Abstract
Background and Aims:
The rainage (PBD) prior to pancreaticoduodenectomy (PD) is controversial. If PBD is required, large bore self-expandable metallic stents (SEMS) are thought to maintain better drainage and have fewer postoperative complications than plastic stents. The confirming evidence is scarce. The aim of the study was to compare outcomes of surgery in patients who underwent PBD with SEMS or plastic stents deployed at endoscopic retrograde cholangiopancreatography (ERCP).
Material and Methods:
This is a retrospective study of 366 patients having had PD during 2000–2009. Preceding endoscopic PBD was performed in 191 patients and nine had had percutaneous transhepatic drainage (PTD). At the time of operation, 163 patients had a plastic stent and 28 had SEMS. Due to stent exchanges, 176 plastic stents and 29 SEMS were placed in all.
Results and Conclusions:
The stent failure rate was 7.4% for plastic stents and 3.4% for SEMS (p = 0.697). A bilirubin level under 50 µmol/L was reached by 80% of the patients with plastic stents and by 61% of the patients with SEMS (p = 0.058). A postoperative infection complication and/or a pancreatic fistula was found in 26% while using plastic stents and in 25% using SEMS (p = 1.000). In unstented patients with biliary obstruction, the bile juice was sterile significantly more often than in endoscopically stented patients (100% vs 1%, p < 0.001). When the stented and unstented patients were compared regarding postoperative infection complications, there was no significant difference between the groups (p = 0.365). Plastic stents did not differ from SEMS regarding the stent failure rate, bilirubin level decrease, amount of bacteria in the bile juice, or postoperative complications when used for PBD. The significantly higher price of SEMS suggests their use in selected cases only.
Keywords
Introduction
Obstructive jaundice correlates with impairment of hepatic function, disturbances in coagulation and development of cholangitis (1). Therefore, preoperative drainage by using stents has been used prior to pancreaticoduodenectomy (PD). In recent years, several studies have revealed routine preoperative biliary drainage (PBD) to show no benefit over early surgery without PBD. At the time, the use of routine PBD is controversial, as there are studies showing no benefit of omitting PBD (2–7). However, some surgical centers, including our own, do not usually possess the resources enabling early surgery entailing a need for PBD. PBD, on the other hand, permits neoadjuvant oncologic therapies or stabilization of the patient, if needed.
Both plastic and, increasingly over the past years, self-expandable metallic stents (SEMS) have been used for biliary drainage. In a recent small study, SEMS was declared as superior to plastic stents, as there was no need for re-stenting when metallic stents were used instead of plastic stents (8). Some studies have revealed higher postoperative complication rates when plastic stents were used (9).
As it has been implied that patients with preoperative SEMS have fewer postoperative complications compared to those drained with plastic stents, the more frequent use of SEMS was suggested at our institution. It was also estimated that the decrease in the bilirubin level might be more substantial when SEMS are used, as their diameter is greater than that of plastic stents. The greater diameter might also correlate with a better bile flow, affecting the bacterial level of the bile juice. As the cost for a SEMS is approximately 40 times the cost of a plastic stent, we decided to look into the matter. The aim of the study was to compare outcomes of surgery in patients who underwent PBD with SEMS or plastic stents.
Methods
The surgical databases at Helsinki University Hospital were retrospectively queried for PDs and total pancreatectomies (TPs) during 2000–2009. At least one of two experienced pancreas surgeons at our institution had taken part in every PD or TP performed. All patients who have had endoscopic PBD were included in the study. Variables such as age, gender, laboratory parameters, comorbidity, and endoscopic retrograde cholangiopancreatography (ERCP)-related and postoperative complications were collected from the patient charts. Microsoft Windows Access® database was used for data collection. The concomitant diseases of patients were calculated using the age-adjusted Charlson’s Comorbidity Index (CCI). CCI takes certain cardiovascular, pulmonary, internal organ conditions, and tumors into account by scoring them according to their severity and the patients age (10, 11).
The ERCPs were mainly performed by three experienced endoscopists (a yearly performance of some 300 ERCPs each) at the Helsinki University Hospital. However, 27 (14%) procedures were performed at other hospitals. The ERCPs were performed under conscious sedation. The plastic stents used were 5–7 cm long with a diameter of 10 Fr and the SEMS were 4–8 cm long, expanding to a diameter of 10 mm. Of the SEMS, 10 were covered and 18 uncovered. The definition and grading of post-ERCP pancreatitis (PEP) and other ERCP-related complications were based on consensus criteria (12).
Data of routine intraoperative bile juice samples for bacterial cultures were obtained in 156 cases. The samples were collected either by syringe aspiration of the bile duct or by sweeping the bile duct after surgical transection. To describe and compare cultures between the stent groups, we created a “bacterial score” (see Fig. 1). This score does not consider the virulence of the microbes.

Calculation of the bacterial score.
The postoperative fistulas were graded in A, B, and C fistulas according to the International Study Group of Pancreatic Fistula (ISGPF) classification (13). Grade A fistulas are transient, asymptomatic fistulas with only elevated drain amylase levels and without need for further interventions. Grade B fistulas are symptomatic, clinically apparent fistulas that require diagnostic evaluation and minor therapeutic management. Grade C fistulas are severe, clinically significant fistulas that require aggressive therapeutic interventions. Patients with Grade C fistulas appear ill and unstable.
The primary endpoint of this study was to determine whether there were differences in stent failure rates between plastic stents and SEMS. Secondary endpoints were the differences between the stent types regarding bilirubin levels, the level of bacteria in the bile juice, and postoperative complications.
As the issue whether to stent patients preoperatively or not has been addressed in the recent years, the secondary endpoints were, moreover, as an incidental supplement, extended to apply to the PD and TP patients operated during 2000–2009 without endoscopic preoperative stenting. The data of these patients were collected similarly to the ones of the actual study group.
Statistics
Data are given as the median (range) or number of cases and percentage. Normality of distribution for continuous data was tested using the Kolmogorov–Smirnov test. Continuous normally distributed data were compared with the two-tailed t-test and data with non-normal distribution with the Mann–Whitney U-test or with the Kruskal–Wallis test. The Spearman correlation coefficient was calculated to find out significant correlations between continuous data. Differences in categorical variables were tested with the Fisher’s exact test or with the Fisher–Freeman–Halton test. Exact 95% confidence intervals (95% CI) are calculated for proportions. Trends in ordinal variables were tested with the linear-association test. Two-sided p-values are used. p-values < 0.05 are considered as significant. Bonferroni correction was used for multiple testing by multiplying the p-values by the number of comparisons made. Statistics were calculated with IBM SPSS (v20, IBM Corporation, New York, NY) or with StatExact (Cytel software Corp., Cambridge, MA).
Results
During 2000–2009, 366 patients including 185 (51%) males had PD (n =
The subgroups of histopathologic diagnoses.
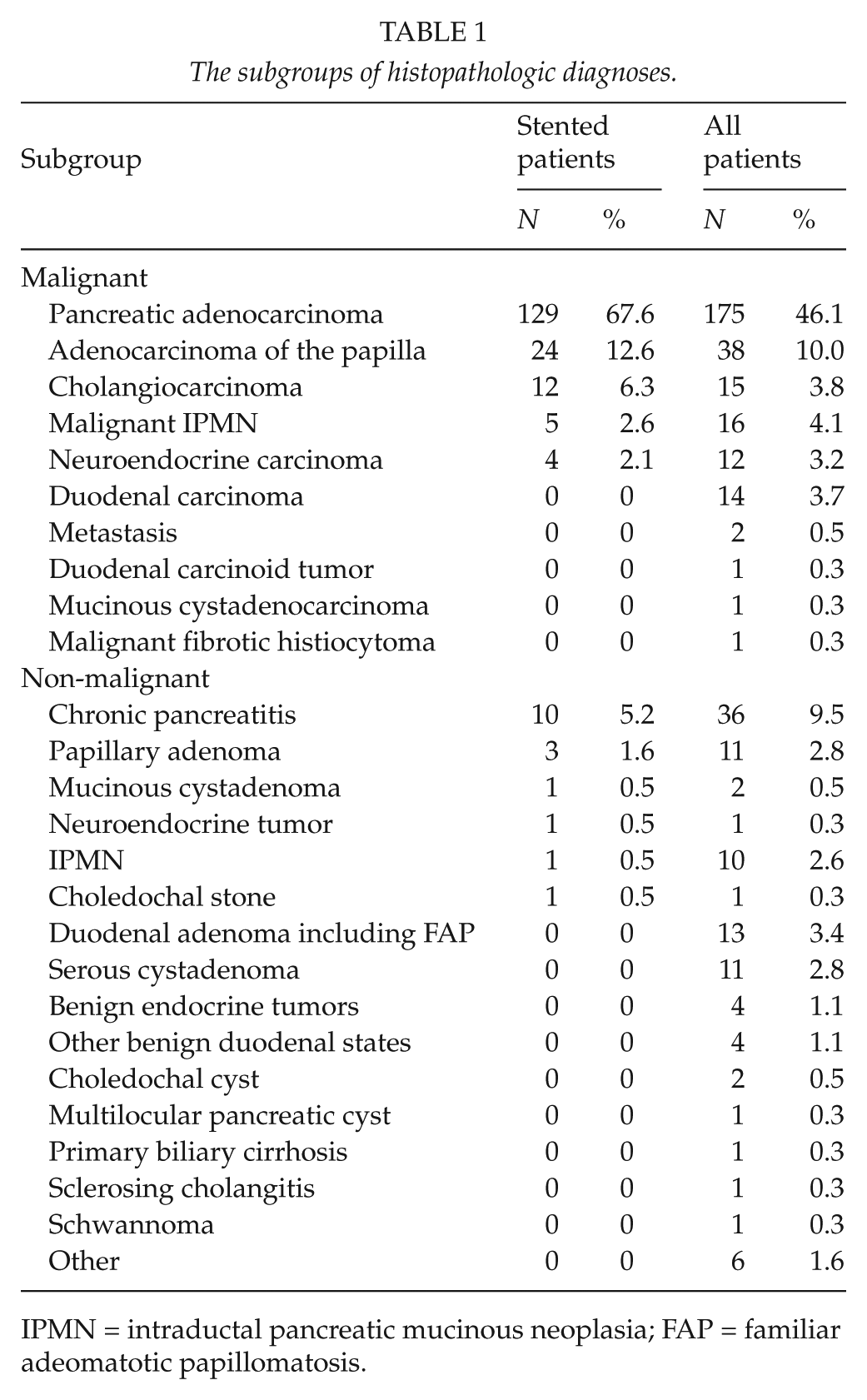
IPMN = intraductal pancreatic mucinous neoplasia; FAP = familiar adeomatotic papillomatosis.
Stent Failure Rate
Of the stented patients, 168 (88%) initially had a plastic stent. Of these, 12 were exchanged because of stent dysfunction and one because of stent migration. Five of these were changed to SEMS. One covered SEMS was exchanged to an uncovered one due to stent dysfunction caused by partial migration toward the duodenum. As a result, plastic stents were placed 176 times and SEMS 29 times. These figures were used to reach the primary endpoint, giving a stent failure rate of 7.4% (95% CI: 4.0%–12.3%) and 3.4% (95% CI: 0.1%–17.7%), respectively. There was no significant difference between the stent groups (p = 0.697, difference: −3.5%, 95% CI: −16.6%–12.9%).
Secondary Endpoints—General Data
The final preoperative stent types were used for grouping when analyzing data for the secondary endpoints. As a result, there were 163 patients (85%) in the plastic stent group and 28 patients in the SEMS group. The results of the secondary endpoints were moreover given for the supplementary patients, grouped as patients with biliary obstruction having had PTD (n = 9), unstented patients with biliary obstruction (UBO, n = 9), and unstented patients with no biliary obstruction (UNO, n = 157). Patient characteristics are shown in Table 2. The distribution regarding malignant versus non-malignant disease and the frequency of preoperative adjuvant therapy were alike both between the two stent groups and when comparing these to the supplementary groups.
Patient characteristics.
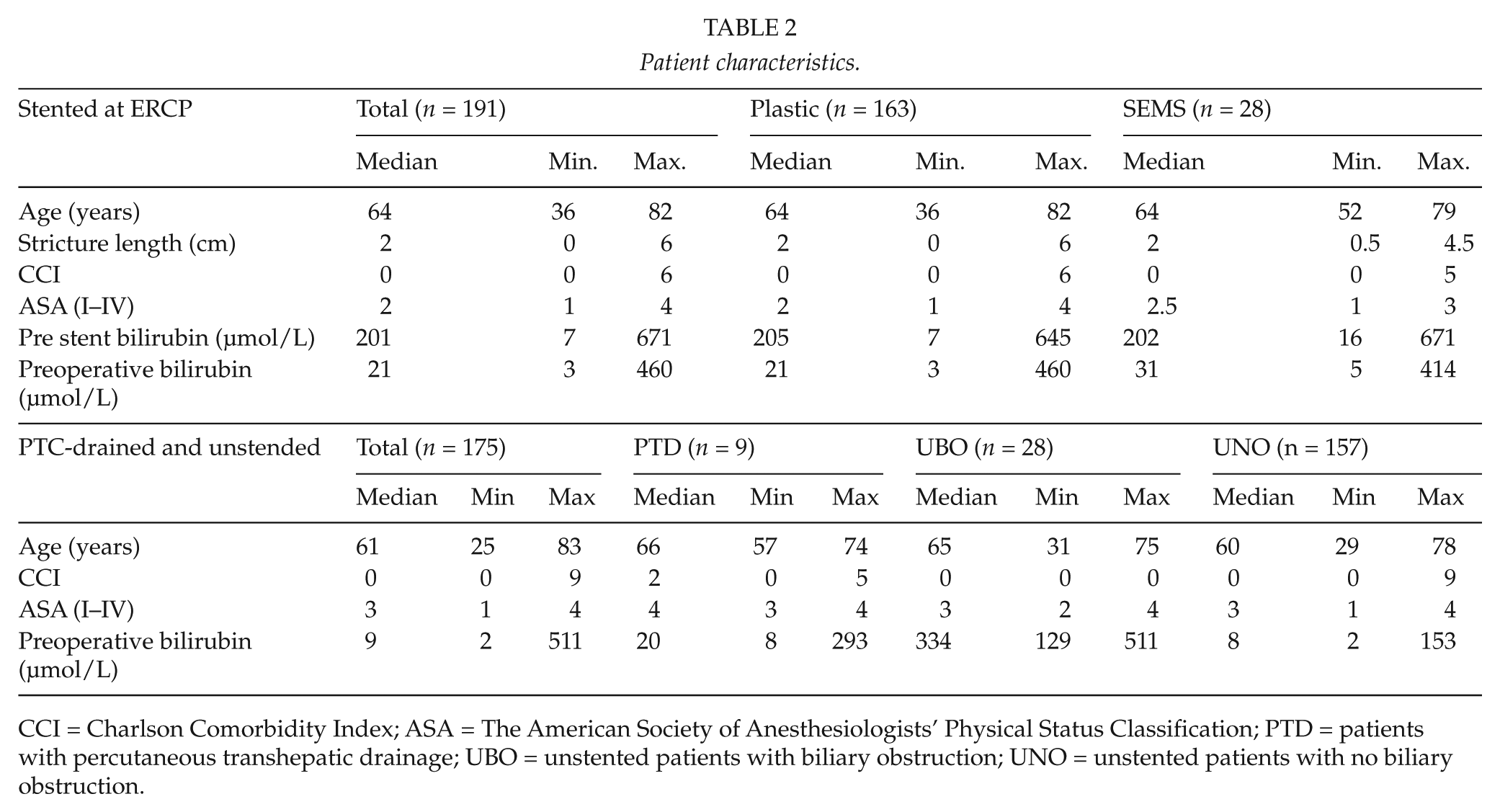
CCI = Charlson Comorbidity Index; ASA = The American Society of Anesthesiologists’ Physical Status Classification; PTD = patients with percutaneous transhepatic drainage; UBO = unstented patients with biliary obstruction; UNO = unstented patients with no biliary obstruction.
Tumor Size and Stage—Neoadjuvant Therapy
The tumor sizes and stages of the different patient groups are shown in Table 3. There was no difference in the tumor size between the plastic and SEMS group (p = 0.931). Of the endoscopically stented patients, 25 (13%) received neoadjuvant therapy. Three of them had a SEMS. None of the patients receiving neoadjuvant therapy needed stent exchange or repeat ERCP in the preoperative course. The median time from stenting to operation was 110 (20–266) days in the neoadjuvant therapy group and 34 (3–102) among the group without neoadjuvant therapy.
The figures of the results for the different groups.
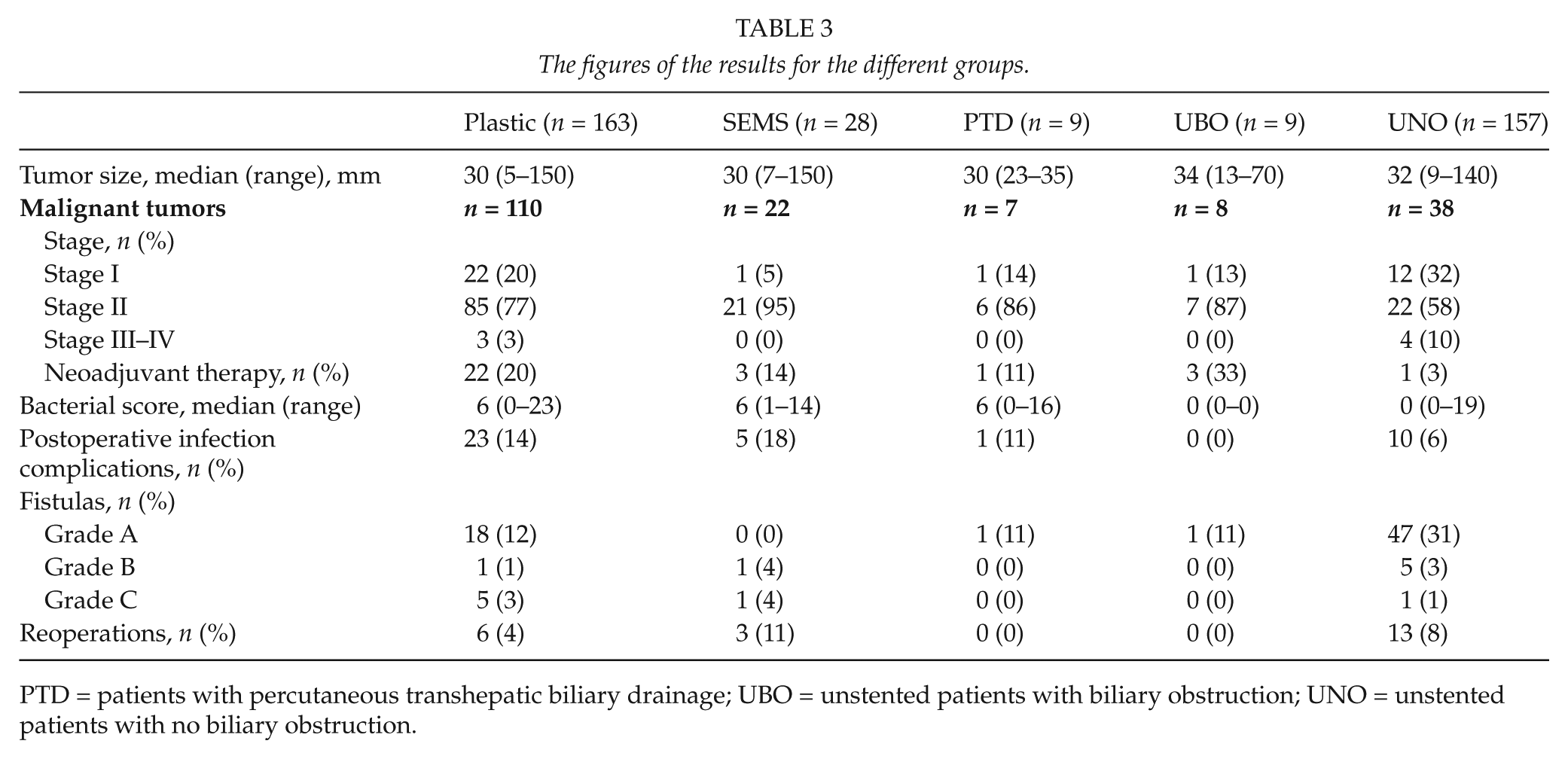
PTD = patients with percutaneous transhepatic biliary drainage; UBO = unstented patients with biliary obstruction; UNO = unstented patients with no biliary obstruction.
Bilirubin Levels
The bilirubin levels are shown in Table 2. The pre-stent levels (p = 0.732) as well as the preoperative levels (p = 0.157) showed no differences between the groups. Documented bilirubin levels for both the pre-stent-point and preoperatively were available in 171 patients. Of these 149 (126 in the plastic and 23 in the SEMS group) had a bilirubin level >50 µmol/L before stent placement. Of these, 45% within the plastic stent and 26% within the SEMS group reached a preoperative bilirubin level of 20 µmol/L or less (p = 0.110). A preoperative bilirubin level of 50 µmol/L or less was reached by 80% of the patients in the plastic stent group and 61% of them in the SEMS group (p = 0.058). The correlation between preoperative bilirubin levels and the time from stenting to operation is shown in Fig. 2. In five cases, the preoperative bilirubin level was higher than the level at the time of ERCP. Plastic stents were used in four of them, giving a “functional stent failure” percentage of 2.5% in the plastic and 3.6% in the SEMS group, having no difference in distribution between the groups (p = 0.551).
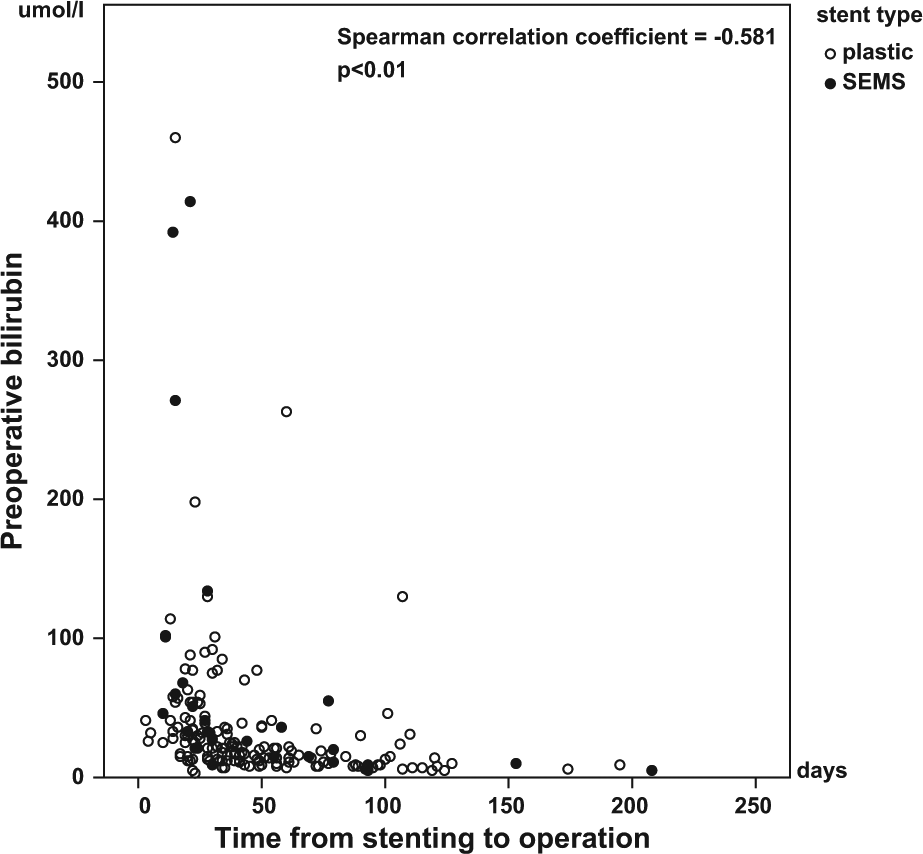
The correlation between the preoperative bilirubin levels and the time from stenting to operation shows that both plastic stents and SEMS decrease the bilirubin levels. The lower the bilirubin levels, the longer the stent has been in place.
Bacterial Score
The median bacterial score of the intraoperatively obtained bile juice samples was 6 (0–23). The scores of the different stent types, which did not differ between groups, are shown in Fig. 3. The mean difference was 0.41, 95% CI was -1.24–2.06. The scores of all groups are shown in Table 3. Only one stented patient (with a plastic stent) had sterile bile juice. In the UBO group, there were seven cases of documented bile samples, all sterile. One PTD patient (11%) had sterile bile. The UNO group had sterile bile samples in 73% of cases. When comparing the proportions of sterile bile juices between the endoscopically stented (n = 1/155 documented samples, 1%) and unstented patients with biliary obstruction (n = 0/7, 0%), there was a significant difference between the groups (p < 0.001).
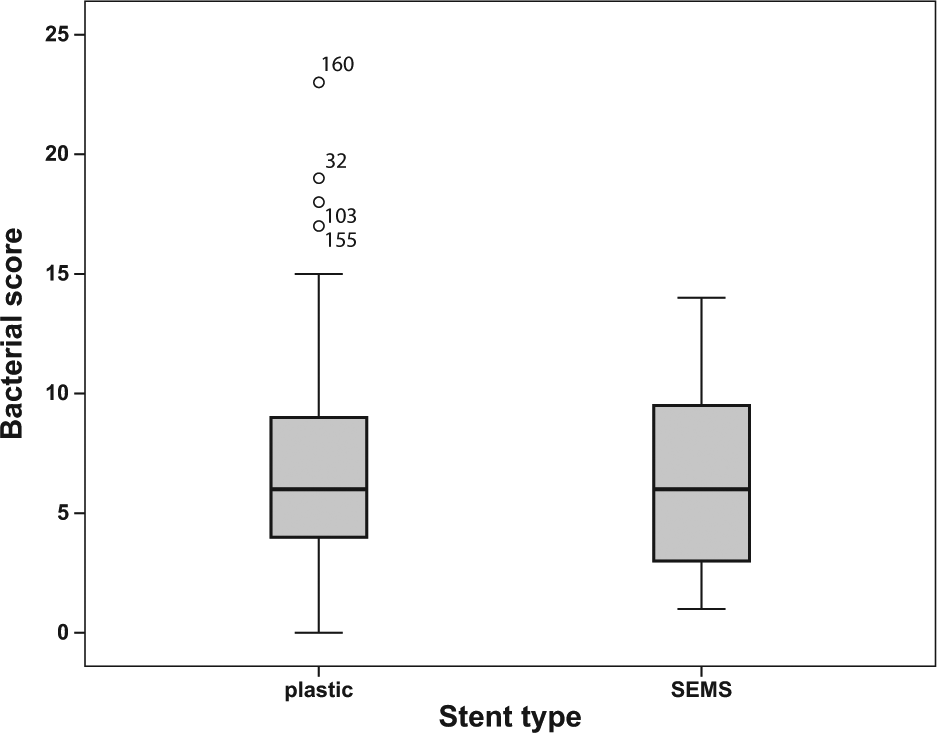
The bacterial scores show no difference between the two stent types.
Infection Complications
Infection complications, including pneumonia (n = 4), urinary tract infection (n = 8), wound infection (n = 9, two of which were at the origin of the vein graft), clostridium colitis (n = 2), intra-abdominal abscess (n = 1), or bacteremia (n = 5), were found in 28 endoscopically stented patients (15%). The figures for all groups are given in Table 3. There was no difference between the plastic and SEMS groups (p = 0.570). When comparing endoscopically stented and UBO patients regarding postoperative infection complications, there was no significant difference between the groups (p = 0.365).
Postoperative Pancreatic Fistulas and Infection Complications
Among the 183 endoscopically stented patients having had PD, 26 (14%) cases of postoperative fistulas were found. The distribution of the fistula grades is shown in Table 3. There was no significant difference between the plastic and SEMS groups (p = 0.072). Nor was there a significant difference in the appearance of Grade A fistulas (p = 0.238 Bonferroni correction), a group that is of minimal interest considering its characteristic as a chemical fistula requiring no further procedures. There was no statistically significant difference in the appearance of pancreatic fistulas between the UBO group and the stented ones (plastic and SEMS altogether, p = 1.000). The distribution of the bacterial score was not different across the different categories of fistulas (p = 0.596) in the endoscopically stented patients. There was neither a difference between the plastic and SEMS groups when evaluating the patients who had some postoperative infection complication and/or a fistula. There were 42 cases (26%) in the plastic stent group and seven (25%) in the SEMS group (p = 1.000).
Reoperations
The number of reoperations performed apart from those indicated by Grade C fistulas are shown in Table 3. There was no difference between the plastic and SEMS groups (p = 0.129). Indications for the reoperations in the endoscopically stented patients were bleeding (n = 2), leakage of the hepaticojejunostomy (n = 3), mechanic stomach retention (n = 1), suspicion of necrotizing fascitis (n = 1), slip of the drain into the abdominal cavity causing peritonitis (n = 1), and thoracotomy due to massive bleeding after drainage of pleural fluid (n = 1). Of those with leakage of the hepaticojejunostomy, two had a plastic stent and one a covered SEMS. When comparing the endoscopically stented (n = 9/191, 5%) and unstented (n = 13/166, 8%) groups for reoperation, there was no significant difference (p = 1.000).
Postoperative Hospital Stay and Mortality
The length of the postoperative hospital stay did not differ between the plastic and SEMS groups (p = 0.750), showing a median of 11 (6–112) days in the plastic and 12 (8–50) days in the SEMS group. The 30-day postoperative mortality rate was 0% both for the stented and the supplementary patients.
ERCP Complications
A total of nine (4.7%) complications connected to ERCP and biliary stenting were found. There were two (1.0%) cases of post-procedural bleeding. Both were conservatively managed, even though one patient had a duodenoscopy by the time the bleeding had stopped. There were seven (3.4%) cases of mild PEP. No cases of severe PEP, procedure-related cholangitis, or perforations were found.
Discussion
At present, preoperative placement of biliary stents in connection with pancreatic surgery is controversial. It seems the disadvantages connected with PBD mainly relate to complications due to the stent placement procedure (6). The ERCP-related complication rate of our material was 4.7%, which is low, compared to figures up to 46% in the literature (2). We did not have any severe complications. Our highly specialized, high volume ERCP unit might explain the low rate of complications. Naturally, due to the retrospective nature of the study, such potential complications that have prevented surgery do not show up in this material.
Cholangitis is regarded as a formal indication of PBD (6). PBD is also advocated in delaying surgery and preceding neoadjuvant therapy.
In recent years, biliary SEMS have been increasingly used for purposes other than palliative. Because of their larger diameter and longer patency rate, the more frequent use of SEMS, when PBD is needed, has been proposed (14).
Preoperative bilirubin reached levels below 20 and 50 µmol/L to a higher percentage in the plastic stent group, with a near to significant (p = 0.058) difference between groups, while setting the bilirubin limit to 50 µmol/L. This is in contrast to the belief that large bore metallic stents might be superior in decreasing the bilirubin levels when compared to narrower plastic stents.
A greater rate of wound infections among stented patients has been reported (9). Reviewing the literature, we found only one study that, among other outcomes, compared PBD using plastic stents and SEMS regarding postoperative complications (8). This study included 12 patients with SEMS and 35 patients with a plastic stent. The overall postoperative complication rates were 15% and 17%, respectively. The authors concluded that SEMS did not add to postoperative complications compared to plastic stents. A recent study presented 79 cases with preoperative SEMS of which 95% underwent neoadjuvant therapy (15). In the preoperative course, 16% of the patients required repeat ERCP procedures due to cholangitis (5%) or an isolated raise in laboratory parameters, compared to none in our material. There were no parallel groups treated with plastic stents or out of stents, but the authors concluded that postoperative complications did not increase in comparison to figures in the literature. As a result, we could not find very strong evidence supporting the superiority of SEMS over plastic stents regarding postoperative complications.
According to Sivaraj et al. (16), the amount of bacteria in the bile juice significantly correlates with postoperative infections. In our study comparing plastic stents and SEMS, there was no difference in the bacterial score between the groups. Nor was there a difference in postoperative infection complications. Kajiwara et al. (17) revealed bacteria in the bile juice on postoperative Day 1 (the samples were obtained using intraoperatively administered external drainage of the hepaticojejunostomy) to correlate with a higher risk of Grade B and C postoperative pancreatic fistulas. In our material, there was no difference between the distribution of the bacterial score across the different categories of fistulas, and no differences between the stent types.
Several studies in the recent years have indicated that preoperative stenting does not give any benefits in comparison to early surgery. In this material, all unstented patients with biliary obstruction had sterile bile. The number of these patients was vanishingly small compared to the stented ones, giving no statistically significant difference between the groups regarding postoperative complications. As mentioned earlier, preoperative stenting has its place in cholangitis, in connection with neoadjuvant therapy, in preoperative stabilization of the patient or if early surgery cannot be arranged for.
The retrospective design of this study and the fairly low number of SEMS restrict highly extensive conclusions regarding preferences in the choice between stent types. Notwithstanding, based on the results of this study, we propose that plastic stents are viable as SEMS when used for PBD. SEMS should be used in selected cases, including suspicion of inoperable disease or patient, failure of previous plastic stent, and, as longer patency for SEMS has been established in previous studies (14), preceding neoadjuvant therapy. In view of the correlation between bilirubin levels and the stenting time of our material, the inferior patency of plastic stents is not, however, unequivocal. The cost benefit using SEMS has been established in palliation of patients with a minimum survival of 6 months. When the stenting period in PBD is expected to be short, the high costs of SEMS should be taken into account.
Conclusively, plastic stents do not differ from SEMS regarding stent failure rate, bilirubin level decrease, bile juice bacterial level, or postoperative complications when used for PBD. The significantly higher price of SEMS suggests their use in selected cases only.
Footnotes
Declaration of Conflicting Interests
The authors declare that there is no conflict of interest.
Funding
The research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
