Abstract
Background and Aims:
Endoscopic submucosal dissection is a minimal invasive method for an en bloc resection of dysplasia or early cancer in the stomach. It was developed in Japan in the 1990s, but thus far has not been widely adopted in Western countries. The aim of this study is to report from the introductory phase of gastric endoscopic submucosal dissection in a small-volume Scandinavian center.
Material and Methods:
The 15 first patients operated on with endoscopic submucosal dissection in the stomach at the University Hospital of North Norway were consecutively registered in a prospective database and evaluated as a quality assurance study. The indications for the procedures were diagnostic or therapeutic.
Results:
Three patients had benign lesions, three had low-grade dysplasia, five had high-grade dysplasia, two had neuroendocrine tumors, and two had early gastric cancers (T1b). The R0 resection rate was 83%. One neuroendocrine tumor was directed to surgery. No recurrences have been detected after the median of 6 months, and with the exception of one delayed bleeding being treated endoscopically, no serious complications occurred. Two perforations were perioperatively closed with clips.
Conclusion:
This is the first report on gastric endoscopic submucosal dissection in Scandinavia. Preliminary results from an introductory phase indicate that endoscopic submucosal dissection for dysplasia or early gastric cancer may be conducted safely and with acceptable results even in a small-volume center, assuming that surgeons and gastroenterologists in a region direct cases to one individual who can learn this skill.
Keywords
Introduction
Open or laparoscopic gastric resections are associated with a significant complication rate in general (1), as well as in Scandinavian hospitals (2, 3). Although the severity of a gastric cancer disease may contribute to the complication rates per se, it is the invasiveness of the surgical procedures that is the primary reason for the complications. Thus, a less invasive laparoscopic approach to surgery on the stomach resulted in fewer complications compared to open surgery (1, 4). Furthermore, for dysplasia and early gastric cancer, an endoscopic resection is an even less invasive procedure compared to open or laparoscopic surgery, thereby representing an alternative to traditional surgery for some patients.
Endoscopic submucosal dissection (ESD) was developed in Japan in the 1990s as a new endoscopic method to help achieve an en bloc resection of superficial lesions in the rectum (5). Gastric ESD is not only a method for the removal of dysplasia but also useful for diagnostic resections of submucosal lesions and the treatment of early cancers.
Due to a high degree of subspecialization, and high patient volumes related to a high incidence of early gastric cancer, this challenging technique is routinely performed in Asia today, with satisfying long-term survival rates and low complication rates (6).
The implementation of ESD in Western countries has been slow, which may be attributed to a lack of structured training (7), an insufficient centralization of patient volumes, and a lower incidence of gastric premalignant lesions in Western countries compared to Japan (8). There have been a few reports on Western experiences with gastric ESD (9–12), but so far none from Scandinavian countries. Here, we report from the startup phase of gastric ESD in a Scandinavian university hospital; an implementation made possible by inter-disciplinary collaboration, evaluation in multidisciplinary teams, and by directing all cases to one surgical endoscopist. The aim of this study is to demonstrate that gastric ESD may be implemented in a small-volume Scandinavian center with acceptable results and safety.
Methods
From February 2011 to December 2014, 15 patients with lesions in the stomach were operated on with ESD and prospectively registered in a database at the Department of Gastrointestinal Surgery, University Hospital of North Norway in Tromsø, Norway. The histology of the preoperative biopsy was inconclusive, benign, or pre-malign, while the selection of patients suitable for endoscopic resection was based on evaluations in multidisciplinary teams. No patient with cancer in preoperative biopsies was enrolled for ESD, and all patients received information on the procedure, with an emphasis on potential complications, including risks of perforation and delayed bleeding. The evaluation of the implementation of gastric ESD, as a procedure already established in other countries, is categorized as a quality assurance project with no requirement for disclosure or consent according to the rules of the Norwegian regional ethical committee.
All patients were operated on under general anesthesia. A gastrointestinal (GI) surgeon (C.R.) with extensive experience in therapeutic endoscopy performed all the procedures with the exception of one procedure, which was performed in collaboration with a second surgeon. The ESD operations were conducted with a gastroscope (EG-3490K; Pentax, Tokyo, Japan), with a separate channel for water flushing. A small volume of a mixture of a colloid solution (Voluven; Fresenius Kabi, Halden, Norway) (100 mL), indigo carmine (1 mL), and adrenaline (0.5 mg) was injected into the submucosa to help elevate the lesion. A 2.0-mm Dual knife (Olympus, Hamburg, Germany) and an It-knife (Olympus) were used for an incision of the mucosa and a dissection of the submocosa (Fig. 1). Hemostatic forceps (Coagrasper; Olympus) were applied for hemostasis, and in a few cases, a diatermic snare (SD-230U-20; Olympus) was applied at the end of the operation if the most central part of the stalk underneath the lesion was difficult to access. The specimens were removed from the stomach with a basket (Roth Net; US Endoscopy, Mentor, OH, USA) or with endoscopic grasper forceps.
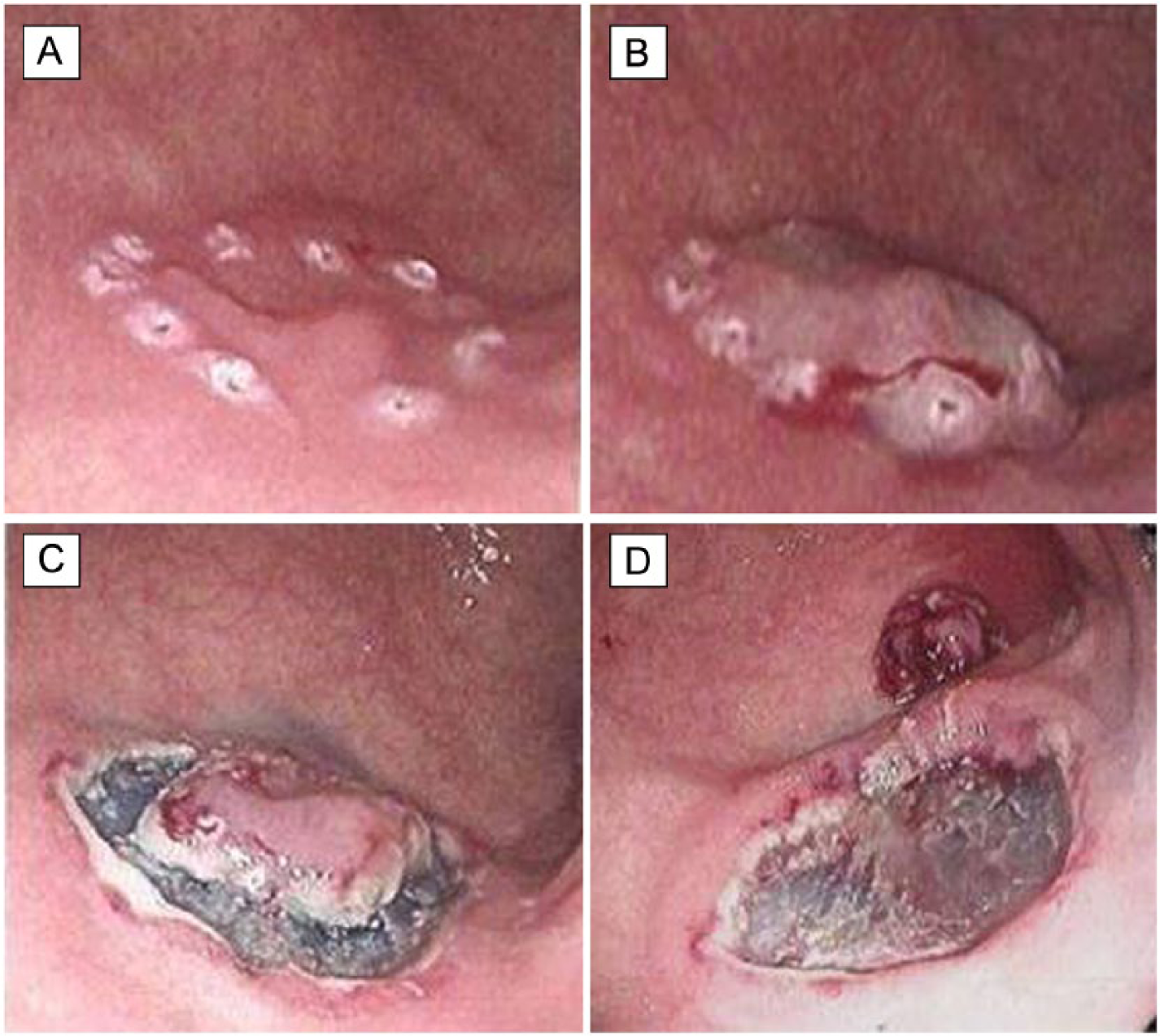
Standard procedure for gastric ESD (third case, Table 1): A) diathermy marking of resection margins around a small lesion with HGD in the antrum; B) elevation of lesion with submucosal injection of a colloid solution with indigo carmine and adrenaline indicates a non-invasive lesion; C) incision of the elevated mucosa with a Dual knife and initial submucosal dissection around the lesion with an It-knife; and D) the resected specimen located in the upper right corner of the picture before it is retrieved in a net. The remaining submucosal resection basis is macroscopically homogeneous with no sign of invasive cancer. Prophylactic hemostasis of submucosal vessels and bleedings is performed at this stage. Histology of the specimen showed an LGD with free resection margins.
ESD-related complications were consecutively registered in the database. Delayed bleeding after gastric ESD is defined as one of the following: hematemesis or melena, unstable vital signs, or hemoglobin loss >2 g/dL (13). Perforations caused by ESD are divided into macro- and microperforations. Macroperforations are noted during the procedure and can be treated with endoclips whereas microperforations are diagnosed by free air on plain X-rays after the procedure, as defined by Jeon et al. (14). Perforations were closed with over-the-scope clips (OTSC-clips; Ovesco, Tübingen, Germany). Prophylactic antibiotics were not routinely administered, but were given as a single dose in case of any perioperative perforation. A single dose of proton pump inhibitor was given to patients with a history of peptic ulcer disease.
Results
In all, 15 patients, 11 males and 4 females, were included in the study. One patient was excluded because ESD was technically impossible due to too much peristalsis. The median age of the patients was 71 years (26–81 years), and the median operation time was 115 min (60–249 min). After the operation, patients were observed either at the hospital hotel or in the ward, with a median of 1 night (0–2 nights).
The pathological and therapeutic results are summarized in Table 1. In total, 11 of the lesions were located close to the pylorus or in the antrum, 3 in the corpus, and 1 in the cardia. According to the Paris classification for superficial neoplastic lesions, the morphology of the lesions was IIb for six, Is for two and Isp for two lesions. Moreover, four lesions were submucosal, while one was superficial and benign.
Anatomy, morphology, histological findings, and clinical outcomes of ESD cases.
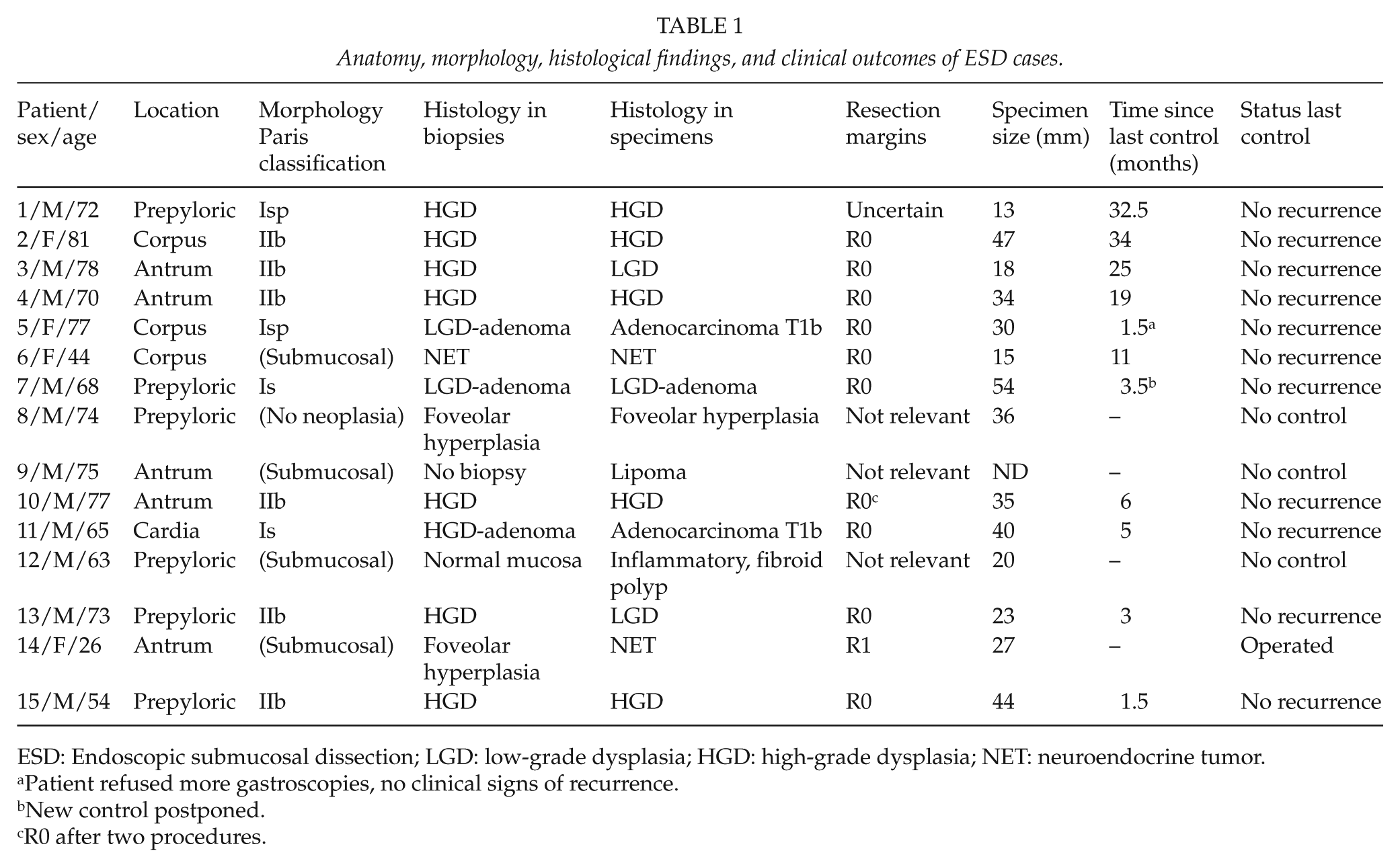
ESD: Endoscopic submucosal dissection; LGD: low-grade dysplasia; HGD: high-grade dysplasia; NET: neuroendocrine tumor.
Patient refused more gastroscopies, no clinical signs of recurrence.
New control postponed.
R0 after two procedures.
The histologic examination of the preoperative biopsies of the polyps and the irregular flat mucosa revealed seven patients with high-grade dysplasia (HGD) and three with low-grade dysplasia (LGD). In the superficial biopsies of the submucosal tumors, there was one neuroendocrine tumor (NET), two foveolar hyperplasia and one normal mucosa. One patient with a submucosal tumor had no preoperative biopsy.
Endoscopic ultra sound (EUS) was performed in 4 of the 15 patients and was inconclusive in 2 patients, whereas there was a lesion with no submucosal invasion in 2 other patients. Eight patients went through a computed tomography (CT) scan, and in five of these the lesion was not detected, two were inconclusive due to diffuse findings, and in only one was it possible to define a submucosal tumor. The patient with a preoperatively diagnosed NET went through an octreotide scintigraphy that was negative.
The median size of the specimens was 32 mm (6–54 mm). In 9 of the 15 patients, the histological diagnosis was the same in the specimens as in the biopsies. In the submucosal tumor without a preoperative biopsy, we found a lipoma (Fig. 2), and the submucosal tumor with normal mucosa contained an inflammatory, fibroid polyp. On the other hand, the histologic examination of one submucosal tumor with foveolar hyperplasia in the biopsy revealed a NET invading the muscularis propria with an unfree resection border, so this patient was therefore referred to laparoscopic resection. However, no tumor cells were detected in the laparoscopically resected specimen containing the basis of the tumor.
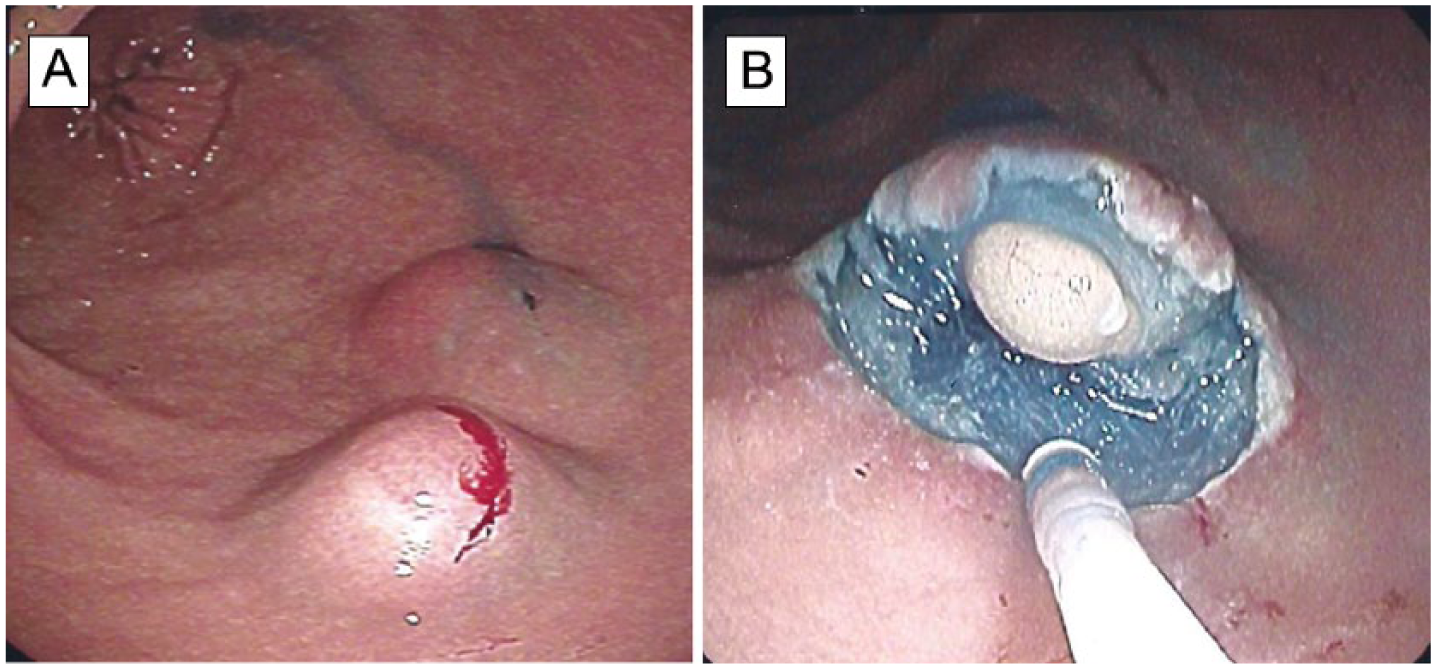
A) Two submucosal tumors in the antrum region after lifting with submucosal injections of the colloid solution (ninth case). B) After semi-circular incision of the mucosa and some submucosal dissection, a lipoma is clearly visible. A histologic examination of the specimens confirmed the diagnosis.
The other patients were R0 resections except for one with microscopically uncertain free margins, but she had no recurrences almost 3 years after the procedure. One patient needed a supplementary ESD procedure in order to obtain R0 status, whereas we resected another patient with “recurrent lesions” two times after the primary ESD and found only hyperplastic tissue (granuloma).
In two patients, one with LGD (5th case, Fig. 3) and one with HGD (11th case) in the biopsies, we detected a T1b adenocarcinoma in both cases. The submucosal invasion depth below the muscularis mucosa was 8.0 and 1.9 mm and the tumor size was 30 and 9 mm, respectively. The first of these patients was not directed to a gastric resection or a gastrectomy because of advanced age and comorbidity, while the second had a serious lung disease.
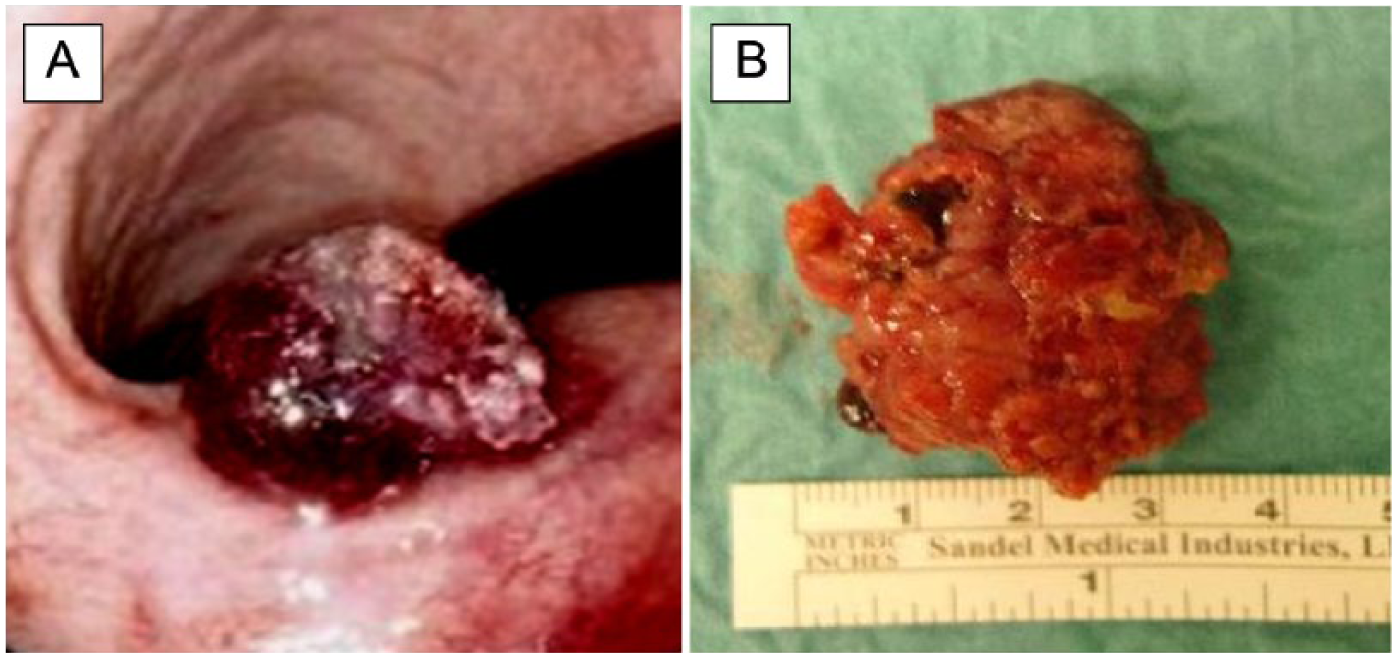
ESD for gastric tumor containing adenocarcinoma (fifth case). The protruding tumor in the angulus region A) before and B) after resection.
All 11 patients with dysplasia or cancer (except 1 with NET who was directed to surgery) have been controlled with gastroscopy and biopsies if necessary, with the last control after a median of 6 months (1.5–34 months). No recurrences have been detected thus far, although the patient with an endoscopically removed T1b cardia cancer later developed an esophageal cancer in his Barrett’s metaplasia.
One of our patients, the fourth case, presented with hematemesis, melena, and a hemoglobin loss of 4.4 g/dL, but with no unstable vital signs the day after the procedure. He was treated with a transfusion of two units of erytrocytes, an infusion of tranexamic acid, and endoscopic injections of adrenaline at a bleeding point at the rim of the resection field. Thereafter, the hemoglobin was stable, the nasogastric tube produced no more blood, and he was discharged the next day without any further bleeding episodes.
Two peroperative macroperforations occurred during the dissection of the two NET lesions (6th and 14th case). Both were closed with large over-the-scope clips at the end of the procedure after finishing the dissection. They received a single dose of prophylactic intravenous (i.v.) antibiotics during the procedure, and based on clinical evaluations, no indications for post-procedural X-rays were found. In the sixth case, a 1- to 2-mm diameter hole was observed, in addition to free air in the abdomen. Free air was evacuated through a needle catheter inserted into the inflated abdomen below the left arcus costalis; the patient expressed no symptoms and was discharged the next day. In the 14th case, a 2-mm hole was detected, although there were no clinical signs of free air in the abdomen. Based on the localization of the tumor on the dorsal part of the antrum, this hole was most probably a perforation to the bursa omentalis, which may explain the lack of free air in the abdomen. She complained of nausea and some abdominal pain while eating in the evening, but did not want any analgetics. She was discharged the next day in good condition, and with no infectious signs.
Discussion
Compared to open or laparoscopic gastric resections, there are obvious advantages to using ESD over traditional surgery for dysplastic and premalignant lesions in the stomach. Except for the overnight observation of some of the patients during this implementation phase, these procedures are essentially same day surgery, thereby requiring significantly less resources than traditional surgery with hospitalized patients. There is no postoperative pain since there are no operating wounds, although some minor discomfort due to the general anesthesia and the scopy procedure itself may occur. However, the most important advantage is that the anatomy and the function of the upper GI tract are preserved since gastric resection or gastrectomy with accompanying worsened digestive functions is avoided (15). Additionally, complications from ESD in the stomach may not be as serious as, for example, anastomotic leaks after a gastric resection or a gastrectomy. In our study with 15 patients, one delayed bleeding was treated endoscopically and two perioperative perforations were closed with clips, which caused no complications.
Post-perforation routines may vary, and others may apply a more conservative approach with post-procedural X-rays, fasting, and longer stays in the hospital. Our approach with early feeding and fast discharge was based on a strict surgical evaluation of the solidity of the closures of the very small holes with the oversized clips we applied, as well as the observed lack of leakage of liquids through the perforations during the procedure. It must also be emphasized that our Department of Gastrointestinal Surgery in Tromsø has a long tradition for early oral feeding and early mobilization and discharge after gastric surgery as a member of the Enhanced Recovery After Surgery Society (ERAS) (16).
A perforation rate of 11% in this study is more than in centers with higher volumes (9, 17). In contrast, both occurred with a dissection of submucosal tumors, with one of these invading the muscularis propria. It is known that dissection of submucosal tumors and dissection in the muscularis propria have a higher rate of perforation (18). In such cases, instead of categorizing a perforation as a complication, it may instead be considered a calculated peroperative incident—as in traditional surgery—or an endoscopic full-thickness resection (19). Furthermore, a Japanese long-term follow-up study of patients with perforation during ESD of early gastric carcinoma did not detect any peritoneal dissemination (20).
EUS was not routinely performed in this study and was inconclusive in two of four patients receiving it. In a recent Cochrane systematic review on the diagnostic accuracy of EUS, it is concluded that EUS performance is lower in diagnosing superficial tumors (T1a versus T1b) (21). As a result, the role of EUS in the preoperative evaluation of superficial gastric tumors suitable for ESD may be a subject for further evaluation.
Of the 12 patients with dysplasia or cancer, 10 patients were R0 resections, 1 was R1, and 1 was judged by the pathologist to be uncertain. The uncertain R0 resection was probably R0 since there were no recurrences after 32.5 months. In a meta-analysis of larger series mainly from Asia, the complete resection rate was 88%–96% depending on indication (6), and in a European multicenter study the R0 rate was 77% (22). Depending on the assessment of our single patient with uncertain margins but negative controls, our R0 rates were in the range of 83%–92%.
In all, 11 of the 15 specimens were resected en bloc. Compared to other Western series (22), an en bloc resection rate of 73% in this study is acceptable and can be improved by always retrieving the specimen in a basket instead of with forceps, as we did in some cases. The reason why the R0 resection rate was higher than the en bloc rate is that two specimens were divided into two parts during retrieval. One of these contained a focal LGD with free margins in each part, while the other one contained a single focus with localized HGD in the anticipated division line since several control biopsies from the resection field were negative. In another case, a benign lesion was brought out in three parts due to fibrosis, which ended with snare resections after a subtotal ESD.
Patients with a curative ESD for dysplasia or adenocarcinoma were controlled with the last gastroscopy after a median of 6 months, without detecting any signs of recurrences. In a meta-analysis from Asia, the recurrence rates after ESD for early gastric cancer are equivalently low within the range of 0.6%–1.5% depending on the indication (6). However, the patient with a T1b adenocarcinoma in the cardia (11th case), who was not directed to surgery because of a serious lung disease, later developed cancer in his Barrett’s metaplasia in the lower esophagus. Based on the new localization of this tumor and the fact that the primary resection was R0, this was most probably a metachron cancer. The other patient with a T1b cancer (Fig. 3, fifth case) was not operated on after the ESD due to advanced age and comorbidity, and she did not want more gastroscopies after the first control gastroscopy 1.5 months after the procedure. Interestingly, after 12 months she was in her habitual condition with no clinical signs of gastric cancer or metastasis.
According to the Japanese classification of gastric carcinoma (23), these two patients with T1b adenocarcinoma are classified as subgroup SM2 since the depth of submucosal penetration is 0.5 mm or more for both. The risk of lymph node metastasis by tumor size for the SM2 subgroup is 19.0% for a tumor ⩽30 mm and 12.8% for a tumor ⩽10, respectively, for the 5th and the 11th case, according to a study on early gastric cancer by Gotoda et al. (24). As outlined in the Japanese gastric cancer treatment guidelines (25), the criteria for an endoscopic resection of gastric cancer as a standard treatment are that the depth of invasion is clinically diagnosed as T1a and the diameter is ⩽20 mm. Clearly, these two patients with T1b adenocarcinoma should have been directed to gastrectomy, but since they were not suitable for surgery, an endoscopic resection was the only option for a potential curative treatment.
Even in an implementation phase and with a small number of patients, our median procedure time of 115 min is similar to the average procedure time of 108 min in 20 European centers (22), but longer than the median procedure time of 47 min in a large Japanese single-center study (26).
With regard to training and practice, the disadvantage of few patients being suitable for ESD in a small-volume center could be partially compensated for by also directing other endoscopic procedures to the same surgeon or internist. The surgeon exerting the ESD in this study obtained basic skills by performing all the endoscopic mucosal resections, as well as endoscopic retrograde cholangiopancreatography (ERCP) and other endoscopic, therapeutic procedures.Moreover, he participated in several ESD training courses on anesthetized pigs at The Intervention Center, Rikshospitalet, Oslo, Norway, in addition to attending theoretical courses and live demonstrations at congresses. Structured training programs for ESD have been proposed both in Japan (27) and in Western countries (28). After obtaining extensive basic endoscopic skills, key elements in such programs are attending courses, congresses, and live demonstrations; watching and assisting experts at work; training on isolated pig stomachs and live pigs; and finally, performing easy human cases under supervision. A Japanese study reports that the experiencing of at least 30 supervised cases is required to gain an early proficiency in this technique (29). However, it is admitted that the Japanese algorithm for learning ESD cannot be directly implemented in the West due to cultural differences in teaching models (30), not to mention a lower case load (8). Small patient volumes in Western countries and the technical difficulties of ESD are arguments for cooperation and common solutions for training programs as well as training centers on a national and even multi-national level.
The limitations of this study are a relatively short median follow-up period of 6 months, a small number of patients, and the fact that the results are based on the skills of a single surgeon. However, the results also encourage the implementation of ESD in small-volume centers on the assumption that one or two surgeons or internists are allowed to specialize in endoscopic resections.
Conclusion
We conclude that ESD in the stomach could be implemented safely and with good results even in a small-volume center when one surgeon or internist with experience in therapeutic endoscopies performed all procedures in order to obtain a maximum of practice. Gastric ESD should be a part of the treatment repertoire for selected lesions in the stomach at regional centers with multidisciplinary teams and with experience in upper GI surgery. Thus, there is a need for structured training programs for ESD in Western countries.
Footnotes
Acknowledgements
None.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
