forest plot.
The results about the duration of operations were controversial. A number of studies supported that laparoscopic gastrectomies last longer than open ones (21, 24, 25, 27 –29), irrespective of the type of gastrectomy (total or partial) (21, 24, 25, 29) or the type of procedure (hand-assisted or totally laparoscopic) (25, 27 –29). On the contrary, other studies suggested that the duration of gastrectomy does not significantly differ in laparoscopic procedures from open ones (22, 26, 30–32), and the same is found when comparing total or partial gastrectomies alone (22, 31, 32) and when comparing hand-assisted or totally laparoscopic operations alone (22, 26, 30 –32). Regarding robotic approaches, one study reported that robotic gastrectomies last longer than open ones (34), whereas another study reported that there is no actual difference between them (33). Moreover, in one study comparing laparoscopic with robotic gastrectomies, robotic operations seemed to last longer than laparoscopic ones (36).
In our meta-analysis, the duration of the operation was longer in cases of minimally invasive surgery than in open gastrectomies, with a mean difference of 46.38 min (95% CI: 19.6 to 73.39, p = 0.0008). However, the evaluated studies (21, 22, 24–27, 30–34) had significant heterogeneity (I2 = 98%, p < 0.00001).
Minimally invasive gastrectomies seemed to have an advantage over the open ones in regard to blood loss. Most studies supported that there is less blood loss in minimally invasive (laparoscopic or robotic) gastrectomies than in open ones (21, 22, 24, 27, 29, 30, 32 –34) regardless of the type of gastrectomy (total or partial) (22, 24, 29, 32, 33) or the type of procedure (hand-assisted or totally minimally invasive) (22, 27, 29, 30, 32, 33). However, one study supported that this difference is not big enough to reach statistical significance (26). According to another survey, only in laparoscopic total gastrectomies there is a significantly less blood loss than in open ones, but this was not the case with laparoscopic partial gastrectomies, which did not seem to differ from open ones (31). Finally, blood loss was even less in robotic operations when compared with laparoscopic operations (36).
Data about the amount of blood loss were provided by 11 studies (21, 22, 24, 26, 27, 29 –34), which were characterized by significant heterogeneity in our meta-analysis. Minimally invasive operations were characterized by significantly lower amount of blood loss compared with open gastrectomies, with a mean difference of −176.78 mL (95% CI: −206.74 to −146.81, p < 0.00001; I2 = 82%, p < 0.00001).
The conversion rates of minimally invasive to open gastrectomy were relatively low. The conversion rates of laparoscopic procedures ranged between 0% and 6.7% in most studies (21 –23, 25, 26, 30, 31, 36, 37). However, two studies reported conversion rates higher than 10%, reaching 15.6% (27) and 27.7% (28), respectively. In studies with robotic procedures, the conversion rates ranged between 0% and 5.6% in most studies (34 –36). However, one study reported a conversion rate of 18.2% (37). In addition, no significant differences were detected when comparing the conversion rates of laparoscopic gastrectomies with those of robotic ones (36, 37). Data about the operative characteristics can be seen in Tables 4 and 5.
Operative characteristics (anastomosis, lymphadenectomy, retrieved lymph nodes, and surgical margins).
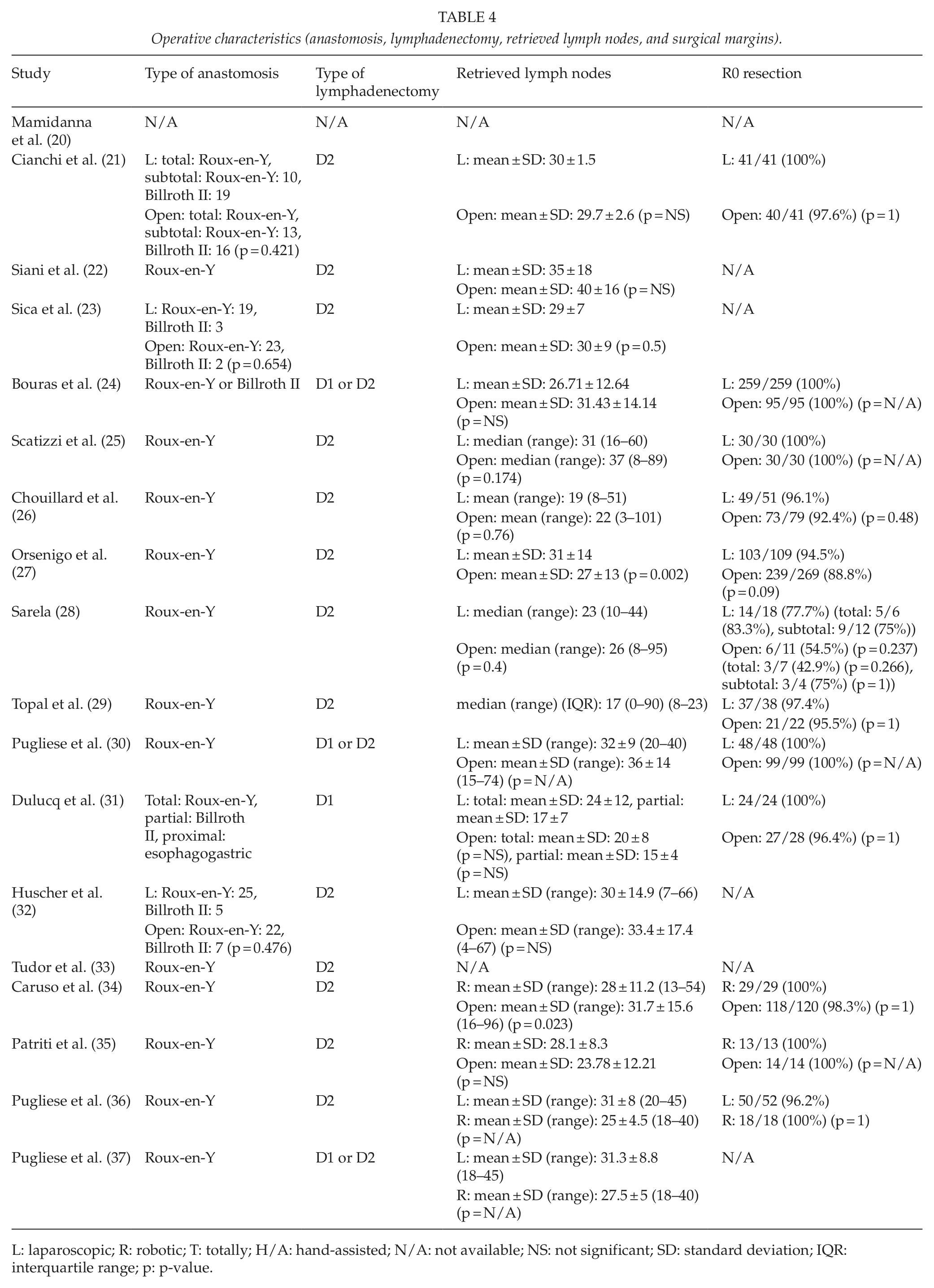
L: laparoscopic; R: robotic; T: totally; H/A: hand-assisted; N/A: not available; NS: not significant; SD: standard deviation; IQR: interquartile range; p: p-value.
Operative characteristics (operative time, blood loss, and conversion to open).
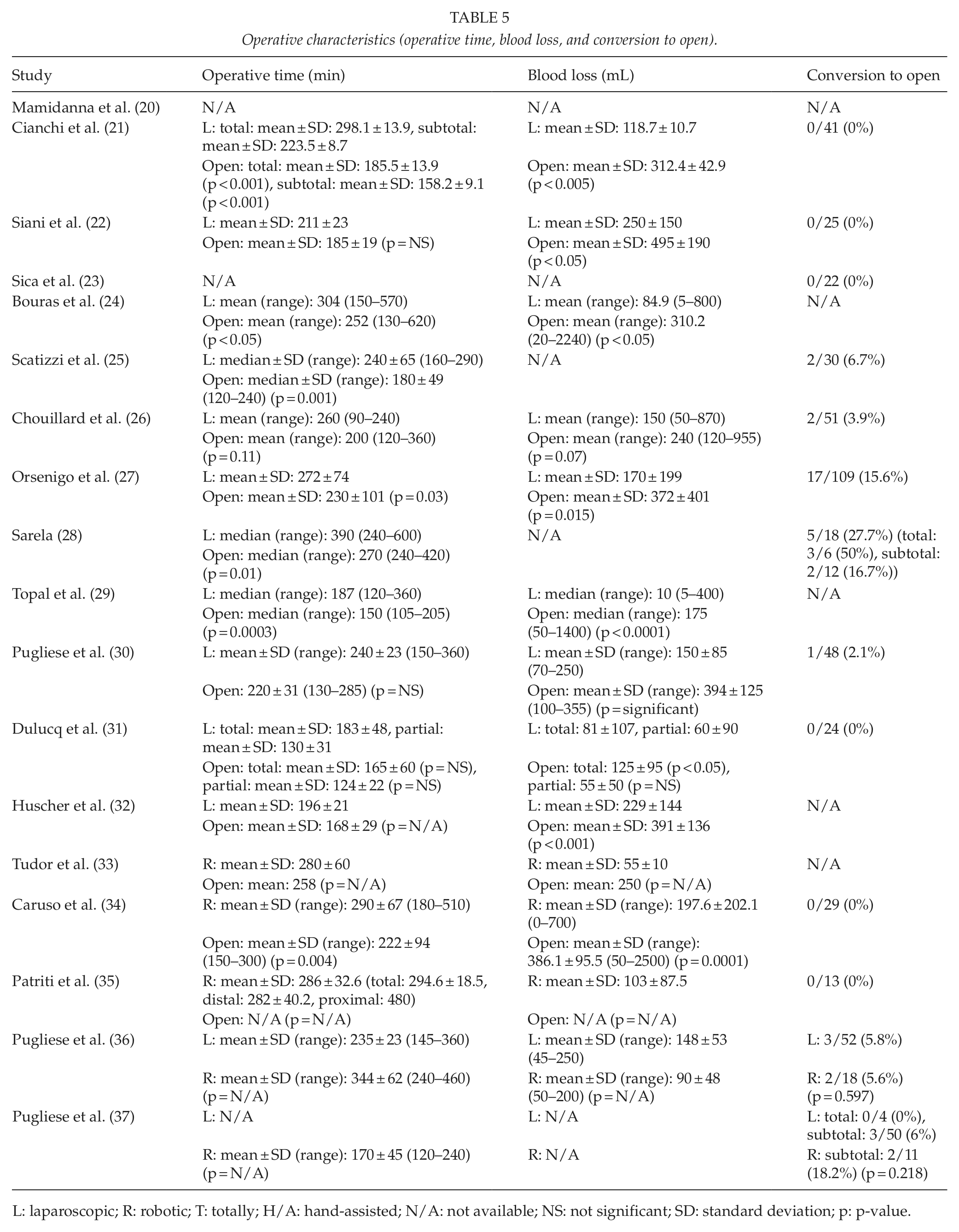
L: laparoscopic; R: robotic; T: totally; H/A: hand-assisted; N/A: not available; NS: not significant; SD: standard deviation; p: p-value.
Complications
No great differences were reported regarding the frequency of anastomotic leakage, duodenal stump leakage, anastomotic stenosis, and postoperative bleeding between minimally invasive and open gastrectomies (21, 22, 24, 25, 28 –34). The rates of anastomotic leakage were 0%–5.6% for laparoscopic procedures (21, 22, 24, 25, 28–30), 0%–3.4% for robotic procedures (33, 34), and 0%–5.8% for open procedures (21, 22, 24, 25, 28–30, 33, 34). Duodenal stump leakage was detected in 0%–18.3% of laparoscopic operations (21, 25, 27, 28, 30–32, 36, 37), 0%–7.7% of robotic operations (34–37), and 0%–9.1% of open operations (21, 25, 27, 28, 30–32, 34). The rates of anastomotic stenosis were 2.4%–5.6% for laparoscopic procedures (21, 28), 0% for robotic procedures (33), and 0%–1.4% for open procedures (21, 28, 33). Postoperative bleeding was observed in 0%–5.6% of laparoscopic operations (21, 25, 28, 30, 37), 0%–7.7% of robotic operations (34, 35, 37), and 0%–4.2% of open operations (21, 25, 28, 30, 34). No significant differences were detected between laparoscopic and open gastrectomies (21, 22, 24, 25, 28–32) or between robotic and open gastrectomies (33, 34), regardless of the type of gastrectomy (total or partial) (22, 24, 25, 28, 29, 32, 33) or the type of procedure (hand-assisted or totally laparoscopic) (22, 25, 28–33). The only exception was a study which reported a significantly higher rate of duodenal stump leakage in the laparoscopic approach (18.3%) than in the open one (5.2%) (27). Furthermore, no significant differences were found when comparing laparoscopic with robotic gastrectomies in terms of duodenal stump leakage (36, 37) and postoperative bleeding (37).
Postoperative morbidity was assessed by the number of complications evident after every type of surgery in our meta-analysis. Comparing the overall morbidity after minimally invasive surgery with that after open gastrectomy, we observed a significant overall RR = 1.96 (95% CI: 1.23 to 3.14, p = 0.005; I2 = 13%, p = 0.31) (Fig. 3), while the funnel plot revealed only mild asymmetry (21, 22, 24, 25, 27–37). In other words, the minimally invasive approach leads to an almost double risk for postoperative complications in comparison with the open approach. Similar results were observed after we excluded the studies by Pugliese et al. in 2009 (37) and 2010 (36), which compared laparoscopic with robotic gastrectomy (RR = 1.90; 95% CI: 1.27 to 2.85, p = 0.002). On the other hand, after excluding the largest study by Orsenigo et al. (27), the comparison of morbidity between minimally invasive and open gastrectomy became non-significant (RR = 1.51; 95% CI: 0.91 to 2.49, p = 0.11; I2 = 0%, p = 0.52). Subsequently, we performed an independent evaluation of morbidity, including all studies describing a laparoscopic approach (21, 22, 24, 25, 27–29, 31, 32, 36) versus an open gastrectomy (RR = 2.20; 95% CI: 1.40 to 3.46, p = 0.0006; I2 = 0%, p = 0.59). This demonstrated again the increased risk of minimally invasive approach. We proceeded by excluding the study by Orsenigo et al. (27), which had received a weight of 48.8%, and observed that the RR of morbidity became 1.41 and was not significant (95% CI: 0.75 to 2.64, p = 0.29; I2 = 0%, p = 0.90), when comparing laparoscopic with open gastrectomy. Finally, we compared studies that evaluated robotic surgery versus open gastrectomy with respect to morbidity (33–35) and observed no significant difference (RR = 1.06; 95% CI: 0.43 to 2.62, p = 0.90).
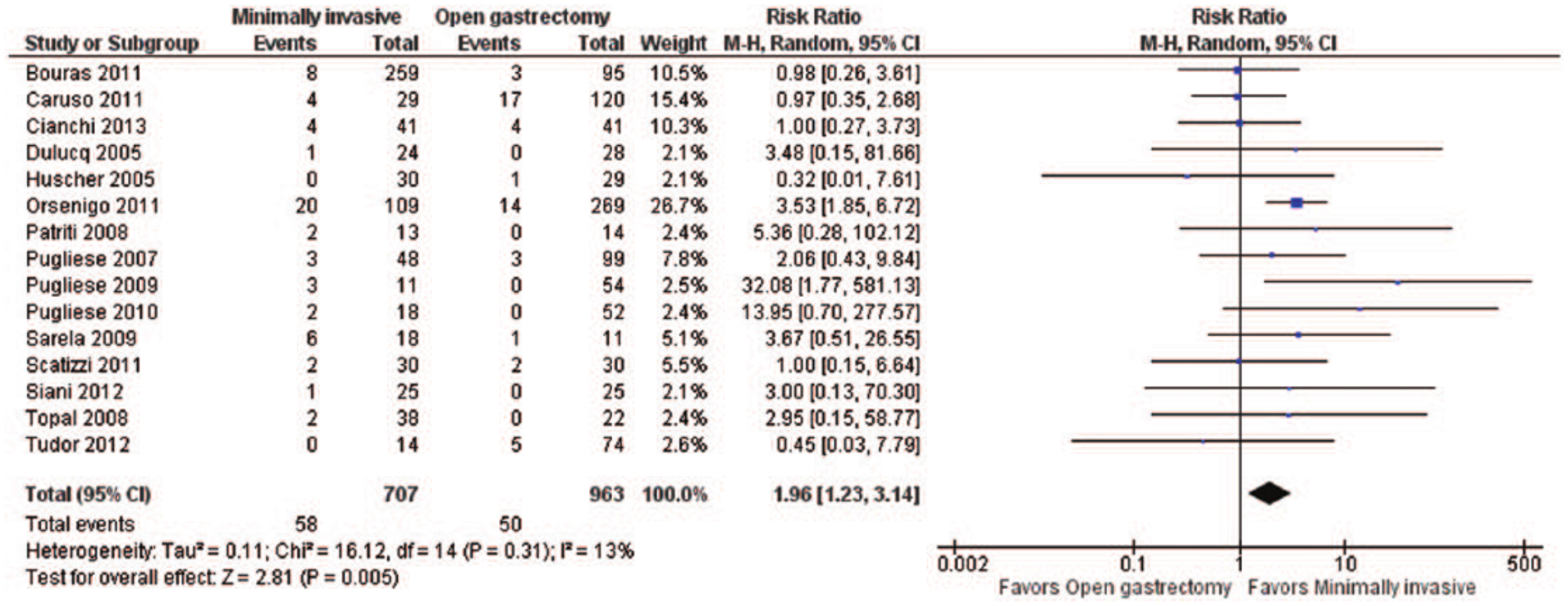
Postoperative morbidity: forest plot.
The reoperation rates were 3.3%–22.2% for laparoscopic procedures (20, 21, 25, 27, 28, 31), 0%–7.7% for robotic procedures (33–35), and 2.2%–18.2% for open procedures (20, 21, 25, 27, 28, 31, 33, 34). No significant differences were observed between laparoscopic and open gastrectomies (20, 21, 25, 28, 31) or between robotic and open gastrectomies (33, 34) in regard to reoperation rates, irrespective of the type of gastrectomy (total or partial) (20, 25, 28, 33) or the type of procedure (hand-assisted or totally laparoscopic) (25, 28, 31, 33). The only exception was one study reporting a significantly higher rate of reoperation rates in the laparoscopic approach (10.1%) than in the open one (2.2%) (27).
With respect to the risk of reoperation, no significant differences were observed between minimally invasive surgery and open gastrectomy when we performed meta-analysis (RR = 1.44; 95% CI: 0.79 to 2.61, p = 0.23; I2 = 31%, p = 0.19), according to the results of the assessed studies (20, 21, 25, 27, 28, 31, 34), which showed moderate heterogeneity (Fig. 4). The exclusion of the study with the largest sample size by Mamidanna et al. (20) did not affect the observed findings (RR = 1.60; 95% CI: 0.70 to 3.67, p = 0.27; I2 = 27%, p = 0.23).
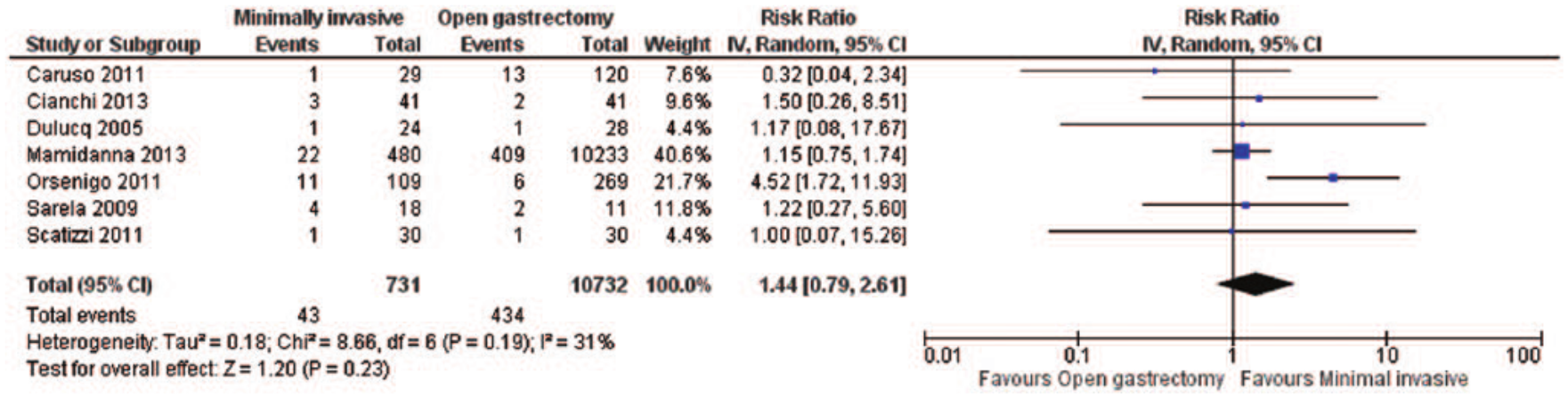
Reoperation risk: forest plot.
No intraoperative death was noted in any laparoscopic, robotic, or open gastrectomy (22, 25, 26, 30, 35–37). However, postoperative deaths occurred in 0%–5.6% of laparoscopic operations (20, 21, 23–30, 32, 36, 37), 0%–9.1% of robotic operations (34–37), and 0%–9.1% of open operations (20, 21, 23–30, 32, 34, 35). No significant differences were detected between laparoscopic and open gastrectomies (20, 21, 23–30, 32) or between robotic and open gastrectomies (34, 35) in regard to postoperative mortality, regardless of the type of gastrectomy (total or partial) (20, 24, 25, 29, 32) or the type of procedure (hand-assisted or totally laparoscopic) (25–30, 32). Moreover, no significant differences were found when comparing laparoscopic with robotic gastrectomies in terms of intraoperative and postoperative mortality (36, 37).
Postoperative mortality was assessed in our meta-analysis on the basis of the number of deaths shortly after the surgery. Relevant data were available in a total of 14 studies (20, 21, 23, 24, 26–32, 34, 36, 37). No significant differences were observed in postoperative overall mortality when comparing minimally invasive with open gastrectomy (RR = 0.89; 95% CI: 0.63 to 1.26, p = 0.51; I2 = 0%, p = 0.97) (Fig. 5). The corresponding funnel plot revealed moderate asymmetry. Postoperative overall mortality did not differ between minimally invasive surgery and open gastrectomy, even after excluding the study by Mamidanna et al. (20), which had the largest sample size (RR = 0.98; 95% CI: 0.51 to 1.87, p = 0.95). In addition, we observed no significant differences, when comparing the rates of postoperative mortality in the laparoscopic approach with those in the open gastrectomy (RR = 0.88; 95% CI: 0.62 to 1.25, p = 0.48; I2 = 0%, p = 0.98) (20, 21, 23, 24, 26–29, 31, 32, 36). The rates of postoperative mortality did not differ between laparoscopic minimally invasive surgery and open gastrectomy, even after excluding the study by Mamidanna et al. (20), which had the largest sample size (RR = 0.95; 95% CI: 0.46 to 1.95, p = 0.89).
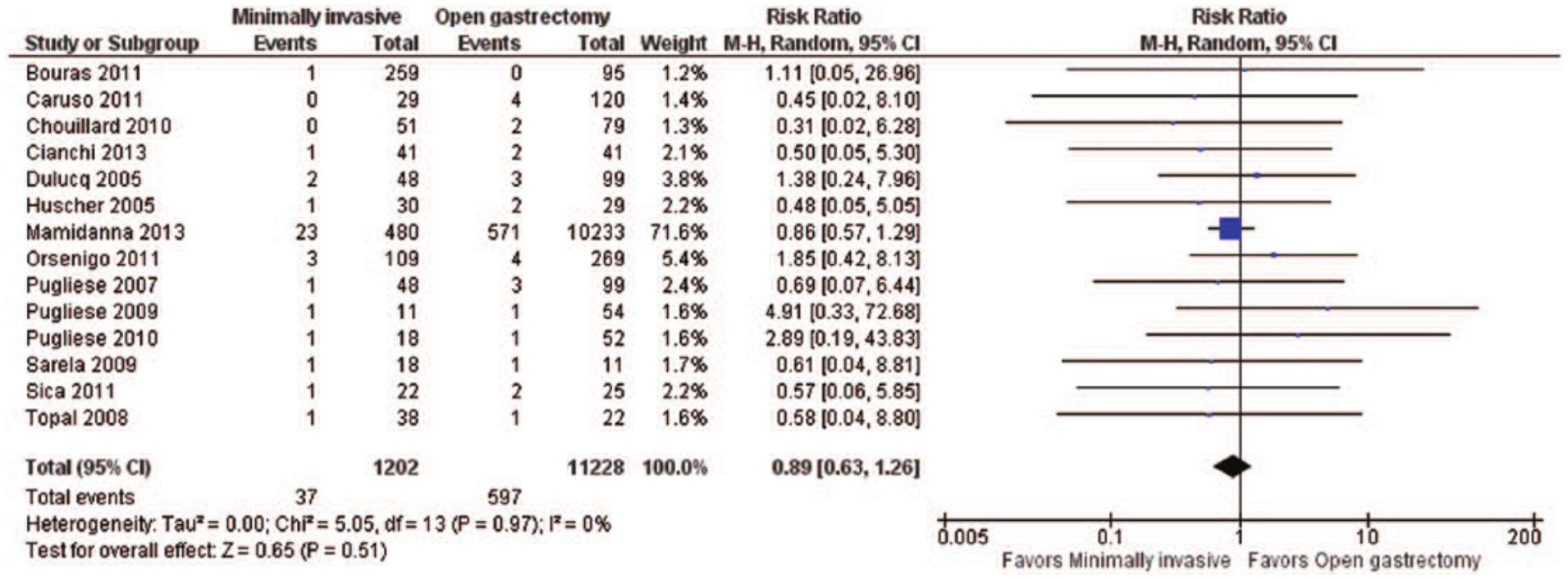
Postoperative mortality: forest plot.
Data about the complications are listed in Tables 6 and 7.
Complications (leakage, stenosis, and bleeding).
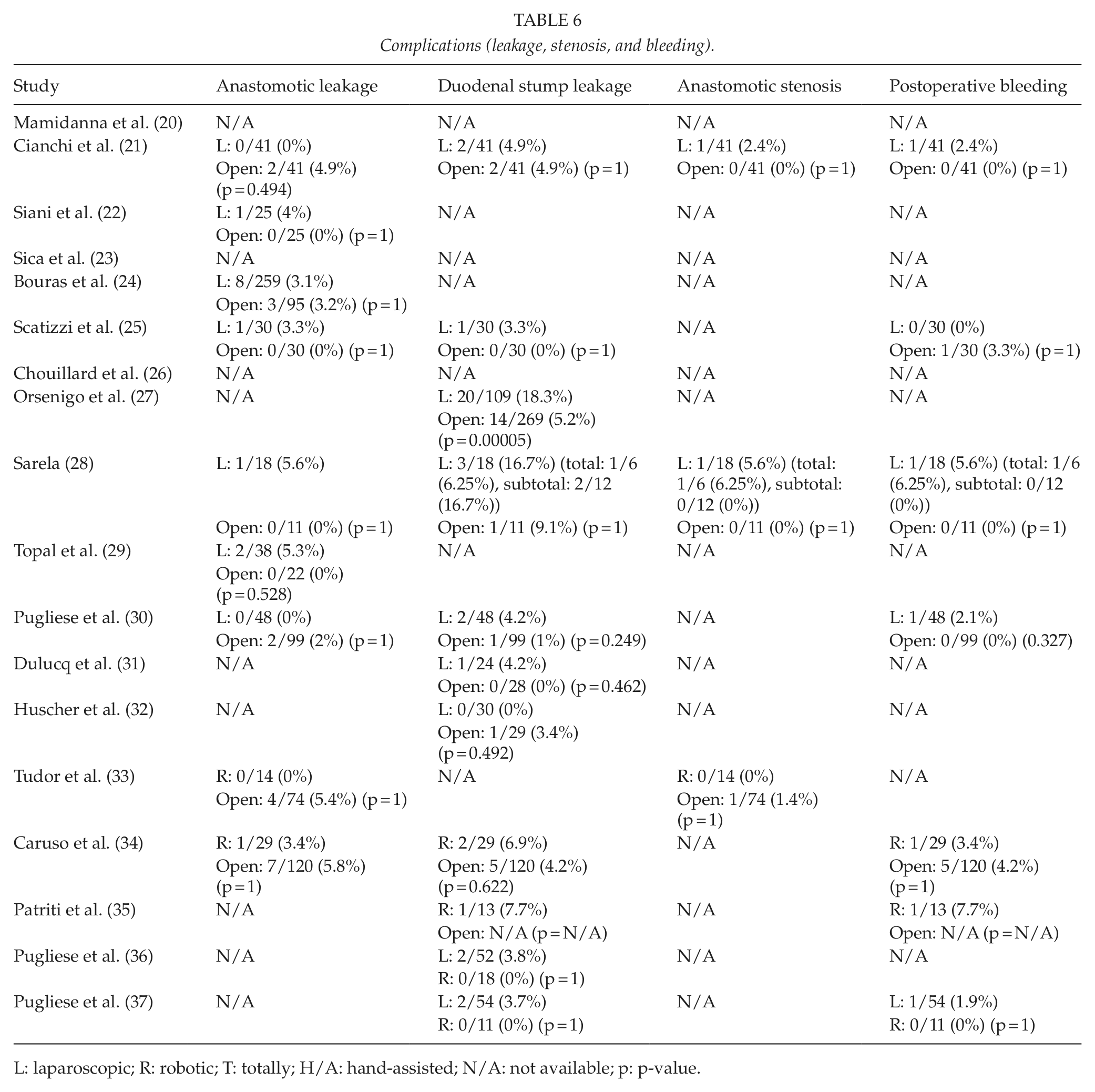
L: laparoscopic; R: robotic; T: totally; H/A: hand-assisted; N/A: not available; p: p-value.
Complications (reoperation and mortality).
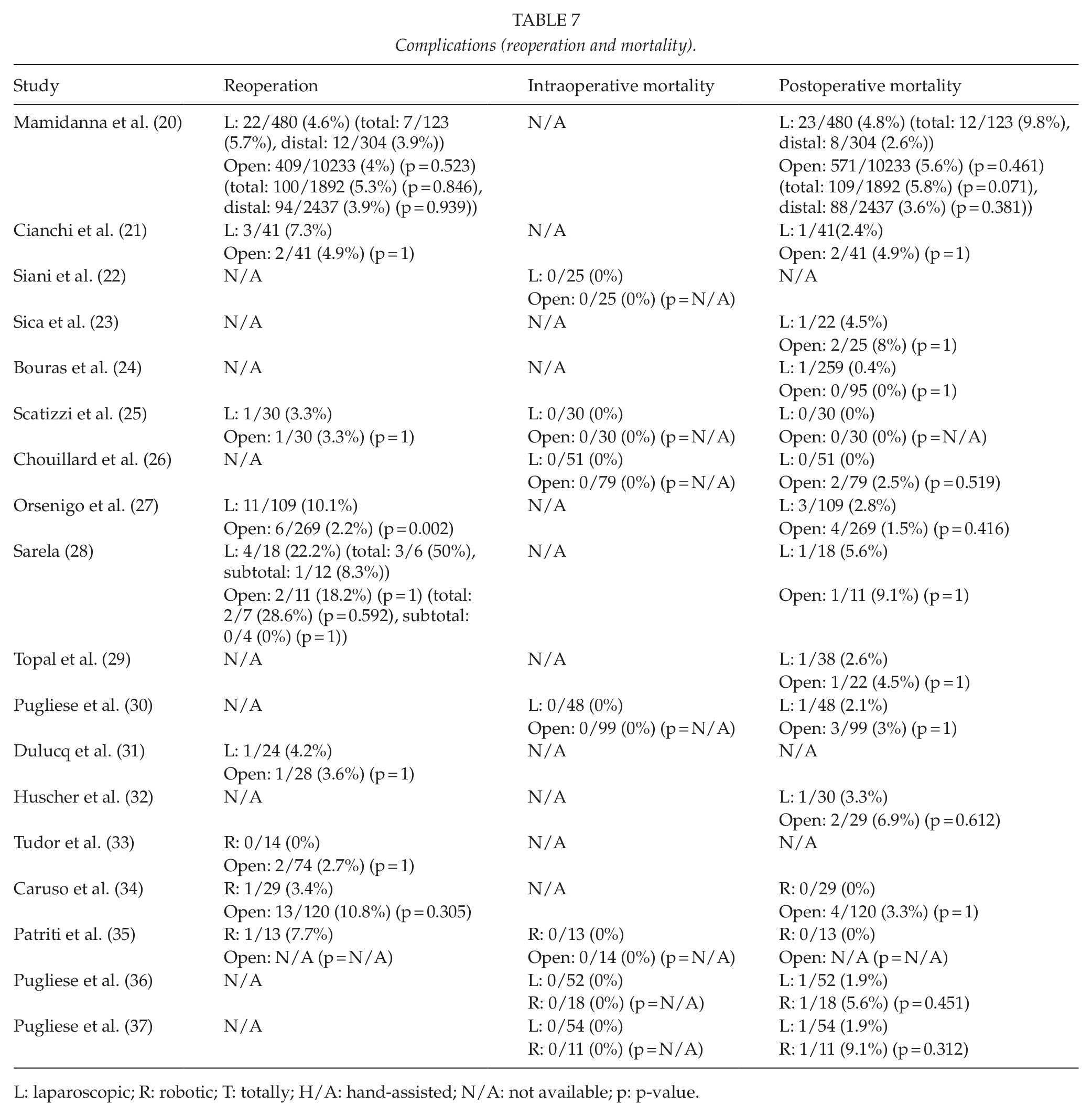
L: laparoscopic; R: robotic; T: totally; H/A: hand-assisted; N/A: not available; p: p-value.
Length of Hospital Stay and Survival
It seemed that patients stayed at the hospital for a shorter period of time in case of laparoscopic and robotic gastrectomies in comparison with open ones (20–22, 25, 26, 30, 31, 33, 34). Patients with gastric cancer having undergone laparoscopic procedures remained for a shorter period of time in hospital than those having undergone open procedures (20–22, 25, 26, 30, 31). The same was found when comparing only total (20, 22, 31) or only partial gastrectomies (20, 25, 31) and when comparing only hand-assisted (30) or only totally laparoscopic gastrectomies (22, 25, 26, 31). The only exception were two studies in which no significant difference between laparoscopic and open approach was detected in regard to the length of hospital stay (27, 28). Moreover, the length of hospital stay was also shorter in robotic operations than in open ones (33, 34), whereas no significant difference was found when comparing laparoscopic with robotic operations (36).
Data regarding the length of hospital stay were provided by a total of seven studies (21, 22, 26, 27, 30, 31, 34), which were characterized by moderate heterogeneity. We observed a significant shorter duration of hospitalization for minimally invasive procedures in comparison with open surgery, with a mean difference of −3.86 days in our meta-analysis (95% CI: −4.70 to −3.01, p < 0.00001; I2 = 56%, p = 0.03) (Fig. 6). Excluding the studies by Dulucq et al. (31) and Pugliese et al. (30), which presented the most extreme results, did not alter the observed association, with the mean difference regarding the length of hospital stay between minimally invasive and open gastrectomy becoming −3.42 days (95% CI: −3.69 to −3.15 years, p < 0.00001; I2 = 0%, p = 0.65), according to the results of the five eligible studies.
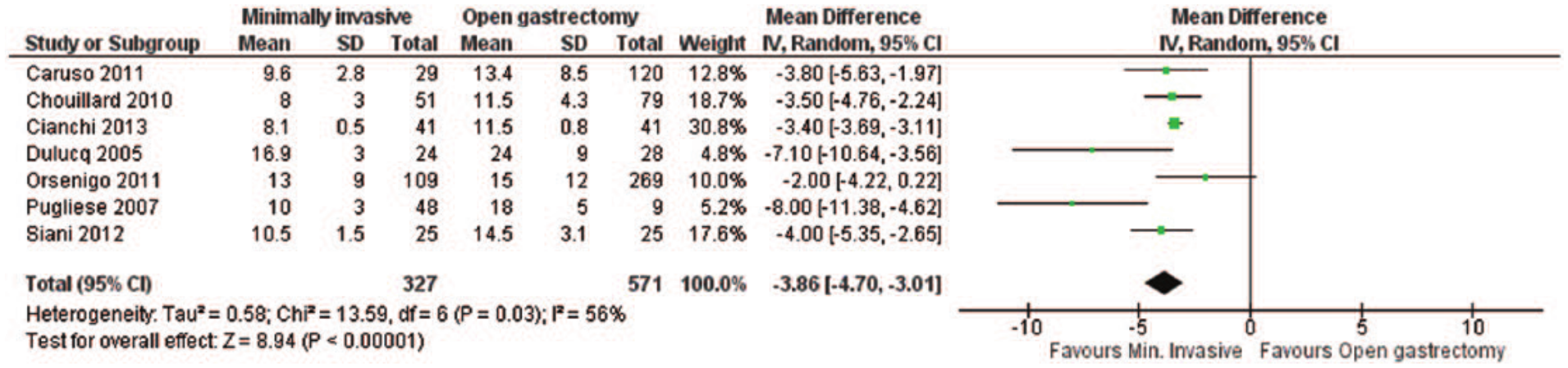
Length of hospital stay: forest plot.
Progression-free and overall survivals were not significantly different when comparing minimally invasive with open gastrectomies (22–27, 32). Patients having undergone laparoscopic procedures had equivalent progression-free and overall survivals with those having undergone open procedures (22–27, 32), irrespective of the type of gastrectomy (total or partial) (22, 24, 25, 32) or the type of procedure (hand-assisted or totally laparoscopic) (22, 25–27, 32). Laparoscopic operations also yielded equivalent overall survival with robotic operations (36).
Data regarding overall survival were available in a total of 10 studies (22–27, 30–32, 34). When we performed meta-analysis, we observed that the minimally invasive approach was associated with a significantly improved overall survival compared with the open gastrectomy (HR = 0.78; 95% CI: 0.64 to 0.95, p = 0.01; I2 = 0%, p = 0.54) (Fig. 7). The visual inspection of the funnel plot revealed asymmetry, implying possible publication bias. However, excluding the studies by Dulucq et al. (31) and Orsenigo et al. (27) as possible outliers, the association became insignificant (HR = 0.84; 95% CI: 0.64 to 1.10, p = 0.21; I2 = 0%, p = 0.99) (Fig. 8), while the final funnel plot became more symmetric.
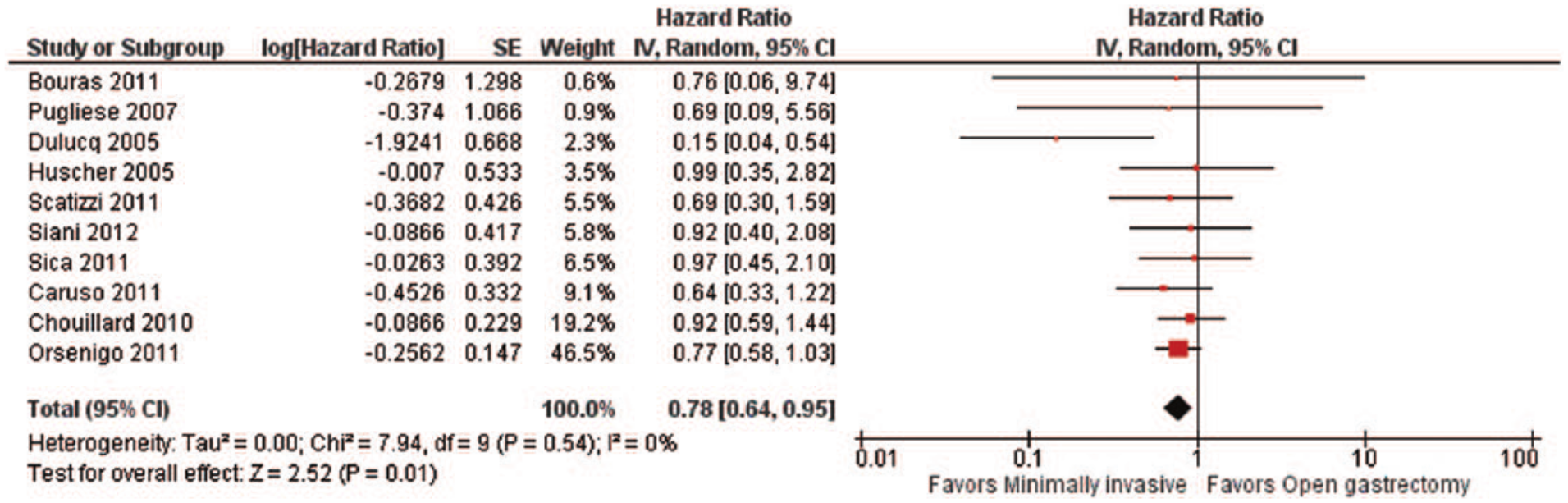
Overall survival: forest plot (10 studies).
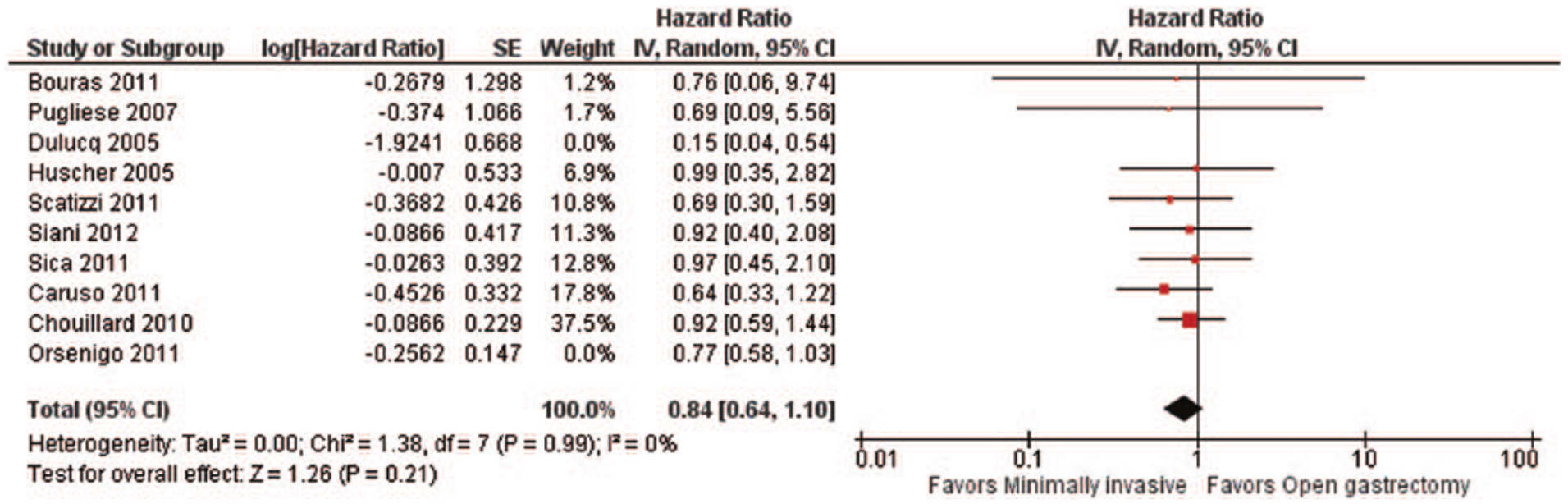
Overall survival: forest plot (8 studies).
Data about the length of hospital stay and survival can be seen in Table 8.
Length of hospital stay and survival.
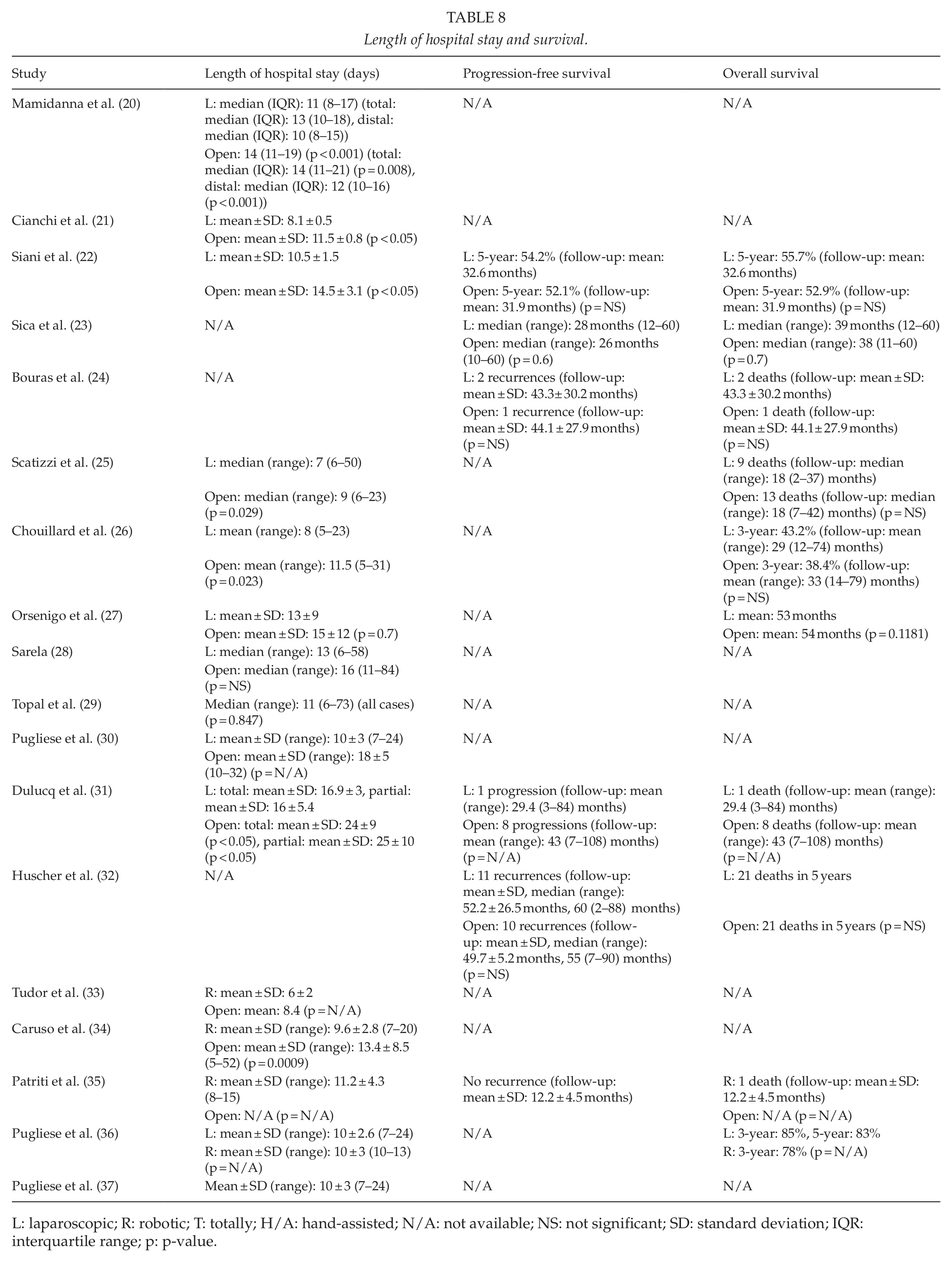
L: laparoscopic; R: robotic; T: totally; H/A: hand-assisted; N/A: not available; NS: not significant; SD: standard deviation; IQR: interquartile range; p: p-value.
Discussion
Laparoscopic procedures have the advantages of decreased blood loss, smaller incisions, reduced postoperative pain, earlier mobilization, faster recovery, and shorter hospital stay over open procedures (2–5). However, laparoscopic operations have the disadvantages of the inadequate tactile feedback and perception of depth, the reduced dexterity due to the handling of rigid tools and the increased operative time. The introduction of robotics in surgery helped surgeons to overcome many of the aforementioned disadvantages, thanks to the stability of tool handling, the 7 degrees of freedom in movement, and the three-dimensional vision that robotic surgery provides (1, 11, 38).
Our review aimed to summarize the European experience in minimally invasive gastrectomies for gastric cancer and to compare this approach with the open one. Minimally invasive gastrectomies did not seem to differ significantly from the open gastrectomies in regard to most tested parameters. Thus, laparoscopic and robotic procedures provided equivalent results with open procedures concerning the distribution of tumor stages (21–23, 25, 26, 28, 29, 31, 32, 34); the type of anastomosis, which was Roux-en-Y in most cases (21–37); the rates of negative resection margins, although our meta-analysis demonstrated a plausible advantage of minimally invasive approaches over the open one (21, 24–31, 34, 35); the frequency of anastomotic leakage, duodenal stump leakage, anastomotic stenosis, and postoperative bleeding (21, 22, 24, 25, 28–34); the reoperation rates (20, 21, 25, 28, 31, 33, 34); the intraoperative and postoperative mortality (20–30, 32, 34, 35); and the progression-free and overall survival, although our meta-analysis yielded a likely improved overall survival in case of minimally invasive approaches (22–27, 32). However, we should be very cautious in drawing conclusions about survival since the studies taken into consideration included patients of different age, tumor stage, and tumor distribution (upper, middle or lower third) who underwent different types of gastrectomy (total, subtotal or distal) with various extent of lymphadenectomy, although there were no significant differences in distribution of stages and tumor location between minimally invasive and open operations in most (21–23, 25, 26, 28, 29, 31, 32, 34), but not in all studies. Moreover, the advantage of minimally invasive procedures over open ones was shown only when we took all the 10 articles with data about overall survival into consideration (22–27, 30–32, 34), which yielded an asymmetry in the funnel plot, implying possible publication bias. When we tried to solve this problem by excluding two certain articles (27, 31), the funnel plot became more symmetric, but the difference in overall survival became insignificant. In regard to the postoperative mortality, it should be mentioned that the study with the largest number of patients (10233 open and 480 laparoscopic gastrectomies), conducted by Mamidanna et al. (20), provided a relatively high rate of mortality for laparoscopic total gastrectomies (9.8%), as well as for open total gastrectomies (5.8%), but without a statistically significant difference between the two approaches (p = 0.071). This finding stresses the need for greater attention to the safety of laparoscopic total gastrectomy. Furthermore, laparoscopic operations did not differ significantly regarding the tumor size (21, 25, 29, 31) and the number of retrieved lymph nodes (21–26, 28, 31, 32). In addition, conversion rates to open for both laparoscopic and robotic approaches were relatively low in most studies (21–23, 25, 26, 30, 31, 34–37).
However, there were significant differences between minimally invasive gastrectomies and open gastrectomies as far as other parameters are concerned. Blood loss was less in minimally invasive procedures than in open ones (21, 22, 24, 27, 29, 30, 32–34). However, our meta-analysis demonstrated only a minor difference of about a half blood unit. Moreover, patients having undergone minimally invasive operations stayed in hospital for a shorter period of time than those having undergone open ones (20–22, 25, 26, 30, 31, 33, 34), which can be considered clinically significant, since our meta-analysis yielded a mean difference of 3–4 days. On the other hand, the results were controversial concerning other parameters. Regarding the number of retrieved lymph nodes, one study reported a small advantage of open over robotic approaches (34) and another study reported no significant difference between them (35). Furthermore, some studies found that laparoscopic and robotic gastrectomies last longer than open ones (21, 24, 25, 27–29, 34), whereas other studies did not find an actual difference regarding the duration of operation (22, 26, 30–33). Our meta-analysis showed that the mean increase in operative time during minimally invasive procedures is about 45 min, but the heterogeneity of the studies was too high for definitive conclusions to be made. Finally, when we examined overall postoperative morbidity in our meta-analysis, we found a plausible advantage of open gastrectomy over the minimally invasive techniques, which provided increased rates of postoperative morbidity in total.
When comparing laparoscopic with robotic gastrectomies, equivalent results were found in most tested parameters. Thus, laparoscopic and robotic approaches did not differ significantly in terms of the rates of negative resection margins (36), duodenal stump leakage (36, 37), postoperative bleeding (37), the intraoperative and postoperative mortality (36, 37), the length of hospital stay (36), and the overall survival (36). However, we have to be very cautious in drawing conclusions about survival since only one study compared overall survival between laparoscopic and robotic gastrectomy (36). On the contrary, robotic operations lasted longer than laparoscopic ones and blood loss was less in robotic procedures than in laparoscopic ones (36).
Nevertheless, we have to keep in mind that most studies comparing minimally invasive and open gastrectomy that come from European countries include a small numbers of patients, are non-randomized trials, and include cases from only one institution. In particular, only 1 eligible study was randomized controlled trial, but it included only 59 patients (32), and only 4 out of the 18 eligible studies had more than 50 patients in each arm (20, 24, 26, 27). Moreover, 15 out of the 18 included studies were conducted in single institutions (21–25, 27–33, 35–37), 2 were carried out with the contribution of two institutions (26, 34), and only 1 included data from all English National Health Service (NHS) Trust hospitals, having a very large number of patients, but it was a non-randomized trial (20). Additionally, many articles included the whole number of minimally invasive gastrectomies performed in an institution, starting from the adoption of the technique and without excluding the cases corresponding to the beginning of the learning curve or even making a comment about them. Furthermore, none of the 18 studies included data for all the parameters tested. Fourteen articles included data for almost all the variables (21, 22, 24–32, 34, 36, 37) and four articles contained information for only a number of the variables (20, 23, 33, 35). Finally, there may be an overlap in the reported cases of three studies by Pugliese et al. (30, 36, 37) because the authors are essentially the same, the institution is the same, and the time periods overlap. However, it is not possible to find the exact cases that are reported in more than one study. It should also be noted that the first study (30) compares laparoscopic with open gastrectomies, whereas the second and the third study (36, 37) compare laparoscopic and robotic ones. Due to the small number of European studies comparing minimally invasive with open gastrectomies and with each other, we decided to include all the available articles that meet our inclusion criteria in order to take into consideration as much data as possible. For all the aforementioned reasons, we have to be very careful in the interpretation of the results of these European studies and we are in need for more prospective randomized controlled trials with large number of patients.
To summarize, laparoscopic and robotic gastrectomies may be considered alternative approaches to open gastrectomies for treating gastric cancer, a conclusion confirmed by the results from several European studies. Minimally invasive gastrectomy has the advantages of the less blood loss and the shorter hospital stay in comparison with open gastrectomy. No other differences are well documented between minimally invasive and open approaches, including the duration of operation, for which the results have been controversial. In addition, robotic procedures have the advantage of the less blood loss over laparoscopic ones, but they last longer than them. No other differences are well documented between laparoscopic and robotic operations. Further European studies, and particularly randomized controlled trials, are needed comparing laparoscopic and robotic gastrectomies with open gastrectomies and with each other. Thus, the advantages and disadvantages of each approach will be clarified and the results from Europe will be compared with those of studies from other parts of the world.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
