Abstract
Background and Aims:
The detection of graft viability is challenging in the multiligament reconstructed knee. Magnetic resonance imaging gives structural information but lacks the capability to assess biological activity of the grafts. 18F-labeled fluorodeoxyglucose positron emission tomography combined with computer tomography is shown to be a sensitive method for imaging tissue metabolism and viability. The aim of the present study was to evaluate the feasibility of fluorodeoxyglucose positron emission tomography combined with computer tomography imaging in the detection of the replacement graft metabolism in multiligament reconstructed knees.
Materials and Methods:
Seven patients (17–44 years) with multiligament reconstructed knee underwent fluorodeoxyglucose positron emission tomography combined with computer tomography to evaluate the biological activity of replacement grafts. The degree of fluorodeoxyglucose uptake reported as standard uptake values from the region of interest was analyzed 3–24 months postoperatively.
Results:
In all patients, the fluorodeoxyglucose positron emission tomography combined with computer tomography showed increased fluorodeoxyglucose uptake in all replacement grafts at different follow-up time points. Furthermore, fluorodeoxyglucose was higher at femoral condyles of operated knees compared to contralateral reference values.
Conclusion:
This pilot study shows a significant increase in tendon graft metabolism during two first years of postoperative healing. The fluorodeoxyglucose positron emission tomography combined with computer tomography imaging seems to be adequate method of assessment of graft metabolism and viability during postoperative healing. The clinical value of fluorodeoxyglucose positron emission tomography combined with computer tomography imaging, however, warrants further evaluation with longitudinal studies with a larger patient population.
Keywords
Introduction
Graft healing after ligament reconstruction of the knee is a complex, partly poorly understood biological process (1). Based primarily on animal models, it is known how replacement grafts undergo different phases of healing in the intra-articular region and at the site of graft-bone incorporation (2–6). In ideal circumstances, these phases lead to graft healing and incorporation into host bone and revascularization.
The clinical healing of multiligament reconstructed knees seems to be well predicted in the vast majority of patients. However, in patients with abnormal postoperative recovery, the diagnosis of graft-healing failure sometimes poses a difficult diagnostic challenge (7, 8). Magnetic resonance imaging (MRI) makes it possible to obtain structural information on the graft, but lacks the capability to assess graft-healing activity and to ensure that the graft healing is biologically active. Positron emission tomography (PET) imaging with 18F-labeled fluorodeoxyglucose (18F-FDG) provides a unique and quantitative method to study tissue in vivo. FDG is transported into cells via facilitated transport, mainly GLUT1 and GLUT4 glucose transporters similarly to glucose. Rather than being specific to any pathological entity, FDG uptake reflects the metabolic activity of target tissue. Clinically, fluorodeoxyglucose positron emission tomography combined with computer tomography (FDG-PET/CT) has been used successfully for imaging of cancer metabolism and for the localization of infection and inflammation. In inflammatory processes, increased glycolytic activity is observed in leucocytes, macrophages, and other inflammatory cells, but the accumulation of 18F-FDG is also attributed to hyperperfusion and hyperemia. In this context, FDG-PET/CT imaging which combines both the functional image of the PET image with the anatomical image of computer tomography (CT) offers a novel functional modality to assess the healing and biological activity of replacement grafts.
The aim of this pilot study was to test the hypothesis that FDG-PET/CT imaging is a feasible imaging method for the evaluation of the graft metabolism after multiligament reconstruction of the knee. The ability to assess tendon graft metabolism could give new insights into tendon healing process. Furthermore, the study aimed to determine the impact of ligament reconstruction procedure on metabolic activity of host bone and intra-articular soft tissues.
Materials and Methods
Study Design
The conducted study was a retrospective registry study. From March 2006 to August 2008, seven patients underwent arthroscopically assisted multiple ligament reconstruction at an average of 12.7 months (range 3–24 months) following their initial knee injury. None of the patients were diagnosed diabetic and were otherwise healthy.
In five patients, the procedures were carried out in one stage, while in two patients in two stages. For these two patients, the first stage included high tibial osteotomy and posterolateral corner (PLC) reconstruction due to varus alignment and second stage included cruciate ligament reconstruction. After surgery, the patients were clinically followed for a period of 3–24 months. During this period, the operated knees (and the contralateral knees for comparison) were scanned once with a 64-row PET/CT scanner (GE Discovery VCT, General Electric, Wisconsin, USA). A static 180-s PET scan of one bed position was acquired 50 min after the injection of 370 MBq of FDG. The scan data were corrected for dead time, decay, and photon attenuation and reconstructed in a 128 × 128 matrix in three-dimensional (3D) mode. The final in-plane full-width at half-maximum (FWHM) of the system was 5–6 mm (9). In addition, low-dose CT images (120 kV, variable mA with the maximum of 80 mA, 3.75 mm slice thickness) were obtained. The radiation dose of used FDG-PET/CT imaging was 5.3 mSv (1.65 times higher than annual background radiation). Maximal standard uptake values (SUVmax) of the PET data were analyzed using GE Advantage Workstation (version 4.5). SUV values were normalized by body weight.
The regions of interest (ROIs) were determined manually to include only the tissues of interest (Fig. 1). The ROI area with a diameter of 7 mm (0.49 cm2) was placed on six different zones as follows: (1) the ligament/graft midsubstance, (2) the femoral and (3) the tibial ligament/graft-bone junction, (4) the Hoffa’s fat pad and the periosteal region of (5) the medial and (6) the lateral femoral condyles. These zones were also used for ROI analysis in intact, contralateral knee as a reference tissue. In visual analysis, these healthy reference regions did not express elevated glucose metabolism (Fig. 1).
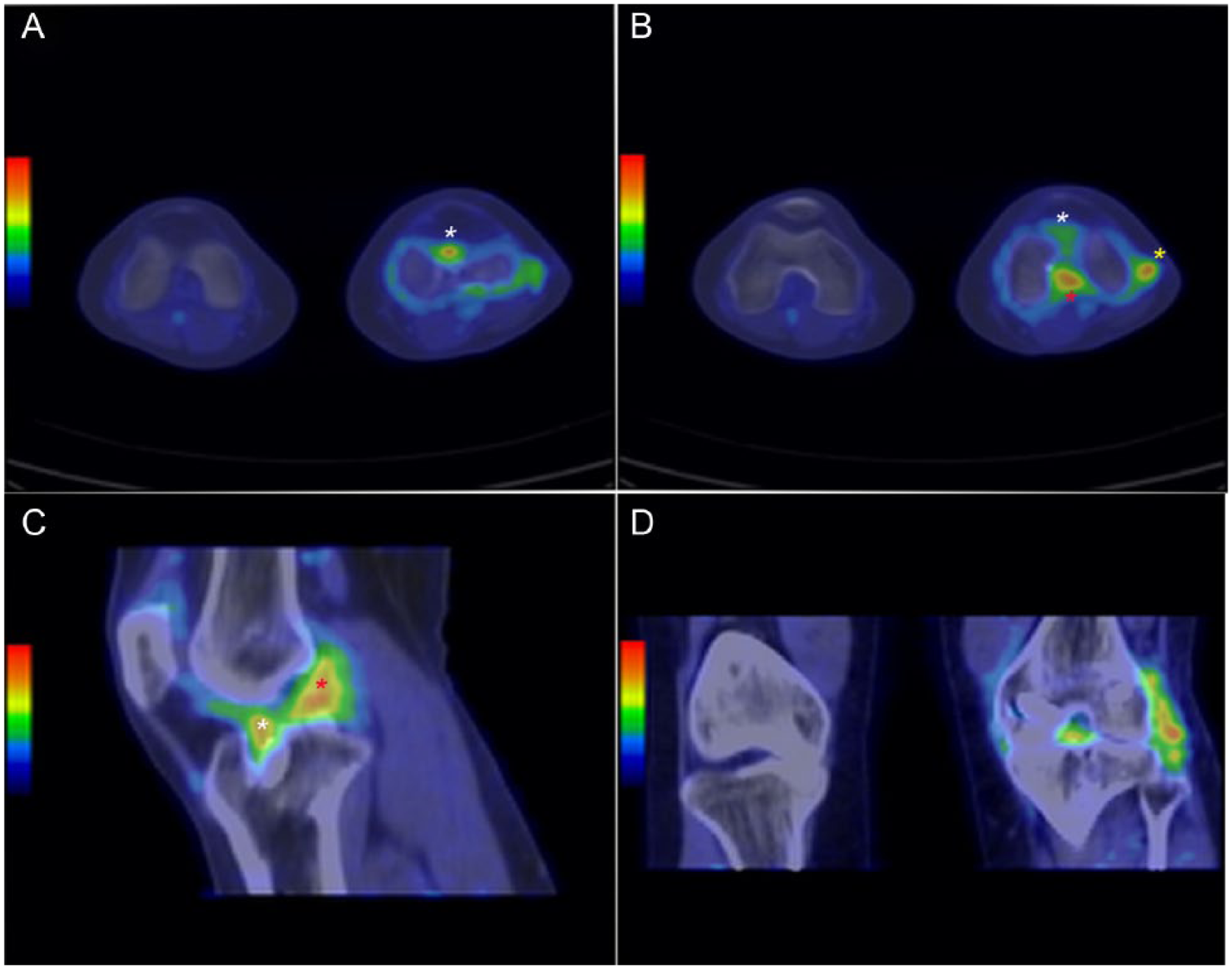
Axial (A,B), sagittal (C), and coronal (D) PET images of postoperative and contralateral healthy intact knee. The regions of interests (ROIs), graft midsubstance (red star), and graft-bone junction (white star) are shown in increased metabolic activity.
Patients were also pooled into two groups (3–12 months and 12–24 months) according to the postoperative time before the PET scan to estimate whether the observed metabolic activity in operated knees decreases over time.
Operative Technique
The anterior cruciate ligament (ACL) was reconstructed using bone–tendon–bone (BTB) allografts and secured by metal interference screw fixation. In the posterior cruciate ligament (PCL), a transtibial, single-bundle reconstruction using an Achilles tendon allograft was performed in all cases. Fixation on the femoral side was achieved with a metal interference screw, and on the tibial side with a bio-absorbable screw.
A figure-of-eight with a two-strand hamstring autograft or tibialis anterior allograft was used for PLC reconstruction. The graft was proximally and distally fixed with a metal interference screw. For the reconstruction of tibial collateral ligaments (medial collateral ligament (MCL)), the gracilis and semitendinosus tendons were harvested proximally and left attached distally. A four-strand graft was proximally fixed with a metal interference screw and distally by suture anchors.
In three patients, procedures of partial meniscectomies (n = 4) and chondral debridements (n = 3) were performed during ligament reconstruction.
After surgery, patients wore a brace at full extension for 4 weeks. Bearing weight on the knee was not allowed for six weeks. A hinged brace was then worn for 3 months.
Statistical Analysis
The above-mentioned SUVmax of ROIs were statistically analyzed and compared using parametrical tests in the operated and contralateral unoperated knee. The normality, homogeneity, and the check for outliers were verified from histograms which made the data suitable for parametric analysis. Repeated measures analysis of variance (ANOVA) with time as repeated factor was used. Least significant difference (LSD) was used as post hoc test in determining the statistical differences between operated and unoperated ROIs. Significant difference was considered when p ⩽ 0.05. PASW 18.0.0 for Windows was used for all statistical analyses.
Results
FDG-PET/CT was performed on seven multiligament reconstructed knees, in four men and three women (Table 1). The mean age of patients was 29.1 years (range 17–44 years). In the FDG-PET/CT-scan, all 20 reconstructed ligament grafts were visible in all seven study patients and were analyzed semi-quantitatively for glucose metabolism using SUVmax as a rate of metabolic activity. Observed activity was compared to intact, symptomless contralateral knee.
Patient characteristics, injury pattern, graft type, and FDG uptake (each graft SUVmax, graft midsubstance, and tunnel aperture).
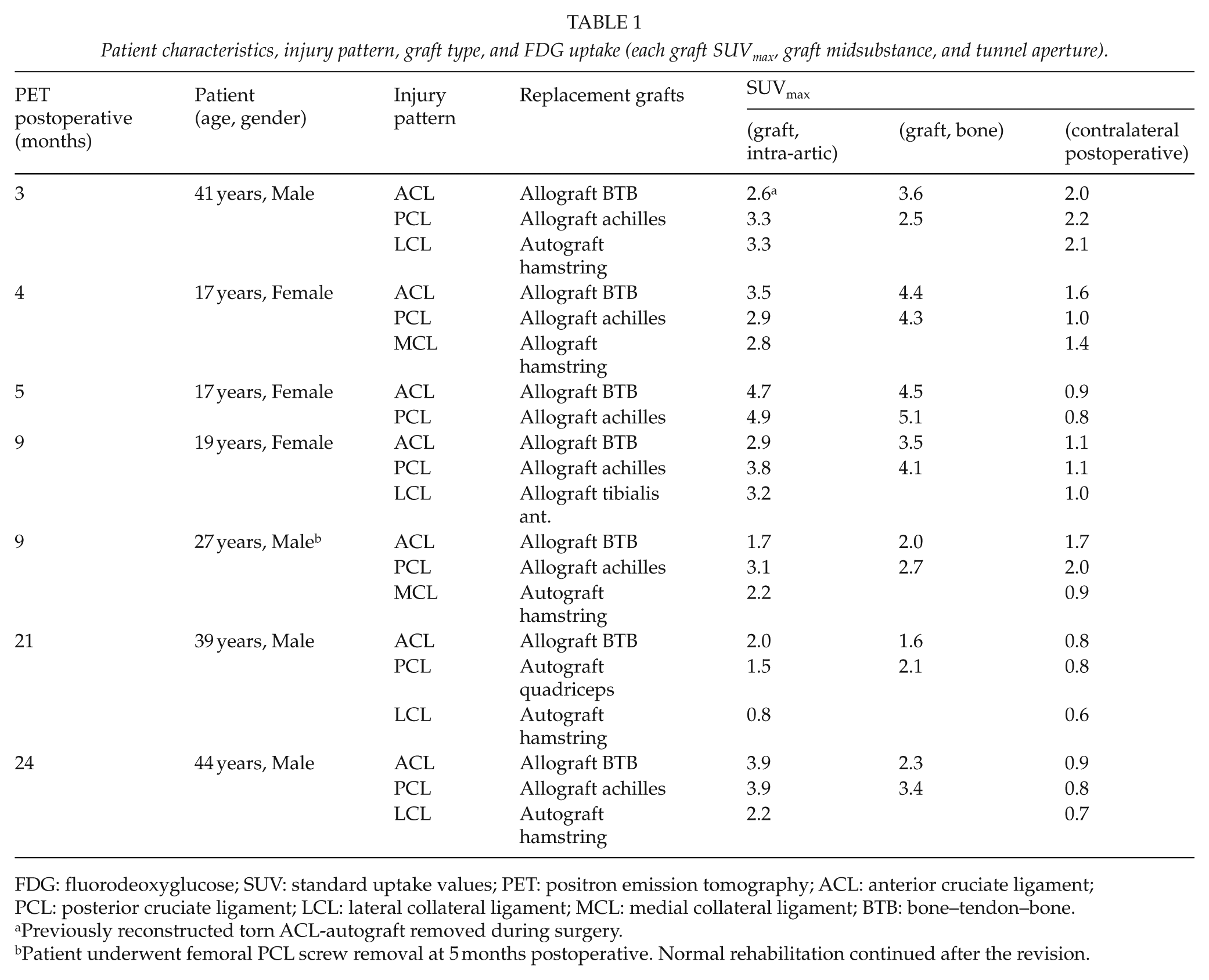
FDG: fluorodeoxyglucose; SUV: standard uptake values; PET: positron emission tomography; ACL: anterior cruciate ligament; PCL: posterior cruciate ligament; LCL: lateral collateral ligament; MCL: medial collateral ligament; BTB: bone–tendon–bone.
Previously reconstructed torn ACL-autograft removed during surgery.
Patient underwent femoral PCL screw removal at 5 months postoperative. Normal rehabilitation continued after the revision.
Patient characteristics are provided in Table 1. Six of seven study patients had clinically normal postoperative recovery. One patient (27 years, male) had acute mechanical locking of the knee at 5 months after reconstruction. The x-ray pictures showed a loose PCL femur interference screw in the knee joint. The screw was removed arthroscopically, and the PCL graft seemed to be initially incorporated. After the screw removal, the rehabilitation continued as planned and the patient was included for further analyses.
The average SUVmax for intact cruciate and collateral ligaments was 1.2 ± 0.1 (range 0.6–2.2), while the SUVmax for replaced cruciate grafts was 2.8 ± 0.9 (range 1.5–4.9). Accordingly, the SUVmax was significantly higher in ACL (p = 0.003), PCL (p = 0.001), and collateral ligaments MCL and lateral collateral ligament (LCL) (p = 0.050 and p = 0.045) in operated knees (Fig. 2). The average SUVmax of reconstructed collateral ligaments was 2.4 ± 0.9, about 2.5 times the values observed in intact collaterals. In addition, we measured the SUVmax at Hoffa’s fat pad and the cortical bone in femoral condyles (Fig. 2). The SUVmax of the medial (p = 0.004) and lateral (p = 0.007) condyles of the operated knees was, on average, 2.4 ± 0.7, while the reference value of contralateral condyles was 1.3 ± 0.8. At Hoffa’s fat pad, the SUVmax was 0.9 ± 0.3 in the operated knee and 0.6 ± 0.3 in the contralateral knee (p = 0.045).
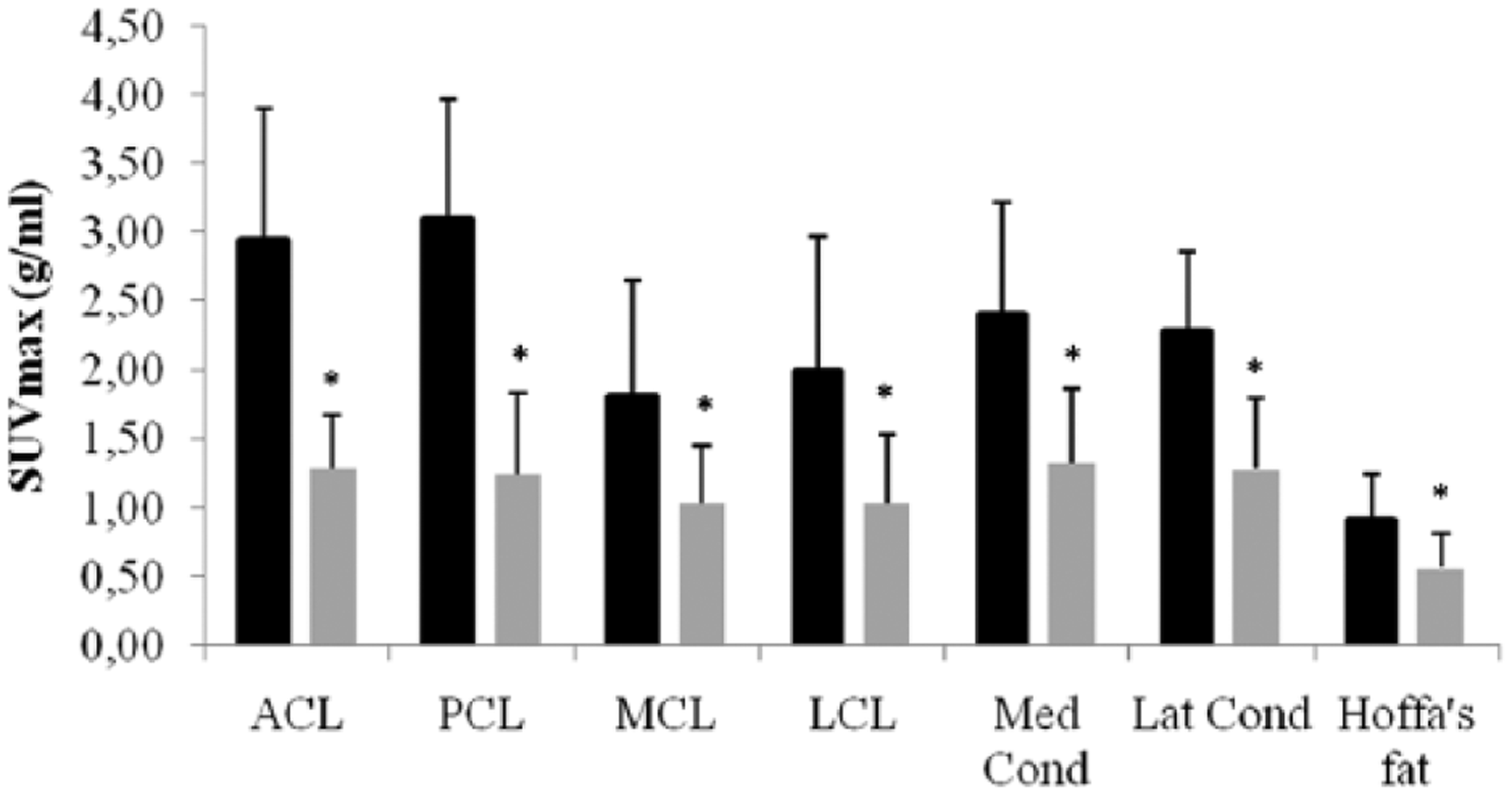
Differences in SUVmax (Mean [SD]) between postoperative (black) and contralateral knees (gray) at different regions of interest.
Both ACL and PCL grafts, their drill holes showed highly elevated activity that correlated to the time elapsed after surgery. Due to heterogeneity of patient population, there was marked inter-individual variation as expected in the observed metabolism in the graft, as well as at graft insertion sites. The metabolic activity in PCL compared to intact tissue was slightly higher than in ACL. The highest value in intra-articular ACL was 522% and 613% in PCL compared to values in intact knee.
SUVmax were higher in patients (n = 5) scanned 3–12 months after the operation compared to patients (n = 2) scanned after 12–24 months, respectively (Fig. 3). However, no statistical differences were seen between the SUVmax in both of these ACL and PCL groups.
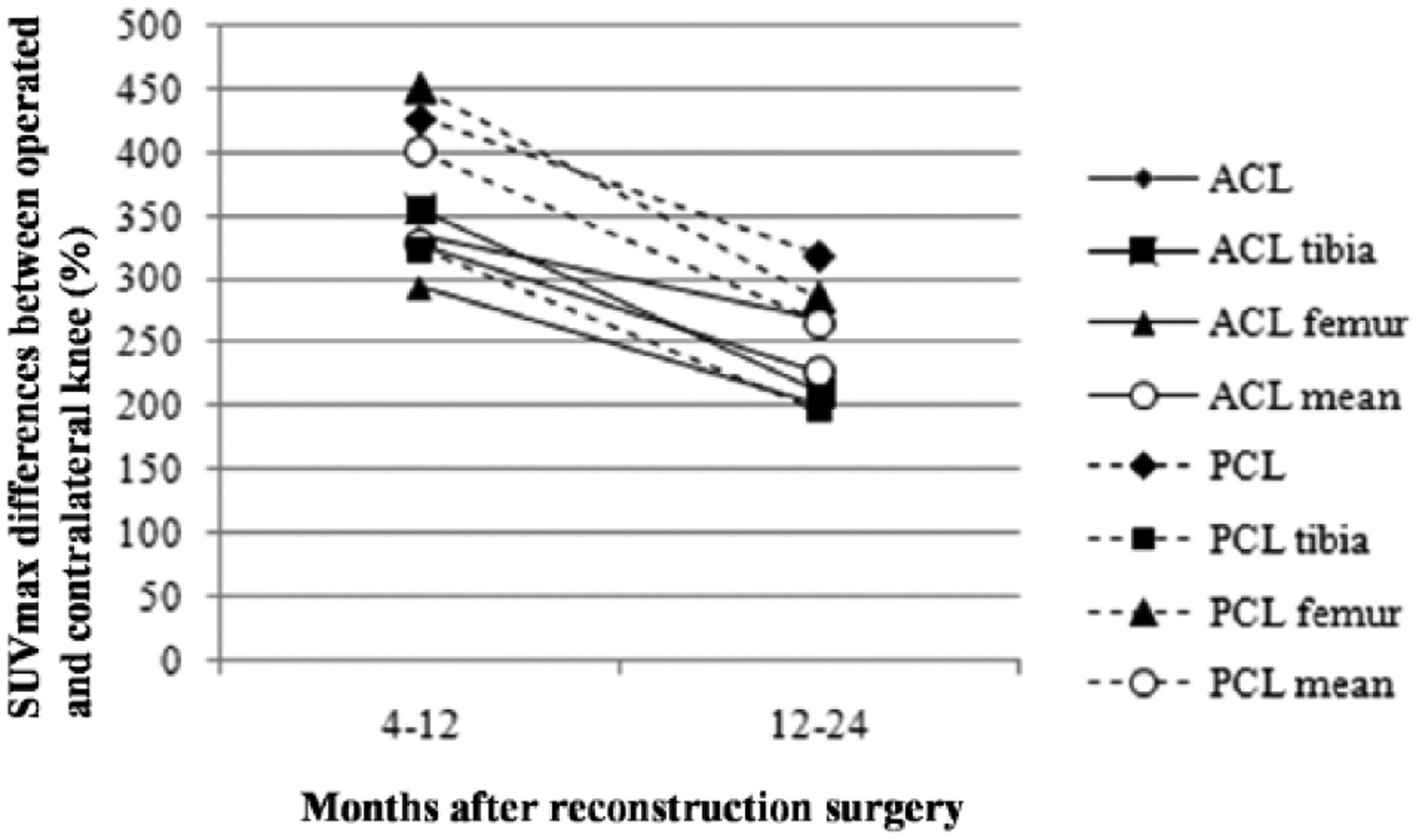
Differences (%) in mean SUVmax values of different regions of interest in ACL and PCL grafts in 3–12 months (n = 5) and 12–24 months (n = 2) postoperatively between the operated and contralateral knee.
Discussion
In this preliminary pilot study, we found that in replacement grafts of the knee, the FDG uptake is increased at the ligaments themselves, and at their fixation points at all follow-up points. Furthermore, at all follow-up points, the FDG uptake was significantly higher in the periosteal region of femoral condyles compared to contralateral reference values.
Due to obvious technical difficulties, knowledge about the graft-healing process after knee ligament reconstruction in humans is limited (10). Based on animal and a few clinical studies, it seems that after ligament reconstruction, replacement grafts have to undergo several different phases of healing resulting in graft-bone incorporation and ligament revascularization. Amiel et al. (11) proposed the term “ligamentization” to explain the histological changes in the graft during this process. Evidence shows that replacement tissues undergo stages consisting of avascular necrosis (0–2 months), cellular repopulation (<1 year), collagen remodeling, and maturation (1–3 years). Finally, after 3 years, the inserted graft becomes quiescent. It has been shown that inflammatory cells and young granulation tissue containing capillary vessels, fibroblasts, and macrophages have significant FDG uptake (12 –14). Previously, two reports have shown that traumatic ligament injury presents elevated glucose metabolism that can be visualized with PET/CT imaging (15, 16). Therefore, it is reasonable to think that with PET imaging, the above-mentioned healing phases of replacement tissue could be followed from the acute inflammatory reaction to the revascularization of the ligament, and finally to complete healing.
Despite the relatively high rate of positive outcome reported (7), graft failures continue to occur. Especially in the case of a painful, loose, multiligament reconstructed knee, the detection of non-traumatic failure of biological graft incorporation is still a challenge. More extensive studies following the normal healing process in multiligament reconstructed knees with FDG-PET/CT might help to identify patients with non-traumatic graft failure. In this context, in the present study, one patient had a revision surgery due to the femoral PCL screw loosening at 5 months after primary ligament reconstruction. After removing the loose PCL screw, the patient’s clinical recovery and rehabilitation continued as planned. Furthermore, the detected SUV values in the area of PCL allograft were elevated compared to values of intact contralateral PCL at 9 months postoperatively. Based on the uncomplicated clinical recovery after screw removal and SUV values that were on the range of other study patients during first postoperative year, it can be assumed that the PCL allograft was integrated into femoral tunnel prior screw loosening and the viability of the graft was maintained despite the unexpected femoral screw loosening.
It has been previously shown that FDG-PET can be used for assessing glucose metabolism in native tendons at rest and during exercise (17). We postulated that the FDG-PET imaging method also possesses possible qualities to evaluate tendon, ligament, or allograft healing by demonstrating increased glucose metabolism which reflects the inflammatory process and the activity of granulation tissue (12, 13). In the present pilot study, the uptake of FDG was increased in all replacement grafts and was higher at all follow-up time points compared to the values of comparable regions of intact contralateral ligaments. Based on earlier data, high early SUV of the grafts are likely to represent postoperative inflammation which gradually fades. The increased glycolytic activity could be a sign of disturbing or non-beneficial healing when surgery is not likely to explain observed metabolism any more (18, 19). Sorensen et al. (20) investigated bone metabolism with 18F-Fluoride and PET. They found increased activity at the bony fixation points of ACL grafts until 7 months after the operation, but normal values at 22 months. They interpreted this as a sign of increased bone turnover related to the incorporation of the graft. However, with this, tracer graft metabolism and its healing process cannot be assessed since 18F-Fluoride accumulates only in bones according to its osteoblastic activity. It could be hypothesized that metabolic activity in tendon grafts decreases during the uncomplicated healing process over time as in bone fixation points. The assessment of this healing process could not be addressed in the present pilot study due to lack of repeated PET scans for the same subjects. However, when comparing patients who have less than a year from surgery (depicting cellular repopulation phase) to patients who have more than a year from operation (collagen remodeling and maturation phase), our data suggest that metabolic activity in tendon grafts seems to decrease over time. Currently, it is not known how long it takes until tendon metabolism reaches the same level as in intact regions and whether the healing pattern is similar in tendon grafts and in bony fixation points. Even during uneventful recovery, FDG avidity in the present study was considerable for at least 21 months in all investigated locations in the knee. To our knowledge, this is the first study to assess graft metabolism with FDG-PET/CT. In the present study, SUVmax were higher in 3–12 month group compared to 12–24 month group postoperatively, which is line with the results of Sorensen et al. (20). However, these results need longitudinal study design with higher participant count, in order to draw more specific conclusions for clear trend of decrease in SUVmax.
Interestingly, also the periosteal region of femoral condyles demonstrated increased SUVmax. It is possible that it reflects the immunologic response to allografts. Theoretically, the multiple drill holes and number of interference screws could act as stress enhancers which again predispose fatigue and periosteal reaction of condyles (21). Studies evaluating how stress distribution around a distal femoral cortical stress evolves over time are also needed.
The main limitation of the study was the small number of patients. Each patient was only imaged once, and values at different time points represent different patients. In addition to limited amount of study participants, the study used both allo- and autografts. Currently, it is not known whether the metabolism in auto- or allografts differs after surgery. It could be hypothesized that allograft would provoke more immune system than autografts and this would cause higher inflammatory reaction and thus higher glucose metabolism around graft. However, this has not been proven, which gives issue for future studies. Given the pilot nature of this descriptive study and above-mentioned limitations, the results of the present study warrant a larger longitudinal study with biopsy confirmation of the pathological and physiological processes involved. However, in this pilot study, our hypothesis was to evaluate the overall feasibility of FDG-PET/CT in detecting graft activity at knee region. For this purpose, the number of pilot patients and the study protocol were, in our opinion, acceptable.
Conclusion
This pilot study suggests that FDG-PET/CT is a novel method to evaluate ligament graft viability and metabolism in multiligament reconstructed knees. However, future longitudinal studies with larger patient populations and biopsy confirmation are needed before definite conclusions about the graft-healing phases in humans can be made.
Footnotes
Acknowledgements
We thank biostatistician Elina Kokkonen for her advice and expertise concerning the statistical analysis.
Declaration of Conflicting Interests
None of the authors have any conflict of interest regarding the study in question.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
