Abstract
Background and Aims:
Idiopathic frozen shoulder can be successfully treated conservatively. Manipulation under anesthesia, in randomized controlled studies, has proven to bring no additional benefit to other treatments. However, manipulation under anesthesia still is a used method mainly among patients with unbearable pain or too longstanding restriction of shoulder motion. Manipulation under anesthesia may lead to significant improvement in shoulder motion and function. However, the best timing for manipulation under anesthesia is still unclear. We therefore investigated whether timing of manipulation under anesthesia has influence on the results of manipulation under anesthesia, concerning range of motion, pain, and function.
Material and Methods:
We followed 57 patients (65 shoulders; 31 in women) at 2–14 years after manipulation under anesthesia evaluating range of motion, pain, and function. Their mean age at manipulation under anesthesia was 54.0 years. A total of 10 patients had diabetes. The time between the onset of symptoms and manipulation averaged 8.8 months. We formed two groups, those mobilized between 6 and 9 months after the onset of symptoms (Group A, 25 shoulders), and the others (Group B, 40 shoulders). We recorded visual analog scale pain, range of motion, Simple Shoulder Test, and Constant–Murley scores.
Results:
Patients manipulated between 6 and 9 months after the onset of symptoms had significantly better abduction and external rotation, less pain at rest and at night, and better Simple Shoulder Test than the others.
Conclusions:
It seems that timing has statistically significant influence on results of manipulation of the idiopathic frozen shoulder. The best time for manipulation under anesthesia, if non-operative treatment has failed to alleviate pain or limitation of shoulder motion is too cumbersome, might be between 6 and 9 months from the onset of the symptoms. However, the clinical importance of results can be questioned.
Introduction
Spontaneous adhesive capsulitis of the shoulder joint, called idiopathic frozen shoulder, is a common, self-limiting disease with an incidence of 3%–5%, and among diabetics reaching even as high as 20% (1–5). Conservative treatment or even thoughtful neglect is considered the treatment of choice, but if non-operative treatment has failed to alleviate pain, or if limitation of shoulder motion is too burdensome, manipulation under anesthesia (MUA) is still being used. Several retrospective studies suggest that duration of symptoms may be reduced by manipulation (2, 6–12), and good outcome is maintained for several decades (13). However, there are randomized controlled trials (RCTs) showing that MUA does not add effectiveness to an exercise program carried out by the patient after instruction (14) or to administration of an intra-articular steroid with distension (15) or hydrodilatation (16). Kivimäki et al. (14) showed in a randomized, controlled trial that MUA with home exercises had not better outcome than home exercise alone in the treatment of frozen shoulder.
The effect of timing of MUA has had surprisingly little attention in the literature to date. We found only four studies reporting on the influence of timing of MUA for idiopathic frozen shoulder. Colville stated that patients who had early intervention, <9 months after the onset of symptoms, had a significantly better Oxford Shoulder Score at final follow-up. Their mobility and visual analog scale (VAS) pain scores were also better than in the late group (1). On the other hand, Thomas et al. (17), in their questionnaire study, found no relation between duration of frozen shoulder and results of MUA. Fazzi and Kelly (18) suggested that MUA can be considered a viable treatment option in the early stages of the disease. One proposal is that MUA should be done at stage III, between 6 and 12 months after symptom onset (11). On the other hand, according to Colville’s findings, results of MUA are inferior if done >9 months after the symptom onset (1).
We thus formed two groups, those manipulated at or after 6 months, but before 9 months (Group A, 25 shoulders, MUA at ≥6 to <9 months), and those manipulated before 6 or at or more than 9 months after onset (Group B, 40 shoulders, MUA <6 or ≥9 months after the onset). We determined whether timing of MUA affects long-term results concerning range of motion (ROM), pain, and function in patients with idiopathic frozen shoulder.
Material and methods
We (H.V., M.V.) performed a retrospective review of a single-surgeon (L.V.), single institution consecutive series of patients from April 1997 to October 2002. Of the 80 patients with a manipulated frozen shoulder identified, 57 (65 shoulders, 71%) were available for follow-up after a mean period of 6 years (range: 2–12 years). Criteria for inclusion were as follows: (1) no or only minor shoulder trauma, (2) marked loss of active and passive shoulder motion (mostly forward flexion <120°, abduction <110°, severe restriction in external rotation), (3) normal findings on a true anteroposterior radiograph of the glenohumeral joint, and (4) diagnosis of frozen shoulder made by an experienced orthopedic surgeon, and (5) MUA. We included patients with diabetes mellitus and thyroid dysfunction and excluded those with (1) any intrinsic glenohumeral disorder such as glenohumeral arthritis or rotator cuff tear, (2) any history of substantial shoulder trauma like motor-vehicle accident, (3) fracture of the ipsilateral extremity, and (4) insufficient patient status records (no mention of preoperative ROM in degrees).
Included were 27 males and 30 females, mean age 54 years (range: 37–69 years) at manipulation. Time delay between onset of the symptoms and manipulation averaged 8.8 months (range: 3–36 months; median: 6.0 months). The right shoulder was involved in 30 patients (56%), 10 patients (18%) had diabetes, and 16 (28%) had a minor shoulder trauma before the onset of symptoms. Mean preoperative flexion was 95° (10°–130°) and abduction 80° (10°–120°). Often no external nor internal rotation was recorded other than “external rotation restricted.” One patient had simultaneous bilateral frozen shoulder; 7 (12%) had a bilateral non-simultaneous one. In addition, 6 patients had experienced MUA of the contralateral shoulder before our inclusion time April 1997. Before MUA, 3 patients had received physiotherapy and 26 patients a mean of 1.9 subacromial corticoid injections.
We formed two groups, those with shoulders mobilized between 6 and 9 months (≥6 to <9 months) (Group A, 25 shoulders), and the others (MUA at 3 to <6 or ≥9 months) (Group B, 40 shoulders). These groups did not differ in age (54.1 versus 53.9 years), gender (p = 0.284), preoperative passive ROM, or follow-up time (6.3 vs 5.9 years), or preoperative treatment (Table 1). In Group B were 16 shoulders manipulated before 6 months and 24 shoulders at or later than 9 months after symptom onset.
Demographics of patients with manipulation under anesthesia (MUA) for spontaneous frozen shoulder.
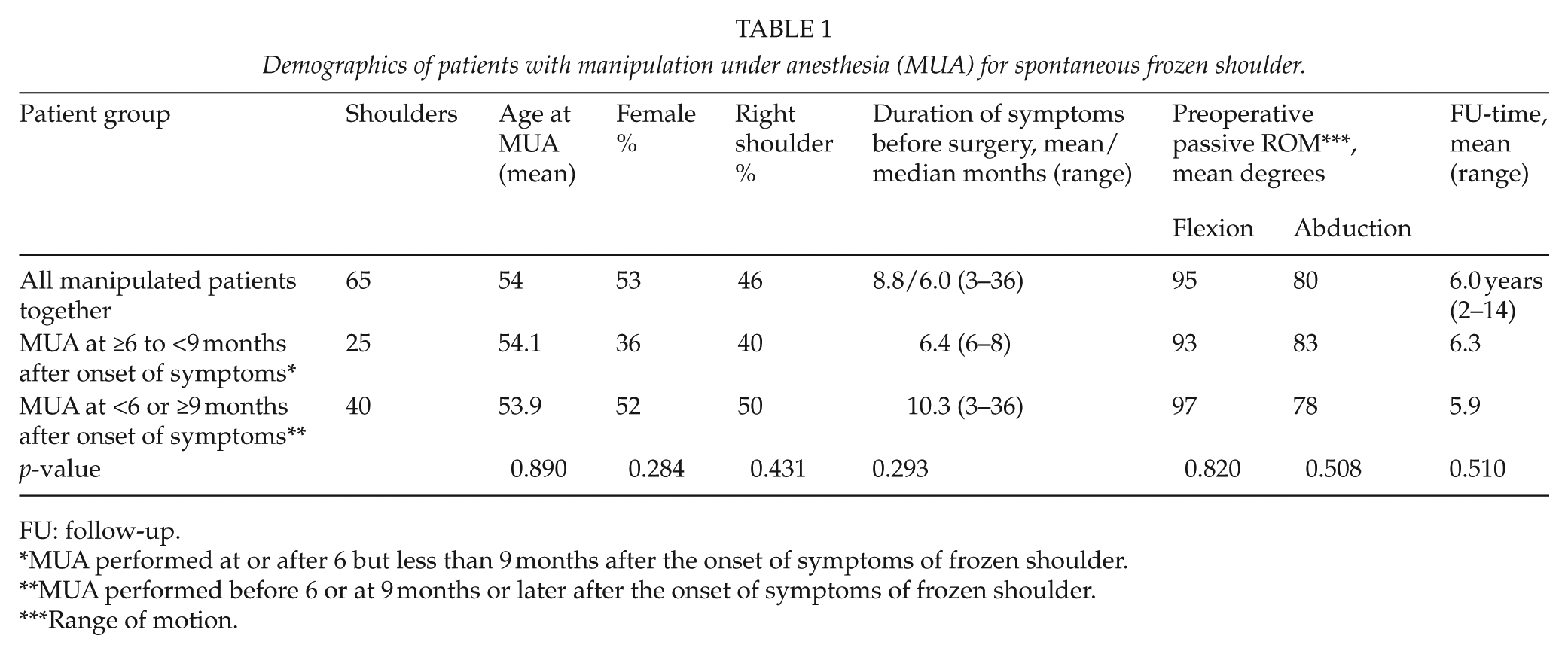
FU: follow-up.
MUA performed at or after 6 but less than 9 months after the onset of symptoms of frozen shoulder.
MUA performed before 6 or at 9 months or later after the onset of symptoms of frozen shoulder.
Range of motion.
A short-acting barbiturate anesthetic was administered by manual mask ventilation with the patient supine, without any special muscle relaxant. The technique for MUA consisted of gradual alternate abduction and forward elevation followed by external and internal rotation, with care taken not to fracture the humerus. The goal was free or almost-free ROM with the surgeon detecting the characteristic feeling of tearing. All patients were offered a postoperative physiotherapy program of passive and active rehabilitation.
Questionnaires (19) were mailed to the patients along with the invitation to the study. The questionnaire comprised questions concerning demographics, subjective satisfaction measured on a 5-point Likert scale (1 = very satisfied and 5 = very unsatisfied), symptoms of the operated shoulder including pain evaluation on a VAS (range: 0–10; 0 = no pain and 10 = maximal, intensive pain). In addition, patients received the Simple Shoulder Test (SST) questionnaire (20). The patients completed these questionnaires at home and returned them at the follow-up.
All patients underwent a physical examination by an independent observer (H.V., M.V.) at final follow-up. We again recorded patients’ medical history, with special attention to estimation of the exact time of symptom onset. Measurement of ROM was as reported earlier (13, 19). Shoulder strength was evaluated with manual muscle testing, except that strength in abduction for the Constant score (21) was evaluated with a Salter spring balance up to 11 kg with increments of 0.2 kg (13). Shoulder function was assessed with the Constant score (21). In addition, a true anteroposterior radiograph in external rotation was taken of both shoulders. The mean duration of follow-up was 6.0 years (range: 2–14.4 years).
Statistical methods
The results are presented as mean values and 95% confidence intervals. All statistical analyses were performed with Statistical Package for the Social Sciences (SPSS) 19.0 for Windows (SPSS Inc., Chicago, IL, USA). Significance level was set at 0.05.
Results
Timing influenced MUA results. Preoperative flexion and abduction did not differ significantly, but at follow-up, clinical and functional outcome was best in Group A (Table 2). Abduction had improved more in Group A than in Group B (177°/167°, p = 0.023) as did external rotation (61°/51°, p = 0.026). Forward flexion was also better in Group A (162°/156°), but not significantly. Active ROM of the affected shoulder had improved to the level of the contralateral shoulder. The final ROM of the affected shoulder did not differ significantly from that of the unaffected shoulder (flexion 160°, abduction 172°, external rotation 48°, p = 0.528–0.926).
Influence of timing of manipulation under anesthesia (MUA) for idiopathic frozen shoulder.
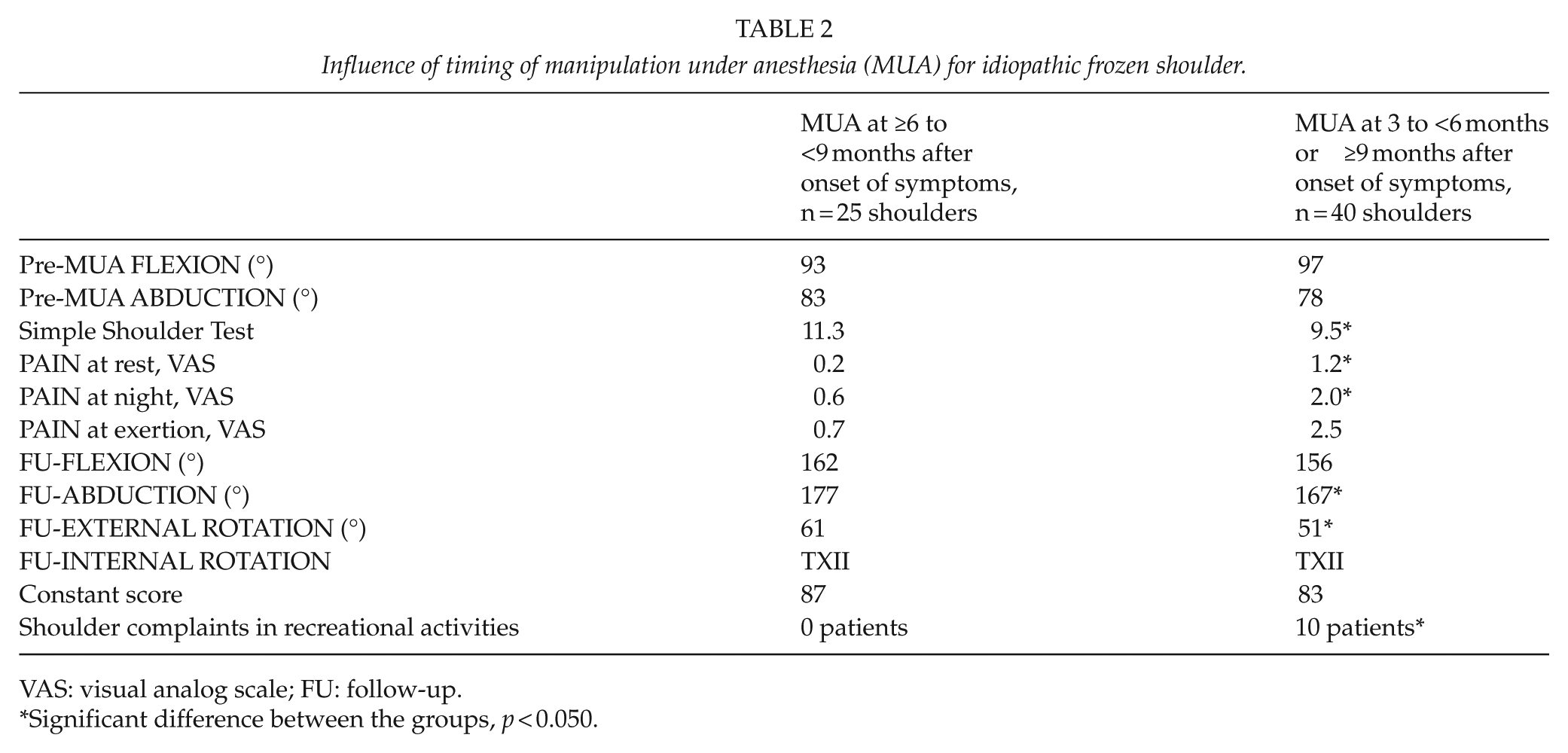
VAS: visual analog scale; FU: follow-up.
Significant difference between the groups, p < 0.050.
Patients in Group A reported significantly less pain on VAS at rest and at night than did patients in Group B. They also had less pain during exertion, but this did not reach significance (Tables 2 and 3).
Outcome in literature on timing of MUA for idiopathic frozen shoulder.
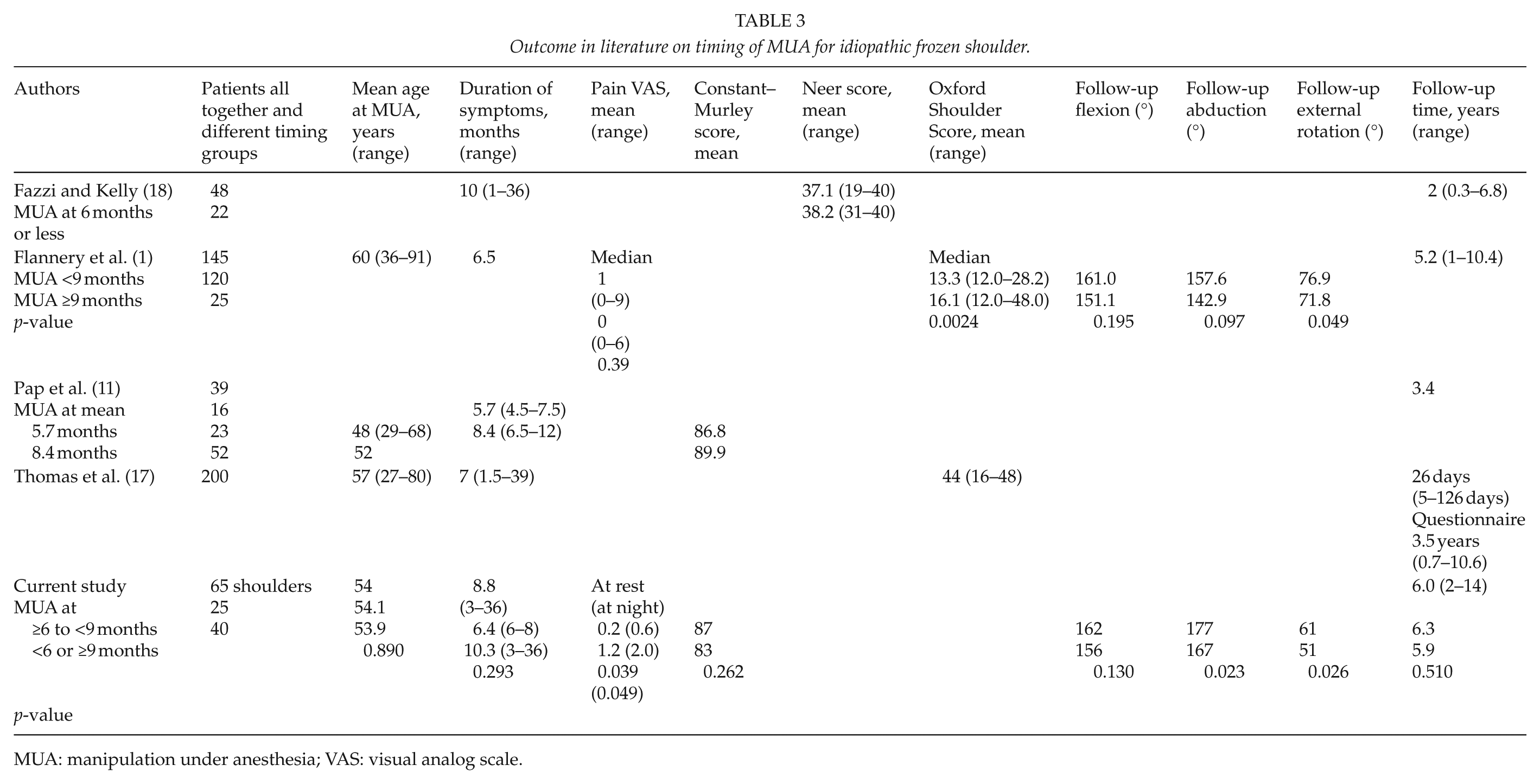
MUA: manipulation under anesthesia; VAS: visual analog scale.
SST figures were better in Group A, 11.3 versus 9.5, p = 0.002. There was also a tendency toward better Constant–Murley score in Group A, 87 versus 83, p = 0.262. The patients in Group A had significantly fewer shoulder complaints during recreational activities (0 versus 10 patients, p = 0.012).
Concerning subjective satisfaction, among five choices, 91% of the patients in Group A were satisfied or very satisfied, in Group B only 72% (p = 0.076). All the dissatisfied or very dissatisfied patients belonged to Group B (MUA at 3, 5, 12, and 19 months after the onset of symptoms).
In Group A, 84% felt that their symptoms improved after manipulation, in Group B 75% (p = 0.084). Of the 57, 26 patients (46%) continued to work, and 28 (49%) had retired, none due to shoulder disorder. Under the same circumstances, 50 patients (88%) would have chosen manipulation again, in Group A 95%, in Group B 75%. Overall, only 8 (14%) were smokers, and 35 (61%) drank some alcohol weekly (no significance between the groups). Concerning general healthiness on a 5-point scale, 36 patients (63%) chose good or very good, 20 partly good, one poor, and one very poor. Post-manipulation sick-leave averaged 1.3 months (range: 0–7 months) with no difference between groups. During the previous year, 9 patients (16%) had suffered shoulder complaints in daily living activities (Group A: 1 patient, 4%; Group B: 8 patients, 20%; p = 0.068).
In clinical examination at follow-up, glenohumeral motion was free in 45 patients (79%) and somewhat restricted in 10 (18%). Of these 10, 9 belonged to Group B, as did 1 patient with severely restricted glenohumeral motion (p = 0.054). We found no significant difference between the groups in the need for remanipulation (five remanipulations in each group), not even when explored between patients manipulated before 6 months versus all the others (p = 0.259).
The results between subgroups MUA at 3–6 months, 6–9 months, 9–12 months, and >12 months did not differ greatly. Clinical and functional outcome was, however, best in Group A (MUA at 6 to <9 months) (Table 2). We found that if MUA was done more than 12 months after the onset of symptoms, the results were significantly inferior to those of Group A. When MUA was done between 3 and 6 months, the SST was inferior to the SST of those manipulated between 6 and 9 months, that is, Group A (p = 0.031).
Discussion
Non-operative treatment is widely considered the treatment of choice for spontaneous frozen shoulder. Kivimäki et al. (14) found in an RCT with 125 patients that during 12 months, MUA does not add effectiveness to an exercise program carried out by the patient after instruction. Quaraishi et al. (16) also found in an RCT with 36 patients that at 6 months those patients undergoing hydrodilatation did better than those who were manipulated. Hsu et al. (15) concluded that patients with frozen shoulder should be treated non-operatively allowing a change for spontaneous improvement, but that MUA and surgical release can often restore motion and obtain pain relief for patients with refractory cases.
For recalcitrant frozen shoulders, most authors agree that MUA reduces duration of symptoms and leads to significant improvement in shoulder motion and function, if pain or limitation of ROM is too cumbersome (5–7, 10, 12, 13, 22). Influence of timing of MUA on the results is still obscure. We wished to know whether timing of MUA has any influence on results. We therefore determined ROM, pain relief, and function in patients with spontaneous, idiopathic frozen shoulder treated by MUA, and studied any correlations with timing.
We acknowledge limitations in our study. First, concerning preoperative ROM, we obtained exact figures for flexion and abduction, while external and internal rotation was mostly stated only as severely restricted. We excluded patients whose ROM was inadequately documented in their medical records, and included only patients with appropriate records for flexion and abduction. Second, we did not obtain Constant–Murley scores or the SST at the time we initially saw the patients. Although we placed great value on longitudinal assessment of outcome measures, only ROM measurements were available. Third, our follow-up varies between 2 and 14 years. This wide range may bring confounding factors during the years that make the comparison between the groups less reliable. Fourth, with larger patient subgroups, statistical significance might be reached more often.
The only four published papers on this topic present conflicting results (1, 11, 17, 18). In one study (1) of 145 patients examined after a mean period of 5 years, 83% had MUA performed less than 9 months from the onset of symptoms. They had a significantly better Oxford Shoulder Score at final follow-up, and mobility and VAS pain scores were also better than among those manipulated after 9 months (Table 3). When Kelly (18) evaluated 48 patients by questionnaire after a mean period of 2 years, Neer scores of the 22 patients manipulated during the 6 months after the onset of symptoms did not differ from the others. Pap et al. (11) examined 39 patients a mean 3.4 years after MUA, and those manipulated at a mean 8.4 months (range: 6.5–12 months) had better outcome than those manipulated mean 5.7 months after symptom onset. On the other hand, Thomas (17) presented 200 patients with MUA evaluated by a questionnaire after a mean period of 3.5 years without any correlation between timing and results (Table 3). The present study detected a significant effect of MUA timing. The best results were achieved from performance of MUA between 6 and 9 months after symptom onset.
Concerning ROM, Flannery et al. (1) showed that patients who were manipulated less than 9 months after onset had better ROM in all directions (flexion 161° versus 151°, abduction 158° versus 143°, and internal rotation 74° versus 69°). However, only the gain in external rotation was significantly better, 77° versus 72°, p = 0.049. According to our long experience in manipulating frozen shoulder, we believe that too early manipulation, in our view before 6 months after the onset of symptoms, may lead to a recurrence. However, this study did not give such an impression when it came to the need for remanipulation. Today, we prefer intra-articular corticosteroid injections at the earlier stages of the disease during the inflammation period. We found that MUA between 6 and 9 months yielded statistically significantly better ROM (Table 3). However, it can be questioned whether a difference of 10° is of clinical importance. Colville provided no figures on patients manipulated between 6 and 9 months, which perhaps would strengthen his findings.
On the other hand, another survey found no significant differences regarding timing of MUA (17). One reason might be differing patient selection. Thomas included patients with preoperative flexion and abduction up to 170° and external rotation up to 70°. It is challenging to make an accurate diagnosis of frozen shoulder in patients with such a good ROM. We also consider that a questionnaire survey is not as relevant as a clinical examination.
In regards to pain, no articles showing the influence of timing exist. Colville used VAS pain intensity score, but the results showed no significant influence of timing (1). Our results showed a significant influence; VAS pain score at rest was 0.2 versus 1.2, p = 0.039, and at night 0.6 versus 2.0, p = 0.049, but the difference in pain with exertion (0.7 vs 2.5, p = 0.076) was not significant. However, it is difficult to say whether these statistically significant VAS differences are clinically significant, although the groups had clear differences concerning activity of daily living (ADL) complaints or complaints during recreational activities. Traditionally, the timing of MUA has been considered to be the phase when pain has gone away or after the inflammation phase. Unfortunately, we had no measurements of pain at the initial consultation, but we only know that some of the patients still complained of pain.
Concerning function, Colville stated that early MUA yielded a significantly better Oxford Shoulder Score than did MUA after 9 months or later, 13 versus 16, p = 0.0024 (1). Our study had similar results and showed that patients manipulated between 6 and 9 months had a better SST score, p = 0.002 (Table 2). However, Tashjian et al. (23) showed that minimal clinically important difference in SST scores after nonoperative treatment of rotator cuff disease is a 2-point change. That may apply also to frozen shoulder, and we had only a 1.8-point change. As regards to Constant–Murley score, there was a slight tendency toward a better score in Group A, 87 versus 83, but Holmgren et al. (24) showed that minimal clinically important difference is 17.
As regards subjective satisfaction, all the dissatisfied or very dissatisfied patients belonged to Group B (MUA at 3, 5, 12, and 19 months after symptom onset). One of those very dissatisfied patients underwent MUA twice, and severe pain lasted for several months and the inability to work lasted 9 months. After 1.5 years, the affected shoulder was symptomless. The other patient had manipulation only 3 months after onset and had a recurrence 1 week after manipulation. After some years, however, the affected shoulder was totally symptom-free.
Why would 6 to 9 months be the most beneficial time to manipulate? It is well known that before 6 months, especially during the first couple of months, the disease is still at the inflammation stage. Believing that inflammation cannot be treated by manipulation, many authors prefer to use intra-articular corticosteroids as the early intervention for idiopathic frozen shoulder. Colville stated that, in general, the longer the stiffness stage, the longer the recovery stage, and that those patients with symptoms of pain and reduced ROM for a longer period of time, being slower to enter the thawing phase, faced a poorer prognosis (1). Our study supports their recommendation to manipulate before, rather than after 9 months after the onset of symptoms. Based on the knowledge of the RCT studies, nowadays, MUA can be considered only as an exceptional treatment. Today, we very rarely manipulate our spontaneous frozen shoulder patients.
Evaluation of long-term results after MUA is difficult, because the natural course of frozen shoulder is mostly beneficial and it mostly resolves in 1.5 years (19, 25). It is possible that MUA only accelerates the recovery of ROM and the disappearance of pain, but this is itself of great value. In this study, MUA was beneficial in most cases. Timing may have an influence on results of manipulation of the idiopathic frozen shoulder. The best time for MUA, if nonoperative treatment has failed to alleviate pain or limitation of shoulder motion is too burdensome, might be between 6 and 9 months from the onset of symptoms.
Footnotes
Acknowledgements
We thank Leena Ristolainen PT, DSc, for statistical advice.
Declaration of Conflicting Interests
None declared.
Research Ethiecs
Each author certifies that his or her institution approved the human protocol for this investigation, that all investigations were concluded in conformity with ethical principles of research, and that informed consent was obtained for participation in the study. They obtained permission to perform this study from the ethics committee of the study’s hospital district (Helsingin ja Uudenmaan Sairaanhoitopiiri (HUS), Helsinki University Hospital, Helsinki, Finland).
