Abstract
Aim:
To review the advent of endovascular treatment of vascular injuries and its controversies.
Methods:
Literature review over the past 13 years using the PubMed search engine.
Results:
The management of vascular trauma has changed dramatically over the last decade. This is mainly due to the advent of advanced endovascular techniques and technologies. There are, however, a limited number of publications with a correspondingly small number of patients and lack of long-term follow-up. In this article, we discuss the injuries to different vessel groups and their respective endovascular approaches. Of the literature available, the endovascular management of aortic injuries still holds the most experience and has shown favorable results.
Conclusion:
Endovascular management of vascular injuries is a new addition to the armamentarium of the surgeon. The fact that it is minimally invasive is a major advantage and the published data are encouraging. However, in order to determine which anatomical sites and circumstances are most appropriate for this method versus open surgery, more reports are necessary.
Keywords
Introduction
The drastic changes in the management of vascular trauma have turned this type of injury into a new and evolving science over the last decade. The reason for this is the advent of endovascular management techniques. The general principles of management are discussed, as well as the evidence for the endovascular approach.
Endovascular treatment for vascular injury is a developing concept. Conclusions are difficult to reach because all the existing studies are small due to the young age of this technology. A look at the US National Trauma Data Bank (NTDB) (1) from 1994 to 2003 reveals that the very first case was in 1997. In this analysis, endovascular cases totaled 3.7%, with gradual annual increase from 2.4% to 8.1% between 2000 and 2003. The endovascular arm had a lower Injury Severity Score (ISS), decreased length of hospital stay (16 vs 20 days), and a significant mortality reduction—particularly with blunt thoracic aortic injury—(3% vs 19%). Recently, an American Association for the Surgery of Trauma (AAST) article (2) also interrogated the NTDB from 2002 to 2008 (2.5 million) and identified 45,220 vascular injuries that required intervention. Of those, 7.3% were endovascular—with 1.0% in 2002 and 10.8% in 2008. These studies highlight the fact that endovascular management is gaining more popularity as time passes.
As endovascular management becomes more common, so do its indications. Improved equipment and grafts allow for stenting of smaller and more tortious vessels. In the past, it was dogma that only hemodynamically stable patients were considered for the procedure. It is now recognized that patients who were initially hypotensive on arrival, or those with a high ISS, could be selectively managed by endovascular methods. While endovascular treatments have become very attractive, one must be aware of not “over-treating” patients simply because a minimally invasive treatment is available. As pointed out by Dennis et al. (3), there has been successful conservative management for minor or occult vascular injuries. These injures have resolved with no invasive intervention whatsoever.
In this article, we will approach specific vessel injuries that can be managed with endovascular techniques and discuss the specific merits in each case.
Subclavian Artery Injury
Subclavian artery injuries carry a significant mortality rate (~34%). In many cases, it is associated with excessive hemorrhage—which requires extensive surgery—and poor outcomes and high morbidity. The complex array of surrounding nerves, and the difficulty of working around the bony structures, add to the complexity. For these reasons, the first experiences of the management of vascular trauma through endovascular techniques were in the area of subclavian artery injury.
In 2012, DuBose et al. (4) reviewed 32 publications of axillo-subclavian vessel injuries. They reported a 97% procedural success rate as well as 84% patency at follow-up (ranging from a week to 70 months). They also cited three comparative articles that show that endovascular repairs had shorter operating times, less blood loss, and less brachial plexus injury than open surgery.
The procedure can be carried out without general anesthesia, the operating time is shorter, and it can also be considered in stable patients who are of poor surgical risk due to co-morbidities. Also, if an active bleed is noticed during conventional angiography, a balloon occlusion technique can be employed, thus reducing bleeding and making surgery or stenting easier and safer (5).
Timing of the stent has consequence. Early stenting is done over a fresh thrombus which is usually soft and poses less of a risk of embolization. The utilization of distal occlusion devices to prevent this complication should probably be examined in this setting.
One must also bear in mind the ramifications of covering a branch of the vessel, particularly the vertebral artery. Coverage of this artery may theoretically compromise cerebral circulation. The theoretical possibility of pressure on the brachial plexus from non-evacuated hematoma (during endovascular surgery) causing neural injury has not been proven to be of concern (6).
In 2008, Du Toit et al. (7) looked at 57 patients with subclavian injuries treated with stent grafts over 10 years. They used covered stents. No anticoagulation or antiplatelet therapy was given. There was a 48-month follow-up (of only half the patients due to poor compliance). Of the patients, 5% had early graft thrombosis (occlusion) which was successfully treated with a second endovascular intervention. At late follow-up (mean 48 months), 20% of the patients presented with >50% graft stenosis and 12% with graft occlusion. The stenotic stents were treated with angioplasty and the occlusions did not require any surgery. There was no limb loss or incapacitating symptoms.
Specific contra-indications to subclavian endovascular management include brachial plexus compression and aerodigestive injuries. A relative contra-indication would be the level of the injury—where easy access to the injury might favor open repair.
Carotid Artery Injuries
Carotid artery injury can occur due to blunt or penetrating trauma. Blunt trauma can occur secondary to a base of skull fracture or in the neck, due to direct contusion or hypermobility of the neck which causes a stretching or shearing effect. In motor vehicle collisions, the incidence of carotid artery injury is alarmingly high; in Moar’s autopsy study of deaths due to motor vehicle collisions, the carotid arteries of the victims were routinely studied, and it was found that up to one-third of all the victims had some degree of carotid artery injury (8). Strangulation has also been shown to cause blunt carotid injury.
Patients with carotid artery injuries may be asymptomatic at first due to an incomplete occlusion, a contained bleed, or a complete occlusion with an intact circle of Willis providing sufficient blood “from the other side.” Therefore, there should be a low threshold for investigating those patients whose mechanism of injury could be related to this pathology. Such injuries are tabulated in the Eastern Association for the Surgery of Trauma (EAST) guidelines (www.east.org).
Treatment of carotid artery injuries is controversial. This controversy stems from the fact that the effect of the injured vessel is unknown, and that the treatment itself may cause catastrophic complications (e.g. the dislodging of a clot distally when mobilizing a carotid vessel, or the risk to nerves when conducting a neck dissection).
Overall, treatment is based on grade of injury. These grades have been defined by Biffl and are sometimes referred to as the Denver Criteria (Table 1).
Biffl grading.

General suggestions are as follows: for blunt injury, management of Grade I and Grade II injuries should be antithrombotic agents (unless there are ischemic symptoms). For this therapy, either antiplatelet agents or heparinization/warfarinization can be used. Biffl himself has conflicting studies where one study showed a clear benefit of heparin over antiplatelet agents in reducing subsequent cerebral thrombotic events (9), and another showed no difference (10). Either way, Biffl has recommended heparinization except in cases where anticoagulation is contraindicated. For Grade III injuries, conservative management is not advised. Certainly, all these injuries should be followed up with repeat imaging and, if seen to be extending, should be managed by surgery or angioembolization. Grade III injuries rarely improve with time if managed conservatively, and these injuries may be best suited for endovascular management from the outset, especially in difficult-to-operate areas. All patients with neurological deficit should be considered for operative or interventional treatment if this can be achieved within 24 h of the onset of neurology. A profound neurological deficit, however, has a poor outcome which is not significantly changed by intervention.
Possible endovascular indications for managing carotid (and vertebral artery) injuries include thrombosis, dissections, transections, pseudoaneurysms, and arteriovenous fistulae (including carotid-cavernous fistula).
There are many studies which have and are examining the efficacy of endovascular management of carotid artery disease. However, very few studies investigate the efficacy of endovascular techniques in traumatic carotid and vertebral artery injury (VAI). Due to this lack of high-powered studies, it is difficult to draw meaningful conclusions from the available data.
Most commonly, endovascular repair has been proposed in blunt vertebral and carotid artery injuries as a means of reducing embolization from, or rupture of, a pseudoaneurysm. Endovascular stenting would be particularly beneficial in areas that are not amenable to surgery, such as the distal internal carotid artery, where open surgery may require subluxation of the mandible or even distal control via a craniotomy. DuBose et al. (11) did show stenting to be an effective treatment for internal carotid artery injuries. There was a follow-up patency of 79.6%, no mortalities related to the procedure, and a low incidence of new neurologic deficits (3.5%).
Other studies show mixed results: Joo et al. (12) (fistulas and pseudoaneurysms) showed no complications and symptom improvement in all of their patients. Their numbers, however, were low (n = 10). Cothren et al. (13) showed different results. In this study (n = 140), the stented group had a higher rate of complications than the “conservative” group treated with anticoagulation alone. Their complications included strokes, subclavian artery dissection, and a 45% arterial occlusion rate (as compared to 5% in the “no-stent” group).
In the case of a pseudoaneurysm, rupture can result in severe hemorrhage, airway compromise, and cerebral ischemia; embolization would result in cerebral infarcts. In these cases, uncovered stents have been used, and the theoretical advantages are that they act as a filter to trap thrombus inside the pseudoaneurysm, decreasing blood flow into the pseudoaneurysm by promoting laminar flow across the artery itself and promoting growth of endothelial cells along the stent matrix (13).
The timing of stenting is controversial and should be balanced between delaying to prevent the risk of embolization while cannulating a damaged vessel and intervening early enough to prevent progression of injury. If one has chosen to embark on a carotid stent, one must realize that the occlusion rate of the stent can be high if the patient is not given antiplatelet agents. The European Society for Vascular Surgery recommends the use of dual antiplatelet therapy (Aspirin and Clopidogrel). There is no consensus in the literature as to how long antithrombotic therapy should be continued.
VAIs
Small studies have begun to appear, but none of the numbers are significant enough to draw any meaningful conclusions. VAI mechanism is associated with hyperextension or hyperflexion of the neck, distraction, cervical facet dislocations, and, the most commonly related injuries, cervical spinal fractures, with some reports stating up to 70% correlation with VAI’s (14).
VAI’s are mostly asymptomatic, but can present with signs and symptoms of basilar territory infarction and even death. One of the most important clues to its possibility is the mechanism of the injury itself. These mechanisms are mostly found in motor vehicle collisions, but VAI can also occur from hangings, sporting injuries, chiropractic neck manipulation, or penetrating trauma.
The natural history of VAI’s is that most occlusions recannalize and stenoses resolve. There are conflicting reports in the literature as to the significance of VAI, with some reports considering it to have a benign course, and others reporting delayed ischemia (15).
Treatment options include observation, anticoagulation, and endovascular and open surgery. Most often, for low grade, asymptomatic lesions, anticoagulation alone will suffice. Cothren et al. (16) strongly advocate anticoagulation to prevent strokes, and they have found no difference between heparin/warfarin and antiplatelet agents. Suggested time for anticoagulation is 3 months, with no real evidence to support it. There are a few studies that show non-significant cerebrovascular accident (CVA) reduction with anticoagulants. The problems with the studies include non-randomization, patients with associated brain injury, and patients where anticoagulation is contraindicated (cerebral bleed, other trauma, etc.). In the case of a concomitant cerebrovascular injury and brain infarct, one does not know if the infarct was due to the trauma or due to the vessel injury. Endovascular management is commonly employed for symptomatic or high-grade lesions. For Grade V lesions, angioembolization is employed, with open surgery a last resort due to the difficult access.
Blunt Aortic Injuries
The management of Blunt Traumatic Aortic Injuries (BTAIs) has been debated over the last 10 years. There has also been a mind-shift toward less invasive management over this time. This has resulted in better short-term outcomes albeit without proof of improved long-term outcomes.
This practice change is best seen in the AAST trials. The original trial (AAST1) (17) had to be repeated (as the AAST2) (18) due to the rapid change in practice management over the short period of time. There has been a shift in the diagnostic investigations from the invasive arteriography, which was the golden rule, to the less invasive and less time-consuming computed tomography angiography (CTA), which has been proven to be nearly as sensitive as formal angiography. Only in the exceptional circumstances that CTA cannot provide the required detailed information can one proceed to formal angiography or other less commonly available investigations (e.g. endovascular ultrasound).
The AAST2 trial also showed a trend toward delayed repairs of the contained injuries (exsanguinating aortic bleeding always requires immediate surgery). The purpose of delaying surgery is to improve the patient’s physiological status, and this has been shown by Demetriades (19) to be related to a decrease in mortality rate (16.5% in early to 5.8% in delayed intervention).
To decrease the possibility of the contained hematoma rupturing into the pleural cavity and causing fatal exsanguination while waiting for surgery, it is prudent to achieve impulse control—lowering the systolic blood pressure and the pulse rate (ΔP/Δt). This is achieved by administering antihypertensives such as Esmolol, Labetalol, or Nitroprusside in refractory cases, aiming at a systolic blood pressure of 120 mmHg. In the case of the elderly patient with known uncontrolled hypertension, a systolic blood pressure of 140 mmHg is acceptable (20).
The AAST2 trial also showed a trend toward endovascular management of these injuries (Table 2). It demonstrated improved survival and reduced rates of paraplegia when compared to open repair. There was, however, a high rate of device-related complications. These complications were noted to be less in centers where endovascular volumes are high, and it is recommended to transfer patients to such centers (if possible) when embarking on the minimally invasive route.
Comparing the AAST1 and AAST2 trials (18).
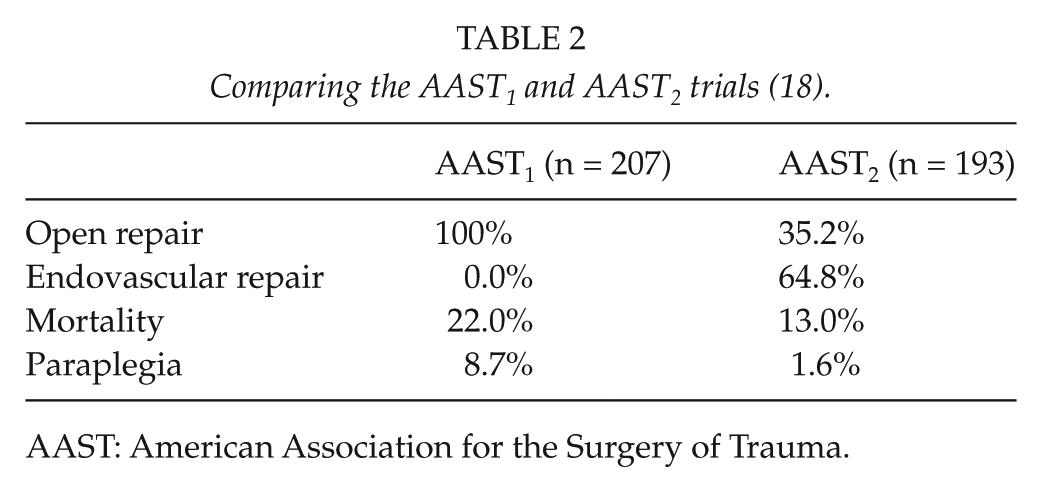
AAST: American Association for the Surgery of Trauma.
Although endovascular repair has by far better mortality results than open surgery, there are not enough long-term data, and there are certain considerations concerning short- and long-term complications:
The unfolding of the aortic arch can cause dislodgement of the graft.
The need for intraoperative coagulation can be detrimental in poly-trauma patients having other sources of bleeding, for example, intracranial hemorrhage.
Incorrect sizing of the graft: the grafts are not purpose-built for the individual patient, thus sizing is often incorrect, and this leads to complications, for example, migration, endoleaks, collapse, fracture, and thrombosis. Overall, accurate sizing is difficult as the currently manufactured grafts are made for older patients who undergo stenting for aneurysmal aortas that are much wider; hence, the available grafts for trauma patients (with non-diseased aortas) tend to be too large. The industry is addressing this problem. Also, the stent deployment devices through the iliac artery are also made for “older/larger vessels,” and thus, the narrower iliac vessels of younger trauma patients can receive considerable damage from access.
Increase in aortic diameter with age: this leads to the aorta becoming too large for the graft and increases the risk of the above complications.
Graft fatigue: current grafts have only been fatigue tested for 10 years, long enough to cover the life expectancy of the old aneurysmal patient, but not enough for the usually young trauma patients. This concern applies to grafting of all vessels, not just the aorta.
Occlusion of the left subclavian artery: when the injury is near to the orifice of the left subclavian artery, as is most commonly the case, there may not be enough “landing zone” and the graft can occlude the artery. This eventuality may be considered as part of the planning process and deemed an acceptable risk as it is usually well tolerated and only a small number of patients require carotid–subclavian bypass to relieve a steal syndrome.
Endovascular patients require frequent follow-up. There is no consensus as to the schedule of timing. The follow-up is done by CTA, and most centers advise follow-up CT 1 month after surgery, twice in the first year and yearly thereafter. There is concern about the cumulative radiation dose and there are suggestions of using magnetic resonance imaging (MRI) instead of CT scan.
Minimal aortic/intimal tear injuries (Starnes modification: Grade I—Intimal tear with length <10 mm; Grade II—Intimal tear with length >10 mm) are recommended to be initially managed conservatively, with no graft. This is safe as long as there is follow-up (21, 22). This option has been further validated by the Stanford study, one of the largest to date (23). The University of Washington Clinical Guidelines recommend follow-up for minimal aortic injury of less than 10 mm by CT angio at 30 days and for those of more than 10 mm at 7 days. If there is progression of the injury, they suggest endovascular repair.
Spinal drainage is not advised in trauma patients undergoing Thoracic EndoVascular Aortic Repair (TEVAR) and debate continues as to the use of post-operative anticoagulation. Standard chemical Deep Venous Thrombosis (DVT) prophylaxis is always advised during the hospital stay. Some use antiplatelet agents post-discharge, while others (24) do not consider it necessary.
There are currently no “prospective” randomized trials directly comparing open to endovascular repair. There probably will not be any for a long time. For now, one would be prudent to follow the guidelines of The Society for Vascular Surgery (Table 3), but one must recognize that the level of evidence is weak. The Society placed a significantly higher value on preventing catastrophic complications of thoracic aortic repair (death, stroke, and spinal cord ischemia) and a lower value on potential adverse events.
Society for Vascular Surgery guidelines on the management of blunt aortic injury.
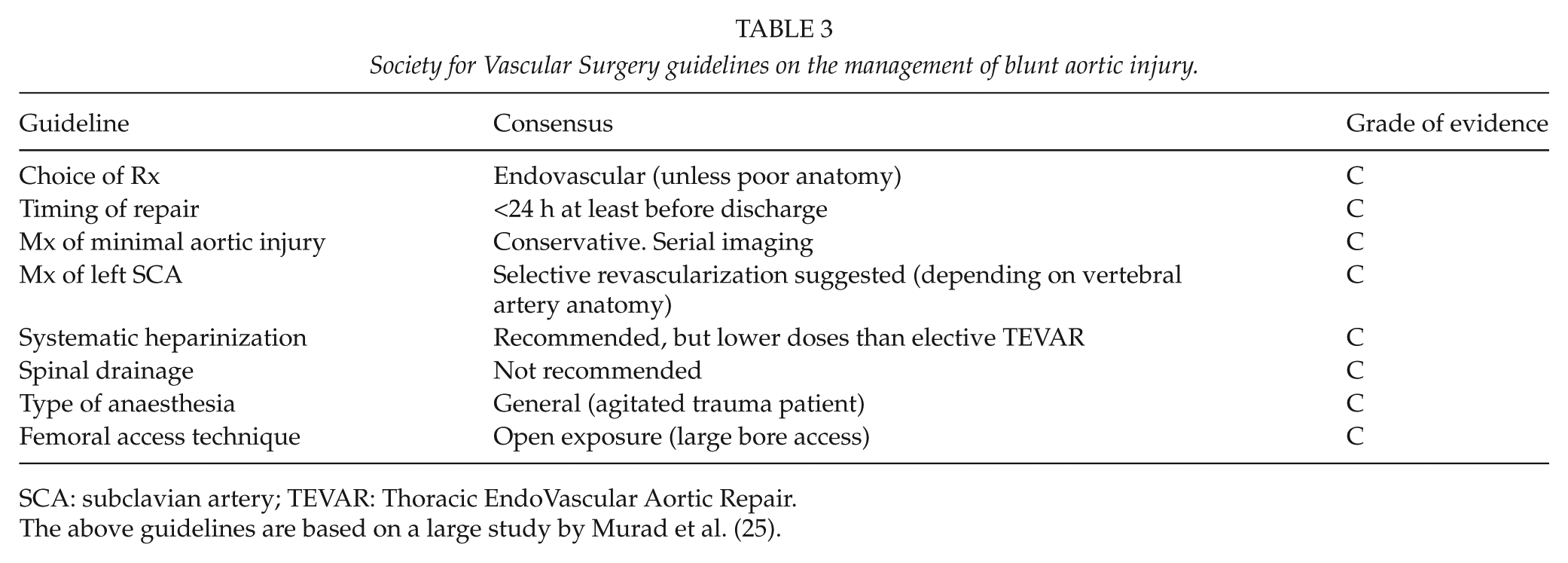
SCA: subclavian artery; TEVAR: Thoracic EndoVascular Aortic Repair.
The above guidelines are based on a large study by Murad et al. (25).
Peripheral Limb Trauma
In the past, there has been debate about the ideal imaging modality for detection of peripheral arterial injury in the absence of hard signs or the need for pre-operative localization. Digital Subtraction Angiography has been the gold standard, but CTA imaging has become popular, due to its ease of use and no need for arterial cannulation. The current EAST guidelines (26) show level 1 evidence that CTA is adequate.
Popliteal Artery
Knee dislocations are well known to be associated with popliteal artery injuries. The associated prevalence is between 23% and 32%. The vessel is tethered proximally as it emerges from the adductor hiatus and distally at the tendinous arch of the soleus. Because of that, it is prone to injury when there is significant displacement of the knee. A distracting injury (such as a knee dislocation; fractures of the distal femur and proximal tibia that encompass the “floating knee”; and a significantly displaced tibial plateau fracture) may therefore cause significant stretching of the popliteal artery and subsequent intimal tearing or other vessel injury.
The knee can reduce spontaneously before arrival to hospital. In this case, the clinician may “miss” the fact that there was a dislocation and may be led into thinking there is no need to investigate the vasculature. In these cases, ligamentous instability may be the only clue to the fact that the knee was dislocated. Vascular injury has even been seen with isolated posterior cruciate ligament injuries. Therefore, in addition to the injuries mentioned above, Posterior Cruciate Ligament injury, any double ligament injury, or other reason to suspect a knee dislocation, should also be investigated for vascular injury.
The genicular arteries are not adequate as collaterals to supply blood to the distal leg, and studies have shown that there is an 86% amputation rate for popliteal arteries that are not restored within 8 h (27).
Several early authors report that ongoing pulse examination alone is adequate to detect popliteal artery injuries. Others suggest that the risk of missing a popliteal injury is too high. One meta-analysis (28) shows that abnormal pulse examination had a sensitivity of 79% and specificity of 91%. These and other authors suggest that we should rather have a liberal policy of angiography and not rely on physical examination. Physical examination has been known to be initially normal in patients who later develop ischemia and require amputation. This is due to small pseudoaneurysms or intimal tears that lend themselves to a normal initial clinical examination. These small injuries may progress and cause ischemic complications with time, when the clinician is not fastidious about the examination, thinking that the patient is out of the danger phase.
Ankle Brachial Index (ABI) Doppler pressures are also controversial. Although there is good evidence to suggest that Doppler pressures (an ABI of <0.9) are sensitive at picking up larger vascular injuries, they may miss occult injuries. Having said this, there are those proponents that claim that these small injuries that cannot be picked up within 48 h have no clinical significance and do not progress (29).
There have been no studies looking at Duplex Doppler studies for popliteal injuries. The debate on whether to do angiography on “all” patients or on selective patients (with any clinical features, decreased pulses, abnormal ABI, etc.) rages on. There are studies that show that all patients should get angiography, regardless of their clinical findings, and then there are studies that suggest that a selective angiogram policy is safe (25). The largest study by Stannard et al. (30) had 126 patients. He showed that patients without hard signs could be managed by frequent clinical examination. Positive predictive value (PPV) was 90% and negative predictive value (NPV) was 100%. Sensitivity was 100% and specificity was 99%. There is no definitive conclusion, and one must follow one’s institution’s protocol.
Delays due to unnecessary investigations cause limb loss. Therefore, if a vascular injury of the popliteal artery is clinically obvious, one should operate immediately. CT angiography/formal angiography should only be done in cases of doubt in diagnosis, and in experienced high-volume centers, need not take longer than 10–20 min.
Endovascular angioplasty and stenting of the popliteal artery have been described with non-traumatic aneurysms and occlusive disorders. There is no significant literature describing its use in traumatic popliteal injuries. Stenting in this area is prone to a high rate of restenosis and thrombosis due to movement around the graft from the mobile joint.
General Endovascular Topics
Placing of Stent Grafts—Technical Tips (31)
Access sites can be either the femoral or the brachial artery, depending on which site is to be used. The femoral artery is often preferred in order to get the larger sheaths in. Stiff wire should be used to get better support during deployment. Self-expandable stents are preferred as they are more flexible, can conform to varying diameters in a single vessel, can be oversized, and have a higher resistance to deformability in mobile areas. Balloon expandable stents can be used in non-mobile areas and have the advantage of greater precision in deployment compared to self-expandable stents.
Angioembolization—Technical Tips
Smaller vessels need to be canalized with microcatheters. Embolization is achieved with microcoils, gel-foam, beads, and glue. Larger vessels require the deployment of larger coils or vascular plugs. Direct comparisons of techniques have not shown one to be superior to another.
Limitations of EndovAscular Management
Hemodynamically unstable patients are usually not candidates for endovascular management. The lack of predictability of the actual embolization would preclude them from these techniques. The time taken for actual hemorrhage control must be factored in. In addition, the time for summons and arrival of specialized staff (radiographers, radiologists, specialized nursing staff, company representatives to supply the correct equipment which may be too expensive to keep on hand, etc.) and the time to cannulate the vessel must also be taken into account. In those centers that run a 24/7 hybrid suite, unstable patients may be considered for endovascular management especially if use of an occlusion balloon is employed to slow bleeding.
Conclusion
Endovascular management is attractive due to its lower morbidity and reduced length of stay. Despite this, there is still very little evidence (due to low case numbers) of its long-term benefit and safety in the trauma population. The reason for this is that these cases are not planned for, expertise is not always available at centers where trauma patients present, follow-up is poor, and most centers only have a handful of cases. Additionally, the young age of the average trauma patient is an ongoing concern as long-term patency rates over their extended life span are unknown. However, in all the literature in existence, the endovascular management of aortic injuries still holds the most experience and has thus far shown favorable results.
Footnotes
Acknowledgements
None.
Declaration Of Conflicting Interests
None.
Ethical Approval
No ethical approval was required for this review.
Funding
No funding was acquired.
Informed Consent
No consent was required.
