Abstract
Background and Aims:
Accumulating evidence indicates that a state of chronic inflammation has a crucial role in the pathogenesis of obesity-related metabolic dysfunction. This study aims to evaluate changes in inflammatory process, iron metabolism, erythropoiesis, and lipid profile associated with weight loss after gastric banding surgery.
Material and Methods:
A total of 46 patients were evaluated before and 3 months after gastric banding surgery. A survey was conducted to record demographic data, body mass index, and presence of comorbidities. Moreover, complete blood cell counts and serum levels of iron, ferritin, transferrin, high-sensitivity C-reactive protein, total cholesterol, low-density lipoprotein–cholesterol, high-density lipoprotein–cholesterol, and triglycerides were performed.
Results:
At a follow-up 3 months after surgery, our patients presented a significant decrease in weight and body mass index, associated with a decreased inflammatory process (decreased high-sensitivity C-reactive protein, neutrophil counts, and neutrophil:lymphocyte ratio), increased iron availability (increased transferrin saturation and a trend to higher iron serum levels), and significant decreased triglycerides and triglycerides:high-density lipoprotein–cholesterol ratio.
Conclusions:
Our results showed a significant decrease in the inflammation process 3 months after gastric banding surgery, associated with adipose tissue loss. This decrease in the inflammatory process is associated with more efficient iron absorption and increased iron availability for erythropoiesis. Moreover, we also found decreased triglyceride serum levels. These changes suggest benefits of weight loss, including decreased risk of cardiovascular disease.
Introduction
Obesity is increasing all over the world both in developed and underdeveloped countries (1, 2) and is known to be associated with inflammatory changes, insulin resistance, and hyperlipidemic state (3). The adipocyte is an important source of cytokines, namely, leptin, interleukin-6, and tumor necrosis factor–alpha, and their levels are significantly higher in the plasma of obese patients (4). Moreover, some inflammatory markers, such as C-reactive protein (CRP), show an independent positive association with body mass index (BMI) (5).
Cardiovascular morbidity and mortality is associated with classic risk factors, namely, dyslipidemia, hypertension, and impaired glucose metabolism. However, more recently, inflammatory mechanisms were recognized as playing a vital role in initiation, maintenance, and progression of atherosclerotic vascular disease. Atherosclerosis is an inflammatory disease with the participation of leukocytes and several cytokines. Among several inflammatory markers that have been shown to predict cardiovascular events, CRP levels have emerged as the most powerful marker (6).
Gastric banding surgery is a safe surgical procedure for the treatment of morbid obesity (7), with 0% of mortality in the perioperative period (8, 9) and a long-term mortality rate from unrelated causes of 3.7% (8). Using laparoscopic surgery, a gradual, continuous, and well-programmed weight loss (10) is achieved, with a postoperative increasing quality of life and reducing comorbidities in over 75% of patients, including systemic inflammatory response (11–15). Korenkov et al. (16) concluded that 8 years after restrictive laparoscopic surgery, the prevalence of type 2 diabetes decreased from 10% to 4% of the patients, pulmonary disease from 15% to 5%, arterial hypertension from 43% to 27%, and knee pain from 47% to 38%. This study aims to evaluate the effects of weight loss achieved after bariatric surgery in inflammatory process, iron metabolism, and lipid profile.
Material and Methods
Patients
In this longitudinal study, 46 patients were evaluated immediately before and 3 months after gastric banding surgery. Surgeries were performed by the same surgical team, under the same preconditions, and following the usual anesthetic and surgical procedures. Patients were randomly selected for this surgical team. Patients included in this study have a BMI more than 33 kg/m2 (43.1 ± 6.2 kg/m2) before gastric band placement (LAP-BAND®, Allergan, Inc., Irvine, CA, USA). None of the patients had abnormal blood loss during operation or serious postoperative complications. No dietary changes were recommended after surgery, neither iron nor vitamin supplementation. This study was approved by the ethics committee of Hospital da Prelada—Dr. Domingos Braga da Cruz, and the patients gave their informed consent.
Assays
A survey was conducted in order to record demographic data, presence of comorbidities, and BMI. Moreover, erythrocyte count, hematocrit, hemoglobin concentration, hematimetric indices (mean cell volume (MCV), mean cell hemoglobin (MCH), and mean cell hemoglobin concentration (MCHC)), red cell distribution width (RDW), reticulocytes counts, and total and differential leukocytes counts were measured using an automatic blood cell counter (Sysmex XT-2000i; Emilio Azevedo Campos, Porto, Portugal). The reticulocyte production index (RPI) was calculated as an appropriate way to measure the effective erythrocyte production, by correcting for both changes in hematocrit (degree of anemia) and premature reticulocyte releasing from the bone marrow (17). Total and direct bilirubin serum levels (Beckman Coulter, Brea, CA, USA) and serum iron (Beckman Coulter) concentration were determined using a colorimetric method, whereas serum transferrin (Beckman Coulter) was measured by immunoturbidimetry, and serum ferritin (Abbott Laboratories, Abbott Park, IL, USA) was measured by microparticle enzyme immunoassay. Transferrin saturation (TS) was calculated using the formula: TS (%) = 70.9 × serum iron concentration (µg/dL)/serum transferrin concentration (mg/dL). Serum high-sensitivity CRP (hsCRP) was determined by immunoturbidimetry (CRP reagent; Beckman Coulter). Triglycerides (TGs), total cholesterol, high-density lipoprotein–cholesterol (HDL-c), low-density lipoprotein–cholesterol (LDL-c) were determined by enzymatic colorimetric assay (all from Beckman Coulter).
Data Analysis
For statistical analysis, we used the Statistical Package for Social Sciences (SPSS version 17.0 for Windows; SPSS, Inc., Chicago, IL, USA). Kolmogorov–Smirnov statistics were used to evaluate sample normality distribution. Measurements are presented as mean ± standard deviation (SD) for parametric values or as median values (interquartile range) for nonparametric value. For comparing data, before and after gastric banding surgery, paired-samples t-test or Wilcoxon test was used. Pearson’s rank correlation coefficient was used to evaluate relationships between sets of data. Significance was accepted at p < 0.05.
Results
Table 1 summarizes the hematological data, iron metabolism, inflammatory markers, and lipid profile, before and 3 months after gastric banding surgery. From the total of 46 obese patients (6 males, 40 females; mean age ± SD = 40.57 ± 10.35 years), 17.39% were consumers of sweets, 39.13% suffered from hypertension, 19.57% were diabetic, 50% had hypercholesterolemia, 4.34% had sleep apnea, and 2.17% had osteoarthroses. In all, 13 of the female patients (32.5%) were premenopausal.
Hematological data, iron metabolism, inflammatory markers, and lipid profile before and 3 months after gastric banding surgery.
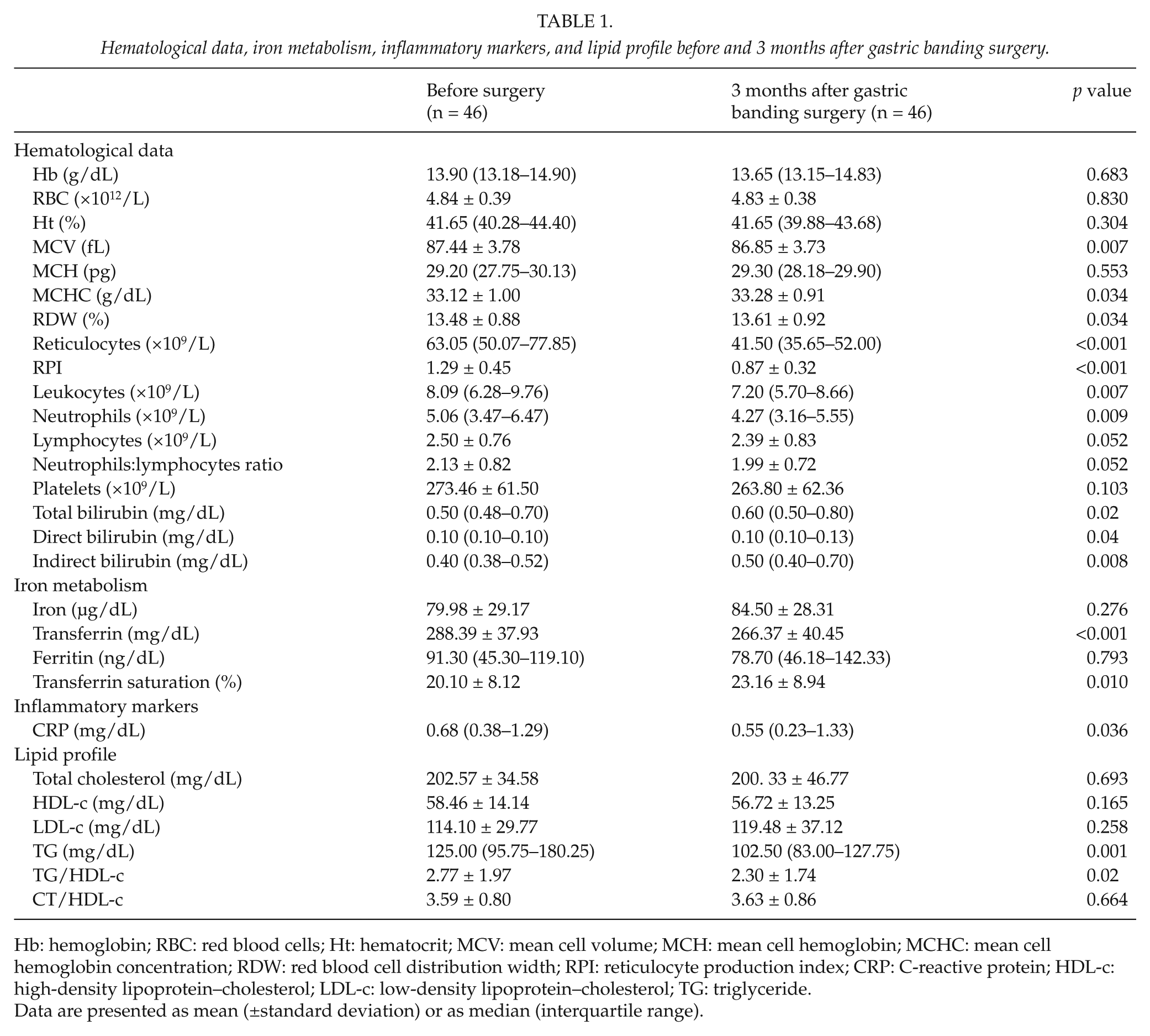
Hb: hemoglobin; RBC: red blood cells; Ht: hematocrit; MCV: mean cell volume; MCH: mean cell hemoglobin; MCHC: mean cell hemoglobin concentration; RDW: red blood cell distribution width; RPI: reticulocyte production index; CRP: C-reactive protein; HDL-c: high-density lipoprotein–cholesterol; LDL-c: low-density lipoprotein–cholesterol; TG: triglyceride.
Data are presented as mean (±standard deviation) or as median (interquartile range).
On follow-up 3 months after gastric banding surgery, our patients showed decreased body weight (115.03 ± 22.39 vs 99.90 ± 19.53 kg, p < 0.001) and BMI (43.10 ± 6.16 vs 37.37 ± 6.28 kg/m2, p < 0.001), associated with decreased MCV, RPI, and reticulocytes, leukocytes, neutrophils, and lymphocytes counts, and a trend toward lower neutrophil:lymphocyte ratio, with an increased total, direct, and indirect bilirubin serum levels. Moreover, we also found changes in iron metabolism (increased TS, decreased transferrin serum levels, and a trend to higher iron serum levels) and inflammatory markers (decreased hsCRP). With regard to lipid profile, a significant decreased TG and TG:HDL-c ratio was observed.
A significant positive correlation was found between BMI variation along the study (BMI 3 months after surgery − BMI before surgery), and hemoglobin concentration variation (r = 0.315; p = 0.033), reticulocyte counts variation (r = 0.444; p = 0.002), and neutrophil counts variation (r = 0.432; p = 0.003).
Discussion
Adipocytes exhibit an active role in metabolism by interacting with the immune system through inflammatory mediators and signaling molecules. In fact, it is well defined that obesity is characterized by the presence of low-grade inflammatory state and that the adipocytes play an important role in the production of inflammatory mediators. Moreover, vascularization of adipocyte tissue became insufficient, leading to hypoxia, which can be a critical factor in inflammatory state (18).
Cytokine production and the conversion of preadipocytes into macrophages results in the accumulation of macrophages with increased expression of proinflammatory molecules that contribute to the inflammatory state. In obese patients, when they reach a significant reduction in BMI, decrease in adipocyte tissue seems to reverse the low-grade inflammation (19). In fact, three months after gastric banding surgery, our obese patients showed significant weight and BMI losses, associated with an improvement in the inflammation process, demonstrated by a significant decreased hsCRP (p = 0.036), leukocyte (p = 0.007), and neutrophil counts (p = 0.009), and a trend toward decrease in neutrophil:lymphocyte ratio (p = 0.052). Moreover, we also found a correlation between BMI variation and neutrophil count variation, which is consistent with the decrease in inflammatory process associated with a decrease in weight loss (20). These results are in accordance with those recently published by Viardot et al. (21), which demonstrated that energy restriction before and after gastric banding surgery attenuates activation of circulating immune cells of innate and adaptive immune system, and with those of Merhi et al. (22) who demonstrated an inverse relationship between changes in T-cells (CD4+ and CD3+) and the amount of weight lost after bariatric surgery.
Recently, a complex regulatory network that governs iron traffic emerged and pointed to hepcidin as a major evolutionary conserved regulator of iron distribution (23, 24). This small hormone produced by the mammalian liver has been proposed as a central mediator of dietary iron absorption. Hepcidin was found to be associated with decreases in both iron uptake from the small intestine and release of iron from macrophages, as well as decreased placental iron transport (23, 24). The synthesis of hepcidin is stimulated by inflammation and iron overload. Since obesity is associated with an increased inflammatory process, it will be expected that these patients show an increase in hepcidin serum levels, a decrease in serum iron levels, and consequently, a decrease in erythropoiesis. Our obese patients showed, 3 months after gastric banding surgery, a trend toward an increased iron serum level and significant increased TS, suggesting more effective iron absorption, perhaps by decreased levels of hepcidin associated with a decrease in the inflammatory process related to weight loss. Thus, our results are in accordance with those published by Tussing-Humphreys et al. (25), who found an association between weight loss and reduced serum hepcidin and inflammation and improved functional iron status.
The decrease in reticulocyte counts and IPR, 3 months after gastric banding surgery, reflects a reduction in erythropoiesis, which is also associated with BMI reduction and weight loss. Thus, weight loss is linked with a reduction in red cell mass, which can justify the increased bilirubin serum levels found in our patients. Moreover, the significant positive correlation between BMI variation, reticulocyte counts, and hemoglobin concentration variations found in our patients corroborates this hypothesis.
We also observed in our obese patients an improvement in lipid profile, as shown by the significant decrease in TG serum levels and TG:HDL-c ratio. It has been documented (26) that there is an increase in adiponectin serum levels after gastric bypass. We do not evaluate adiponectin levels in our patients; however, an increased adiponectin level associated with weight loss was expected. Therefore, considering the protective effects of adiponectin, it seems reasonable to hypothesize that the increase in adiponectin level is likely to explain, at least in part, the modifications in TG metabolism associated with weight loss.
In conclusion, our results showed a significant decrease in the inflammation process 3 months after gastric banding surgery, which can be associated with adipose tissue loss. This decrease in the inflammatory process is associated with more efficient iron absorption and with increased iron availability for erythropoiesis. Moreover, we also found decreased TG serum levels. These changes suggest benefits from weight loss after gastric banding surgery, including decreased risk of cardiovascular diseases.
