Abstract
Background and Aims:
Self-expanding metallic stents are increasingly used in the management of malignant and benign colorectal obstructions. We aimed to identify relevant predictive factors for stent failure and stent-related complications.
Material and Methods:
We conducted a retrospective single-center analysis of 204 consecutive patients who underwent emergency colorectal stenting procedures because of symptomatic bowel obstructions from 1996 to 2011 at the Sisters of Charity Hospital Linz, Austria.
Results:
A total of 204 patients (median age 74 years) with 36 (17.7%) benign and 168 (82.3%) malignant obstructions were included in the study. Technical success was achieved in 92.5% and clinical success in 86.8% of the cases. Major complications occurred in 2.9% and minor ones in 19.6%. Overall mortality during a median follow-up period of 4.3 years was 73% (149 patients). Relevant predictors of increased risk of complications were extracolonic obstruction (p = 0.001), complete obstruction (p = 0.066), and inflammatory bowel disease (p = 0.05). Stent localization at the splenic flexure, a stenosis of >8 cm in length, and the need for endoscopic guidance were associated with higher rates of technical and/or clinical stenting failure.
Conclusion:
Colorectal stenting is less invasive than other means of emergency treatment for large bowel obstruction; it is generally safe and effective in different types of colorectal obstruction. However, relevant rates of failure and complications were recorded and predictors could be determined.
Keywords
Introduction
Colorectal cancer is one of the most common types of malignancy and clinically relevant obstruction is encountered in 29% of affected patients (1). Self-expanding metallic stents (SEMSs) were introduced by Dohotmo (2) in 1991. They offer a minimally invasive option for relief of malignant bowel obstruction and resemble an alternative management to emergency surgery, which is associated with mortality rates of up to 50% (3), especially in elderly and mulitmorbid patients (4). The use of SEMS has gained acceptance as a “bridge” to surgical treatment. The immediate restoration of luminal patency prior to resection allows for optimization of the patients’ state of health, for bowel preparation and colonoscopy, gives time for tumor-staging, and makes single-stage, elective surgery possible (5, 6). Thereby stoma creation can be avoided in up to 90% (7) while oncological outcome and long-term survival seem to be equal compared to elective surgery (8). Moreover, it is possible to use SEMS in long-term palliative treatment of selected patients who are at high risk of adverse events during surgery, who present with unresectable tumors or advanced metastatic malignant disease (9, 10). Special indications for stenting are colorectal obstructions caused by large extracolonic tumors and carcinosis. In these cases, the role of SEMS is currently discussed controversially (11–13). Furthermore, SEMSs have been used for endoluminal decompression in cases of benign obstructions caused by inflammatory bowel disease or anastomotic strictures (14, 15).
Previous studies have described a substantial rate of re-obstructions through tumor ingrowth or fecal obstruction—especially in uncovered stents. This must be seen as major drawback compared to surgery (16, 17). SEMSs have been reported to be used less frequently in the setting of proximal colonic obstructions. Nonetheless, successful decompressions there have been reported (18).
The aim of the present study was to evaluate to what extent the use of SEMS can avoid emergency surgery as the traditionally mandatory treatment option in cases of severe colorectal obstructions by providing a “bridge to definitive surgery,” and to what extent SEMS can provide durable patency without a need for any surgery in palliative patients. The focus was on the detection of predictive factors for stent-failure and device-related complications.
Material and Methods
We performed a retrospective single-center analysis of all consecutive patients who underwent stenting procedures due to acute symptomatic colorectal obstructions at the Sisters of Charity Hospital Linz, Austria between November 1996 and December 2011. All patients suffered from different types of high-grade stenosis or complete bowel obstruction. The standard protocol for emergency treatment of left-sided colonic or rectal obstruction in our hospital consists of immediate stenting to restore luminal patency. Immediate surgery is only considered if stenting fails or cannot be performed (e.g. because of a tumor in the lower part of the rectum). Data were collected from the surgical and radiological institutions’ medical records (SAP®) and from the patients’ medical files, and authors from two other mentioned surgical Departments (Salzburg and Neuwerk) have made substantial contributions to the study’s concept and design with advisory activity for collection, analysis, and interpretation of data as well as through review of the intellectual and scientific content of this work.
Patient Categorization
Patients were divided into two groups with either benign or malignant obstructions. The second cohort was subdivided into a group with an indication for “bridge to surgery” in accordance to the classification of the International Union against Cancer seventh edition 2009 and a group for palliative treatment. Obstructions caused by locally progressed and/or metastasizing extracolonic tumors of visceral, urological, or gynecological origin were included into the palliative group. Benign obstructions in the first cohort comprised colorectal obstruction due to inflammatory bowel disease—including diverticulitis, colitis, and anastomotic strictures.
Technical And Clinical Stenting Success And Failure
Technical success was defined as a completed stenting procedure, clinical success as a sufficient decompression through one or more SEM(S) as well as adequate bowel movement within 48 h of the procedure.
Stenting Technique
We examined type of SEMS and insertion technique with endoscopic guidance in colonic obstructions and without endoscopic guidance in more highly located rectal obstructions. Endoscopy was required to pass the rectosigmoideal junction and distal parts of the colon up to the distal end of the obstruction in order to safely position the guide wire under visual control. All stenting procedures were performed by two interventional radiologists in the same technique: either via the working channel of the endoscope and with additional guidance by a pivotable fluoroscopic X-ray unit or without a need for endoscopy but threaded over a flexible, maneuverable guide wire with a bent, soft tip and subsequent deployment under radiographic control. The successful passage of the guide wire through the obstruction under radiographic control to ensure its correct, intraluminal position is the key point of the procedure—regardless of whether or not endoscopic assistance was employed. Stenting was never attempted without previous correct guide wire insertion. The length of the stenosis and the exact location of its proximal end were defined by endoluminal injection of iodine-containing contrast medium. This was important for choosing the adequate length of the SEMS and for its placement because the stent’s funnel-shaped oral opening should not stand out over the proximal end of the obstruction in order to avoid iatrogenic obstructions through the stent itself. All SEMSs were inserted under sedation without the need for general anesthesia.
Complications
Stent-related complications were divided into minor (i.e. stent dislocation /migration or occlusion with conservative, endoscopical, or surgical management) or major complications (i.e. need for emergency surgery because of perforation, bleeding, or uncontrollable pain). They were additionally grouped according to Clavien–Dindo’s classification of surgical complications (post-interventional in our cases) and time of occurrence within or beyond 30 days after implantation. Stent occlusion during the follow-up period was an issue mainly in the palliative settings. It was defined as re-obstruction by tumor ingrowth with or without solvable fecal impaction. Predictors for technical and/or clinical stenting failure and complications were determined by multi- and univariate regression analyses.
Follow-Up
Follow-up data were collected for all patients with malignant disease in accordance with defined oncological aftercare quarterly during the first 2 years and 6-monthly thereafter for up to 5 years. In palliative patients without recent follow-up data or without confirmation of death, a telephone interview with the patient, his next of kin or with the attending family doctor was conducted in June 2013. Non-surgical patients with benign conditions (inflammatory bowel disease or anastomotic strictures) and scheduled conservative treatment were followed up by endoscopic control and stent removal between 2 and 6 weeks after stenting.
Statistics
Statistical analysis was performed using SPSS—Statistical-Analysis Software Version 20 (SPSS Inc. Chicago, IL, USA). Population homogeneity was conducted using independent t-test or Mann–Whitney U-Test. Multivariate regression analysis for categorical data to determine predictors of stent failure and complications was performed. For correlated proportions in the marginals of a 2 × 2 contingency table, McNemar’s test and, in some cases, descriptive statistics were used. A p-value of less than 0.05 was regarded statistically significant.
Results
Overall Results
Within an observation period of 15 years, overall 204 patients were included (98 female). The median age was 74 years (range: 40–96). Patients’ disease characteristics and management is depicted in Fig 1. Median length of hospital stay was 20.8 days (range: 1–131). Of the 204 patients, 30 (14.7%) needed one or more re-stenting procedures due to stent dislocations, inadequate stent expansion, or colorectal re-obstructions during the follow-up period. Overall 255 stents were used in these 204 patients. Table 1 shows the types and number of stents employed, their main characteristics, and the indications for placement. The overall technical success rate of the procedures was 92.5% (236 of 255 stents), and clinical success as defined was achieved in 86.8% (177 of 204 patients). These numbers do not add up to the immediate surgery cases because of the opportunity of re-stentings in early dislocations and due to a number of stent-in-stent procedures in order to increase radial force for adequate stent expansion. Of overall 255 stenting procedures, 169 SEMSs (66.3%) were inserted with endoscopic guidance in 131 of 204 patients (64.2%). In view of complications, it did not make a difference whether or not endoscopy was required for stent placement (31/169 vs 15/86; p = 0.88). However, technical and clinical stenting failure occurred more frequently in the group of endoscopically assisted stent placements—chiefly due to poor accessibility of more proximal colonic segments (technical failure: 16/169 vs 3/86, p = 0.08; clinical failure: 22/131 vs 5/73, p = 0.04).
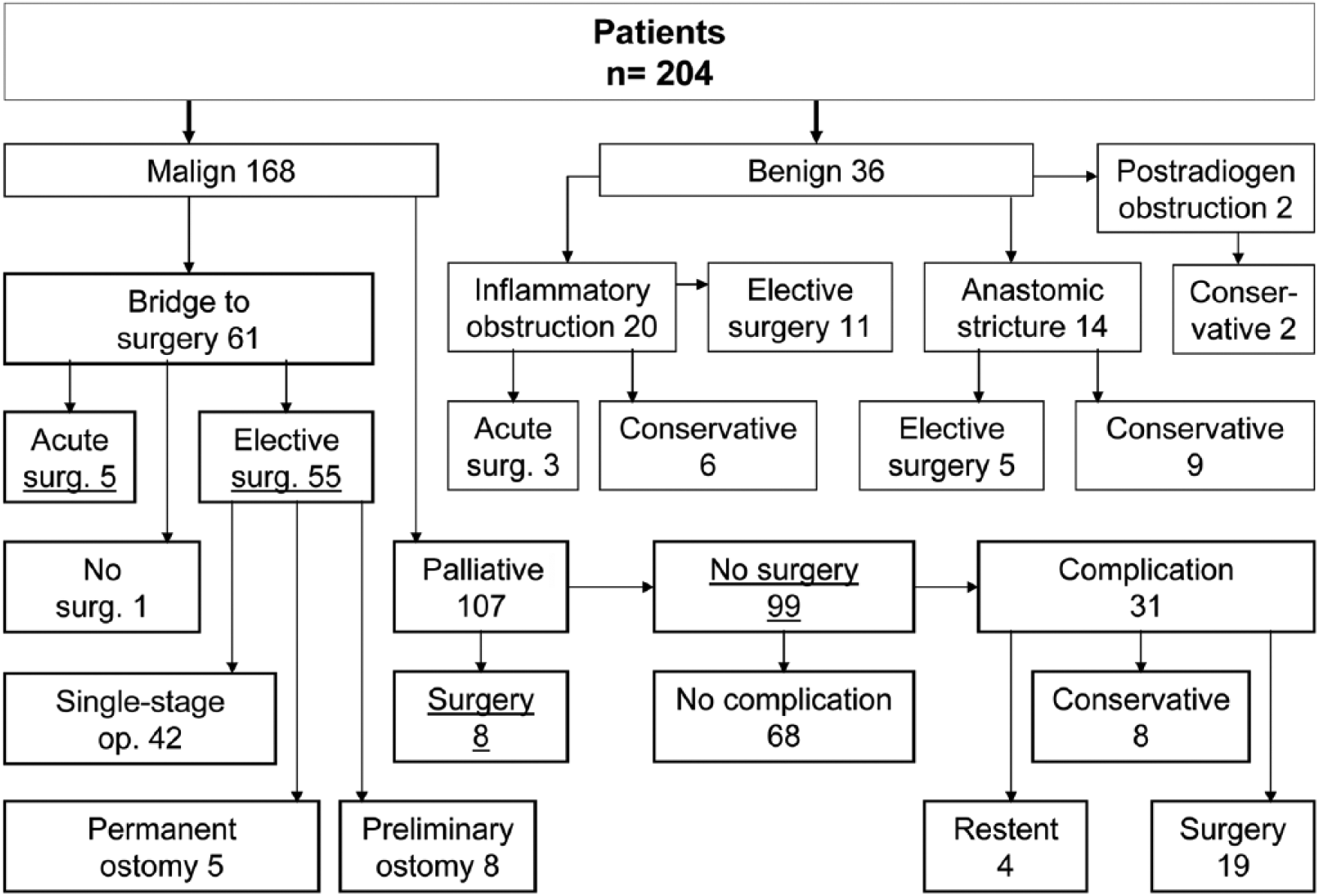
Patients disease characteristics and management.
SEMS characteristics and indications.
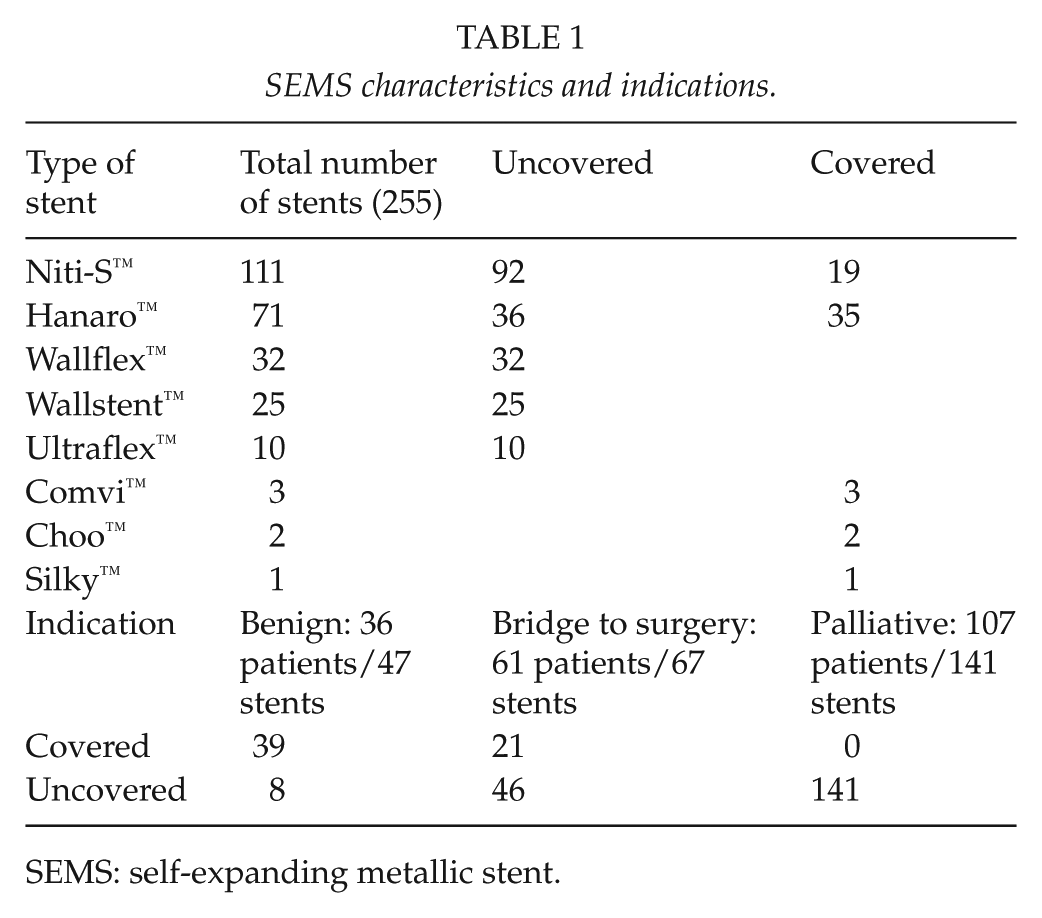
SEMS: self-expanding metallic stent.
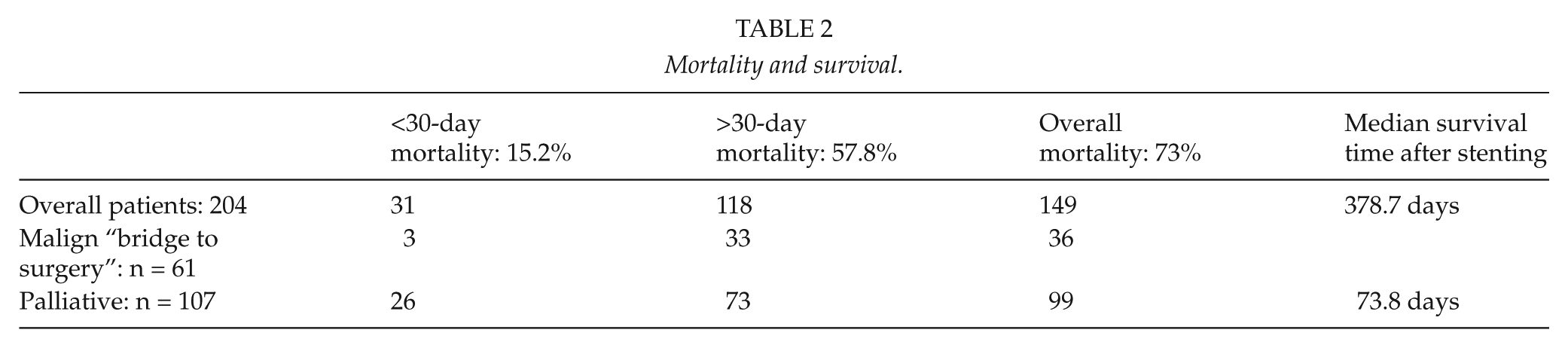
Follow-up was median 4.3 years, ranging from 1 to 15 years. Overall follow-up data were made available for 196 of 204 patients (96%). Median survival time after stenting for malignant indications, overall mortality, and 30-day mortality are shown in Table 2.
Mortality and survival.
Overall Complications
Complications were divided into minor (40/204, 19.6%) and major (6/204, 2.9%) events. A total of 32 (69.5%) complications occurred in the palliative group, 5 (10.9%) in the bridge to surgery group, and 9 (19.6%) in the benign group. The rate of adverse events was connected to the duration of stenting. Complications according to the Clavien–Dindo’s classification are outlined in Table 3. All six major complications were perforations and occurred in obstructions caused by extracolonic tumors (n = 3) or obstructions following diverticulitis (n = 3). No perforation occurred in obstructions resulting from primary colorectal cancer. No death was related to stenting complications.
Clavien–Dindo’s classification of surgical complications (19).
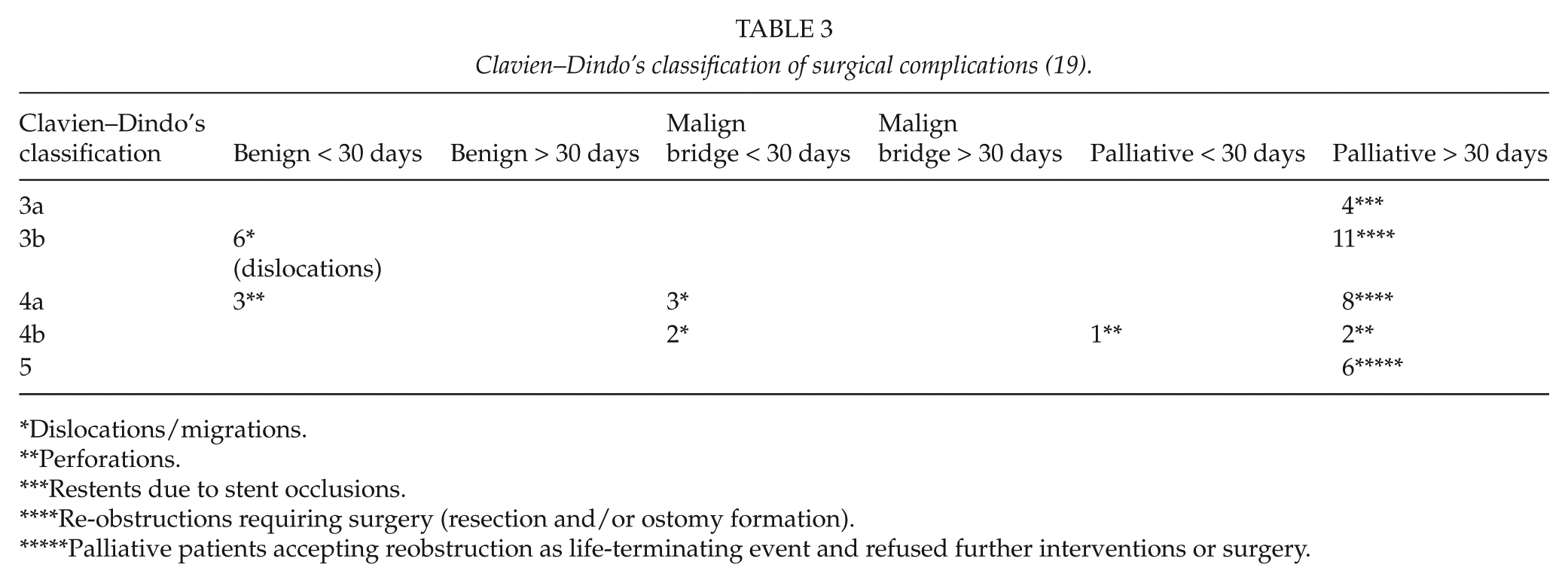
Dislocations/migrations.
Perforations.
Restents due to stent occlusions.
Re-obstructions requiring surgery (resection and/or ostomy formation).
Palliative patients accepting reobstruction as life-terminating event and refused further interventions or surgery.
Benign Obstruction Results
From overall 36 patients, 20 patients suffered from inflammatory bowel disease. Immediate surgery was required due to perforations (n = 3), and elective surgery (n = 11) due to stent dislocations and/or persistent or recurrent stenosis. The six remaining patients were treated conservatively. Two patients with obstructions after radiotherapy were successfully stented. Of 14 patients with anastomotic strictures, 9 found long-term cure through stenting.
“Bridge to Surgery” Results
From 61 “bridging” patients, 55 (90.2%) underwent elective surgery and 42 of 55 (76.4%) were subjected to a single-stage operation with primary anastomosis and without ostomy.
Palliative Stenting Results
Out of 107, 7 (6.5%) palliative patients underwent elective bowel resection, whereas only 1 (0.9%) patient had to undergo emergency surgery. During the follow-up period, 31 (31.3%) of our 99 nonsurgical, palliative patients required re-admission because of stent occlusions by tumor ingrowth with or without fecal impaction. These occlusions occurred after a median time of 61.3 days (range 0–605). A total of 4 patients underwent re-stenting procedures, and 19 patients underwent surgical bowel resection and/or ostomy creation. Eight patients were treated conservatively (six patients refused further invasive treatment and two patients only required enema/laxatives). In summary, 72 of 107 palliative patients (67.3%) had received a durable palliation through stenting until the end of the observation period which was defined either by the patient’s documented death or in all other cases by a telephone interview in June 2013.
Predictors of Stenting Failure Regarding Technical and Clinical Success and Complications
The localization of the obstructions is shown in Fig 2. Obstructions of the splenic flexure were associated with a remarkably higher rate of technical failure compared to other localizations (5/13, 38.5% vs 22/191, 11.5%; p = 0.017, odds ratio (OR) = 4.80, 95% confidence interval (CI) = 1.44–15.98) (Table 4).
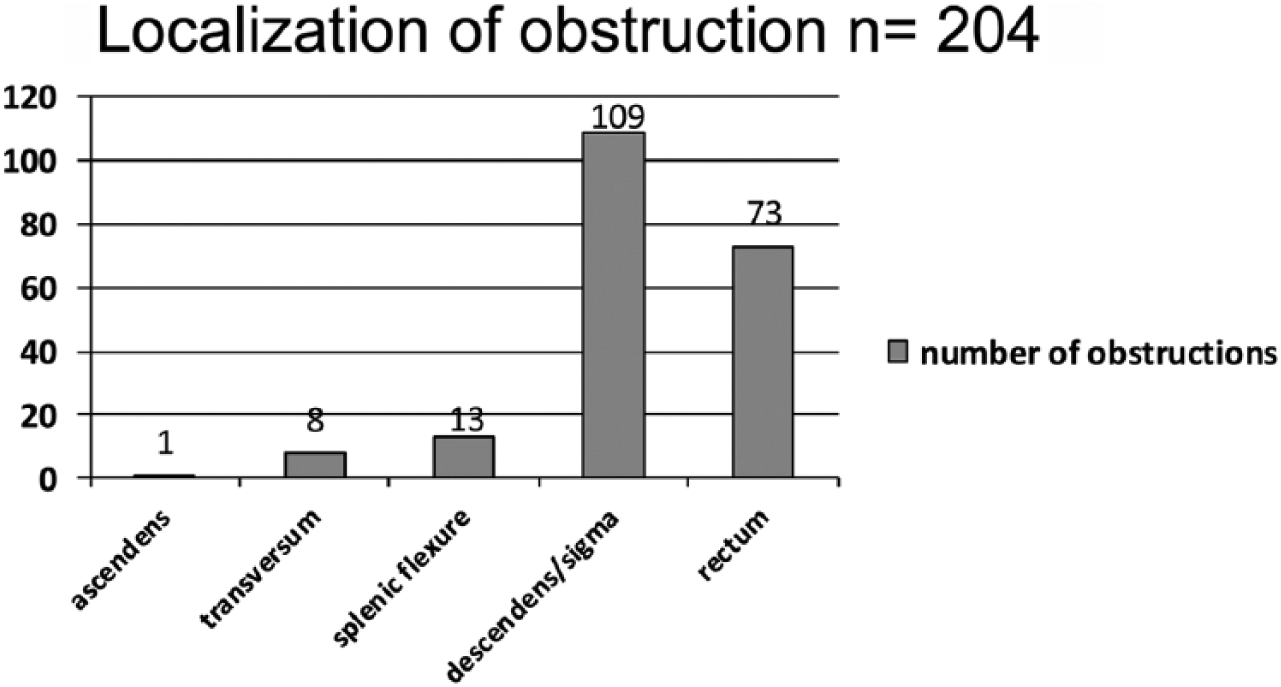
Localization of obstruction.
Predictors of stenting failure and complications.
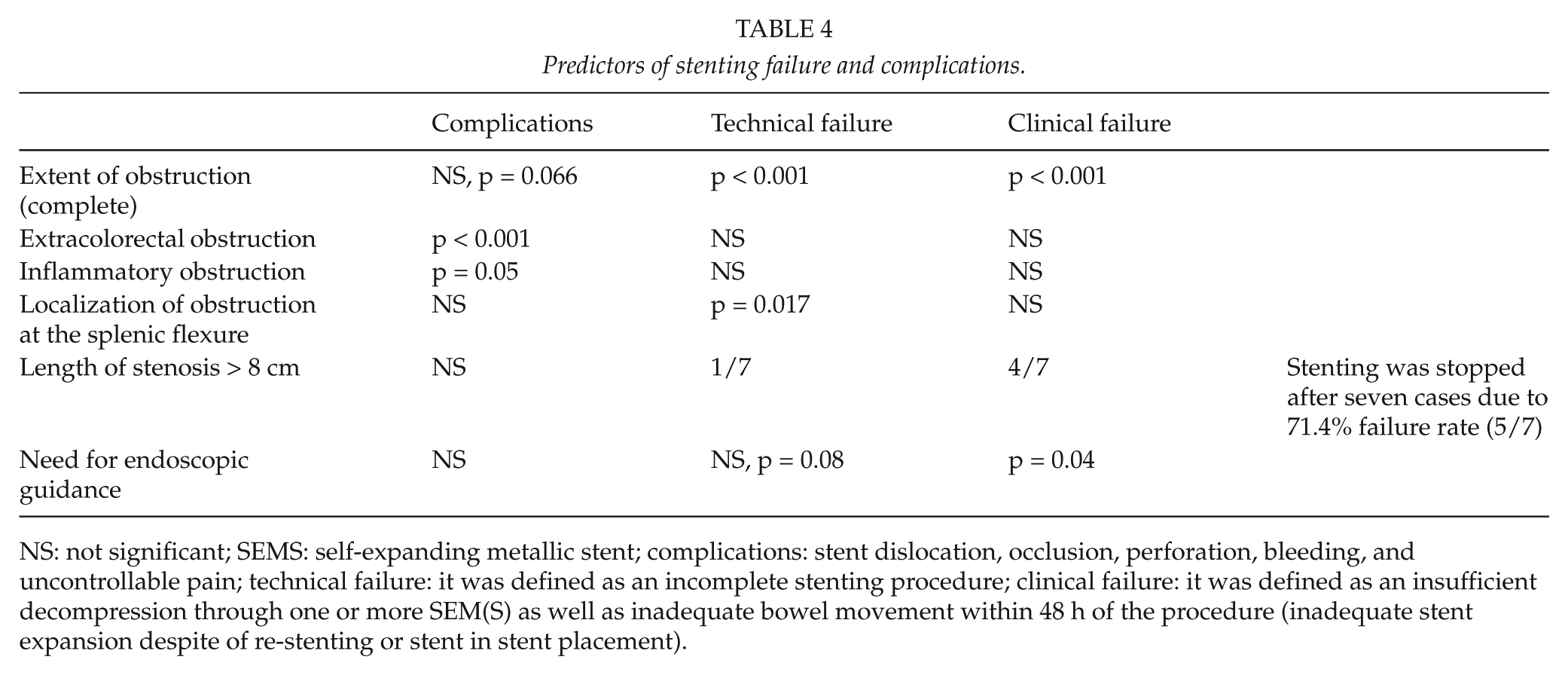
NS: not significant; SEMS: self-expanding metallic stent; complications: stent dislocation, occlusion, perforation, bleeding, and uncontrollable pain; technical failure: it was defined as an incomplete stenting procedure; clinical failure: it was defined as an insufficient decompression through one or more SEM(S) as well as inadequate bowel movement within 48 h of the procedure (inadequate stent expansion despite of re-stenting or stent in stent placement).
Of further interest in the study was the group of patients with obstructions caused by extracolonic obstructions. These affected 26 of our 204 patients (12.7%). All 26 belonged to the palliative group and were compared with the remaining 81 palliative patients. Complications significantly more often occurred in this subgroup of our study cohort than in palliative patients suffering from colorectal cancer (15/26, 57.7% vs 17/81, 21%; p = 0.001, OR = 5.13, 95% CI = 1.99–13.19). Three perforations occurred in the group of extracolonic obstructions (3/26, 11.5%) and none (0/81) in the colorectal cancer group (p = 0.013, OR = 4.52, 95% CI = 3.15–6.48).
With regard to benign conditions, overall complications were significantly more frequent in inflammatory diseases than in anastomotic strictures (8/20, 40% vs 1/14, 7.1%, p = 0.05, OR = 8.66, 95% CI = 0.93–79.95).
The extent of obstruction showed a significant influence on the likeliness of stenting failure. Complete obstructions occurred in 42 of 204 patients (20.6%). Technical success in these patients could be achieved in 31/42 patients (73.8%) and clinical success was documented in 28/42 patients (66.6%). In incomplete bowel obstructions (162 of 204 patients, 79.4%), the results were generally better with a technical stenting success in 154/162 (95.1%) and clinical success in 149/162 (92%) cases (technical success: p < 0.001, OR = 0.15, 95% CI = 0.05–0.39; and clinical success: p < 0.001, OR = 0.17, 95% CI = 0.07–0.41). Upon comparison of complications, we did find that they occurred more frequently in the group with complete obstructions, but we could not document statistical significance (14/42, 33.3% vs 32/162, 19.8%, p = 0.066, OR = 2.03, 95% CI = 0.96–4.29).
With regard to stenting success, the length of stenosis was evaluated further and a cut-off value of 8 cm was determined. Only in seven of our early cases, we noticed a stenosis length of more than 8 cm. Because of an overall technical or clinical failure rate of 71.4% (5/7), stenting procedures in less common cases of a present stenosis longer than 8 cm were stopped in 2001. We found that malignant obstructions in our patients do hardly exceed 8 cm.
Discussion
The safety and efficacy of endoscopic colonic stenting as a “bridge to surgery” in the management of intestinal obstruction is currently discussed controversially (20). It has been suggested that surgery should be favored due to a higher rate of clinical success (21). In contrast, other authors suggest that initial stenting eventually allows for a higher rate of primary anastomosis, reduces morbidity, and cuts length of hospital stay short (5, 6). In our experience, patients can be effectively resuscitated and tumor-staging (including colonoscopy through the stent to rule out a further colonic pathology) can be completed (22). Patients can then undergo elective surgery. Therefore, stenting is our therapy of choice. We aim to delay surgery for a minimum of 10 days in order to allow for a sufficient recovery of the intestinal wall’s tonicity.
The use of SEMS in palliative patients is more commonly accepted as a reliable alternative to surgery because of its lesser invasiveness with shorter associated hospital stay and less morbidity and mortality (23). Our study results confirm this observance with a high rate of durable palliation, but they also support the results published by De Gregorio et al. (24), who reported stent-related long-term complications of a considerable 42% in a multicenter trial in 1998.
Stent management in completely obstructing colorectal cancer is also discussed controversially. Lamazza et al. (25) conclude that stenting in this subgroup seems to be feasible and safe with high technical and clinical success rates, but a need for repeated colonoscopy due to re-obstruction is given about one-third of the cases. Van Hooft et al. (26) report high perforation rates, which is why that trial was stopped early. Stenhouse et al. (27) suggest that complete obstruction is not a contraindication for stenting but state that success is less predictable. More recent data of our study suggest that stenting in cases of complete obstruction is associated with lower rates of technical and clinical success. This is because of inefficient guide wire insertions and/or insufficient stent expansion.
Another interesting and discordant question is, whether or not stenting of obstructions caused by extracolonic tumors leads to different results compared to colorectal tumor obstruction. Some authors do not support the routine use of SEMS in this subgroup because of the low rate of success and increased number of complications (13, 28). In contrast, Kim et al. (11) did not notice differences in clinical success, complications, and stent patency when colorectal and extra-colorectal obstructions were compared. In our cohort of patients, stenting of extracolonic obstructions was afflicted with a significantly higher rate of complications. We currently favor a tailored therapeutic approach with more restrictive indications for stenting in this particular subgroup. In addition, all treatment options have to be evaluated in an interdisciplinary “tumor board.” However, the emergency character of obstructions frequently calls for an urgent, interdisciplinary, and individualized board-meeting.
The evidence for using SEMS in the treatment of benign bowel obstructions is scarce. Forshaw et al. (15) presented 11 patients with stenting in benign colorectal obstruction and demonstrated satisfactory results for anastomotic strictures, but recommended that SEMS should be avoided in acute disease because of a higher incidence of complications. Day et al. (14) reported that temporary stent insertion is a safe procedure in benign colorectal anastomotic strictures, but surgery will be necessary in a considerable number of patients due to primary or secondary failure of stenting. The results of our study are consistent with these findings. Complications occurred significantly more often in patients with inflammatory bowel disease. All three cases of stent perforations in benign indications occurred after stenting of obstructions caused by the effects of diverticulitis. They all occurred within 7 days after stenting, chiefly because of the intestinal wall’s weakness secondary to inflammation.
To our knowledge, only one study investigated the role of the length of obstructions in the context of SEMS placement and reported poor results for SEMS longer than 8 cm (29). Our early data suggest that stenting in this subgroup is afflicted with an unacceptably high failure rate and should therefore be avoided.
Another point of interest is the localization of the obstruction in relation to clinical outcome. Distal obstructions are generally reported to provide better outcomes with stenting (29). We evaluated the obstruction’s localization in an angled colonic area with regard to stenting success and complications. We noted a significantly poorer outcome as soon as obstructions were localized at the splenic flexure. In this context, the influence of the investigator’s and the industry’s (purveyor of new devices and improvement of stent material properties) learning curve can be discussed to increase stenting success in angled colonic areas as well as in each other localization.
The influence of SEMS as a “bridge to surgery” with regard to oncological outcome is elusive: Knight et al. (8) summarized from their retrospective analysis that long-term survival shows no differences in groups of primary stenting compared to surgery. Other authors also noted that stenting was without adverse oncological consequences (5, 6). A comparative analysis between SEMS used in obstructive colorectal cancer and elective surgery due to nonobstructing colorectal cancer showed that bridge to surgery stenting is possibly associated with adverse oncological outcome (30). A recent study is concerned with stenting versus emergency resections and shows that stenting is associated with a significantly higher local recurrence rate (31). Further randomized studies are warranted to evaluate the oncological safety of this technique and potential differences in overall survival rates in comparison of stenting and surgery.
A limitation of our study was the single-arm retrospective observational design. Points of strength were the inclusion of a large number of consecutive patients, who underwent colorectal stenting procedures over a long period of time, the tenacity through which we gained a long follow-up period, and the standardized and consistent routines for stent insertion. Our analysis also extends to areas of interest regarding predictors of complications and failure for which until now only rare data were available.
In conclusion, the employment of stents for different types of emergency colorectal obstructions is commonly accepted as a less invasive, safe, and effective alternative to surgery. The high rate of elective single-stage surgery in “bridge to surgery” patients and the high rate of durable palliation without the need for any classical surgical intervention in palliative patients are beneficial. The use of SEMS in patients who present with stenosis of certain characteristics, including complete or long segment obstructions, inflammatory or extra-luminal colorectal obstructions, or localization at the splenic flexure, is associated with a higher risk of complications and technical and/or clinical stenting failure.
Footnotes
Acknowledgements
The kind statistical support by Mag. Michael Stumpner is greatly acknowledged
Declaration of Conflicting Interests
Gernot Köhler, Stavros A. Antoniou, Michael Lechner, Franz Mayer, Johann Mair, and Klaus Emmanuel have no conflicts of interest or financial ties to disclose. All authors do disclose all institutional or corporate/commercial relationships to all the time of the data collection going back 18 years and longer.
