Abstract
Background and Aims:
Recent advancements in manufacturing technology have enabled more precise tolerances and surface finishes using metal-on-metal bearing surfaces in total hip arthroplasty. The aim of this study was to compare the level of metal ions in urine after implantation of a 28-mm metal-on-metal bearing manufactured from high-carbon wrought alloy and a 28-mm metal-on-polyethylene bearing.
Material and Methods:
A total of 92 total hip arthroplasty patients were prospectively randomized into two groups: those receiving metal-on-metal bearings and those receiving metal-on-polyethylene bearings. Chromium, cobalt, and molybdenum ion levels in urine were measured preoperatively and at 1 year and 2 years postoperatively.
Results:
In the metal-on-polyethylene group, there was a slight increase in mean chromium and cobalt concentrations at 2-year follow-up compared to the preoperative level (p = 0.02 for both chromium and cobalt). In the metal-on-metal group, there was a 15-fold increase in chromium and a 26-fold increase in cobalt at 2-year follow-up compared to the preoperative level (p < 0.001 for both chromium and cobalt). However, the quantity of chromium and cobalt in urine from the metal-on-metal group was not higher at 2-year follow-up than at 1-year follow-up (p = 0.5 and p = 0.6, respectively).
Conclusions:
The 28-mm metal-on-metal bearings yield chromium and cobalt concentrations in urine that can be higher than those recommended for occupational exposure. However, our results also indicate that a steady state in wear and ion production using metal-on-metal total hip arthroplasty can occur.
Introduction
Cobalt chrome (CoCr) was first used for metal-on-metal (MOM) bearing surface applications of total hip arthroplasty (THA) in 1958 in designs such as the Ring, Müller, McKee-Farrar, and Sivash. The McKee-Farrar prosthesis was manufactured from Co-Cr-Mo alloy and had head diameters of 32–42 mm. There was a poor fit between the head and socket in early versions; however, quality improved with time, and the long-term survivorship of early MOM designs is comparable to that of the metal-on-polyethylene (MOP) design of Charnley (1, 2). Steady-state wear rates of these early designs were just a few micrometers per year (3). Satisfactory survival results for the McKee-Farrar prosthesis have been reported with an implant survival of 74% after 28 years (4).
First-generation MOM prostheses were abandoned in the 1970s because results for Charnley-type low-friction arthroplasties appeared to be better. There were also issues with the frictional torque and concerns about carcinogens and metal sensitivity. Recent advancements in manufacturing technology have enabled more precise tolerances and surface finishes and designs that provide an increased range of motion. A wrought alloy offers the unparalleled advantages of being very hard and having a very low surface roughness enabling better lubrication and minimizing friction and wear. However, an increased number of MOM bearing surface wear complications (adverse reaction to metal debris (ARMD)) has been recently detected using large-head MOM articulations (5–7). So far, the ARMD problem has been related solely on MOM articulations with femoral heads of more than 36-mm diameter.
The aim of this prospective, randomized study was to compare the level of metal ions (Co, Cr, and Mo) in urine, after implantation of a 28-mm MOM bearing THA manufactured from high-carbon wrought alloy and a 28-mm MOP bearing THA.
Patients and Methods
A total of 92 patients at two centers were prospectively randomized into two groups: those receiving THA with MOM bearings and those receiving THA with MOP bearings. Except for the acetabular insert, the implants in the two groups were similar. All implants were manufactured by Biomet. The indications for surgery were osteoarthrosis in 70 cases and rheumatoid arthritis in 16 cases; collagenosis, Legg–Calve–Perthes disease, necrosis of caput femoris, and epiphyseolysis of caput femoris accounted for 6 cases. Demographic data are presented in Table 1.
Demographic data and clinical results.
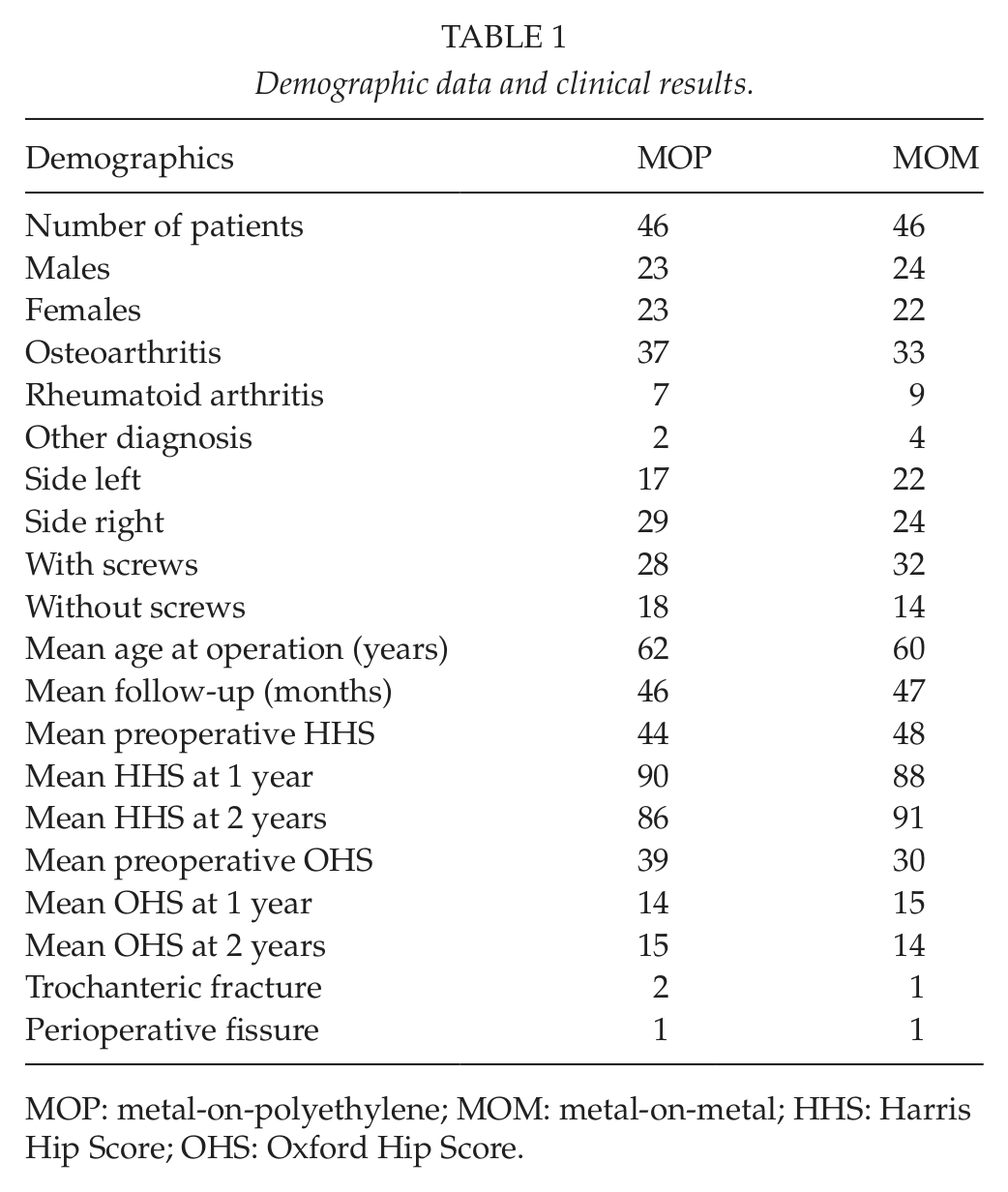
MOP: metal-on-polyethylene; MOM: metal-on-metal; HHS: Harris Hip Score; OHS: Oxford Hip Score.
At 1-year follow-up, ion measurements were performed for 41 patients in the MOM group and for 35 patients in the MOP group. At 2-year follow-up, ion measurements were performed for 29 patients in the MOM group and for 25 patients in the MOP group. Clinical and radiological analyses were performed for all patients.
In the MOM group, radial clearance between the head and liner was 25–75 µm (diametrical clearance 50–150 µm). The subtended angle of liner bearing surface was 177°. The liner inner diameter surface finish was 0.005 µm and femoral head surface finish was 0.009 µm. The femoral head and liner sphericity was <5 µm. The femoral head and MOM liner were manufactured from wrought high-carbon cobalt–chromium–molybdenum per American Society for Testing and Materials (ASTM) F-1537. The maximum range of motion achieved between head and acetabular insert was theoretically 115°. The MOM acetabular insert is seen in Fig. 1.

The m2a metal-on-metal liner.
The Harris Hip Score (HHS) and Oxford Hip Score (OHS) as well as radiographic evaluation were used to assess the outcome. Cup inclination was determined by the method of Murray (8), and cup anteversion was determined by the method of Widmer (9). Urine samples were collected from each patient preoperatively and then at 1 and 2 years postoperatively. The relative density of samples was measured using a refractometer (Atago, Japan). Analysis was performed by electrothermal atomic absorption spectrometry (EAAS, Varian SpectrAA 880 Zeeman, Australia) for urinary Cr (10) and by inductively coupled plasma mass spectrometry (ICP-MS, Varian UltrAA 700, Australia) for urinary Co and Mo. All the analyses were performed against the set of urine standards (11). The Finnish Institute of Occupational Health (FIOH) carried out all metal ion urine analyses. It participated in quality control schemes organized by Centre de Toxicology du Quebec, Canada and Friedrich-Alexander University, Erlangen-Nuremberg, Germany. All methods used to measure metal ion levels were accredited by the Centre of Metrology and Accreditation in Finland. (Laboratory T013).
Mixed test was used to analyze time trends for MOM and MOP groups by estimating and comparing means. Paired-comparison t-test was used for testing the differences of means between different time points in equal groups. Paired t-test was also used for testing difference of metal concentrations between implants groups at different time periods. The level of statistical significance was set at p < 0.05.
Results
Clinical And Radiographic Evaluation
Clinical and radiological results of the study groups are seen in Table 1. Slight radiolucency (<1 mm) around the stem was observed in Gruen zone 1 in 5 cases, in zone 2 in 1 case, in zone 4 in 1 case, in zone 6 in 2 cases, and in zone 7 in 6 cases. Slight radiolucency (<1 mm) around the cup was observed in DeLee and Charnley zone 1 in 15 cases, in zone 2 in 6 cases, and in zone 3 in 3 cases. There were more radiolucencies in the MOM group but they were all <1 mm. X-rays taken at the 2-year follow-up showed no change in stem or cup position. All implants were well fixed at 2 years. Trochanteric fracture occurred postoperatively in 3 patients: 2 of them were treated with cables and 1 with a plate. Two perioperative fissure femoral fractures occurred and they were treated with cables.
Metal Ion Levels in Urine
The upper reference limits (URLs) for Co, Cr, and Mo concentrations in Finland are presented in Table 2. The number of subjects studied in detecting reference limits varied between 100 and 200 (12).
The reference values of urinary metal ions (Co, Cr, and Mo) with and without occupational exposure in Finland.

The urinary metal concentrations in the study groups are presented in Table 3. Paired t-test results of urinary metal concentrations between different time periods in implants groups are presented in Table 4. Paired t-test results of urinary metal concentrations between implants groups at different time periods are presented in Table 5. The concentration differences between the MOM and the MOP groups were statistically significant for urinary chromium concentrations (p = 0.002) on different years (Table 3). For urinary chromium concentrations in the MOM group, differences were observed between preoperation sample and 1-year sample (p < 0.001) and 2-years sample (p < 0.001), but no significant difference was observed with results after first and second years (p = 0.5) (Table 4).The concentration differences between the MOM and the MOP groups were statistically significant for urinary cobalt concentrations (p = 0.005) on different years (Table 3). For urinary cobalt concentrations in the MOM group, the differences were statistically significant between preoperation sample and samples at first and second years (p = 0.002 and p < 0.001, respectively). No significant difference was observed with results after first and second years (p = 0.6) (Table 4). The urine chromium, cobalt, and molybdenum concentrations in the MOM and the MOP groups during the study period are also presented in Fig. 2A to C.
The preoperative, 1-year, and 2-year metal concentrations in urine7.
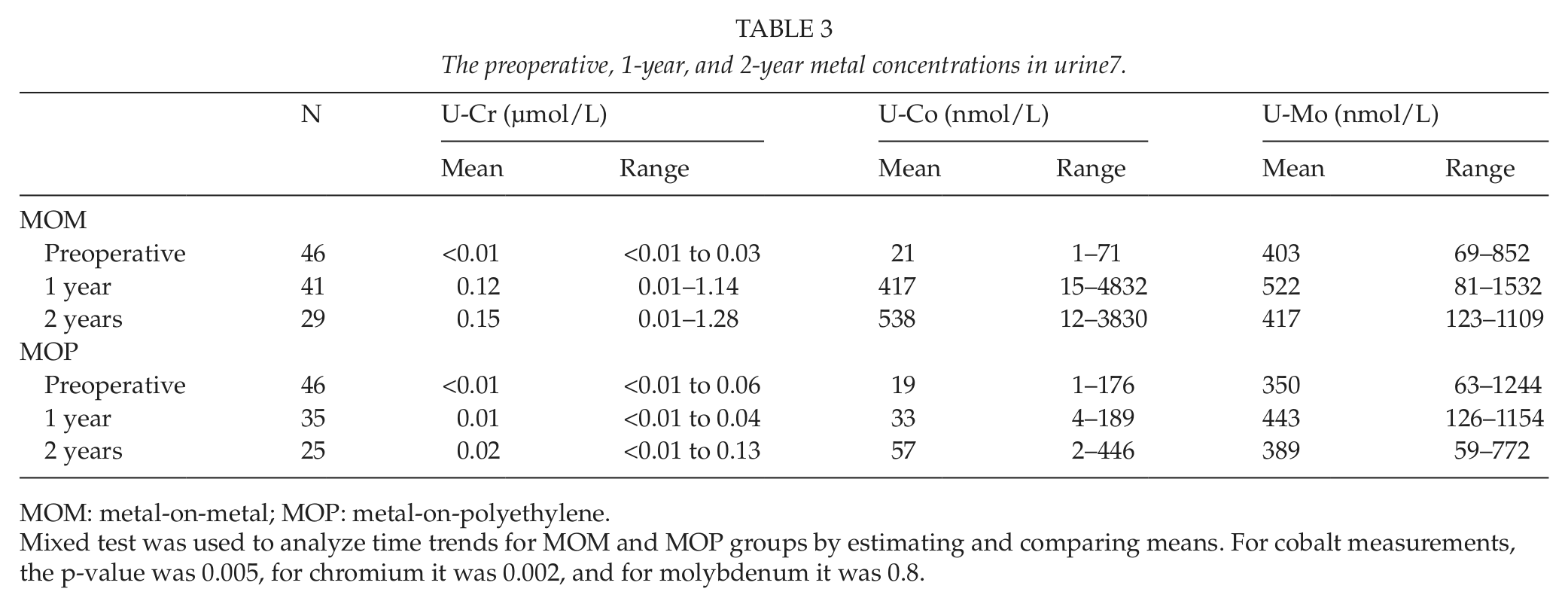
MOM: metal-on-metal; MOP: metal-on-polyethylene.
Mixed test was used to analyze time trends for MOM and MOP groups by estimating and comparing means. For cobalt measurements, the p-value was 0.005, for chromium it was 0.002, and for molybdenum it was 0.8.
Paired t-test results of urinary metal concentrations between different time periods in implants groups.
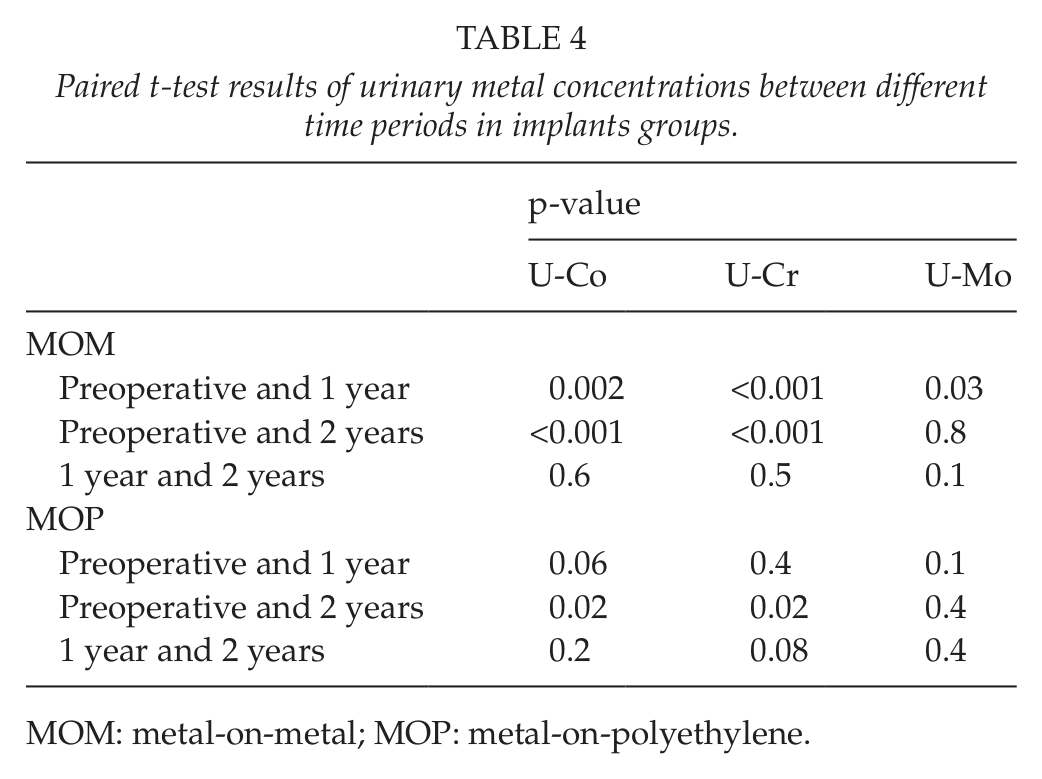
MOM: metal-on-metal; MOP: metal-on-polyethylene.
Paired t-test results of urinary metal concentrations between implants groups on different time periods.
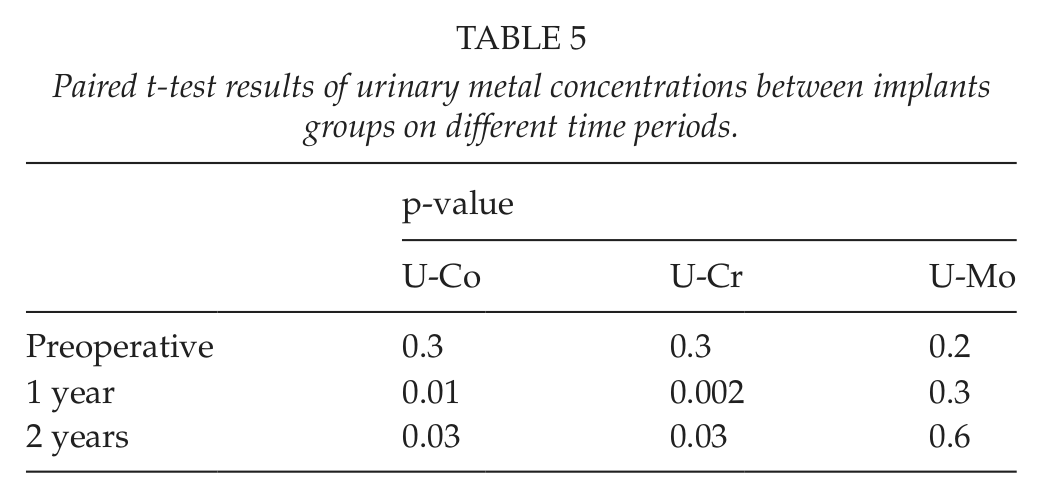
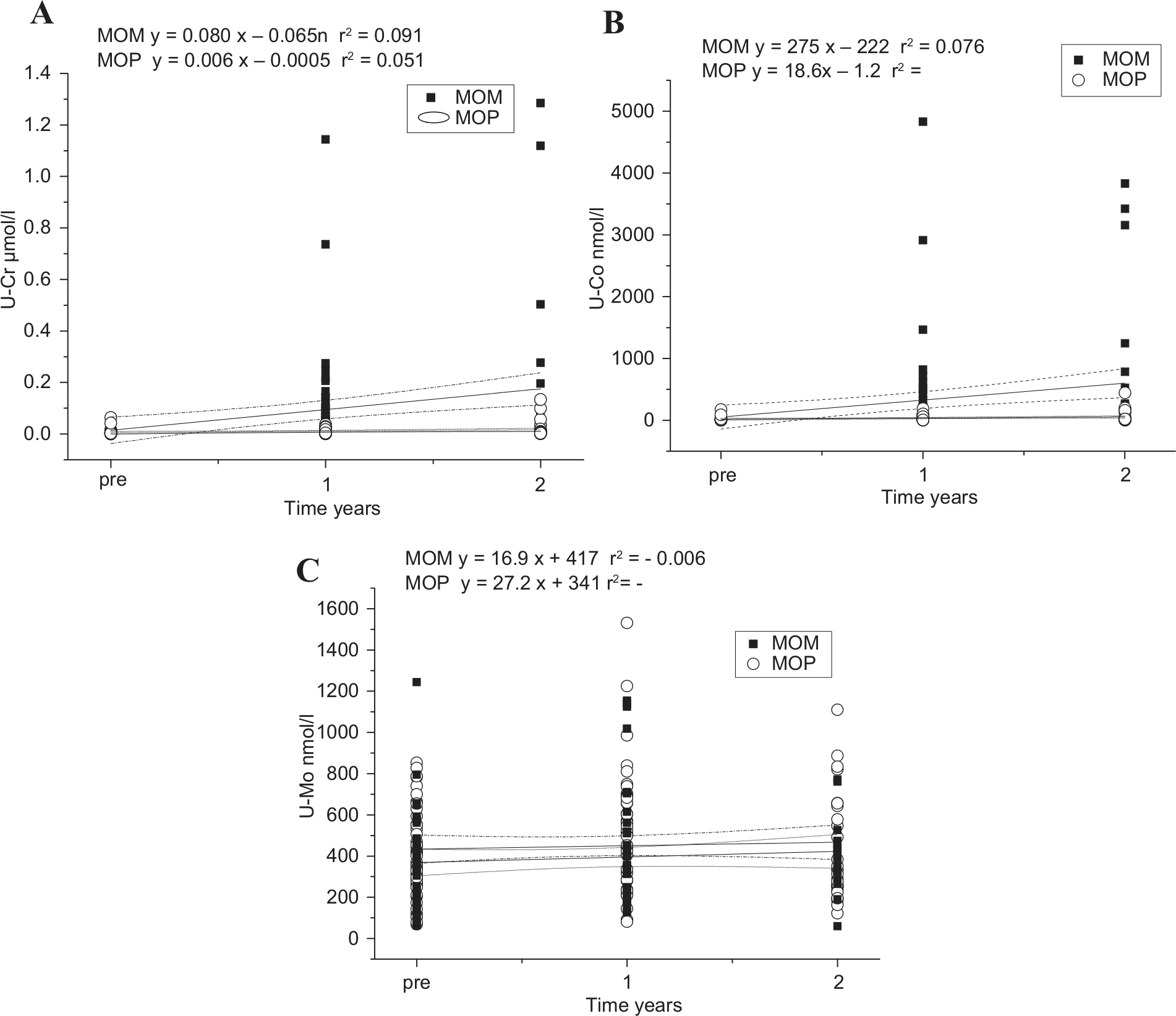
A) The urine chromium concentration in the MOM and the MOP groups during the study period. B) The urine cobalt concentration in the MOM and the MOP groups during the study period. C) The urine molybdenum concentration in the MOM and the MOP groups during the study period.
Discussion
We found out that 28-mm MOM bearings in THA yield chromium and cobalt concentrations in urine that are higher than those recommended for occupational exposure. However, our results also indicate that a steady state in wear and ion production using MOM bearings can occur.
We acknowledge that this study has some methodological shortcomings. The metal concentrations in this study were measured only from urine, not from blood. We measured the metal ion levels in urine as they are representative of the total body load. In addition, changes in their concentrations are more easily detected than in either blood or serum samples. Blood samples are taken with a metallic needle, and metal ions from the needle can significantly raise the level of the sample (13). Serum analysis is also prone to mistakes. Measuring high metal ion concentrations is not problematic, but at low levels errors can be 100-fold. Analytical problems at low metal ion concentrations are common for both serum and blood samples, and in many cases, small changes in metal concentrations are difficult or impossible to measure (14). Cigarette smoke contains Co that can cause transient changes in blood and serum concentration, and there is a lack of reliable data on the normal Co, Cr, and Mo ion levels contained in serum. However, the present consensus on measuring metal ion release is whole blood analyses.
The follow-up time of this study was relatively short. We aim at following the study groups also for a longer term. Furthermore, the number of drop-outs was relatively high. However, we think that it did not have a remarkable effect on our message.
Cobalt and chromium are essential trace elements. Cobalt is needed for the synthesis of cyanocobalamin (vitamin B12). Excessive administration of cobalt is believed to produce goiter and possibly cardiomyopathy. Chromium is part of the metabolic pathway that produces a cellular response to insulin and the uptake of glucose. It is therefore essential for the energy functions of the cell. The possible carcinogenic properties of these metals have been discussed since MOM bearing surfaces were introduced. Cobalt and chromium wear particles have been shown to induce carcinoma in animal models (15). Elevated levels of chromium and cobalt have been found also in human tissues surrounding orthopedic implants and tissues at remote sites (16). There has been concern that MOM bearings may pose a higher risk of malignant degeneration because of an increased exposure to metal particles and ions (17). Recent studies have raised the possibility of increased DNA (18) and chromosomal changes (19) occurring in patients with both MOM and non-MOM devices (19). As MOM bearings are used in young patients, there is concern that mutagenic or teratogenic effects could be manifest in children conceived by these patients.
A study of the transplacental transfer of metal ions in patients with MOM hip devices led to the conclusion that the elevated ion levels in maternal blood do not lead to raised levels in the umbilical cord blood and that the placenta acts as an effective barrier to metal ions released from these devices (20). This would imply that the rapidly dividing and differentiating tissues in the developing fetus are not exposed to the elevated metal ion levels. However, according to Ziaee et al. (21), both cobalt and chromium cross the placenta, both in normal subjects without metal implants and in patients with MOM hip devices. The placenta exerts a modulatory effect on the rate of metal transfer. At low maternal metal ion levels, there is a higher transfer rate than at higher maternal level.
Levels Of Metal Ions After Small- And Large-Diameter Mom Tha
Theoretically, the production of wear debris and thus metal ion release would be less after large-diameter-head THA and hip resurfacing arthroplasty (HRA) than after THA using small MOM heads. This is based on the fluid film lubrication and reduced potential for the production of wear debris (22). However, there is some discrepancy in the published results. Clarke et al. (23) showed that serum levels of cobalt and chromium using HRA were higher than those after 28-mm MOM THA. In the study of Antoniou et al. (24), the median cobalt and chromium levels in blood were significantly lower in the 36-mm MOM THA group compared to that of the 28-mm MOM THA group and the HRA group at 6 months postoperatively. At 1 year, however, neither the cobalt nor the chromium levels were significantly different among the three MOM groups. It should be emphasized that ion levels are influenced by several reasons. There are differences in the radial clearances and other manufacturing parameters between the implants. Metal ions can be generated from surface corrosion as well as from wear particles. Our study device with small MOM heads has been clinically successful in Finland. Recently, bigger MOM heads have become more common in our country. However, an increased number of MOM bearing surface wear complications (ARMD) has been recently detected using large-head MOM articulations (5–7). So far, the ARMD problem has been related solely on MOM articulations with femoral heads of more than 36 mm in diameter.
In the study of De Haan et al. (25), there were higher levels of metal ions in patients with steeply inclined components. Steeply inclined acetabular components with abduction angles greater than 55° combined with a small size of components are likely to increase serum levels of cobalt and chromium ions. Unfortunately, we were not able to present data concerning association of cup positioning and ion production in this study.
We did not find any increase in the Cr level from the preoperative level to 2-year level for the MOP group. In Finland, the URL for Cr in urine for unexposed people is 0.01 µmol/L, and it is held as target value for chromium exposure. This level is based to derived no-effect level (DNEL) and for exposure via breathing in occupational exposure (12). This level was exceeded in 1- and 2-year samples in MOM group and, in few cases, in 2-year samples in the MOP group. Co level for the MOP group increased from a mean preoperative value of 19 nmol/L to 37 nmol/L at 1 year postoperatively and to 57 nmol/L at 2 years postoperatively, which is above the URL of 25 nmol/L for people not exposed to chemicals at work. The reason for the increase at 1 year and 2 years is not clear, but the consumption of greater amounts of vitamin B12 may explain it. The bronchoalveolar lavage (BAL) for Co is 130 nmol/L based on the occupational exposure limit of 0.010 mg/m3, and the mean value was not reached in the MOP group (26). The Mo concentration in urine for the MOP group remained fairly constant throughout the study and was well below the URL of 1340 nmol/L for unexposed people in Finland (12).
In the MOM group, there was a 10-fold increase (1 year postoperatively) and a 15-fold increase (2 years postoperatively) in mean urine Cr compared to the preoperative Cr value, and it exceeded the target level of 0.01 µmol/L. The mean Co concentration in urine corresponded to almost a 20-fold increase (1 year postoperatively) and a 26-fold increase (2 years postoperatively) in urine cobalt level, and Co levels were above those of unexposed Finns and also clearly exceed the BAL of 130 nmol/L. This value was reduced during 2012 because of evidence that cobalt and its inorganic compounds cause cancer (27, 28). The mean amount of Mo in urine from the MOM group did not differ during the study period. None of these values exceeded the URL for Mo of 1340 nmol/L for unexposed people in Finland (12).
Wear studies have shown that MOM implants often exhibit an accelerated wear rate during the initial 1 to 2 years of clinical use (29). After lubrication, self-polishing effects, and oxygenation, this increased wear period is followed by a lower steady-state wear. During the steady state, linear wear rate is less than 5 µm for several years, which corresponds to a volumetric wear rate of less than 0.2 mm3/year (30). We tried to define maximum values for cobalt and chromium levels. This is difficult because any direct toxicity associated with the release of metal ions from MOM bearings has not yet been demonstrated (31). Vendittoli et al. (32) compared blood ion levels of the patients having 28-mm diameter head Metasul bearing with those having large-diameter-head Metasul bearing. No significant difference was found at 2 years postoperatively for cobalt and chromium. A steady state was reached at 1 year in the resurfaced group and after 3 months in the total hip replacement group. However, the large-diameter-head Durom cup with Metasul bearing studied by Vendittoli has recently been associated with ARMD and early loosening (33).
Further studies are required to measure metal levels over longer periods of time and to evaluate whether they have any clinical significance following MOM hip implantation. MOP bearings seem to be a safe option for THA patients compared to MOM bearings with regard to metal ion levels.
Footnotes
Funding
This study was sponsored by Biomet UK Limited.
