Abstract
Background:
Postoperative respiratory complications often arise in lung cancer patients after lung resection, although these are often difficult to predict. We sought to identify reliable predictors of early-onset postoperative respiratory complications in lung cancer patients who had moderate-to-severe preoperative respiratory impairment.
Methods:
This was a prospective observational study that included 107 consecutive lung cancer patients with forced expiratory volume in 1 s <60% of predicted who were scheduled for thoracotomy and lung resection. Preoperative functional assessments included pulmonary function testing by spirometry, single breath diffusion capacity of lung for carbon monoxide, and cardiopulmonary exercise testing. Risk factors for early-onset postoperative respiratory complications that occurred within 30 days postoperatively were sought from among these pulmonary function testing and cardiopulmonary exercise testing results.
Results:
By multivariable logistic regression, peak oxygen uptake (V′O2max%; p < 0.001) and the transcutaneous pulse oxygen saturation difference during load exercise (ΔSPO2%; p < 0.001) were independent predictors of postoperative respiratory complications. A receiver operating characteristic curve had an area under the curve of 0.846 for the combination of V′O2max% and ΔSPO2%, while the area under the curve with V′O2max% only was 0.726. From this, the probability of postoperative respiratory complications was
Conclusions:
For lung cancer patients with forced expiratory volume in 1 s <60% of predicted, in addition to common preoperative tests, V′O2max% and ΔSPO2% may be an aid for predicting early-onset postoperative respiratory complications.
Introduction
Surgical resection remains the most effective treatment for operable non-small cell lung cancer (NSCLC) with 5-year survival rates ranging from 24% (stage III A) to 73% (stage IA) (1). However, many potentially resectable NSCLCs occur in individuals with moderate-to-severe pulmonary function impairment due to cigarette smoking or other concomitant factors, such as chronic obstructive pulmonary disease (COPD), history of previous thoracotomy, and chemotherapy and radiotherapy for lung cancer. All of these factors might contribute to an increased risk of postoperative complications, including death. To maximally improve their survival, surgical treatment should be provided for every suitable NSCLC patient.
To this end, a protocol should be established based on a series of careful assessments with regard to surgical tolerance, postoperative respiratory complications (PRCs), cardiovascular complications, or other possible complications. Traditional algorithms established by the British Thoracic Society (BTS) (2), the American College of Chest Physicians (ACCP) (3), and guidelines of the European Respiratory Society and the European Society of Thoracic Surgeons (ERS/ESTS) (4) suggest stepwise approaches to evaluate candidates for lung resection, although these approaches have some differences. Pulmonary function testing (PFT) is recommended first to include spirometry, particularly %predicted forced expiratory volume in 1 s (FEV1), single breath diffusion capacity of lung for carbon monoxide (DLCO), and nuclear differential perfusion scan–based measurements. Cardiopulmonary exercise testing (CPET) is used only for those who have inadequate results from these assessments (2,3). In contrast, ERS/ESTS guidelines place more emphasis on CPET evaluations (4). Alternative algorithms have been proposed in which earlier exercise peak oxygen uptake (V′O2max) might eliminate unnecessary radionuclear scanning for some patients and is more cost-effective than accepted algorithms (5,6).
Therefore, in this prospective study, we evaluated the results of preoperative PFT and CPET in order to identify those variables that were most reliable and valuable for predicting the risk of early PRCs in lung cancer patients. For this purpose, we chose patients who were at relatively “high risk” for these complications (7). These were lung cancer patients who showed preoperative moderate-to-severe pulmonary function impairment based on spirometry test results (i.e. FEV1 < 60% of predicted).
Methods
Subjects and study design
This prospective study was conducted from July 2005 to July 2010 at the Sun Yat-sen University Cancer Center. During this period, patients with potentially resectable lung cancer first underwent PFT, including spirometry and DLCO. Subsequently, patients with forced FEV1 <60% of predicted, defined as functionally moderate-to-severe impairment (8), were eligible for enrollment. Additional eligibility requirements included adequate laboratory results (hemoglobin, PaO2, PaCO2), an electrocardiogram (ECG) that was not suggestive of any serious cardiac abnormalities, and no serious medical illness that would limit survival to less than 2 years (these included serious dysfunctions of vital organs, such as heart, liver, kidney, which would contraindicate surgery or cardiopulmonary function assessments).
After their registration, patients underwent symptom-limited CPET. The minimum entry criteria for thoracic surgery included both preoperative V′O2max of ≥15 mL/min/kg and a multiplier of predicted postoperative (ppo) FEV1% and ppo DLCO% of ≥1650%. However, some patients with either a multiplier of ppo FEV1% and ppo DLCO% of <1650% (n = 14) or a preoperative V′O2max of <15 mL/min/kg (n = 1) were allowed to proceed to surgery based on their surgeon’s assessment that such treatment would offer them the best chance for long-term survival.
This study was approved by the Ethics Committee of our Hospital Clinic, and written informed consent was obtained from each participant. A total of 107 patients with moderate-to-severe pulmonary function impairment and who were expected to have more than a lobectomy were ultimately selected for this study.
Preoperative Pulmonary Function Tests
Arterial blood gases were determined while on room air. Spirometry measurements were vital capacity (VC), FEV1, and maximum ventilatory volume (MVV). Single-breath DLCO was corrected for the serum hemoglobin level using a standardized equation. Tests were performed using accepted standards (9–11) with a Quark PFT 4 (Cosmed, Milan, Italy). Results were expressed as percentages of the predicted values based on European Respiratory Society guidelines of 1993 (12). The ppo value for FEV1 was estimated by either an anatomic method based on counting the number of segments removed during lobectomy or a technetium-99 macroaggregated albumin (99mTc-MAA) lung scan method for pneumonectomy, as described previously (13).
Preoperative Cardiopulmonary Exercise Tests
Symptom-limited incremental ramp workload exercises were done using a cycle ergometer 900 (Cosmed). After a 2-min warm-up period at 20 W, a ramp protocol with a 10–20 W·min−1 workload increase was begun. The exercise test was stopped when the patient was exhausted based on either a Borg rating of >16 (14) or the occurrence of any limiting symptoms (15). The variables measured included peak oxygen uptake (V′O2max), anaerobic threshold (AT), oxygen pulse (V′O2/HR), and transcutaneous pulse oxygen saturation difference during load exercise (ΔSPO2%). V′O2max was normalized by patient mass and also expressed as a percentage of the predicted value based on the equation of Wasserman and Whipp (16). ΔSPO2% was the difference between the initial value and the lowest observed value. When determining the lowest SPO2% value, if SPO2% did not decrease rapidly within a short period, the value was required to be maintained for more than 1 min. However, if SPO2% declined swiftly within a short period, the SPO2% value at the end of CPET was chosen.
Surgical Procedures and Postoperative Care
All patients underwent thoracotomy (pneumonectomy via a posterolateral incision + mediastinal lymph node dissection), and none underwent video-assisted thoracoscopic surgery (VATS). Routine postoperative care for general thoracic surgery was provided after surgery. This included the following. (a) Routine oxygen inhalation and arterial blood gas monitoring. The flow rate and time of oxygen inhalation were adjusted appropriately (17). (b) Patients were encouraged and assisted in coughing and expectoration. Oral and nasal secretions were removed by suction if necessary. (c) Changes in vital signs (blood pressure, pulse, respiration, body temperature, transcutaneous oxygen saturation, etc.) were observed. (d) The quality and quantity of fluid drained from the chest cavity were observed. The occurrence of chest bleeding was closely observed. (e) Early ambulation was encouraged and gradual increased activity was expected. (f) Routine rehydration support was used. Phlegm-removing methods (including aerosol inhalation, intravenous injection, or oral intake of the expectorant) were also used. Antibiotics were administered to prevent infection.
Postoperative epidural analgesia was not used for any of the patients. Analgesics were administered if necessary (oral painkillers or intramuscular injection of analgesics).
Extent of Lung Tissue Resection
We estimated lung tissue loss as follows. Similar to the formulas used for estimating postoperative lung function, the percentage of lung tissue lost was calculated as the percentage of segments removed assuming a total of 19 segments. For example, a patient undergoing a right upper lobectomy would have three segments removed for an estimated tissue loss of 3/19 = 15.8% (18).
PRCs
PRCs were limited to the hospitalization period after surgery or a 30-day postoperative period and defined as follows (19): (1) lung atelectasis requiring bronchoscopy, (2) prolonged mechanical ventilation for >24 h or requiring re-intubation during the first 24 h, (3) acute respiratory failure requiring mechanical ventilation (patients in this study underwent invasive ventilation), (4) pneumonia (temperature >38 °C, compatible chest radiograph, and purulent sputum), and (5) death due to respiratory failure. Some patients had more than one complication.
Statistical Methods
Results for categorical variables are given as numbers (n) and percentages (%) and were compared between groups by chi-square or Fisher’s exact tests. Results for continuous variables are given as means ± standard deviations (SDs) and were compared between groups by independent sample Student’s t-tests. Univariate logistic regression analysis was used to identify risk factors for PRCs with results reported as odds ratios (ORs) and their corresponding 95% confidence intervals (95% CIs). Factors with P-values of < 0.05 in univariate analysis were selected for multivariate logistic regression using the forward selection method. A receiver operating characteristic (ROC) curve was generated, and the area under the curve (AUC), sensitivity, and specificity were determined. A Youden index was used to determine the cutoff values for these factors. Statistical analysis used SPSS 16.0 statistics software (SPSS Inc, Chicago, IL). A two-sided P-value of <0.05 was considered significant.
Results
A total of 178 patients were initially registered at the Sun Yat-sen University Cancer Center from July 2005 to July 2010. However, 71 were excluded for further study, 24 were considered inoperable by attending physicians because of poor cardiopulmonary function, 38 underwent less than a lobectomy, and 9 refused surgical treatment. The remaining 107 patients (104 males) were selected for this study. Their demographic and clinical characteristics (including any co-morbid conditions), preoperative functional evaluation results, surgery types, and pathological diagnosis and staging are shown in Table 1.
Demographics, pathologic characteristics, and PFT and CPET results.
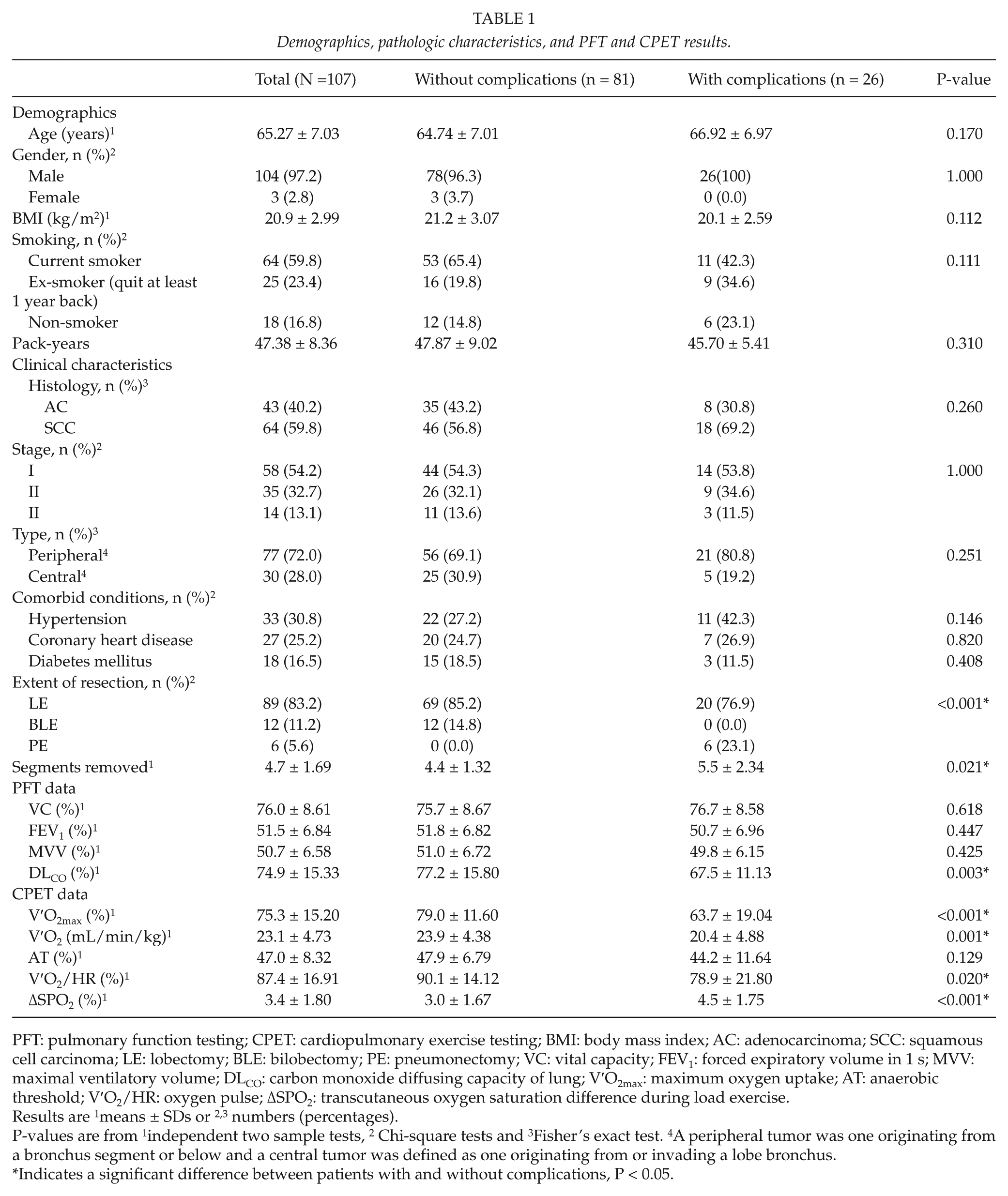
PFT: pulmonary function testing; CPET: cardiopulmonary exercise testing; BMI: body mass index; AC: adenocarcinoma; SCC: squamous cell carcinoma; LE: lobectomy; BLE: bilobectomy; PE: pneumonectomy; VC: vital capacity; FEV1: forced expiratory volume in 1 s; MVV: maximal ventilatory volume; DLCO: carbon monoxide diffusing capacity of lung; V′O2max: maximum oxygen uptake; AT: anaerobic threshold; V′O2/HR: oxygen pulse; ΔSPO2: transcutaneous oxygen saturation difference during load exercise.
Results are 1means ± SDs or 2,3 numbers (percentages).
P-values are from 1independent two sample tests, 2 Chi-square tests and 3Fisher’s exact test. 4A peripheral tumor was one originating from a bronchus segment or below and a central tumor was defined as one originating from or invading a lobe bronchus.
Indicates a significant difference between patients with and without complications, P < 0.05.
PRCs
Only 11 (10.3%) of these patients experienced mild postoperative cardiac complications of arrhythmias of <2 days duration. Thus, we focused on PRCs. Overall, for a 30-day postoperative period, PRCs occurred in 26 (24.3%) patients (Table 1). These included 11 (42.3%) who had more than one PRC and 5 (4.7%) who subsequently died after surgery. Their causes of death were as follows: two cases of postoperative pneumonia associated with atelectasis and prolonged mechanical ventilation, one case of acute respiratory failure, and two cases of acute respiratory failure and prolonged mechanical ventilation. The surgeries performed for these five patients were pneumonectomy (3), bilobectomy (1), and lobectomy (1).
Preoperative Indices and Risks of PRCs
As shown in Table 1, there were no significant differences between patients with and without PRCs for age, gender, body mass index (BMI), smoking habits, histology, pathological stage, tumor type, co-morbid conditions, airflow parameters (VC%, FEV1%, and MVV%), or exercise results (AT%). However, there were statistically significant differences between these groups for other variables. As shown in Table 2, by univariate logistic regression analysis, these variables included the number of segments removed during resection, DLCO%, V′O2max%, V′O2max/kg, AT, V′O2/HR, and ΔSPO2% during exercise (all P < 0.05). These variables were selected for multiple forward stepwise logistic regression analysis; however, because FEV1 versus ppo FEV1, DLCO versus ppo DLCO and V′O2max versus ppo V′O2max were collinear, we excluded ppo FEV1, ppo DLCO, and ppo V′O2max from the final model.
Results of univariate and multivariate logistic regression analyses.
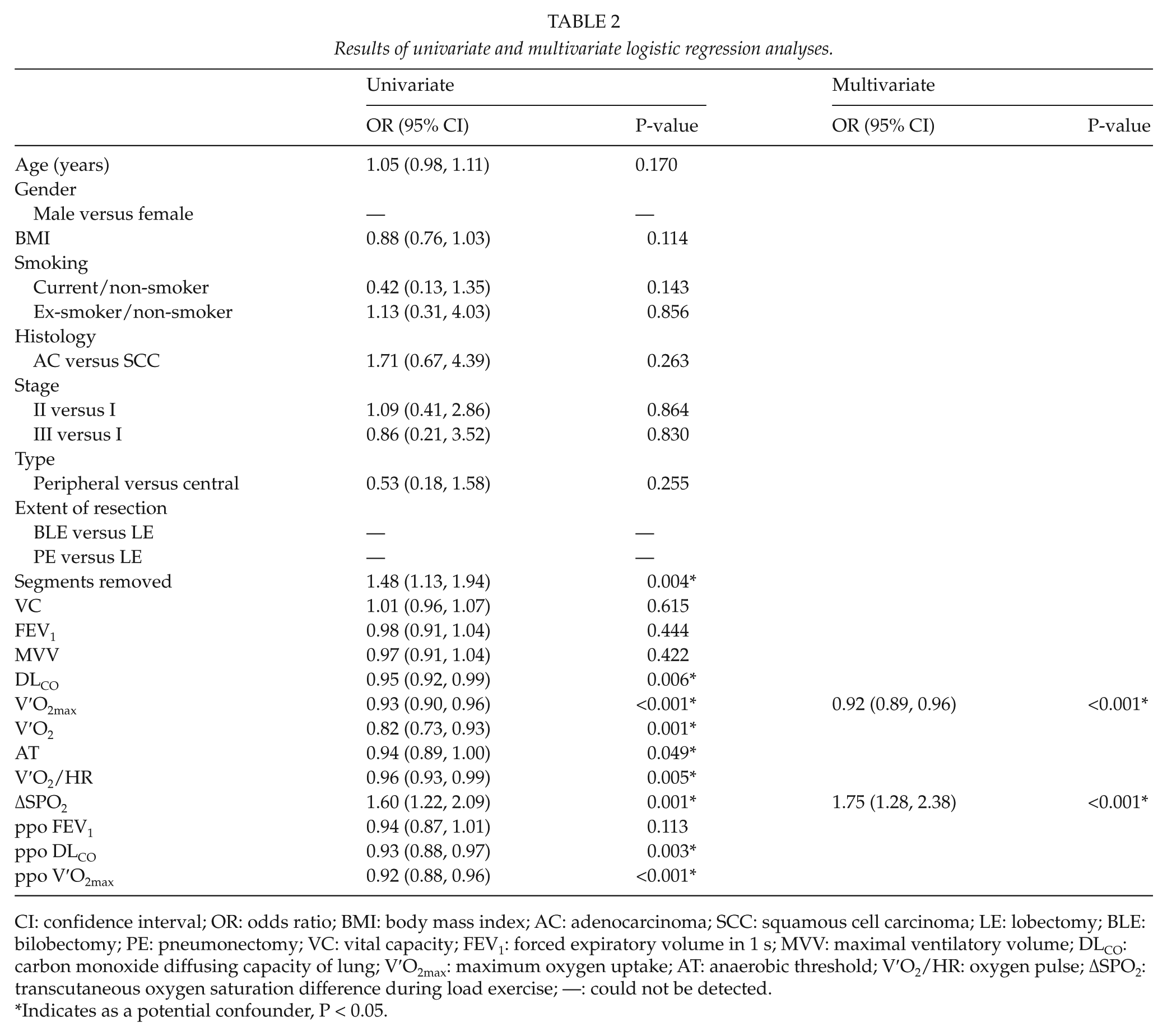
CI: confidence interval; OR: odds ratio; BMI: body mass index; AC: adenocarcinoma; SCC: squamous cell carcinoma; LE: lobectomy; BLE: bilobectomy; PE: pneumonectomy; VC: vital capacity; FEV1: forced expiratory volume in 1 s; MVV: maximal ventilatory volume; DLCO: carbon monoxide diffusing capacity of lung; V′O2max: maximum oxygen uptake; AT: anaerobic threshold; V′O2/HR: oxygen pulse; ΔSPO2: transcutaneous oxygen saturation difference during load exercise; —: could not be detected.
Indicates as a potential confounder, P < 0.05.
As also shown in Table 2, two of these were identified as independent risk factors for PRCs: V′O2max% (OR = 0.92; P < 0.001) and ΔSPO2% during exercise (OR = 1.75; P < 0.001). These OR’s were based on incremental changes of one percentage point in the respective variables. Due to the small number of cases, no analysis was done using death as the dependent variable.
Sensitivity and Specificity of Independent Predictors for PRCs
We used the variables identified by multivariate logistic regression to construct the following prediction model:
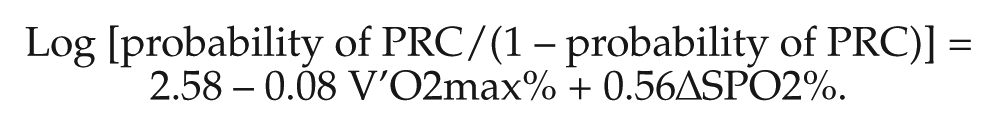
Fig. 1A shows the ROC curve from which the cutoff point was chosen. The AUC was 0.847 (95% CI: 0.762–0.931; P < 0.001). We decided upon a cutoff probability point of 0.202. For a probability of >0.202 that a patient would have a PRC, the sensitivity was 80.8% and the specificity was 81.5%. The accuracies of predicting PRCs by two combined variables (V′O2max% and ΔSPO2%) and one variable (V′O2max%) were tested by determining the AUCs for their corresponding ROCs (Fig. 1A, B). There was a significant difference in the AUCs for these two ROC curves (0.847 vs. 0.729, respectively; P = 0.001). Table 3 shows some representative examples for predicting PRCs based on these results.
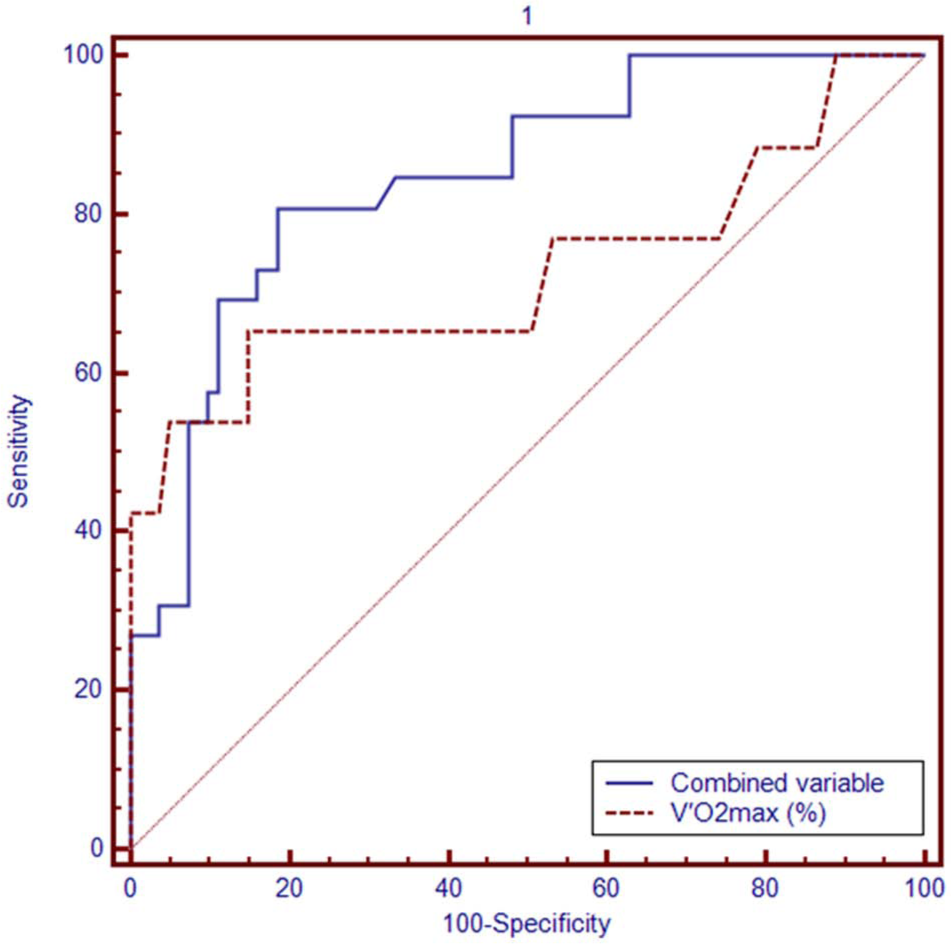
ROC curves for A) the combined variables V′O2max% and ΔSPO2% and B) the single variable V′O2max% for the occurrence of postoperative respiratory complications. The AUC for A) is 0.847 and the AUC for B) is 0.729; these AUCs are significantly different (P = 0.001).
Examples for predicting PRC probabilities.
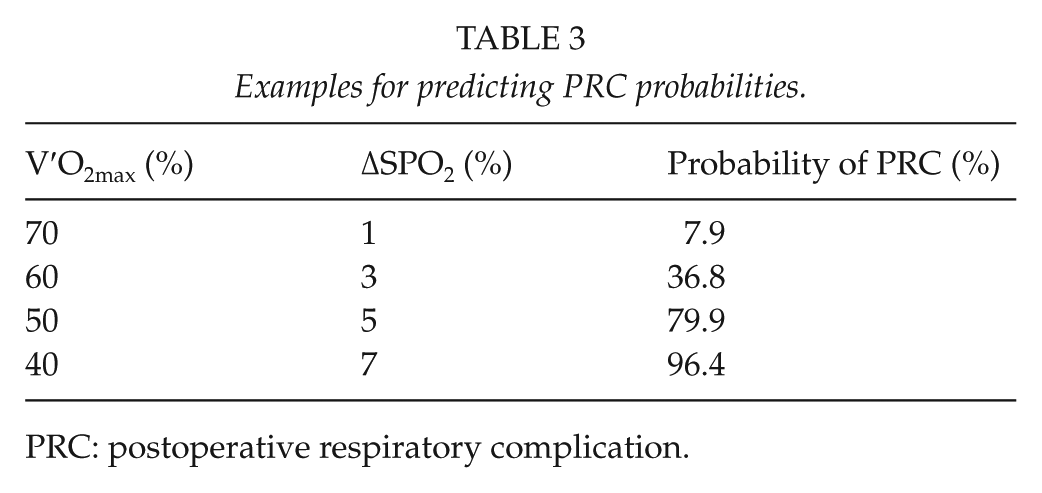
PRC: postoperative respiratory complication.
Discussion
Lung resection may decrease the ventilation volume and the size of the pulmonary vascular bed. Significant impairments in preoperative pulmonary function will increase the risk of postoperative complications, including death. In our patients with low FEV1 values, preoperative static lung function results were not useful for predicting early-onset PRCs because these data did not provide reliable information regarding cardiopulmonary reserve and postoperative pulmonary function. However, preoperative exercise capacity, reflected by V′O2max% rather than V′O2max/kg, was an independent predictor of early PRCs based on multiple logistic regression analysis; this finding was in agreement with previous studies (2,3). Therefore, efforts must focus on establishing a reliable preoperative risk assessment to increase the likelihood that lung resection can be performed safely for these patients.
For this study, we purposely selected lung cancer patients who were at “high risk” of postoperative complications (7,8), as they exhibited moderate-to-severe preoperative pulmonary functional impairment (i.e. preoperative FEV1 < 60% of predicted). Furthermore, we only focused on relatively early postoperative complications (i.e. those that occurred within 30 days postoperatively). During this observation period, 26/107 (24.3%) patients developed PRCs for which the best predictors were V′O2max% (OR = 0.92; P < 0.001) and ΔSPO2% during exercise (OR = 1.75; P < 0.001) based on preoperative CPET. None of the preoperative spirometry results or DLCO was a reliable predictor of early-onset PRCs.
Current guidelines for preoperative physiologic assessments of lung resection for lung cancer patients proposed by the ACCP (3) are based on stepwise evaluations to first include PFTs, followed by the likely postoperative pulmonary reserve, and finally CPET results. These assessments begin with spirometry to determine FEV1. If diffuse parenchymal lung disease is found on radiographic studies or if dyspnea occurs on exertion that is clinically out of proportion to the measured FEV1, then DLCO should be considered. For patients with FEV1 or DLCO <80% of the predicted value, the likely postoperative pulmonary reserve should be estimated either by a perfusion scan method for pneumonectomy or an anatomic method by counting the number of segments removed during lobectomy. When the estimated postoperative FEV1 and DLCO are both <40% of their respective predicted values, then CPET for V′O2max should be performed; a V′O2max of <15 mL/kg min indicates an increased risk of postoperative complications (3). However, due to differences in tumor sites, sizes, growth patterns, and the relationship between cardiopulmonary blood vessels or the pericardium and the presence of cardiopulmonary diseases, static pulmonary and exercise (kinetic) cardiopulmonary functions results may present diverse characteristics and have relatively low consistency for assessing pulmonary disability. For example, some patients who have abnormal pulmonary function may present with normal exercise capacity, while others who have normal pulmonary function may present with abnormal exercise capacity (20,21). In addition, preoperative pulmonary assessments to predict postoperative residual pulmonary volume may not coincide with the actual “range” of surgical resection that is actually performed (more or fewer resected segments). Therefore, both PFTs and CPETs should be performed before surgery.
In addition, radionuclide lung scanning (for predicting postoperative function) is not always available at the majority of institutions in developing countries due to the high cost of the equipment, lack of specialized personnel, and the fear of potential harm from radioactive materials. Thus, to overcome these limitations, we investigated a combination of PFT, DLCO, and CPET, from which we expected that these data would provide valuable information for evaluating the surgical risk for patients with low pulmonary function.
In this study, patients at high-risk for PRCs after lung resection were evaluated by conventional methods, and their mortality rate did not exceed that generally reported after thoracotomy (22). This suggested that surgical treatment was a reasonable choice for these patients with acceptable morbidity and mortality rates.
More recent investigations also suggest that exercise testing is more reliable than PFT results for determining lung function capacity (23,24). Exercise testing evaluates multiple organs; it incorporates an evaluation of systemic conditions, such as muscle power and nutritional status, as well as the cardiopulmonary reserve. Under a given load exercise, both cardiopulmonary stress and oxygen demand are increased, which are similar to the stress placed upon patients by lung resection. Therefore, exercise capacity indirectly reflects the capacity to endure surgery. However, the absolute value of V′O2max can be affected by many factors, such as age, gender, height, and weight. We believe that this explains why V′O2max% was more accurate than V′O2max/kg for predicting the risks of surgery.
Transcutaneous pulse oximetry may indirectly reflect the actual arterial O2 saturation during exercise. Our results showed that this was an independent predictor of postoperative complications. However, an arterial catheter for pulmonary gas exchange measurements during exercise in high-risk patients is recommended if PFT results cannot be obtained, and transcutaneous pulse oximetry provides an unreliable assessment (25).
DLCO is used to assess disease that affects the alveolar-capillary bed or the pulmonary vasculature (26) and has been shown to be a useful predictor of morbidity and mortality for patients undergoing lung resection (27,28). In the present study, univariate analysis also revealed a significant difference in DLCO% between the groups with and without early PRCs, whereas a change in arterial O2 saturation during exercise is only weakly correlated with DLCO% (25). Unlike ΔSPO2% during a load exercise, DLCO% was not an independent predictor for early PRCs using multiple logistic regression analysis. A likely explanation is that postoperative complications occur when the impairment in DLCO is severe enough to cause an obvious change in ΔSPO2%. Thus, ΔSPO2% rather than DLCO% was shown to be a more accurate predictor of early PRCs.
Furthermore, the combined variables V′O2max% and ΔSPO2% had a greater AUC than that of the single variable V′O2max%. This suggested that the integrated indices of V′O2max% and ΔSPO2% provided a greater accuracy than that of a single index (V′O2max%) for predicting postoperative complications. The logistic regression equation generated in this study that incorporated these two variables could aid in predicting the probability of PRCs with high accuracy.
There were some limitations to our study. First, we evaluated only “high–risk” lung cancer patients who had moderate-to-severe preoperative lung function impairments. Although these patients would be most likely to have early-onset postoperative complications, we cannot determine if our results could be generalized to patients with less severe pulmonary impairments. Furthermore, although we found a good AUC value in our risk assessment analysis, this was base on the same data set used for multivariable regression. A larger, independent study cohort will be needed to confirm our results. In addition, the patients we examined had not received induction therapy either with radiotherapy or chemotherapy, which could affect postoperative lung function (29). Also, the vast majority of our patients (97%) were males, which means that we cannot generalize our results to females. This high rate may be because our study focused on moderate-to-severe preoperative respiratory impairment, a higher risk condition. After reviewing the medical records, we found that all of our patients in this study were combined with COPD (FEV1/FVC% < 70%). Also, in China, most COPD patients are male. Finally, for the purposes of this study, we only evaluated relatively early-onset complications (i.e. those that occurred within 30 days postoperatively); thus, we did not determine whether the predictors we identified would also translate to predicting complications over a longer term.
In summary, we concluded from this study that for patients with resectable NSCLC, if FEV1 is <60% of the predicted value, then CPETs may be a valuable aid in predicting complications prior to lung resection. As independent predictors for early PRCs, V′O2max% and ΔSPO2% should be integrated into the preoperative assessment decisions regarding lung resection.
Footnotes
Acknowledgements
We thank Professor Liu Qing of the Statistical Analysis Department of Sun Yat-sen University Cancer Center for assistance with data processing. We thank all of the patients who participated in this study and the surgeons in the Thoracic Surgery Department for their generous cooperation.
Declaration of Conflicting Interests
The authors declare that there are no conflicts of interest associated with this work.
Funding
This study was supported by the Medical Science Research Foundation of the Guangdong Provincial Health Bureau (A2009208).
