Abstract
Background and Aims:
There is an increasing trend toward performing liver resections in the setting of metastatic disease. Renal cell cancer liver metastases are associated with poor survival. The indications for and the short- and long-term outcomes of liver resection for renal cell cancer liver metastases remain not well defined.
Material and Methods:
A focused, structured literature review on PubMed, EMBASE, and Google Scholar was performed to identify primary research articles, on short- and long-term outcomes and prognostic factors of patients undergoing liver resection for renal cell cancer liver metastases. Only studies with a sample size equal or larger than 10 patients were included.
Results and Conclusion:
A total of 10 studies met inclusion criteria. Median overall survival ranged between 16 and 142 months. Major morbidity was rare while 30-day postoperative mortality was less than 5%. A disease-free interval of more than 2 years from nephrectomy to evidence of liver metastases and a radical, microscopically negative surgical resection (R0) were the most consistent prognostic factors that, in turn, could be used as potential selection criteria to identify patients who can benefit the most from liver-directed surgery. Liver surgery for renal cell cancer liver metastases can be performed with low mortality, acceptable morbidity, and promising survival benefit in carefully selected patients. Studies that can assess the impact of modern, targeted regimens in the preoperative setting and liver-directed surgery and in turn shape new selection criteria are warranted.
Introduction
Liver surgery has evolved substantially and the morbidity and mortality in specialized, high-volume academic centers approach rates of 15%–50% and <5%, respectively, today (1). Those advances can be attributed to a refinement of surgical techniques and improvement in anesthesia and perioperative management such as the implementation of enhanced recovery after surgery (ERAS) programs that were shown previously to have efficacy in colorectal surgery (2). More importantly, the safety of performing liver resections has led to a wide expansion of indications for hepatic resections of metastatic disease (3, 4).
Among the non-neuroendocrine, non-colorectal secondary liver tumors, renal cell cancer liver metastases (RCCLM) resection has arisen as an increasingly commonly performed surgical procedure (5). Liver is the third most common site of metastasis in renal cell cancer (RCC) after lung and bones with an incidence of round 20% (6). RCCLM have been associated with worse overall survival (OS) and recurrence-free survival (RFS) in multiple studies including the largest series in literature (7). In an effort to interpret the poor long-term outcomes in patients with RCC and synchronous or metachronous liver metastases, it has been postulated that the unique organ microenvironment of the liver may be selective for a more aggressive clinical phenotype of the disease (8, 9). However, in contrast with colorectal liver metastases (CRLM), the exact indications for surgery, the long-term outcomes after metastasectomy, and the clinicopathologic factors that determine actuarial long-term prognosis in that particular cohort of patients remain not well defined. Moreover, previous reviews mostly focused on overall non-neuroendocrine, non-CRLMs and did not explicitly investigate patients with RCCLM (5, 10, 11). Given that, we sought to review the current literature by including all available studies of patients who underwent curative intent liver resection for RCCLM.
Material and Methods
Literature Search Strategy
From May to June 2015, a focused, structured literature review on PubMed, Google Scholar, and EMBASE was performed to identify articles involving patients who underwent liver resection for hepatic metastases from RCC. All relevant studies written in German or English language were included. Articles were identified using the search string “renal cancer,” “liver metastases,” “hepatic metastases,” “resection,” and “surgery” that were input either independently or in various multiple combinations in order to retrieve the maximum of search-specific results. The results were cross checked to exclude overlapping series or double entries. All studies investigating the impact of hepatic resection on long-term outcomes as well as potential prognostic factors are described in a chronological manner.
Selection Criteria and Data Extraction
All studies in humans with publication dates from 1990 to 2015 were considered for analysis. Studies including more than 10 patients, either as part of renal cancer patient populations or as part of non-colorectal, non-neuroendocrine liver metastases patient populations were implemented. The minimum number of 10 patients was selected in order to reduce the risk of confounding from small clinical series and the impact of the unit’s low volume experience. Conference abstracts that did not proceed to publication in peer-reviewed journals were excluded from further consideration. Double entries and overlapping study populations were filtered and excluded. For each study article included in the review, data pertaining to study setting, time period, authors, and patient characteristics were extracted. Data were also extracted for short-term clinical outcomes such as 30-day morbidity and mortality, long-term outcomes including median, 5-year and 1-year OS and finally prognostic factors deemed significant on multivariable analysis.
Results
RCC represents 2%–3% of all types of malignancies, and its incidence is increasing at a rate of 2% annually. A subset of 20%–30% of all patients diagnosed with RCC have synchronous liver metastases while another 20%–40% of patients will develop metachronous metastases after undergoing curative intent nephrectomy for the primary tumor. Unfortunately, only in around 5% of all metastatic cases is the liver the only organ affected by metastases. The OS of stage IV RCC is dismal, with a 1-year survival ranging from only 10% to 15%. Patients with RCCLM overall have a particularly poor prognosis, which is even worse than patients with RCC metastases to other organs, such as lungs and bone. For those patients with technically resectable disease, the role of liver metastasectomy has been investigated in several small, single institutional retrospective studies.
In 1997, Stief et al. (12) were the first to report survival outcomes of patients who underwent liver resection for metachronous solitary RCCLM, in a cohort larger than 10 patients. They studied a cohort of 13 patients and showed a median survival of 16 months. However, they did not report on 3- or 5-year survival rates. More importantly, while an R0 resection was achieved in most patients (85%), surgery in these cases was associated with dismal short-term outcomes, with a mortality of 31%. The authors concluded that while OS was improved when compared with a contemporary cohort of four patients (median OS: 4 months) with similar disease characteristics who did not undergo resection, surgeons should strive for a very careful patient selection since the operation was associated with very high rates of morbidity and mortality.
In the late 1990s, another German group performed a retrospective analysis of patients who underwent liver resection for non-colorectal, non-neuroendocrine liver metastases (13). Among these patients, a total of 13 patients with RCCLM were offered surgical treatment. An R0 resection was feasible on 80% of the patients. The group reported a median survival of 24 months for the entire cohort, and 25 and 14 months for the R0 and R1/R2 groups, respectively. The 5-year OS was 8% and 10% for the entire cohort and the patients who underwent an R0 resection, respectively.
In 2003, Alves et al. (14) from Paul Brousse hospital in France published the third retrospective study about liver metastasectomy in RCCLM. They had a small cohort size of only 14 patients. Of note, extrahepatic disease (lung metastasis) was not considered a contraindication for RCCLM surgery. Consistent with the previous studies an R0 resection was achieved in most of the patients (84.6%). There was no mortality within 60 days from surgery. The median survival of their surgical cohort was 26 months, with a 3-year OS of 26%. The curative pattern of hepatectomy (2-year survival, 69% vs 0%; p = 0.001), a disease-free interval over 24 months (2-year survival, 71% vs 25%; p = 0.05), a maximal tumor size less than 5 cm (2-year survival, 83% vs 17%; p = 0.006), and the possibility of achieving a repeat hepatectomy in the case of recurrence (2-year survival, 100% vs 21%; p = 0.02) were associated with a better OS after liver resection. The number of metastases did not impact significantly on survival outcomes. Of note, disease-free interval was a significant prognostic factor along with a complete tumor extirpation (R0). The authors recommended an aggressive surgical treatment of RCCLM as the best chance of cure for those patients. It should be noted that this study was the first to report on prognostic factors.
The largest study so far was conducted by Adam et al. (15) in 2006 and assessed 85 patients undergoing liver resection for metastatic renal cancer (Table 1). The 5-year OS was 38% and the median OS was 36 months. In that study, renal cancer was among the primary tumors that displayed favorable prognosis, even when metastasizing to the liver, compared to primary tumors such as melanoma and pancreaticobiliary malignancies. Unfortunately, the authors did not conduct multivariable analysis to identify prognostic factors specifically valid for RCCLM.
Morbidity and mortality data.
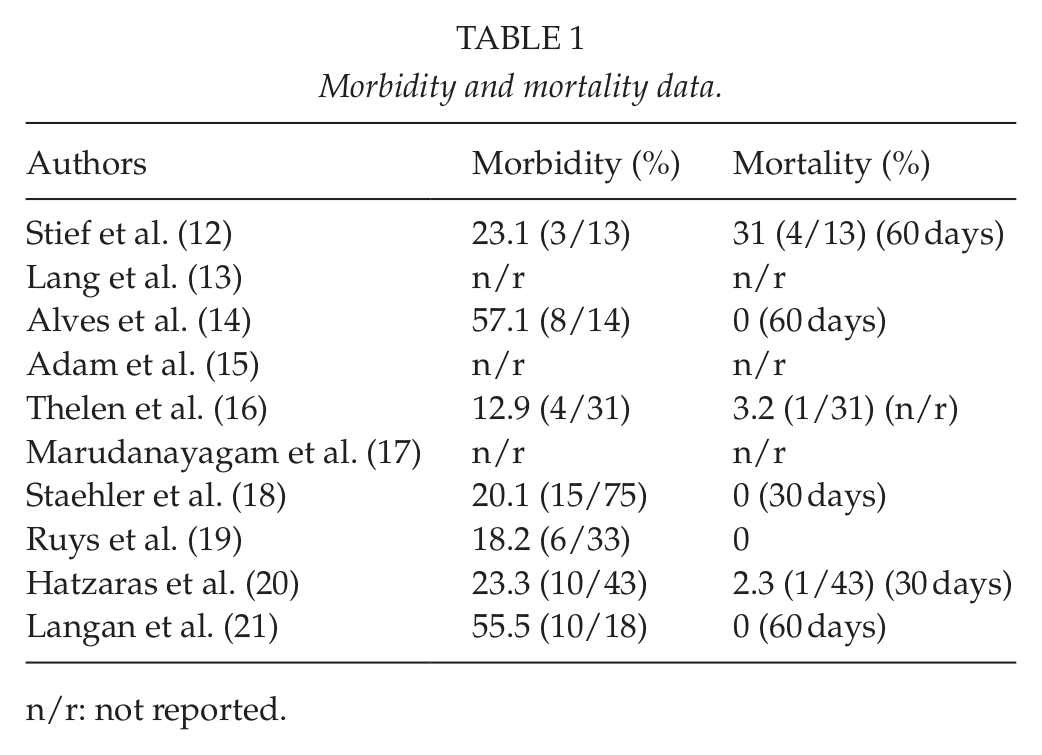
n/r: not reported.
In 2007, Thelen et al. (16) from Charite, Berlin, investigated a cohort of 31 patients who underwent curative intent resection for RCCLM. Of all patients, 77.4% underwent a microscopically negative resection (R0), confirming previous studies. The authors demonstrated a 5-year OS of 38.9% with a median follow-up of 29 months. While disease-free interval and resection margin were deemed significant on univariable analysis, on multivariable analysis only positive resection margins (R1/R2) were associated with a worse OS (p = 0.005). The authors reported also on the RFS and the recurrence patterns. A 5-year RFS was 26.1% and the most common site of recurrence after liver surgery was the liver. The authors compared their long-term outcomes with historic outcomes in CRLM resection and concluded that their outcomes were not inferior to survival rates achieved by liver resection in patients with metastases from colorectal cancer. Their insight was that liver resection represents the best chance for long-term survival in that cohort of patients and microscopically negative surgical margin was of utmost importance in terms of long-term survival after liver surgery.
Marudanayagam et al. (17) from Canada evaluated 24 patients with RCCLM within an overall cohort of 65 patient treated with liver resection for non-colorectal, non-neuroendocrine, non-sarcomatous liver metastases. RCCLM, diagnosed in 24 patients, were the most common tumors in the entire cohort and were also associated with a median survival of 22.5 months. Specific about the rate of R0 resections in the RCCLM cohort were not reported. No perioperative mortality was reported in that cohort. The authors reported that R0 resections were significantly associated with a better OS in the entire cohort. They also confirmed previous studies showing that kidney and breast tumors carry a better prognosis when compared to gastrointestinal primary tumors. RCCLM-specific prognostic factors were not evaluated.
In a separate study, Staehler et al. (18) examined a cohort of 68 patients who underwent liver metastasectomy and compared them to a contemporary control cohort of 20 patients who declined surgery for reasons not related with the underlying disease or the provider’s decision. Interestingly, in that study, patients were offered liver resection even when the tumor burden exceeded the 10 liver lesions. Authors did not report the rate of R0 resections. The receipt of perioperative chemotherapy was comparable between the two groups. On multivariable analysis, the authors reported that patients with high grade tumors (hazard ratio (HR): 2.568, confidence interval (CI): 1.238–5.329, p = 0.011), those with worse ECOG (Eastern Cooperative Oncology Group) performance status (HR: 3.763, CI: 1.777–7.972, p = 0.001) and those who did not undergo hepatectomy (HR: 2.23, CI: 1.054–4.719, p = 0.036) had a significantly higher risk of death. The strength of the study, besides being the second largest study explicitly focusing on RCCLM, was that it involved a control group. This is the only study in RCCLM literature that used a specific control group instead of historic controls. A 5-year OS after metastasectomy was 62.2% versus only 29.3% in the control group. Concerning the indications for liver resection the authors concluded that a resection should be attempted as long as complete tumor extirpation can be achieved.
In 2011, Ruys et al. (19) investigated 33 patients who underwent curative intent liver resection for RCCLM enrolling patients from all academic centers of liver surgery in the Netherlands. The surgically treated cohort comprised only 0.5%–1% of all patients diagnosed with RCCLM in the same period of time. On histopathologic analysis, 76% of the patients had R0 resection. Among them, the number of RCCLM lesions was as high as 19, and 14 patients (43%) had at least 2 metastatic liver lesions. The outcomes of this population-based study included an OS of 43%. On multivariabe analysis, metachronous metastases (n = 23, p = 0.03) and microscopically negative resections (R0) on final histopathology (n = 19, p>0.001) were the only factors that remained significant and determined prognosis. Except for the worse OS in patients with synchronous liver disease, a disease-free interval greater than 24 months conferred better prognosis when compared to disease-free interval less than 24 months, even if it was borderline significant (p = 0.051). Consistent with the aforementioned studies, tumor-related factors such as number of lesions and size of lesions did not impact significantly on OS.
Interestingly, Hatzaras et al. (20) reported on a cohort of 43 patients with RCCLM who underwent curative hepatic resection. Moreover, they compared that cohort to matched controls of patients (1:3 match) who underwent liver resection for CRLM. On final histology, surgical margin was negative in 95.3% of the patients. The authors showed that the overall median survival was equivalent in both cohorts (p = 0.582). Given that, CRLM is considered the most amenable surgically treated liver metastasis, it is obvious from this study that RCCLM is more than justified in selected patients. The authors attributed the excellent 3-year OS of their cohort that reached 62.1%, to the very stringent resection criteria that were applied. On multivariable analysis, they identified prolonged disease-free interval greater than 12 months (p = 0.022) and the absence of extrahepatic disease (p = 0.024) as the only two statistically significant prognostic factors. Interestingly and consistent with the results from Staehler et al. multiple hepatic lesions versus solitary lesion did not impact significantly on OS and RFS. That reinforces the notion that metastasectomy should be pursued as long as complete tumor extirpation can be achieved irrespective of other adverse tumor characteristics.
Finally, in 2012, Langan et al. (21) from the NCI reported on 18 patients who received liver-directed treatment for RCCLM. Among these, 10 underwent curative intent hepatectomy while 8 underwent radio frequency ablation (RFA). The R0 resection rates were not mentioned. The 5-year actuarial OS all patients receiving any kind of liver-directed treatment was 27%. Median survival for those who underwent surgery was 24 months, and for those who underwent RFA was 15.6 months (p = 0.56). On multivariable analysis, only metachronous liver metastasis was associated with a better long-term survival (p = 0.02). Specifically, median survival was 155 months with metachronous metastases compared to only 29 months in those belonging to the synchronous group. Interestingly, previous study findings were confirmed since number of liver lesions, disease-free interval (DFI) and the presence of extrahepatic disease did not impact significantly on long-term outcomes.
Discussion
Operating on patients with non-colorectal, non-neuroendocrine liver metastases was a no man’s land for many years. It is evident now that the surgical landscape is changing and a constantly growing number of patients with liver metastatic disease are offered surgical treatment. In our review, the first study was conducted in 1997 by a German group; all previous reports on the surgical management of RCCLM were anecdotal reports with sample sizes less than 5 patients. The 5-year survival rates reported from all studies included in the current review range between 8% and 62.2% (Table 2). Importantly, a 5-year survival reported in a cohort of patients that did not undergo liver resection of RCCLM was only 10%. In addition, postoperative morbidity and mortality were reported to range in 12.9%–57.1% and 0%–31%, respectively (Table 1). Of note, only the first study that was conducted in 1997 reported a high mortality rate of 31%. More importantly, from the chronological order that was selected it is apparent that both short-term and long-term outcomes have substantially improved during time. Thus, liver resection in that occasion is a safe procedure and is indeed justified from an oncological standpoint in appropriately selected RCCLM patients. However, the importance of careful selection cannot be overstated. We searched the literature in an effort to identify significant prognostic factors that in turn, can shape and establish potential selection resection criteria. The most consistent favorable ones based on our review of the literature included a disease-free interval of more than 2 years from nephrectomy to evidence of liver metastases, and a radical, microscopically negative surgical resection (R0) (Table 3). According to literature tumor size, number and laterality should not preclude from a curative intent hepatectomy. Of note, all previous studies have suffered from small sample sizes that limit their clinical usefulness in defining selection criteria that could guide surgeons. In fact, when assessed collectively with other secondary liver tumors RCCLM make up less than 7.5% of all non-colorectal, non-sarcoma, non-carcinoid resected liver metastases (15, 22).
Survival data.
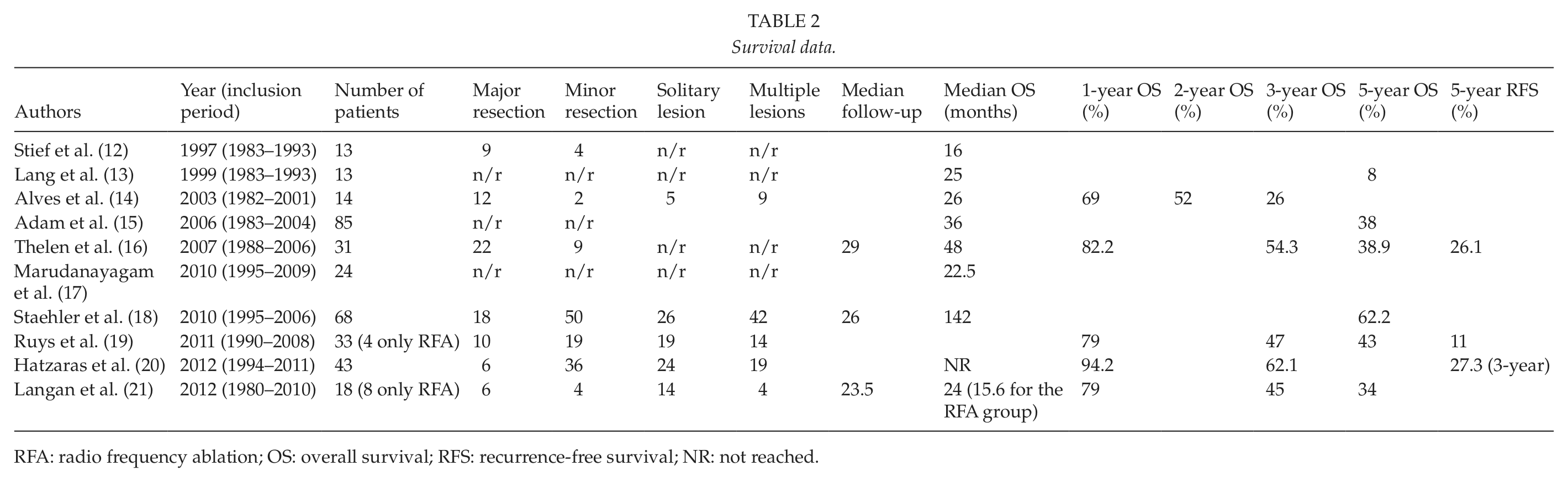
RFA: radio frequency ablation; OS: overall survival; RFS: recurrence-free survival; NR: not reached.
Favorable prognostic factors as reported in the studies overviewed.
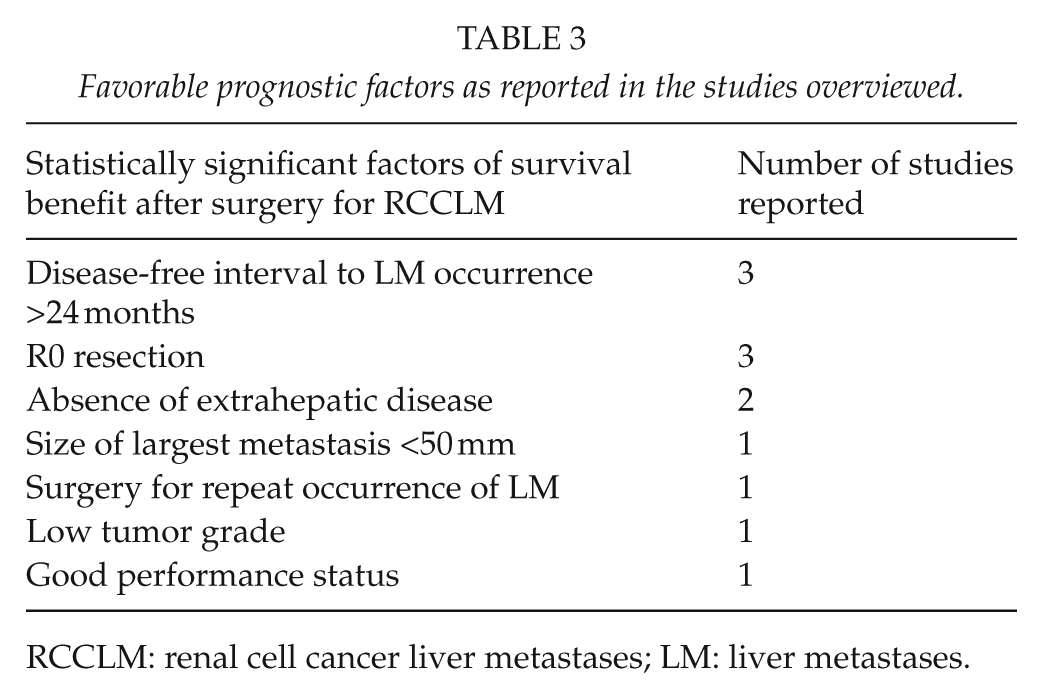
RCCLM: renal cell cancer liver metastases; LM: liver metastases.
Toward the direction of establishing specific resection criteria, the role of preoperative chemotherapy in testing tumor biology and selecting tumors with a favorable biologic behavior should be further elucidated (23). The advent of modern, molecular agents in the last few years will perhaps improve the patient selection by identifying patients bearing tumors with favorable biologic behavior and further improve the prognosis of those resected for RCCLM. A limitation of current studies is that data on modern targeted regimens such as agents that inhibit the vascular endothelial growth factor (VEGF) and the mTOR signal transduction pathways are lacking as most studies lag behind the modern era of targeted therapies. Since almost all published studies about the surgical treatment of RCCLM span earlier periods of immunotherapy and chemotherapy, modern studies that will assess the efficacy of the combined implementation of hepatic resection and molecular modalities are warranted. Of note, liver metastases of RCC are solitary in only 2–4% of cases. As such, future studies should address the value of resection of limited extrahepatic disease along with the liver metastasectomy in an effort to increase the pool of surgical candidates and further refine selection criteria.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors would like to express their gratitude to Ms Nefeli Bampatsikou for excellent technical support.
