Abstract
Background and Aims:
Prior to bariatric surgery, a preoperative weight-reducing regimen is usually adhered to in most centers. The clinical effects of such a regimen are yet to be determined.
Material and Methods:
We reviewed the current literature by searching in PubMed for publications reporting clinical effects resulting from a preoperative weight loss regimen prior to bariatric surgery published from January 1, 1995 to April 30, 2014.
Results:
In total, we identified 23 original publications and 2 review articles which met all inclusion criteria. These were included and fully analyzed with regard to effects of preoperative weight loss. In general, for parameters such as operating time and intraoperative complications including blood loss and recovery, inconsistent data were reported. Most studies included low number of patients and with heterogenic designs, and the results could not form the base for recommendations. However, for outcomes such as postoperative complications and weight development over time, data from large-scale studies and randomized controlled trials suggest beneficial effects following adherence to weight loss prior to bariatric surgery.
Conclusion:
Although a large amount of data in the current literature on the effects of weight loss prior to bariatric surgery are inconsistent for many outcome parameters, recently published results regarding effects on postoperative complications and weight development over time strongly suggest that such a regimen should be recommended. Whether a certain degree of weight loss should be mandatory before being accepted for bariatric surgery is, however, still controversial.
Keywords
Introduction
During the last decades, the “epidemic of obesity” has become an increasing health concern in the Western world. Obesity is associated with comorbidities such as metabolic disease (in particular, type 2 diabetes mellitus), cardiovascular and joint disease, and certain types of cancer as well as increased mortality over time (1). Moreover, obesity is associated with marked reductions in self-reported quality of life and well-being. Unfortunately, results with conservative treatment for morbid obesity are often unsatisfactory, particularly in the long term. On the contrary, bariatric surgery has been demonstrated to be associated with not only sustainable weight loss but also resolution of comorbidities as well as reduced mortality over time (2). Due to these encouraging results, the annual number of bariatric surgical procedures performed worldwide has increased dramatically from 146,000 to some 340,000 procedures between years 2003 and 2011 (3). In Europe, the relative increase is even more pronounced during the same period (from 33,000 to 113,000) (3).
Today, bariatric surgery is typically performed laparoscopically, which has been shown to be safe and effective in terms of achieving sustainable weight loss, as well as improvement or resolution of obesity-related comorbidities (4–6).
In most centers performing bariatric surgery, a preoperative 2- to 3-week period of low-calorie diet (LCD) intake is usually recommended. Such a regimen has been shown to be associated with a reduction in liver and intraabdominal fat volume (7, 8). Potential/suggested clinical advantages include improved visibility during the operation with reduced technical complexity of the procedure, reduced risk of intra- as well as postoperative complications, “preparation” of patients for postoperative diet regimens, and improved postoperative weight development. Indeed, some insurance companies in the United States require preoperative weight loss in the range of 5%–10% before surgery is approved, and some bariatric centers prescribe mandatory successful adherence to weight loss programs in order to accept patients for operation (9). However, potential negative effects associated with a preoperative low-calorie program include patient discomfort, increased costs, denial of proper treatment, and increased morbidity associated with undergoing surgery in a catabolic state.
There are conflicting data in the literature as to the true effect of a preoperative weight loss regimen on various aspects of clinical outcome after bariatric surgery. In a systematic review published in 2011, it was concluded that “little evidence is available to support or refute the routine use of preoperative weight reduction in bariatric surgery” (10). One main reason for the conflicting results might be the heterogeneity of study protocols and the relatively small number of included patients in most studies.
The aim of this study was therefore to give an updated review of the literature in order to identify current evidence supporting the use of weight reduction prior to bariatric surgery in terms of clinical effects.
Material and Methods
We searched PubMed with the terms obesity and, bariatric or surgery or gastric bypass or sleeve or Roux en Y, and weight loss and preoperative and complications from January 1, 1995 to April 30, 2014. A consort diagram illustrating the inclusion process of publications is given in Fig. 1. There were a total of 2010 articles from the combination of the terms above. We excluded publications based on the following criteria: included patients <18 years, sample size <20, not English language, no support in the heading for the pre- or postoperative weight loss, or no support in the heading for postoperative outcome or complications. All study designs were accepted.
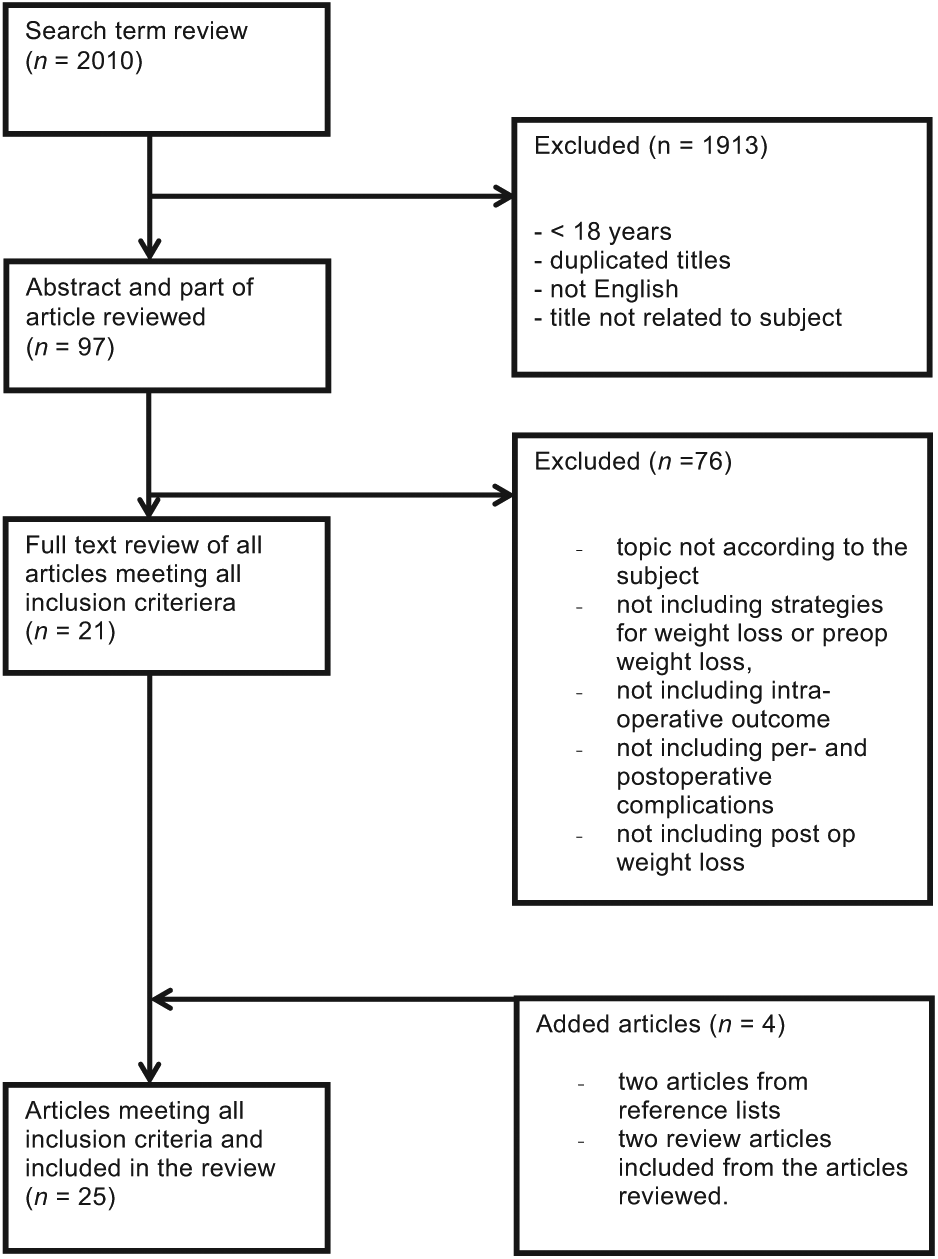
Consort diagram illustrating the selection process of publications included in the current review.
A total of 97 articles were found where abstract and, if necessary, parts of the articles were analyzed according to the above criteria. An additional 76 publications were excluded based on criteria given in Fig. 1. This resulted in 21 publications. These were supplemented by two additional articles on the basis of reference lists in included articles, which gave us a list of a total of 23 original publications. In addition, two review articles were included and used as support in the various areas for this review. An overview of included publications is given in Table 1.
Overview of publications included.
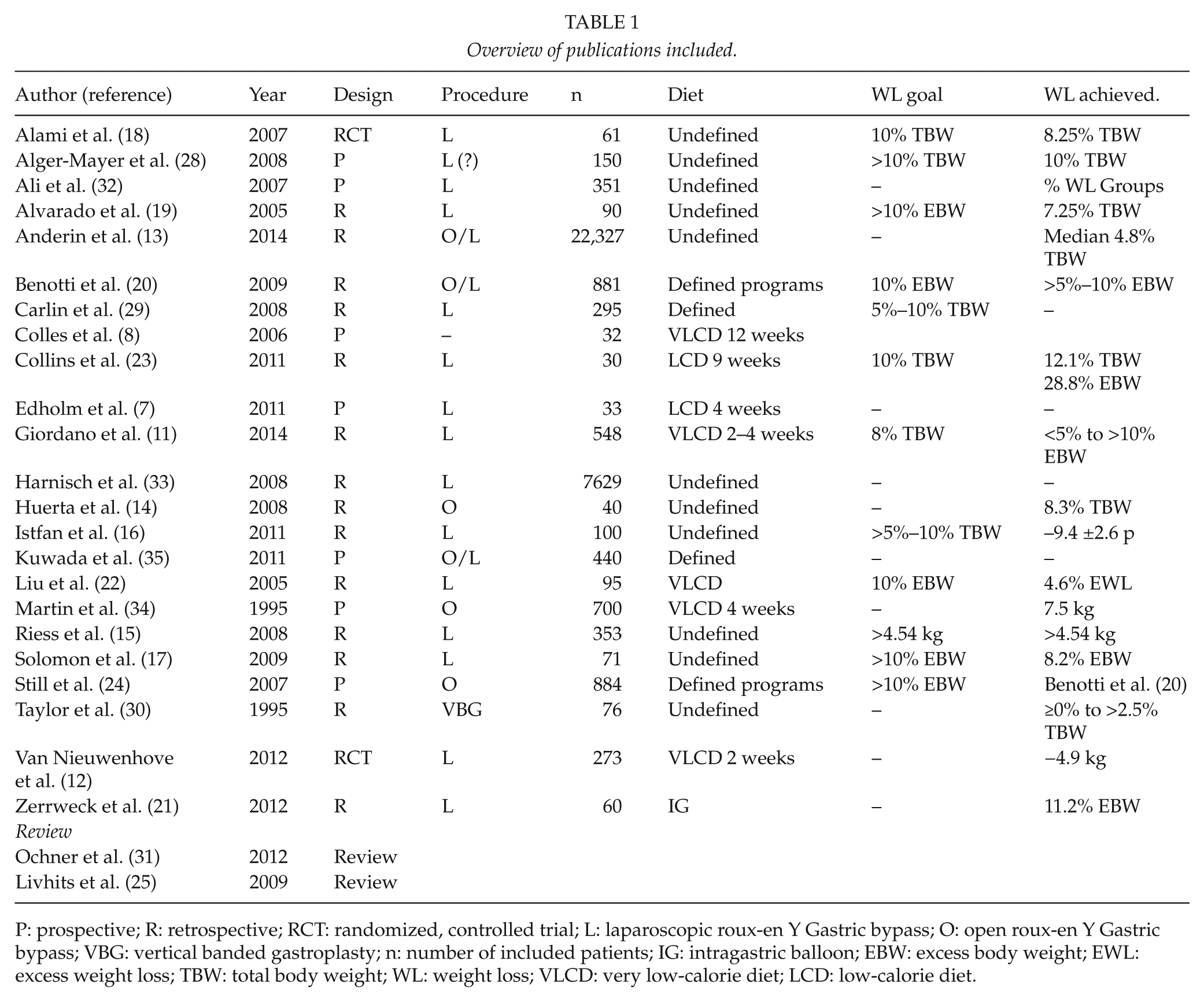
P: prospective; R: retrospective; RCT: randomized, controlled trial; L: laparoscopic roux-en Y Gastric bypass; O: open roux-en Y Gastric bypass; VBG: vertical banded gastroplasty; n: number of included patients; IG: intragastric balloon; EBW: excess body weight; EWL: excess weight loss; TBW: total body weight; WL: weight loss; VLCD: very low-calorie diet; LCD: low-calorie diet.
Based on factors suggested to be influenced by preoperative weight loss, each article was analyzed and results are reported according to the following parameters:
Strategies used for achieving preoperative weight loss;
Preoperative weight loss achieved;
Intraoperative outcomes;
Intraoperative complications;
Length of stay (LOS);
Postoperative complications;
Postoperative weight loss.
Since a large majority of studies were observational and uncontrolled, we did not perform any formal grading of evidence.
Results and Discussion
Strategies used for Achieving Preoperative Weight Loss
Very low-calorie diet (VLCD) intake and LCD intake are usually defined as 800 kcal/day and 800–1200 kcal/day, respectively. In a landmark study from 2006, using serial magnetic resonance imaging (MRI) in obese individuals, Colles et al. (8) demonstrated that VLCD for 12 weeks was associated with a reduction in liver volume by 20%. Interestingly, 80% of this loss was achieved already during the first 2 weeks of treatment, a period during which it could be assumed that most patients should be able to comply with such a protocol. Therefore, in most studies reporting results including a defined preoperative weight loss regimen, a 2- to 4-week period of VLCD or LCD has been prescribed. This regimen has been used in some of the studies (7, 11, 12), whereas no clear definition of preoperative dietary protocol was given in others (13–17). In some studies, no restrictions were given (18, 19), and in one (20), a 6-month weight loss program was applied. Finally, in one study, intragastric balloon was used for a period of 155 ± 62 days for achieving preoperative weight loss (21).
Preoperative Weight Loss Achieved
In most studies in which any “treatment goal” was defined, loss of 10% of excess body weight (EBW; defined as weight exceeding the weight corresponding to body mass index (BMI) 25) (17, 19, 20, 22) or total body weight (TBW) (16, 18, 23) has been aimed at. The reported weight loss that was actually achieved varies within broad ranges in the included studies, from loss of 8.2% to 12.1% and from 4.6% to 28.8% of TBW and EBW, respectively (Table 1).
In the study with the most pronounced weight loss (23), very few patients were included (n = 30) in a 9-week program with an intake of 800 kcal/day in combination with regular professional medical counseling/surveillance.
In terms of preoperative weight loss actually achieved, the duration of the program does not seem to be crucial per se. In the study including a 6-month weight loss program (24), almost 50% of included patients achieved a TBW loss of >10% and over 50% lost between 5% and 10% of TBW, demonstrating that programs of longer duration could be successful as well. It seems, therefore, as if a well-structured design of preoperative weight loss program is at least as important as the amount of energy provided or the duration of the preoperative weight loss period.
Intraoperative Outcomes
The question whether better visualization during surgery could be achieved with preoperative weight loss was only possible to specifically address in two studies in which controls that were not prescribed preoperative weight reduction were included (7, 12). In both of these, participating surgeons scored visibility as being improved after weight loss, although these were blinded to treatment allocation in only one study (12). Moreover, Alami et al. (18) reported that estimated liver volume was reduced in patients who were successful in losing weight preoperatively, which further support the potential for this regimen to reduce technical complexity during bariatric surgery.
It could be assumed that a reduction in complexity through a better visualization should have the potential to result in a shortening of operating time. Accordingly, this was also reported in several of the included studies (11, 14, 18, 21, 22). In a meta-analysis from 2009 (25) including “the only three high-quality studies that reported this finding,” a reduction in mean operating time of 23 min (95% confidence interval (CI) 13.8–32.8) was reported. However, there was a large heterogeneity between studies (p = 0.07), and mean operating times were long, varying from 105 to 220 min.
Interestingly, in the only randomized, blinded study (12), although visibility was scored higher, no reduction in operating time was reported in patients who lost weight preoperatively, which was also true for patients with higher BMI (>48 kg/m2). The authors suggested that this might be due to the fact that only surgeons with vast experience from laparoscopic bariatric surgery participated, who were able to perform the procedure without consuming more time also in patients where visibility was reduced. This might also explain the fact that there was a difference in operating time between groups in the other published randomized controlled trial (RCT) (18) with exceptional long operating times, suggesting that participating surgeons were still at their learning curve. However, from the available data, it could not be excluded that preoperative weight loss might affect operating time in patients with very high BMI.
Intraoperative Complications
For other intraoperative outcomes, the current evidence is relatively sparse and inconsistent. Although recognized as a difficult variable to estimate accurately, some studies reported data on intraoperative blood loss. In one study, Liu et al (22) reported reduced blood loss (72 ± 40 vs 102 ± 65 mL, p = 0.03) in patients who lost weight preoperatively. In Riess et al.’s (15) study, although the amount of intraoperative blood loss was not given, preoperative “weight loss” patients were in need of postoperative blood transfusions in 1.4% compared to 4.7% in non-weight loss patients. Somewhat surprisingly, however, the total number of complications was reported higher for patients who lost weight prior to surgery.
In both of the two randomized studies published so far (18, 12), no difference in estimated blood loss was found between groups. Moreover, increased blood loss was reported by Edholm et al. (7) in patients with MRI-documented reduction in liver volume due to LCD intake preoperatively compared to non-restricted patients (130 vs 75 mL, p < 0.01).
Other more unconventional perioperative outcome parameters have been evaluated as well, such as “deviation from standard procedure” (conversion to an open procedure, intensive care unit (ICU) care > 2 days, length of hospital stay >2 weeks) which was reported more uncommon in patients who lost weight preoperatively with the use of an intragastric balloon (8.6% vs 35.1%, p = 0.031) (21).
Los
Length of hospital stay has also been used as a parameter evaluating outcome after preoperative weight loss, and as for intraoperative parameters, conflicting data are reported. In a retrospective analysis of 884 patients, Still et al. (24) found that patients who lost >5% EBW preoperatively were more likely to have a shorter LOS compared to those with less pronounced weight loss. In a systematic review, Cassie et al. (10) pooled data from five studies with heterogeneous designs and reported, based on their analysis, that the mean LOS after preoperative weight loss was 3.34 days compared to 3.98 days for the non-preoperative weight loss patients (p < 0.05). In these uncontrolled studies, LOS as a parameter obviously suffers from several biases, some of the most important being the time relationship between changes in routines for postoperative hospital stay and preoperative dietary recommendations, increased hospital volume and surgical skills. Accordingly, in several studies, the authors have not been able to find any differences in LOS in patients who did versus those who did not lose weight preoperatively (7, 14, 15, 18, 21, 22). In the only randomized study in which LOS was commented on (18), no beneficial effect could be demonstrated between groups.
Postoperative Complications
The potential of preoperative weight loss to reduce postoperative complications has been evaluated in a number of studies, most of them observational and uncontrolled. In some of these (11, 15, 20), the findings suggest a reduction in complication rates in response to preoperative weight reduction, whereas other report no beneficial effect (7, 14, 18, 19, 21). In addition to the conflicting results, these studies suffer from the weaknesses of the non-randomized design and/or limited number of included patients.
In a small randomized study including 61 patients, Alami et al. (18) reported no reduction in postoperative complication rates in patients who achieved 10% preoperative weight loss compared to those who did not. The small number of patients included and/or the fact that participating surgeons did not seem to be beyond the learning curve might explain this lack of difference. However, in a later, single-blinded, multicenter RCT comprising 298 patients, a statistically significant reduction in overall complication rates was reported in patients allocated to preoperative VLCD compared to non-weight-reduced patients (12). The total number of complications was 8 and 18 in the VLCD and non-VLCD groups, respectively, and the main reduction seemed to be for complications of infectious origin.
Recently, Anderin et al. (13) further evaluated the relationship between preoperative weight loss and postoperative complications in a large population-based cohort including over 22,000 patients from the Scandinavian Obesity Registry (SOReg). Patients underwent gastric bypass (96.5% laparoscopic) between 1 January 2008 and 30 June 2012 and were divided into percentiles based on preoperative weight loss. No standardized protocol for weight loss was used, and median preoperative total weight change was 0.5%, −4.7%, and −9.5% in the 25th, 50th, and 75th percentiles, respectively. At follow-up 6 weeks after surgery, any complication was noted in 9.1% of all patients. When comparing patients in the 75th to the 25th percentile of preoperative weight loss in multivariate analysis adjusted for covariates, the risk of any complication was reduced by 13% (odds ratio (OR) 0.87, 95% CI 0.82–0.94). For specific complications, the corresponding risks were reduced for anastomotic leakage by 24% (OR 0.76, 95% CI 0.64–0.91), deep infection/abscess by 37% (OR 0.63, 95% CI 0.43–0.93), and minor wound complications by 54% (OR 0.46, 95% CI 0.33–0.64). For patients in the highest range of BMI (>45.8 kg/m2), the risk reduction associated with preoperative weight loss was statistically significant for all analyzed complications—OR ranging from 0.28 (minor wound complication) to 0.55 (postoperative bleeding)—whereas corresponding risk reductions were only occasionally encountered and less pronounced in patients with lower BMI. Although not generated from an RCT, these data, based on the very large number of patients included, strongly suggest that weight loss prior to bariatric surgery is associated with marked reduction in the risk of postoperative complications. Interestingly, the degree of risk reduction seems to be related to the amount of weight lost, and patients in the higher range of BMI are likely to benefit most from preoperative weight reduction.
The precise mechanisms for the reduced rate of postoperative complications after preoperative weight loss remain to be defined. One possible explanation might be that patients exposed to preoperative VLCD could be expected to have a better glucose control in the postoperative period. It is amply demonstrated that a period of hypocaloric nutrition in obese, insulin-resistant subjects will improve insulin sensitivity, mainly due to depletion of glycogen stores and increased glucose uptake (26). Therefore, patients with improved insulin sensitivity before operation are likely to have better glucose control postoperatively, which has been convincingly shown to be associated with improved outcome after surgery (27).
Postoperative Weight Loss
As for postoperative complications, a number of studies have addressed the possible relationship between preoperative weight loss and postoperative weight development over time. Similarly, in observational studies, a positive relation has been reported in some (19, 28), while no relation could be confirmed in others (29, 30). Interestingly, a negative correlation was reported in one study (31) in which preoperative weight loss was associated with less postoperative weight loss.
In studies in which preoperative weight loss was required, non-consistent findings have been reported as well. In most studies, retrospective analysis was performed and protocols as well as degree of weight loss achieved varied. Again, some authors reported improved weight loss at time points varying between 6 and 24 months postoperatively (15, 32, 33). In other, no benefit in terms of postoperative weight development could be demonstrated in patients who were submitted to mandatory weight loss prior to bariatric surgery (30, 34, 35). In one of very few RCTs, Alami et al. (18) reported no difference in excess body weight loss (EBWL) at 6 months postoperatively in patients assigned to 10% preoperative weight loss requirement compared to patients with no weight loss requirement. In a reanalysis with 12-month follow-up of the same study, Solomon et al. (17) reported statistically significant improvement in body weight, BMI, and EBWL when patients who lost >5% EBW preoperatively were compared with those who did not. In the other RCT published, Van Nieuwenhove et al. (12) reported no difference in weight loss 3 months after gastric bypass in patients randomized to preoperative VLCD (Modifast® 800 kcal/day) compared to patients allocated to no preoperative diet regimen. However, 3 months is too short follow-up in order to make any conclusions, and the study was not designed to address this issue.
Due to these inconclusive data and lack of controlled data from studies with sufficient power, it is not entirely clear whether preoperative weight loss predisposes to better weight development after bariatric surgery. Intuitively, it seems reasonable to believe that patients who lose weight on their own before surgery create a selection bias by being more motivated to take responsibility for their new lifestyle and, thus, being more successful in achieving and sustaining satisfactory weight loss over time.
Summary and Conclusion
Although the routine to recommend preoperative weight loss prior to bariatric surgery has been common in most centers for many years, high-quality evidence for the clinical effects of its use has been lacking. Indeed, this update of the current literature illustrates the difficulties to, based on available data, give definite recommendations as to whether mandatory weight loss should be requested in all patients. For parameters such as operating time and intraoperative complications including blood loss and recovery, available data are inconsistent and could not be used as arguments for mandatory adherence to weight loss regimens.
For clinically highly relevant parameters, however, such as postoperative complications and weight development over time, data from robust studies such as RCT and population-based registry studies are emerging. Based on these, it seems justified to state that the available evidence strongly suggests that a preoperative weight loss regimen should be recommended at least in order to reduce the risk of postoperative complications.
Whether the ability to comply with preoperative weight loss regimens could be used in order to “select” patients who will benefit most from bariatric surgery is, however, still not clear. As for all surgical decisions, the potential benefits associated with a procedure need to be weighed against the risks. In particular for bariatric surgery, this evaluation should be performed within multidisciplinary team collaborations which should include the patient as the most important participant.
It seems reasonable to believe that if patients realize that preoperative dietary preparation should be looked upon as an opportunity to improve postoperative outcome, most of them should be able to be successful in this respect. If so, results after bariatric surgery could be expected to improve even further in the future, at least regarding postoperative complications and weight development over time.
Footnotes
Declaration of Conflicting Interests
The authors declare that there is no conflict of interest.
Funding
Financial support for this study was provided through the Erling-Persson Family Foundation and through the regional agreement on medical training and clinical research (ALF) between Stockholm County Council and Karolinska Institutet.
