Abstract
Background and Aims:
Patient-rated outcome measures have become increasingly important in clinical research. They provide research and clinical tools which can be utilized in the assessment of patient recovery and treatment efficacy. The purpose of our study was to translate and validate the original version of the PRWE form into Finnish.
Material and Methods:
We conducted the translation of the PRWE questionnaire according to standardized guidelines. Patients (N = 119) with an acute distal radius fracture were recruited, and they completed the PRWE and QuickDASH questionnaires at 2 months and 4 months after the wrist injury.
Results:
The mean answering times were 52 days (standard deviation [SD] 9.8 days) and 116 days (standard deviation [SD] 14.8 days), respectively. Both the internal consistency (Cronbach’s alpha) of 0.976 and the intraclass correlation coefficient (ICC) of 0.992 (95% CI 0.966–0.998) showed excellent reliability for the total PRWE score. The correlation coefficients between the total score, the subscales, and for improvement over time for PRWE and QuickDASH were excellent. The responsiveness was good with an effect size of 0.83 and a standard response mean of 1.22.
Conclusion:
Our study shows that the Finnish version of the PRWE is reliable, valid, and responsive for the evaluation of pain and disability after distal radius fracture.
Keywords
Introduction
During past decades, we have clearly seen an increasing trend toward the operative treatment of distal radius fractures (DRF) without rigorous evidence of an improved functional or patient-reported outcome over casting in an elderly population (1–3). Instead of treating patients according to the best clinical practice and evidence, the prevailing trend appears to lead the current treatment guidelines. Therefore validated patient-rated outcome measures (PROMs) are needed to complement the clinical outcome assessments to better understand and measure the effects of the interventions.
The patient-rated wrist evaluation (PRWE) questionnaire developed by McDermid in 1998, like the Disabilities of the Arm, Shoulder, and Hand (DASH) and the QuickDASH questionnaires, provides the clinicians with a standardized outcome measure that is easy to administer and score in clinical practice. The former is designed to be wrist-specific and therefore the recommended tool for measuring the outcome for wrist injuries, whereas the latter considers the upper extremity as one functional unit (4). PROMs are considered the best subjective measure for the patient’s perception of their state of health and the importance of PROMs in research and clinical practice is shown in many studies (5).
The purpose of this study was to translate, cross-culturally adapt, and validate the original version of the PRWE into Finnish. Few upper extremity PROMs have been validated in Finnish (6) and to our knowledge, PRWE is the first wrist-specific PROM that will be translated and validated into Finnish. The Finnish version of the PRWE will provide a valuable tool for clinicians and researchers.
Material and Methods
The Prwe Questionnaire
The PRWE is a PROM consisting of 15 questions and is designed to measure wrist pain and disability in daily activities. Five questions measure pain, whereas the 10 remaining questions measure wrist function in specific or usual daily activities. If the patient rarely does a task questioned, an estimate is encouraged. If the patient cannot perform a task because of the condition of the wrist, the worst possible outcome is answered. If the patient never does a task questioned, the question is left blank. Missing question is replaced with the mean score of the subscale. A score of 100 is then calculated (pain score + function score: 2), where a score of 100 represents the greatest disability.
Clinical Utility
It is critically important for a questionnaire to be patient friendly in order to minimize additional stress in patients and to minimize non-response bias. The PRWE provides the clinician with a standardized patient-reported outcome measure that is easy to administer and score in clinical practice. It takes approximately 5 minutes to complete the one-page PRWE questionnaire. The questions are clear, concise, and easy to understand.
Translation and Adaptation Process
We did the translation and adaptation process according to the guidelines by Beaton et al. (7) (Fig. 1). We used language services of the University of Helsinki Language Center for the translation of the English version of the PRWE questionnaire. Flowchart depicting the translation and adaptation process is shown in Fig. 1. The final Finnish version is presented in this article as a supplement.
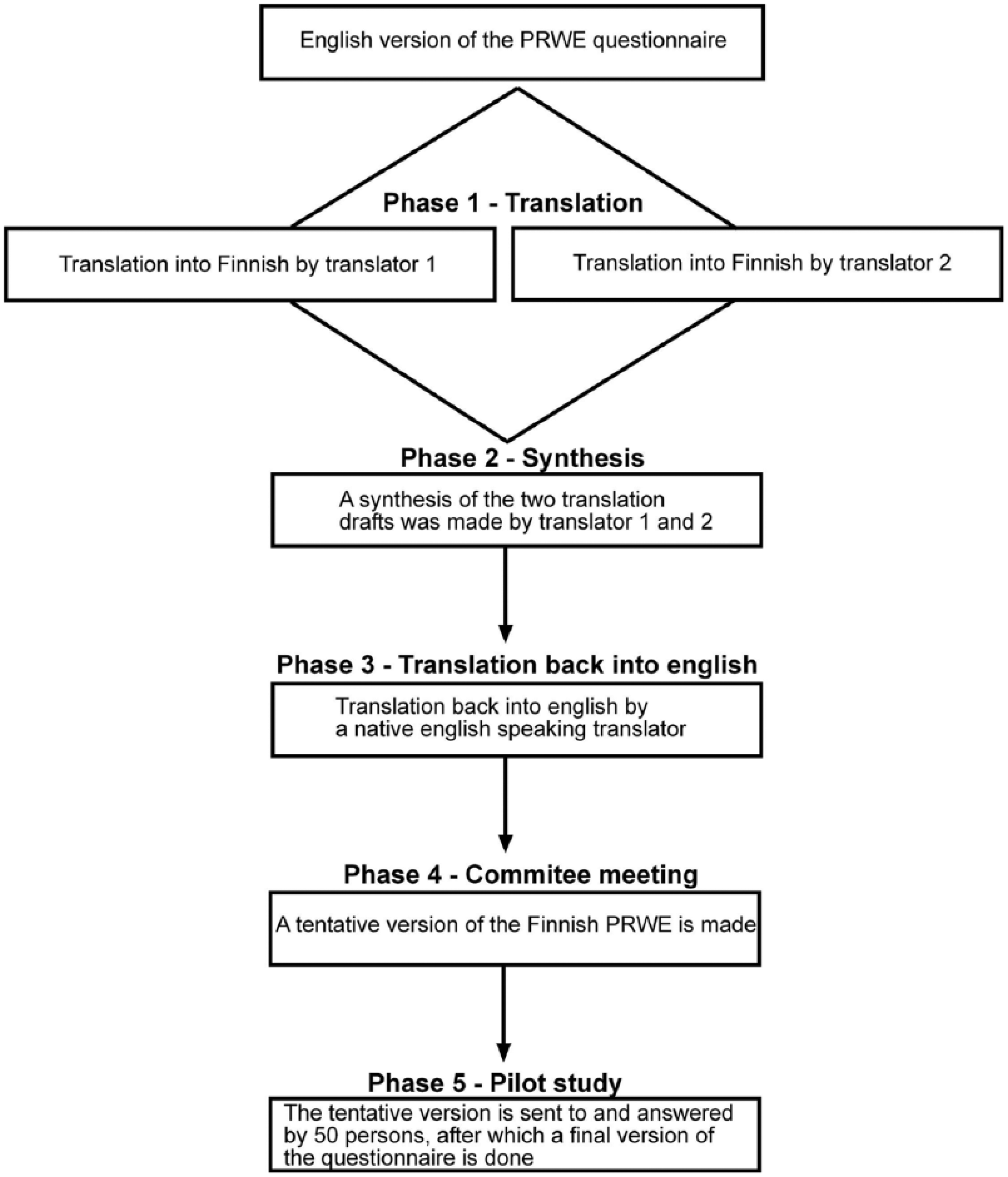
The translation and adaptation process. In Phase 1, two native Finnish-speaking translators (KS and LS) independently made a translation draft each. In Phase 2, the two drafts were merged to form a synthesis by translators 1 and 2. In Phase 3, an English native speaking translator (WH) translated the synthesis back to English to evaluate the preciseness of the translation of the Finnish version compared with the original English version. In Phase 4, a committee consisting of two orthopedic surgeons (HS, TH), both with experience of treating distal radius fractures and excellent English language skill, evaluated the contents of the questionnaire and made a tentative Finnish version. In Phase 5, we sent the tentative Finnish version to 50 patients recovering from a distal radius fracture for commenting, after which the final version of the questionnaire was done according to the comments of the patients in the pilot study.
Patients
We retrospectively identified 336 patients with a DRF in three different hospitals and sent them the two questionnaires once at 2 months and 4 months after the injury accompanied by information about the study, informed consent, and a postage-paid reply envelope. No questionnaires were administered over the phone. Altogether, 136 patients recovering from DRF answered both the PRWE and the QuickDASH questionnaires. Sixteen of these patients were excluded, resulting in 119 patients for the final analyses. Exclusion criteria were insufficient answers, both wrists affected, concomitant fractures, no DRF identified, or dementia (Fig. 2). We classified the radiographs according to the Arbeitsgemeinshaft für Osteosynthesefragen (AO) classification (8), where A-type fractures are extra-articular, B-type fractures partially articular, and C-type fractures intra-articular. Fracture classification was conducted by two researchers (H.S. and T.H.).
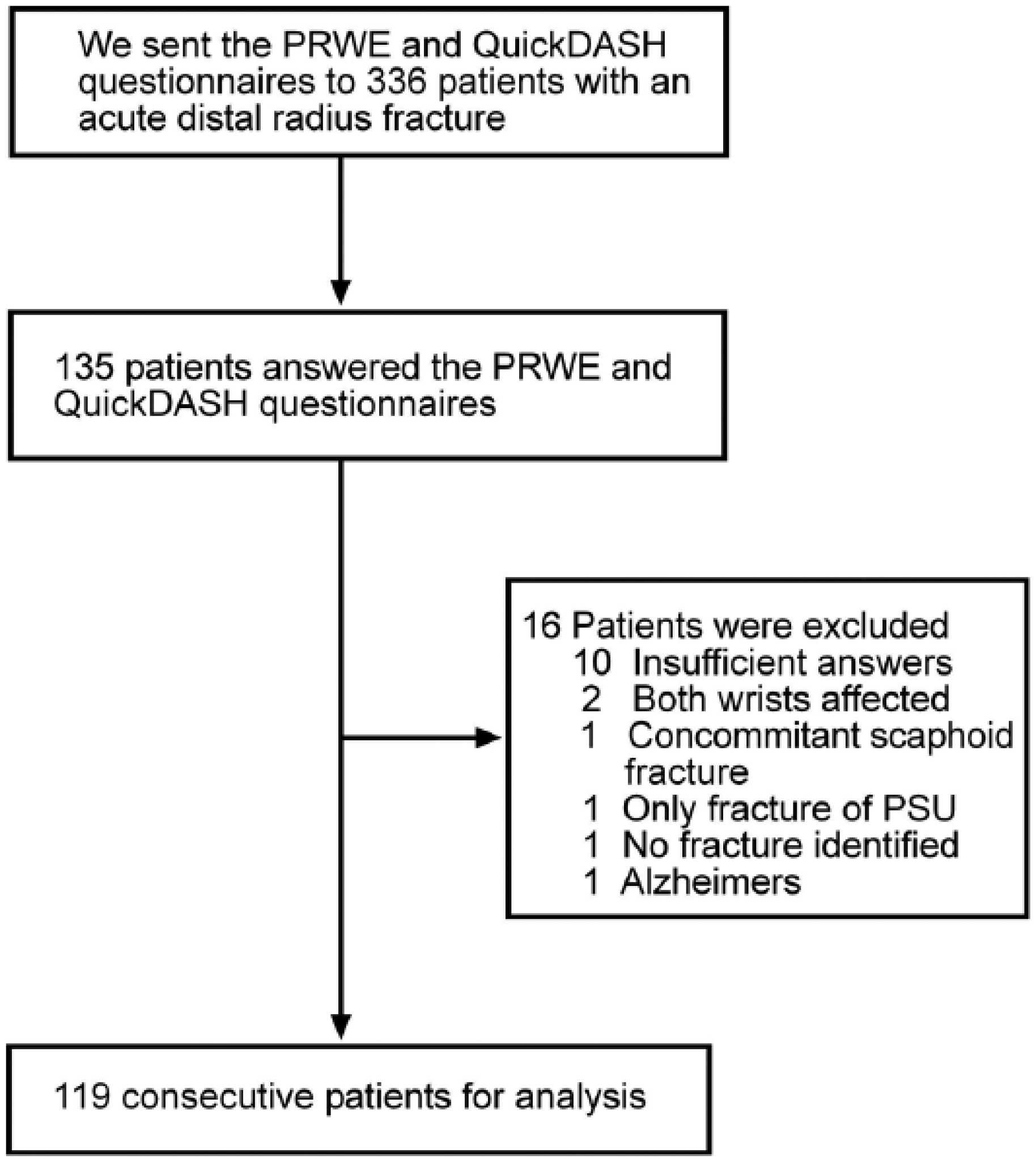
Retrieval of patients.
Reliability
We measured
Validity
The definition of validity is the extent to which the questionnaire measures what it intends to measure. Validity is divided into content, construct, and criterion validity.
Responsiveness
With responsiveness, we mean the ability of the outcome measure to change as the clinical status of the patient changes. We assessed the responsiveness by standard response mean (SRM) and Effect size (ES). SRM is defined as the mean of the difference in scores divided by standard deviation of the difference in scores. ES is defined as the mean of the difference in scores divided by the standard deviation of the initial scores. A benchmark of 0.8 or greater was considered large for both SRM and ES (15).
Statistics
All data were analyzed by an independent biostatistician (H.H.) with the Statistical Package for the Social Sciences (SPSS) for Windows version 22.0 (IBM SPSS corp, Armonk, NY). Values of p < 0.05 were regarded as statistically significant.
Ethical Approval Details
Study was conducted in three hospitals located in two different hospital districts. The permits for the study were obtained from the Ethics Committee of University of Helsinki (D.nr. 133/13/03/02/2008) as well as from the ethics committee of University of Tampere (D.nr. R11057). The study was conducted according to the Declaration of Helsinki. Data handling was performed according to Finnish data protection legislation.
Results
Translation and Adaptation Process
We translated the instruction of the PRWE questionnaire precisely without changes. The bolded parts of the text we kept as in the original version, but we changed the upper case to normal because in our opinion it gives a too imperative feel to the patient, which in turn can effect negatively on the response rate. We also translated the instructions for the pain and function section precisely without changes, but we combined the instructions for specific and usual activities into one paragraph as we thought it was unnecessary to have it stated twice. We left the sample scales off because we thought they were confusing for the patient and could be mistaken for a question. As in other translations, the first question of the function section caused us some problems because door knobs are not common in Finland. We therefore change the question to “turn a door handle” as door handles are widely used in households in Finland. At the end of the questionnaire, we added two questions asking for the affected side and the arm dominance since the original version lacks this important information. The addition of information about laterality to the questionnaire does not affect the scoring of the PRWE. In the pilot study, the questionnaire was considered both patient-friendly and clinician-friendly.
Patients
During the 6-month period, we enrolled 119 patients with a mean age of 60.6 years (range 18–94) in the study. One hundred (84.0%) were female and 91 (76.5%) patients were conservatively treated. In 60 (50.4%) patients, the dominant wrist was injured. The patients answered the PRWE and QuickDASH questionnaires at a mean of 52 days and 116 days. Demographic data about the patients can be seen in Table 1.
Patient demographics.
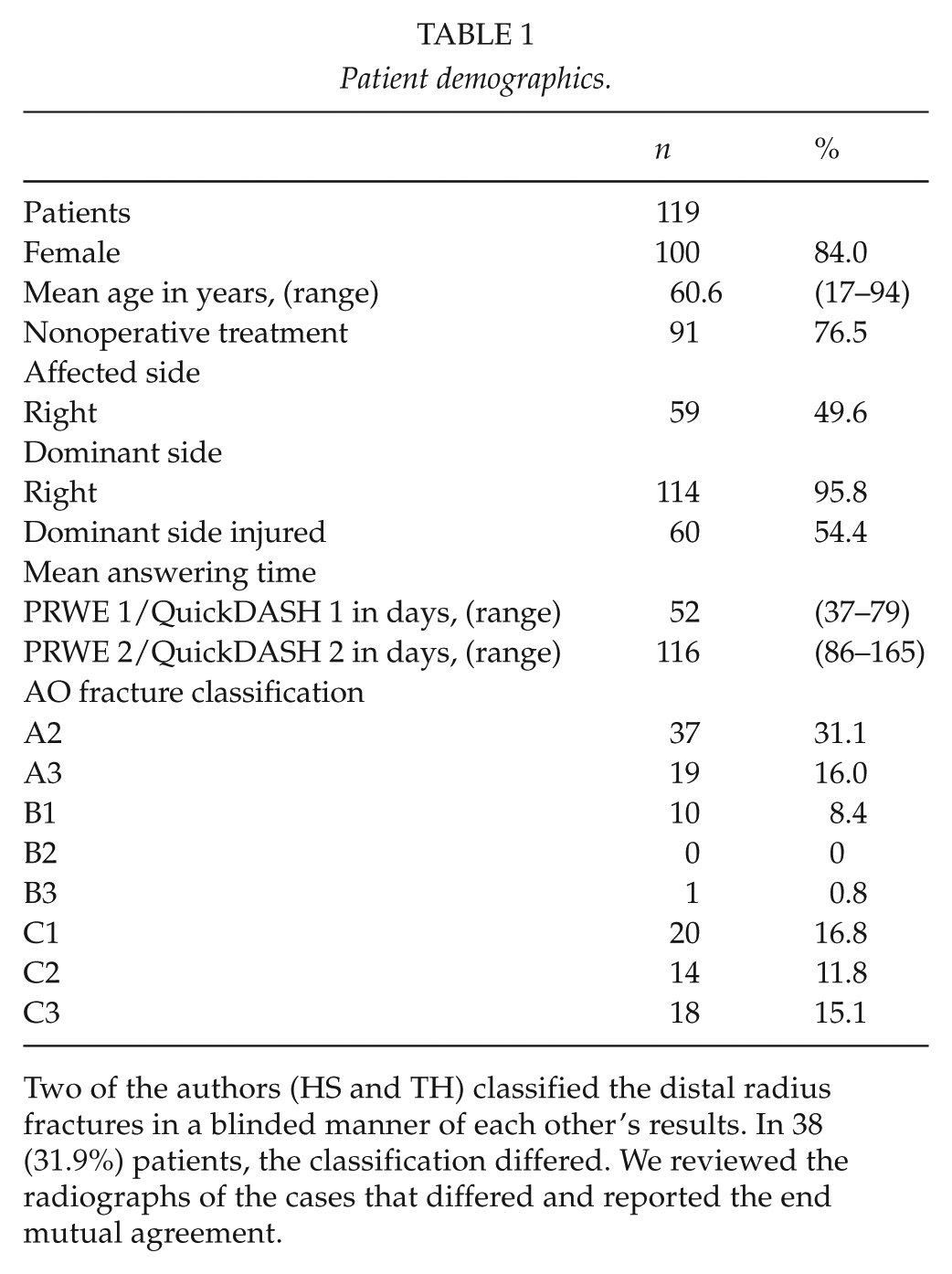
Two of the authors (HS and TH) classified the distal radius fractures in a blinded manner of each other’s results. In 38 (31.9%) patients, the classification differed. We reviewed the radiographs of the cases that differed and reported the end mutual agreement.
Reliability
The
Validity
Content validity
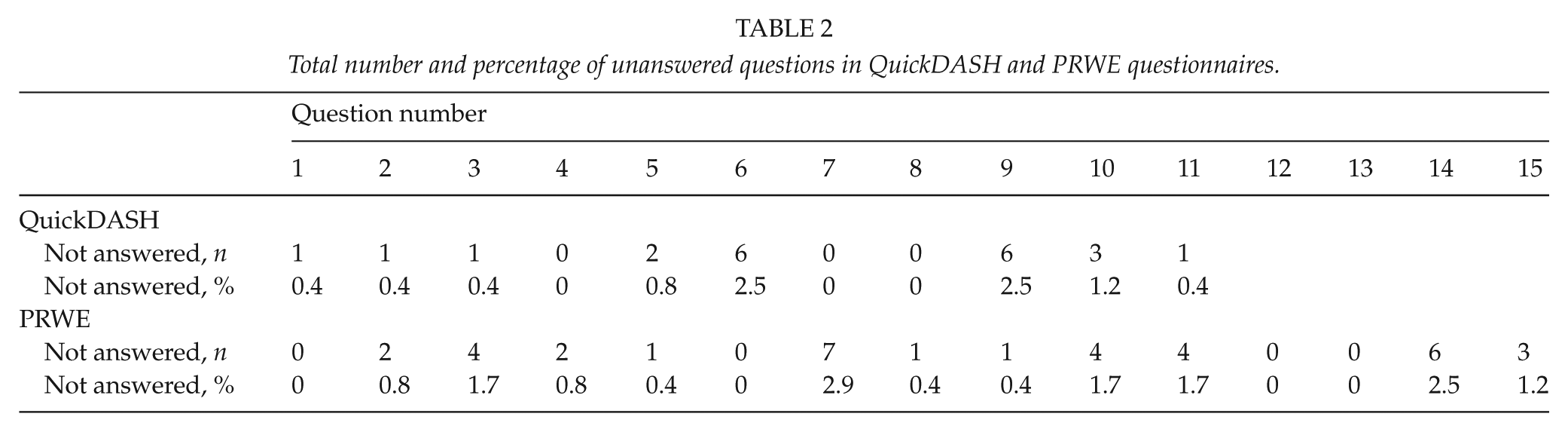
Although both PRWE and QuickDASH are considered patient-friendly, the response rate was low (35.4%; Fig. 2). Question numbers 6 and 10 in the QuichDASH were left unanswered by 2.5% of the patients. Questions 7 and 14 in the PRWE were left unanswered by 2.9% and 2.6% of the patients, respectively. The other unanswered questions in both questionnaires distributed evenly (Table 2). Ten patients (7.4%) were excluded due to unanswered questions or questionnaires. Of these 10 patients, five patients (4.2%) left one or more of the four questionnaires unanswered and were hence excluded, whereas five patients (4.2%) were excluded for leaving more than one question unanswered in one of the questionnaires. For patients failing to follow the instructions, the mean age was 83.9 years (range 49–87). We saw no floor or ceiling effect at the first time point at 2 months, but at 4 months, we saw a slight floor effect with six patients (5%) getting a minimum score of zero (best possible score).
Total number and percentage of unanswered questions in QuickDASH and PRWE questionnaires.
Construct validity
Correlation between the different subscales for PRWE and QuickDASH are shown in Table 3. As expected, the correlation was highest between the PRWE function and the QuickDASH function subscale (convergent validity) and lowest between the PRWE function and the QuickDASH symptom subscale (divergent validity).
Construct and criterion validity. For construct validity measurement, we compared the subscales of the questionnaires (PRWE and QuickDASH) with each other at 2 and 4 months. For criterion validity, we compared the questionnaires total score with each other.
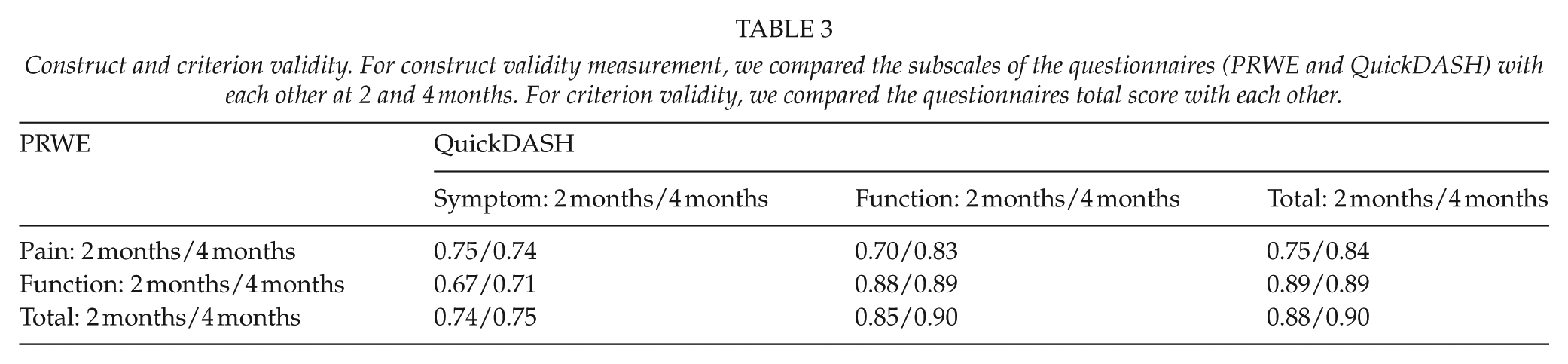
Criterion validity
We saw a significant improvement both for the PRWE and QuickDASH between the two time points. Pearson’s rank correlation coefficient for the total score was 0.88 at 2 months and 0.90 at 4 months (p < 0.01 for both; Table 3).
Responsiveness
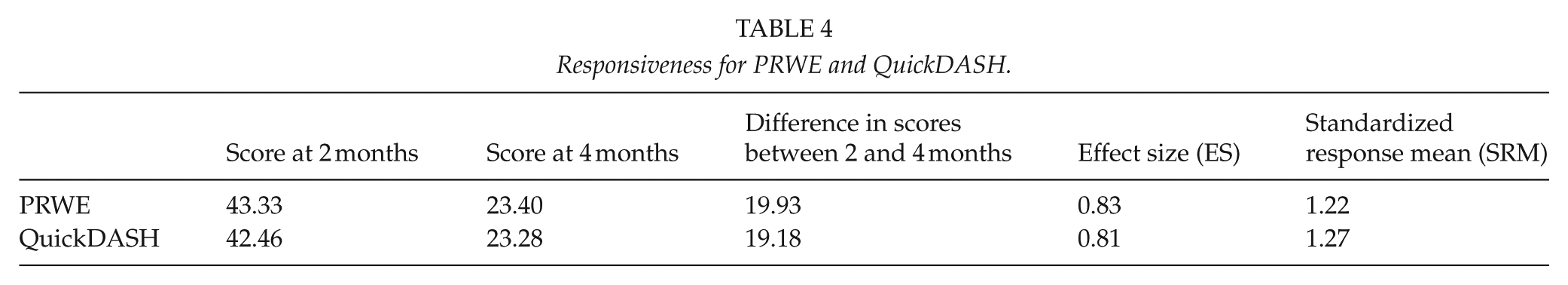
The responsiveness of the Finnish version of the PRWE indicated a large clinical change in the status of the wrist in patients with acute DRF between the two time points. The ES for PRWE was 0.83 and for QuickDASH, 0.81. The SRM for PRWE was 1.22 and for QuickDASH 1.27. Scores, effect size, and standardized response mean for PRWE and QuickDASH can be seen in Table 4.
Responsiveness for PRWE and QuickDASH.
Discussion
The translated and validated Finnish version of the PRWE proved to be reliable, responsive, and the scores strongly correlated with the QuickDASH. DASH and QuickDASH PROMs are widely used questionnaires to measure upper extremity pain and disability. The DASH and QuickDASH consider the upper extremity as one functional unit and are potentially influenced by concomitant upper limb problems (4), whereas PRWE is designed to be wrist-specific and therefore the recommended tool for measuring the outcome for wrist injuries. Moreover, Walenkamp et al. (16) recently determined the minimum clinically important difference (MCID) for PRWE among DRF patients to be 11.5 points, helping researchers and clinicians interpret the scores correctly and to estimate the required sample size in studies. Because of this, and a lack of validated wrist-specific PROMs in Finnish, we chose to translate and validate the PRWE. We chose to validate it against QuickDASH because the questionnaires have similar length and subscales. We preferred the QuickDASH over the DASH because it is shorter and the responsiveness and clinical utility are thus better (17, 18).
As door knobs are unusual in Finland, we changed the first question of the function section to “turn a door handle” instead. Turning a door knob is very similar to opening a jar, a question found in the QuickDASH function section. To investigate whether the question, “turn a door handle,” measures what it is supposed to measure, we assessed the correlation between the PRWE question, “turning a door handle,” and the QuickDASH question, “opening a jar,” and found the correlation to be strong, proving the alternative translation to be sufficient.
We chose the 2-month time point for the earlier assessment because we estimated that it was the first possible time point late enough for casts and ex-fixes to have been removed. However, the fracture healing and functional disability are still usually noticeable. We chose the later time point according to previous studies in the literature and for the longitudinal study design in order to see a significant improvement and healing between the two time points, which we saw.
There was a slight floor effect at the 4-month time point where 5% of the patients reported best possible score. This is well below the tolerance threshold of 15% (19). At a later stage, the floor effect would probably have been more significant, affecting the quality of the content validity. We detected only small differences in the percentage of unanswered questions in either of the two questionnaires during the study indicating a good content validity. The exclusions due to unanswered questions or questionnaires concentrated to the older part of the patient population. The average age of these patients was over 20 years more than DRF patients successfully answering the questionnaires. We suspect that this most likely reflects the reduced cognitive performance and/or increased co-morbidity (e.g. dementia) of these patients rather than content validity issues of the questionnaire. For the construct and criterion validity, the correlation was strong for the subscales and strong to very strong for the total score.
Some limitations of our study require discussion. We used the Finnish translation and cross-cultural adaptation of the QuickDASH in the validation process. The QuickDASH translation has been done according to proposed guidelines by Guillemin and accepted by the Institute for Work and Health (IWH) (20). However, it has not been validated against other outcome measures earlier (21).
Despite PRWE and QuickDASH being both patient-friendly and clinician-friendly, we saw a surprisingly low response rate of 35.4%. In our pilot study, we were able to reach significantly better response rate up to 72.0%. However, during that study, we sent the questionnaires to non-responsive patients up to three times and thus were able to reach such high response rate. Due to the multicenter study design, we were not able to do this within the time limits in the final study. The response rate during the first round of our pilot study was 44.0%. Also, in the pilot study, 32.0% of the patients were treated operatively compared with 23.5% in this study. Operatively treated patients had a significantly higher overall response rate (82.1%). Also, Schamber et al. (22) showed that completion rates are lower among older patients with comorbidities, factors which are typical for patients with DRFs. Interestingly, electronic data capture methods increased overall participation in their study. The idea to embed PROMs in the electronic health record and routinely start using PROMs in clinical practice offers an interesting opportunity to improve outcome and assess the effectiveness of the treatment (5, 23, 24).
In conclusion, we showed that the Finnish version of the PRWE is clinically practical, reliable, valid, and responsive. PROMs complement the traditional clinical outcome measures and have become increasingly important in addressing the effectiveness of treatments in an era of value-based healthcare. The validated Finnish version of the PRWE will provide an anticipated important tool for clinicians and researchers treating wrist injury patients.
Footnotes
Acknowledgements
We thank Kati Salo, Leila Soikkonen, and William Hellberg for excellent technical assistance with the translation of the questionnaire.
Supplementary Data
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Details of Informed Consent
Informed consent was obtained from all patients.
Funding
The work was supported by grants from Päijät-Häme central hospital.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
