Abstract
Background and Aims:
A systematic review and meta-analysis was performed to investigate the risk factors associated with periprosthetic fracture after total hip arthroplasty.
Material and Methods:
We searched potential studies in the following databases: MEDLINE, Embase, Web of Science, SCOPUS and Cochrane CENTRAL up to December 2013. Newcastle–Ottawa Scale was used to evaluate the methodological quality, and Stata 11.0 was used to perform all the analyses.
Results:
Seven studies altogether, including 1069 cases of periprosthetic fractures and 74,776 controls, were included in the meta-analysis. Compared to those absent following demographic or medical conditions, patients involved with female gender (odds ratio, 1.534; p < 0.001), advanced age (>80) (odds ratio: 4.203; p < 0.001), revision (odds ratio: 4.398; p < 0.001), rheumatoid arthritis (odds ratio: 2.503; p < 0.001), osteonecrosis (odds ratio: 1.563; p = 0.009), and implant type of Exeter (odds ratio: 1.511; p = 0.017) were more likely to sustain periprosthetic fractures. Osteoarthritis (vs not) (odds ratio: 0.449; p < 0.001) was identified a protective factor for periprosthetic fractures after total hip arthroplasty. The other factors, including lower ages, American Society of Anesthesiologists ≥ 3, and other implant types, were not significant risk factors for periprosthetic fractures.
Conclusions:
These medical conditions as reminder should be kept in clinicians’ mind and close follow-up should be implemented in patients involved for preventing the occurrence of periprosthetic fractures after total hip arthroplasty.
Introduction
As a devastating complication of total hip arthroplasty (THA), periprosthetic fracture (PF) is not rare with the reported incidence from 0.1% to 4.1% (1–4), and has been increasing due to the age-profile and population structure (1, 5–7). Consequently, PF patients have higher mortality rates of 7.3% (8) and 11.0% (9) within the 6-month and 12-month postoperatively, respectively, which were almost similar to that after treatment for hip fractures. Furthermore, patients who survived after PFs suffered functional deterioration, limited mobility, and even had fourfold higher risk of hospitalization for postoperative complications, especially in the first year postoperatively (2, 8). Therefore, it is vital to identify risk factors to prevent the occurrence of the PFs in THA patients.
A great number of risk factors have been identified for PFs, including advanced age (10–12), female gender (13, 14), implant type (15, 16), prior diagnosis of osteoporosis (10, 17, 18) and rheumatoid arthritis (RA) (10), revision (4, 18, 19) and aseptic loosening (Tsiridis et al. (7) and Incavo et al. (20)). However, some limitations had to be mentioned in these studies such as a small sample size and the inclusion of a single or very few potential risk factors. In addition, intense controversies remain regarding these risk factors for PFs in literature. For example, being female was considered being associated with a decreased risk for PFs in some study (10) but an increased risk in the other studies (19, 21, 22). Franklin and Malchau (23) conducted a systematic review of studies on PFs in 2007, with a qualitative description rather than a quantitative meta-analysis on the potential risk factors. From then on, more recent clinical studies were performed on the identification of risk factors for PFs after the treatment of THA. Given the limitations above, efforts to both update and quantitatively analyze are warranted to improve risk factor screening in THA patients and to avoid the occurrence of PFs.
Accordingly in this study, we performed this meta-analysis using data available from previous original studies to summarize these factors and expect to provide an evidential base, from which surgeons could better determinate at-risk patients and target them with better prevention and intervention strategies to prevent the occurrence of PFs
Methods
Search Strategy
A computerized search was performed on MEDLINE, Embase, and Cochrane central database (all through December 2013) for the potentially relevant studies according to the main key words as follows: “factor” or “predictor” or “risk” AND “Periprosthetic fracture.” All articles identified as potentially relevant were obtained and reviewed by a research assistant. Also, a manual search of references by this assistant was performed in the identified articles and systematic reviews for any additional eligible articles.
Two reviewers (Chen and Liu) independently evaluated the titles and abstracts of the identified articles. Only full-text articles were eligible and included in this meta-analysis. The inclusion criteria were as follows: (1) only subsequent fracture around the prosthesis (rather than femoral stem fractures) in patients with THA was considered PF; (2) a study was performed to explore risk factors for PFs, and cases and controls were defined based on the presence of PFs (with or without); (3) sufficient data were published for estimating an odds ratio (OR) with 95% confidence intervals (CIs).
Data Extraction
The two reviewers (Chen and Liu) independently extracted seven publications for meta-analysis to a standard form. The following variables were extracted from each study: first author’s name, publication year, country, follow-up periods, significant risk factors, the number of case and control groups, and the number of citations for each potential risk factor for PFs. Any disagreement was settled by discussion and a consensus was reached for all data.
Quality of Included Studies
The two reviewers (Chen and Liu) independently evaluated the quality of the included studies using the Newcastle–Ottawa Scale (NOS) (24) based on the three main items: the selection of the study groups (0–4 points), the comparability of the groups (0–2 points), and the determination of either the exposure or the outcome of interest (0–3 points), with a perfect score of 9.
Meta-Analyses
We estimated ORs and 95% CIs and pooled from across studies to assess the associations between potential risk factors and PFs with a p < 0.05 indicating significance. Heterogeneity between studies was tested qualitative by Q-test statistics with significance set at p < 0.10 (25) and quantitatively tested by I2 statistics, with I2 more than 50% indicating large inconsistency. A random-effect model was used to calculate pooled ORs in the case of significant heterogeneity (p < 0.10 or I2 > 50%); otherwise, a fixed-effect model was used (26). The outcomes of meta-analysis for risk factors were summarized using a forest plot. Publication bias was assessed by Begg’s test and a funnel plot, a p < 0.10 was considered significant. All analyses were performed using the software Stata 11.0 (Stata Corporation, College Station, TX).
Sensitivity Analysis
Furthermore, to explore causes of heterogeneity and make the result more reliable, sensitivity analysis was performed based on the following factors: assessment quality of included studies, areas of participants, and publication year of studies.
Results
Literature Search and Basic Characteristics of Studies
A total of 92 initially selected references were retrieved, of which seven studies (all were case-control studies), altogether including 1069 cases and 74,776 controls were identified as eligible (Fig. 1). Two studies were designed as control-matched, thus the remaining five studies, including 996 PFs and 75,210 THAs from registry databases could be appropriate for calculating the accumulated incidence (1.32%). The mean time after the THA operation for the PFs was 5.9 years, and 53.4% of PFs occurred within 5 years postoperatively. The basic characteristics of included studies and participants are summarized (Table 1). Data on 10 potential risk factors for the PFs were abstracted from the included studies and are presented in Table 2 along with the number of citations for each risk factor.
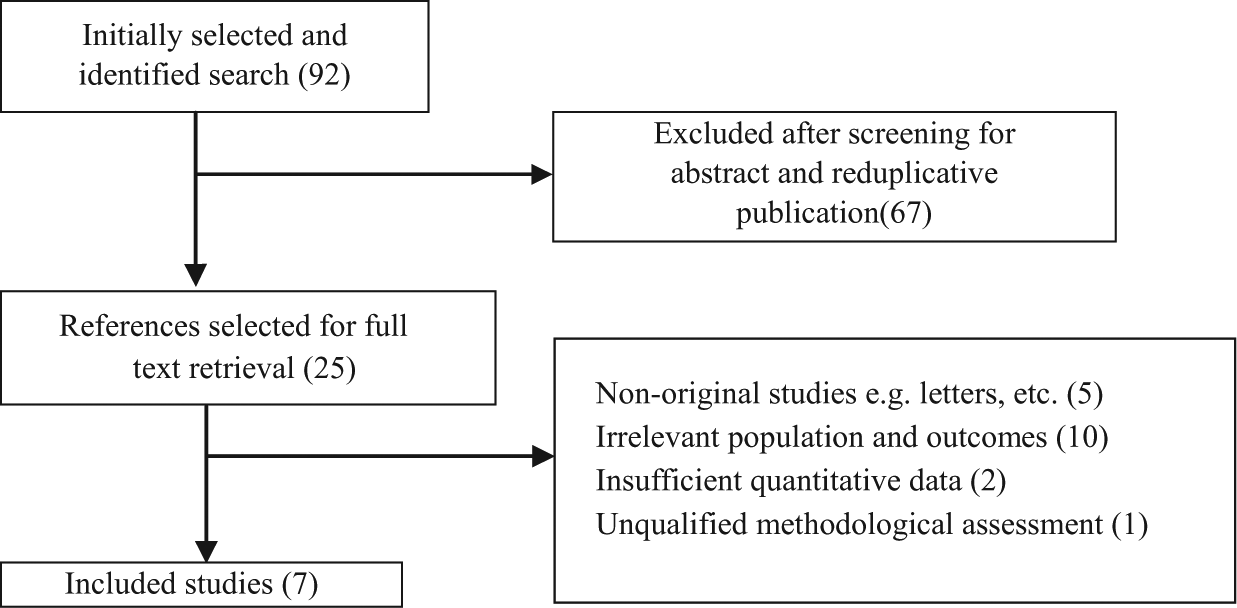
Flow diagram of literature search.
The detailed information on basic characteristics of seven included studies and participants.
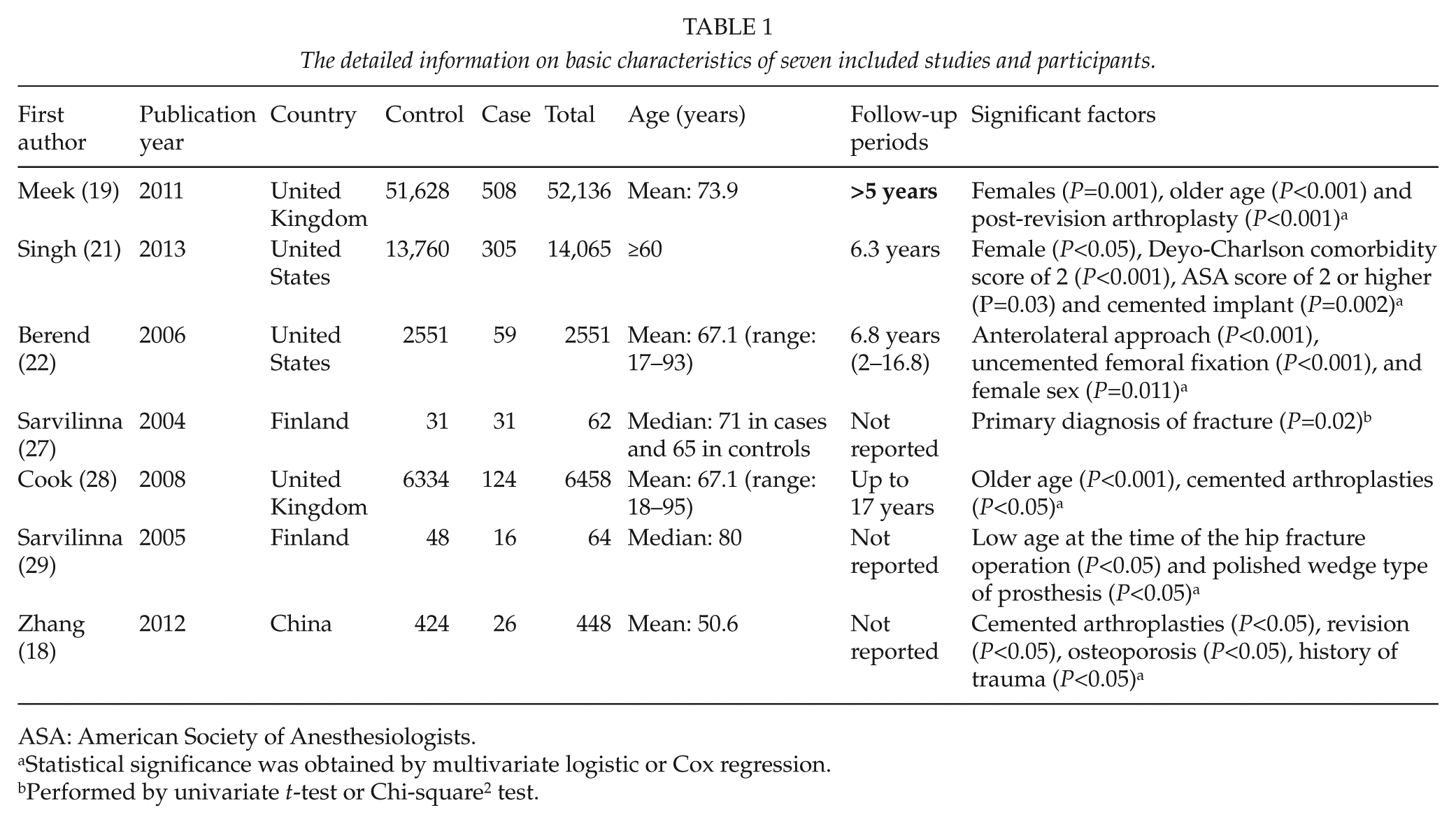
ASA: American Society of Anesthesiologists.
Statistical significance was obtained by multivariate logistic or Cox regression.
Performed by univariate t-test or Chi-square2 test.
Detailed data on 10 potential risk factors and the pooled outcome of meta-analyses.
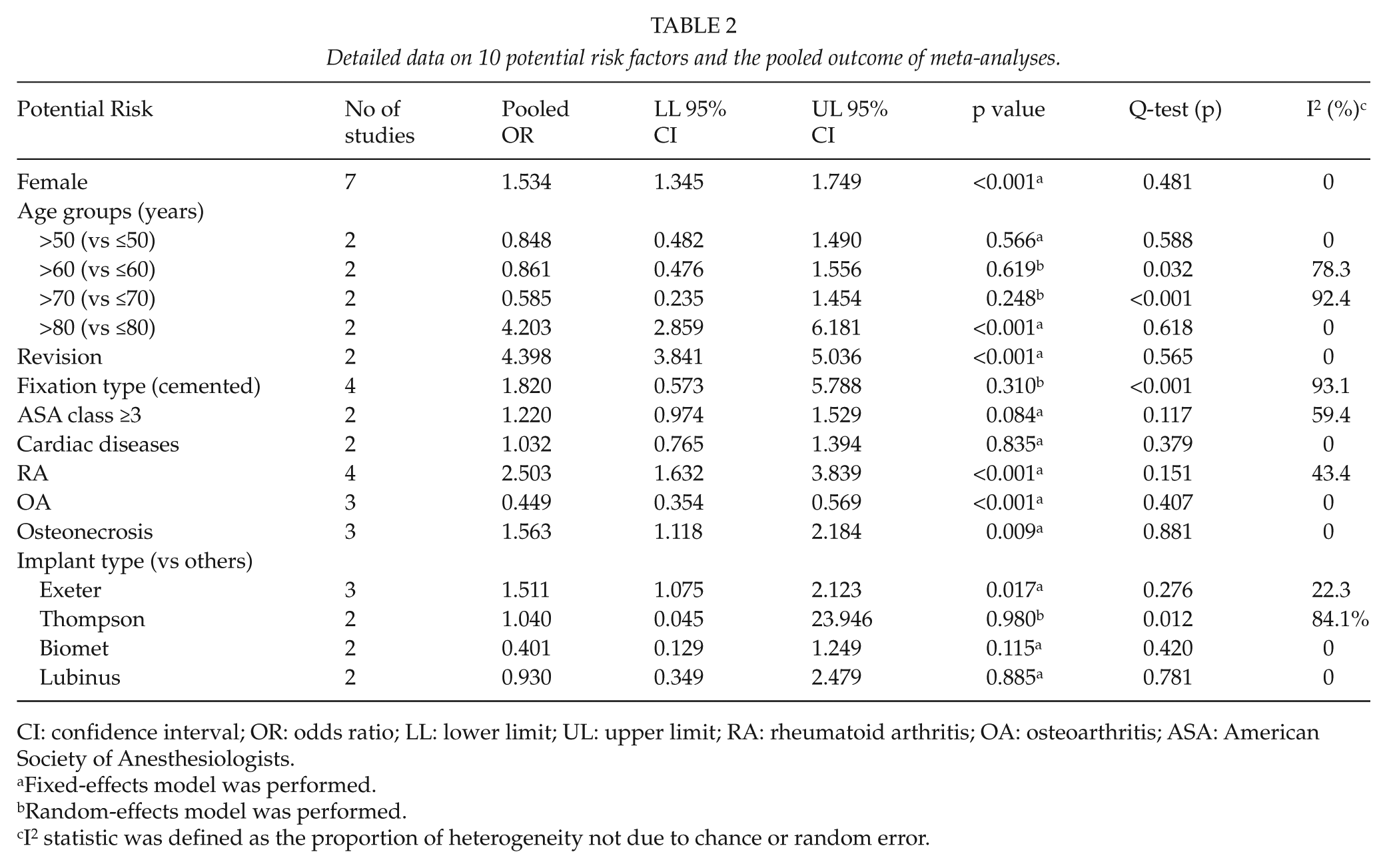
CI: confidence interval; OR: odds ratio; LL: lower limit; UL: upper limit; RA: rheumatoid arthritis; OA: osteoarthritis; ASA: American Society of Anesthesiologists.
Fixed-effects model was performed.
Random-effects model was performed.
I2 statistic was defined as the proportion of heterogeneity not due to chance or random error.
Methodological Quality Assessment
The outcome of quality assessment for included studies was as follows: one study scored 9 (19), four studies (18, 21, 22, 28) scored 8, and two studies (27, 29) scored 7.
Pooled Analyses of Risk Factors
A meta-analysis of combinable data was conducted to analyze the risk factors for the PFs, and the main results were shown in Table 2. The combined OR ranged from 1.511 to 4.398. Significant heterogeneity was observed among studies evaluating as risk factors for fixation (I2: 93.1%). Compared to those absent following demographic or medical conditions, patients involved with female gender (OR: 1.534; 95% CI: 1.345–1.749), age (>80 years) (OR: 4.203; 95% CI: 2.859–6.181), revision (OR: 4.398; 95% CI: 3.841–5.036), RA (OR: 2.503; 95% CI: 1.632–3.839), osteonecrosis (OR: 1.563; 95% CI: 1.118–2.184), and implant type of Exeter (OR: 1.511; 95% CI: 1.075–2.123) were more likely to sustain PFs after THA. Osteoarthritis (OA) was considered to be a decreased risk factor associated with PFs (OR: 0.449; 95% CI: 0.354–0.569). Fixation type of cemented, higher American Society of Anesthesiologists (ASA) class of 3 and 4, ages (50, 60, and 70 years, respectively) and implant type of Thompson, Biomet and Lubinus were identified not the risk factors for the PFs (p > 0.05). The outcome of analysis for advanced age (>80 years), RA and OA as significant factors are presented by forest plots (Fig. 2
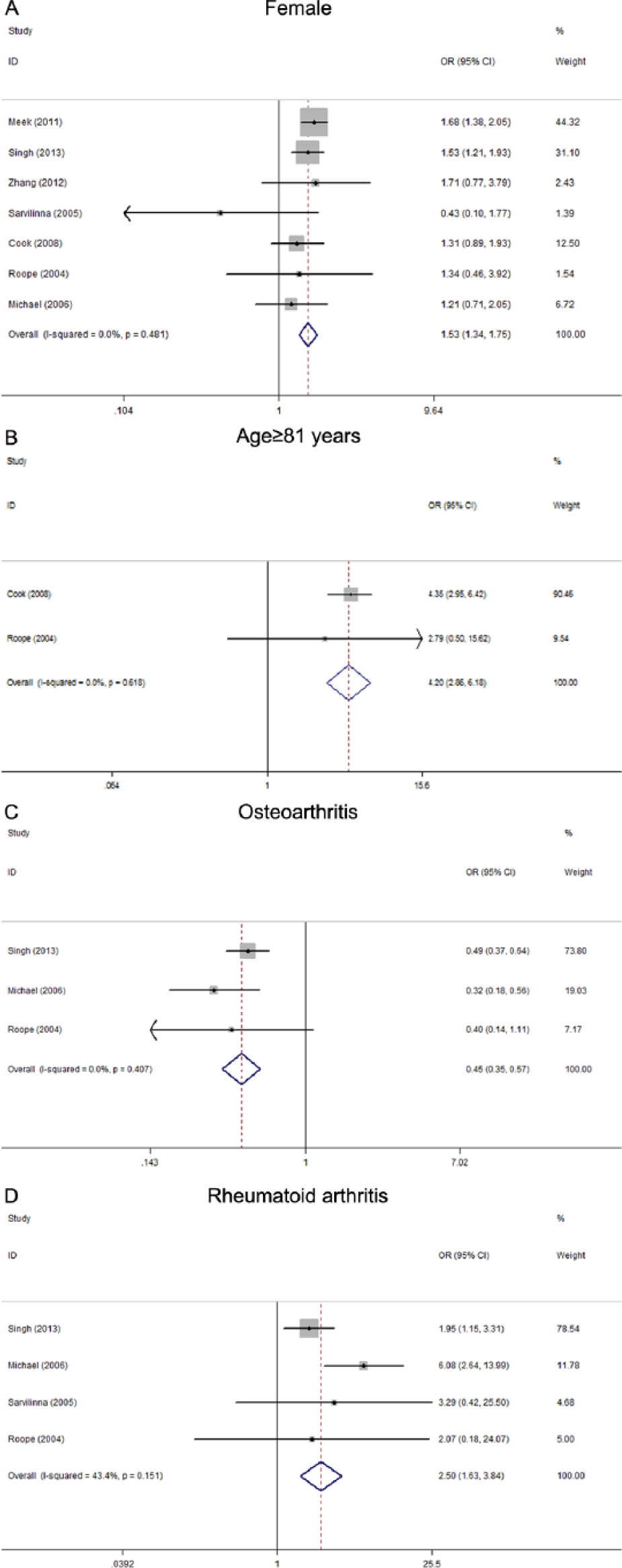
Forest plots of the meta-analysis of (A) female, (B) advanced age (>80 years), (C) osteoarthritis and (D) rheumatoid arthritis and as significant factors for the periprosthetic fractures after total hip arthroplasty. The width of the horizontal line represents the 95% CI of the individual studies, and the square proportional represents the weight of each study. The diamond represents the pooled OR and 95% CI.
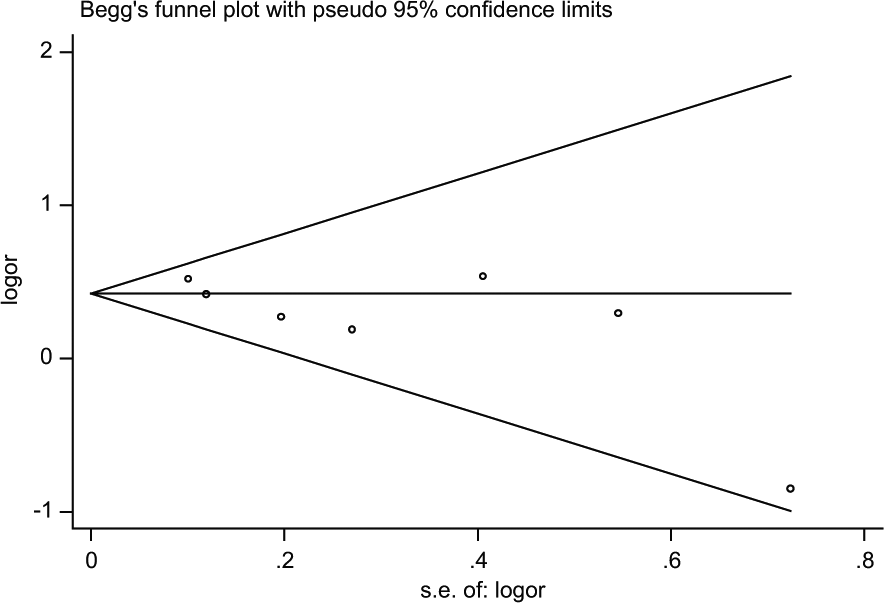
Begg’s funnel plot for publication bias (with 95% pseudoconfidence limits) of the case-control studies that investigated female as a risk factor for PFs (p = 0.133). (continuity corrected).
Sensitivity and Subgroup Analysis
The results of sensitivity analysis revealed, after excluding the study (22) with lower quality, it did not affect the statistical significance of fixation type (cemented) as potential risk factor (I2, 43.5%; p for heterogeneity, 0.828); hence, the meta-analytic association reported in this review for the factor of fixation type was robust to the extent and the outcome was reliable.
Discussion
PF after the THA is not uncommon in elderly patients, and the incidence of the PFs was reported to be 0.1%–4.1%; most of them occurred at proximally 6 years postoperatively (27, 28). Given the higher rate of mortality, more complex complications, and poorer clinical outcome, identification of the risk factors leading to PFs is quite important. The outcome of pooled individual studies (from registry databases) in this review revealed an incidence rate of 1.32% (996/75,210) for PFs. Meta-analysis of combinable individual results suggests that multiple risk factors are associated with PFs after the THA. Statistically significant associations were identified for female gender, advanced age (>80 years), revision, prior diagnose of osteonecrosis and RA, implant type of Exeter. The prior diagnosis of OA was demonstrated to be a decreased risk for sustaining a PF. In consideration of the clinical significance associated with PFs in aging patients (>80 years), efforts to limit individual patient risk should be increasingly important with the aging population, especially in females. Besides, clinical risk factors including revision and prior diagnoses of RA and osteonecrosis were identified significantly to be associated with PFs. Thus, this will undoubtedly improve the ability to predict the occurrence of PFs and contribute to comprehensive understanding of these risk factors for PFs to develop more effective prevention and treatment strategies.
The cemented fixation type has been discussed in many studies (18, 22, 28), but its potential role as a risk factor for PFs was inclusive and even contradictory. It is probably that significant differences in methodology, including sample sizes and a wide range of follow-up periods, resulted in significant inconsistencies in the analytical results. In this review, combinable results suggest a non-significant risk factor for the cemented type imparted to the PFs, and exclusion of the study (22) with lower quality did not affect the result, indicating the meta-analytic outcome is reliable.
In this review, OA was identified to be the only protective factor associated with PFs, which was consistent with the study by Lindahl (10) from Swedish Register. The mechanism linking OA and the protective effect for PFs was unclear. The potential impacts exerted by OA may be explained by patients’ lower activity level due to advanced age and pain from knee or hip joint inflammation. More prospective studies of large size are required to identify the protective mechanism that links OA and PFs.
It is notable that, the majority of above-mentioned risk factors could not be modified (age, gender, or a history of RA, revision, and osteonecrosis), however, the selection of implant type (Exeter) was potentially amenable. As a most common implant type used clinically, Exeter stem was designed as polished, straight, double-tapered, and itself had many inherent limitations, such as inappropriate positioning and usability to afford an adequate cement mantle (15, 16). Reports from the Swedish hip arthroplasty register have shown that the early survival of uncemented THA is inferior to cemented fixation, partly due to early PFs (10). Therefore, in clinics, surgeons might select more appropriate cemented implant type (depending upon the underlying reason for its use) such as the system, including anatomically adapted SPII prosthesis stem (cemented) and Lubinus acetabular cup both generated by LINK corporation, which was reported to be associated with a lower PF incidence and a higher survival rate even after 16 years (13).
In addition, low-energy falls from sitting and standing height result in the occurrence of more than 90% of PFs (13, 30), and preoperative diagnosis of osteoporosis has been identified as an independent risk factor for PFs (31, 32) although they were not pooled in this study due to significant heterogeneity or inconsistent data forms. Therefore, it is reasonable for clinicians to assess all patients treated by THA for bone mass deficiency and commence supplementation of vitamin D or bisphosphonates as indicated (33). Meanwhile, postoperative close follow-ups with radiography to monitor for bone mass change would seem advisable to help prevent the occurrence of PFs, which has been proved to be a cost-effective method compared to the substantial costs for the treatment of an acute PF or a planned elective revision for loosening (34). In summary, routine follow-up for patients involved with some identified significant risk factors especially for those with more combined risk factors is likely cost-effective for preventing the occurrence of PFs after THA.
In this study, some risk factors could not be combined between studies due to inconsistent data forms and a broad range of definitions or were reported in a single study. Prior diagnosis of fracture for THA was considered to be a significantly increased risk factor for PFs in the study by Sarvilinna et al. (27), and history of trauma was identified a risk factor associated with PFs, whether trauma was specified to be the hip fracture was unknown, because we could not obtain the original raw data. Other potential risk factors such us body mass index (BMI), surgical approach, prior diagnosis of dementia, lung disease, hypertension, diabetes mellitus, and congenital dysplasia were reported in a single study, hence, could not be pooled in this meta-analysis.
This study has some strengths, including the comprehensive literature search, the application of heterogeneity measures, and the careful evaluation of methodological quality. Furthermore, more than 95.1% of the PFs and 99.9% of controls were obtained from the national arthroplasty register in this meta-analysis; hence, the accuracy and cover of these were found to be more reliable. In this sense, the level of evidence in this study was higher compared to those studies designed as matched case-control or cohort in a single traumatic center. Above all, our study updates and extends upon previous systematic review performed by Franklin and Malchau, (23) and first, quantitatively evaluates the risk factors for PFs after THA, which reflects the current state of the available evidence for risk estimates.
Certain limitations in this meta-analysis had to be mentioned. First, a weakness inherent in this analysis is that only study-level data were obtained as available in the published literature, rather than raw data for each study included in our study. Second, all the included studies were retrospective case-control studies unavoidable of recall and interviewer biases, which might affect the associations between the risks and PFs. Third, the measurement of the various risk factors differed substantially from each other, either by standard grading forms or by patient questionnaires. Similarly, follow-up periods ranged broadly from several months to more than 10 years and several risk factors were discussed by no more than two studies. Therefore, a significant heterogeneity was unavoidable in this review. However, after sensitive analysis for fixation type as risk factor, I2 values lowered to below than 50%, indicating a robust analysis and a reliable result. Finally, all the subjects in the seven articles were from China, United States, United Kingdom, and Finland, although on behalf of a large number of populations, our conclusions might not extrapolate to populations of various countries and regions.
Conclusion
Although with limitations more or less, this systematic and meta-analysis provides the current estimates of the magnitude of effects. Statistically significant increased risk factors of female gender, advanced age, (>80 years), revision, RA, osteonecrosis, and implant type of Exeter were identified associated with PFs. These medical conditions as reminder should be noted by clinicians and close follow-ups should be implemented in patients involved, to prevent the occurrence of PFs after THA. OA as a protective factor for PF remains to be confirmed, and its underlying mechanism requires investigation by more studies.
Footnotes
Acknowledgements
We are grateful to J.C. and Z.W. of the Department of Orthopedics, and to X.C. and G.W. of the Department of Statistics and Applications for their kind assistance.
Declaration of Conflicting Interests
All the authors declare that they have no conflict of interest with any organization that sponsored the research.
