Abstract
Background and Aims:
Complex nontraumatic foot lesions often lead to major lower-limb amputation in diabetic patients. We aimed to evaluate outcome of free flap transfer in such cases.
Materials and Methods:
A total of 11 consecutive diabetic patients, hospitalized between 2007 and 2012 at a university central hospital for a free flap transfer, were followed until September 2013. Amputation-free survival, patient survival, and complete wound healing were defined as primary endpoints. Healing time of tissue lesions was analyzed. All pre- and postoperative data were retrospectively collected from hospital charts.
Results:
Complete tissue healing at 6 and 12 months after free flap transfer were 55% (6/11) and 82% (9/11), respectively. The median time to complete tissue healing was 123 days (range, 45–207 days). Overall limb salvage, survival, and amputation-free survival rates at 12 months were 90%, 91%, and 82%, respectively. The clinically important endpoint, namely, amputation-free survival with completely healed wounds, was attained in 9 of 11 patients at 1 year.
Conclusion:
Free tissue transfer enables successful wound healing and limb salvage rather than amputation in selected diabetic patients with difficult-to-heal wounds. Complete healing of tissue lesions is slow even after successful surgery. Preoperative assessment of the condition of patients is the key to success.
Keywords
Introduction
At present, there are estimated to be 55 million people in Europe with diabetes (1). The lifetime risk of developing foot ulcer can be as high as 15%–25% among diabetics, although long-term epidemiological data are lacking (2, 3). Diabetic foot ulcers result from varying degrees of peripheral neuropathy, infection, and ischemia (4). With specialist multidisciplinary care, some two-thirds of patients with diabetes who develop an ulcer will eventually heal their foot (5, 6). One-third of patients’ wounds will not heal and these patients are at high risk of amputation. Diabetic patients account for 40%–70% of all lower-limb major amputations (7). Globally, this results in one major amputation every 30 s with over 2500 limbs lost per day (8). Studies have demonstrated that the five-year mortality rate in patients with diabetes following a major amputation is significant and even greater than many major forms of cancer (9). The Eurodiale (The European Study Group on Diabetes and the Lower Extremity) study clearly highlighted how more emphasis should be placed on developing strategies to improve the poor outcome of diabetic patients with a neuroischemic foot ulcer, and, in particular, in the patients with peripheral arterial disease (PAD) and infection (10).
Arterial revascularization should be performed whenever feasible to repair perfusion and to achieve ulcer healing (11). Patients with large tissue defects, deep space infection, osteomyelitis, or exposed tendon or bone may need free tissue transfer to heal their wounds and save their legs (12).
Surgeons have used free tissue transfer for a range of defects including breast reconstruction, cancer, and trauma since the late 1970s (13). However, their use in patients with diabetic foot ulcers has been less well documented. These procedures seem to be concentrated in highly specialized units (12). It is estimated that 2%–25% of diabetics with large tissue defects would benefit from microvascular free flaps (14, 15). Improved survival after successful limb salvage using microvascular free flap transfer has also been demonstrated (16).
The aim of this study was to analyze the results of free tissue transfer for complicated diabetic foot lesions with regard to wound healing, limb salvage, survivorship, as well as the risks and complications of the procedure.
Material and Methods
Patient Selection
All the records of free tissue transfer procedures performed at our hospital between January 2007 and December 2012 were reviewed. Our unit serves as the academic plastic surgery center for a catchment area of 1 million inhabitants and is the second largest university hospital in Finland. Trauma patients were excluded. Only patients with diabetes mellitus (DM) and chronic lower extremity wounds were included. The follow-up lasted until the death of the patient or until 30 September 2013, whichever occurred first.
The study cohort consisted of nine men (82%) and two women aged 33–65 years (median 54 years). All patients had diabetes (type I/II). Patient characteristics are described in Table 1. All patients had been ambulant before the manifestation of the tissue lesions, as only patients capable of regaining functional ambulatory status were considered as suitable candidates for operation. One patient had already had contralateral major lower-limb amputation. His mobility and independence was threatened by the loss of the other leg.
Patient demographics.
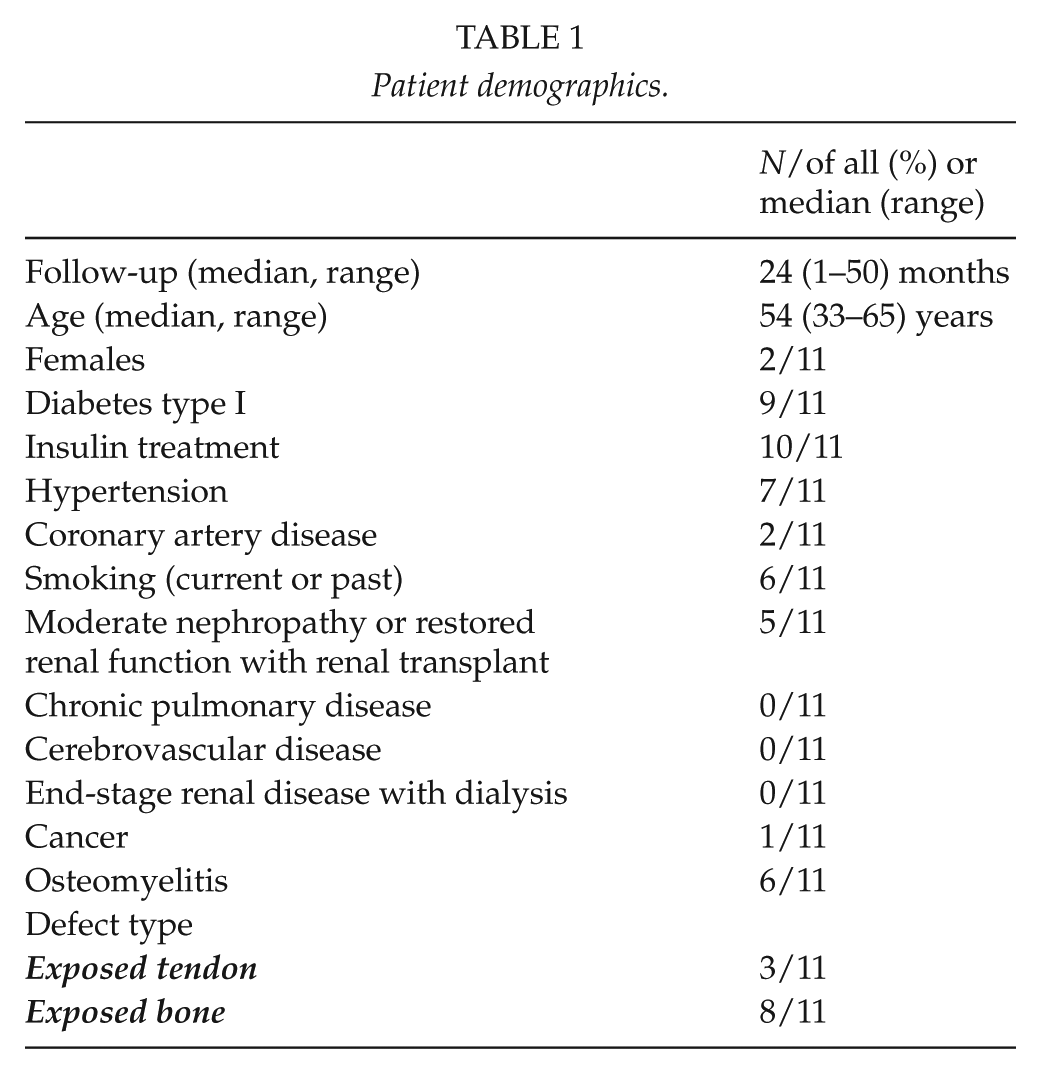
All patients included had a large soft-tissue deficit (exposed bone and/or tendon). Lesions were located most often in the plantar or heel area (Table 2). The most common bacteria in the cultures taken from the wound was Staphylococcus species (in six of 11 cultures). Osteomyelitis was detected in six legs.
Surgical variables.

TMT: transmetatarsal; LD: latissimus dorsi; ALT: anterolateral thigh; ATA: anterior tibial artery; ATP: posterior tibial artery; ADP: dorsalis pedis artery.
We defined tissue healing time as the time (in days) needed to achieve complete epithelialization of the lesions and surgical wounds. Major amputations were regarded as nonhealed tissue lesions.
Successful ambulatory function was defined as the ability of the patient to walk alone or with a cane or walker. Patients requiring a wheelchair for mobility or using their leg only for pivot functions were considered as nonambulatory.
Procedure
Vascular status of all patients was examined before the procedure. Preoperative angiography (or magnetic resonance angiography; MRA) was performed for all patients to evaluate the extent of possible arterial occlusions and to select appropriate inflow vessels.
Muscle flaps accounted for nine of 11 free flaps (Table 2), latissimus dorsi flap being the most common flap used in six legs. All muscular flaps were fixed and covered with split-thickness skin grafts. Only two patients had a lower-leg revascularization procedure alongside free tissue transfer, and this was a femoro-tibial bypass operation with autologous vein graft in both cases. In these two legs inflow to the free flap was taken directly from the bypass graft. None of the patients had angioplasty before the free flap operation.
Postoperative meticulous monitoring of free muscle flap was achieved by hourly Doppler examination and temperature control and by tissue pO2-measurements.
Data Collection and Statistics
The follow-up lasted until 30 September 2013 or until the death of the patient, whichever occurs first. The median follow-up was 24 months (range, 1–49 months). All pre- and postoperative data were obtained from patient files at our department.
Data were analyzed using the SPSS 20.0 statistical software (SPSS Inc., Chicago, Ill, USA). Limb-salvage, survival, amputation-free survival, and amputation-free survival with complete tissue healing were calculated using the Kaplan–Meier survival analysis. This retrospective study with prospective follow-up complied with the Declaration of Helsinki and was approved by the institutional review board of our hospital.
Results
No patients (0%) were lost to follow-up before reaching any of study endpoints or complete healing of the tissue lesions. The median hospital stay was 18 days (range, 9–30 days).
Healing of Tissue Lesions
In 9 of the 11 legs, the wound healed completely. Healing rate of the tissue lesions was 55% at 6 months and 82% at 12 months. The median time to complete healing of tissue lesions was 123 days (range, 45–207 days). Once the ulcers had healed, none of the patients had reulceration problems during the follow-up period.
Complications (Minor/Major)
Three patients had minor complications: wound hematoma in two flaps and partial flap necrosis in one flap, which healed after revision. Three more patients had major complications: one patient had arterial thrombosis, which was successfully revised and the flap survived completely. Another patient had major amputation. He had heel ulcer, osteomyelitis, and persisting infection with Methicillin-resistant Staphylococcus aureus (MRSA) (type FIN-16 (125:IA)) in the bacterial cultures. Despite successful free flap transfer, the infection and osteomyelitis persisted and the leg had to be amputated (below the knee). The third patient had both total flap loss and died because of sepsis, leading to multiorgan failure and death 26 days postoperatively. This patient suffered from metastatic malignancy (pancreatic cancer) as his pre-existing condition. Thus, the in-hospital mortality (30-day mortality) was 9%.
Limb Salvage and Survival
The overall cumulative limb salvage rate was 90% (95% confidence interval (CI) 71–100) at 12 months. Two patients lost their leg. The cumulative survival rate was 91% (95% CI 74–100) at 12 months. All 10 surviving patients returned home. The amputation-free survival rate was 82% (95% CI 59–100) at 12 months. Furthermore, the amputation-free survival with completely healed wounds was also 82% at 12 months (Fig. 1). All the patients who retained their legs were ambulatory.
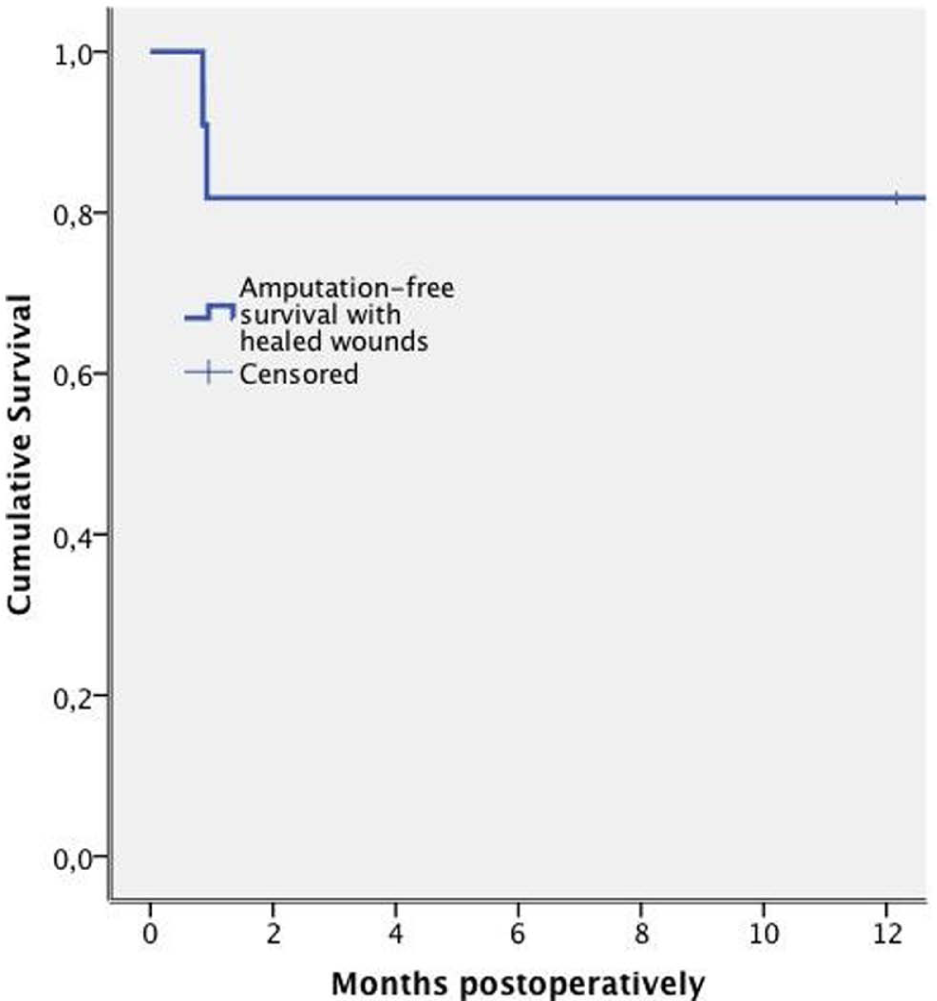
Amputation-free survival with completely healed wounds. The survival curve was derived from the Kaplan–Meier analysis.
Discussion
In this study, free tissue transfer achieved successful wound healing in patients with diabetes and difficult-to-heal wounds that would have otherwise required amputation. One year after the operation, all patients who were alive with their leg were ambulant and had returned home.
Patient loss to follow-up is common (13%–41%) in retrospective data and registry (17–19). In our small cohort, we managed to record complete data as we followed each patient at our outpatient clinic (multidisciplinary diabetic foot team) at least a year or until death. Our study, however, has a few limitations. The number of patients in our series is relatively small. However, there are only four studies on this issue with large patient population (14, 15, 20, 21). All others report the number of patients to be 9–26 (12). There are no randomized controlled studies on the subject. None of the previous studies on free tissue transfer for diabetic foot used validated wound-scoring systems for the preoperative assessments of wound (12) as is the situation also in our series making a true comparison of the different studies difficult. One problem is the lack of an accepted classification system for neuroischemic ulcers because most studies focus on a wound classification not related to the decreased perfusion of the diabetic foot (11).
In large cohort studies of patients with diabetes and foot ulcers, the outcome has often been measured as avoiding major amputation at or above the ankle (11). The information regarding the probability of complete wound healing after surgical procedures in diabetic patients is limited (19). Hoffmann et al. (22) systematically reviewed the literature (1985–2005) and identified 1915 papers on revascularization in critical leg ischemia (CLI) with tissue lesions. Complete tissue healing for surgical procedures was reported in only six studies. In our study, the overall healing rate of tissue lesions at 12 months was 82% being higher than in many studies on critical limb ischemia (22). Naturally, study population differs from ours even if diabetes has been reported to be significant risk factor to delay tissue healing also among patients with CLI (17, 19). Nevertheless, none of the patients in our study would have gained complete wound healing with conservative methods as is the situation with some CLI patients (23).
In many centers, free flap candidates are evaluated and treated through a collaborative effort of the vascular and plastic surgical services (12, 14, 21, 24). Fizgerald O’Connor et al. (12) report in their systematic review of free tissue transfer that 66% patients had concomitant revascularization with bypass surgery and 3,8% angioplasty. In our series, the proportion was only 18%. At our hospital, plastic surgery ward is part of the orthopedic unit with an active podiatric team, which may affect our patient population. Since 2012, however, all diabetic patients with foot lesions have been treated at a multidisciplinary wound center at our hospital including plastic, orthopedic, and vascular surgical services, not to forget experts from other disciplines as well.
Because of the complexity of the diabetic patient with a foot ulcer, the operative intervention should always be considered as an integrated part of a multidisciplinary strategy with several components. Treating infection, improving metabolic control, improving general condition, improving perfusion, controlling pain, and off-loading require multidisciplinary team approach. These ulcers must always be considered as a sign of multiorgan disease.
The proportion of patients who might benefit from free flaps to heal their wounds is the top of an iceberg and remains uncertain. There are, however, 10,000 major amputations performed every year in the United Kingdom for diabetes (25). At present, the numbers of free flaps performed worldwide are low and appear to be restricted to enthusiastic, highly specialized centers operating on an average of six to seven patients a year (12). The data from Hong (15) suggested that even 25% of patients with complex diabetic foot ulcers may be suitable for free flap surgery. Including a plastic surgeon with expertise in free flap reconstruction in the multidisciplinary diabetic foot team, as is the situation now at our hospital, might save limbs previously deemed unsalvageable. The use of negative pressure wound therapy (NPWT) has increased markedly in the management of all wounds worldwide, and also in our department. We regularly use NPWT in the treatment of patients with diabetic foot ulcers. In this study, however, none of the patients with complex wounds were amenable to NPWT only.
All previous studies report limb salvage rate after free tissue transfer. The average was 83.4% (95% CI 77.1–89.7) over a follow-up period ranging from 1 to 68 months (12). Our 90% rate compares well to this. In-hospital mortality, however, was 3.9% (95% CI 1.5–62) in the previous reports, thus being much lower than in our study (9%) (12). The patient who died in our study had a cancer with metastasis in his liver. Although he was then considered as a suitable candidate for free flap procedure, we now would consider presence of untreatable malignancy as a contraindication to this procedure, highlighting the learning curve and also multidisciplinary team work in patient selection. The 1-year survival rate 91% in our study, however, compares favorably to the previous reports.
Latissimus dorsi flap has traditionally been the most frequently used flap in our practice. The large muscle mass offers well-vascularized tissue to the defect area. Moreover, the donor site seldom has serious postoperative problems, the flap can be harvested simultaneously with revision and preparation of the vessels, the pedicle is long, and thoracodorsal artery is not severely affected by the arteriosclerotic disease. It is also possible to harvest the flap as a nerve-sparing partial muscle flap, retaining the function of the latissimus dorsi muscle partially (26). The “split” latissimus dorsi flap was used in two cases of this series of patients.
As limb salvage has a clear impact on quality of life and probably survival (16), we think that primary amputation is only indicated in patients who are bedridden, who have severe neurological impairment, and who have a poor survival or poor predicted functional outcome after free flap transfer.
Conclusion
Free tissue transfer achieved successful wound healing in selected patients with diabetes and difficult-to-heal wounds that would have required amputation. Complete healing of tissue lesions is slow even after successful surgery, and the patients need meticulous monitoring and controlling at the diabetic outpatient foot clinic.
Footnotes
Declaration of Conflicting Interests
No conflict of interest.
Funding
This study was financially supported by Diabetestut-kimussäätiö.
