Abstract
Background:
The aim of this study was to investigate the impact of transfusion of blood products on intermediate outcome after coronary artery bypass surgery.
Patients:
Complete data on perioperative blood transfusion in patients undergoing coronary artery bypass surgery were available from 2001 patients who were operated at our institution.
Results:
Transfusion of any blood product (relative risk = 1.678, 95% confidence interval = 1.087–2.590) was an independent predictor of all-cause mortality. The additive effect of each blood product on all-cause mortality (relative risk = 1.401, 95% confidence interval = 1.203–1.630) and cardiac mortality (relative risk = 1.553, 95% confidence interval = 1.273–1.895) was evident when the sum of each blood product was included in the regression models. However, when single blood products were included in the regression model, transfusion of fresh frozen plasma/Octaplas® was the only blood product associated with increased risk of all-cause mortality (relative risk = 1.692, 95% confidence interval = 1.222–2.344) and cardiac mortality (relative risk = 2.125, 95% confidence interval = 1.414–3.194). The effect of blood product transfusion was particularly evident during the first three postoperative months. Since follow-up was truncated at 3 months, transfusion of any blood product was a significant predictor of all-cause mortality (relative risk = 2.998, 95% confidence interval = 1.053–0.537). Analysis of patients who survived or had at least 3 months of potential follow-up showed that transfusion of any blood product was not associated with a significantly increased risk of intermediate all-cause mortality (relative risk = 1.430, 95% confidence interval = 0.880–2.323).
Conclusions:
Transfusion of any blood product is associated with a significant risk of all-cause and cardiac mortality after coronary artery bypass surgery. Such a risk seems to be limited to the early postoperative period and diminishes later on. Among blood products, perioperative use of fresh frozen plasma or Octaplas seems to be the main determinant of mortality.
Keywords
Introduction
Transfusion of blood products is a cornerstone treatment to improve oxygen delivery and promote hemostasis during major surgery. Despite this, an increasing burden of evidence suggests the negative impact of blood products on the immediate postoperative outcome (1). This is particularly true in coronary artery bypass surgery (CABG) as the need of blood product transfusion is rather high due to the invasiveness of the procedure and perioperative use of potent antiplatelet drugs (2). There is some evidence that blood transfusion may affect also late outcome, but this issue is controversial and also the reasons behind such possible negative late effects are unknown. Furthermore, much research has been done on the impact of transfusion of red blood cells (RBCs) (3–8), but the prognostic impact of transfusion of platelets, fresh frozen plasma (FFP), and solvent-/detergent-treated plasma (Octaplas®; Octapharma AG, Lachen, Switzerland) on patients undergoing CABG is unknown. These issues have been investigated in this study.
Material and Methods
This study includes a consecutive series of patients who underwent CABG from June 2006 to April 2011 at the Oulu University Hospital, Finland. During the study period, 2234 consecutive patients underwent CABG at our institutions. Complete data on the perioperative use of blood products such as RBC, platelets, FFP, and Octaplas were available from 2001 patients who are the subjects of the present analysis. Patients’ characteristics are summarized in Table 1.
Baseline characteristics and operative data of 2001 patients who underwent coronary artery bypass surgery.
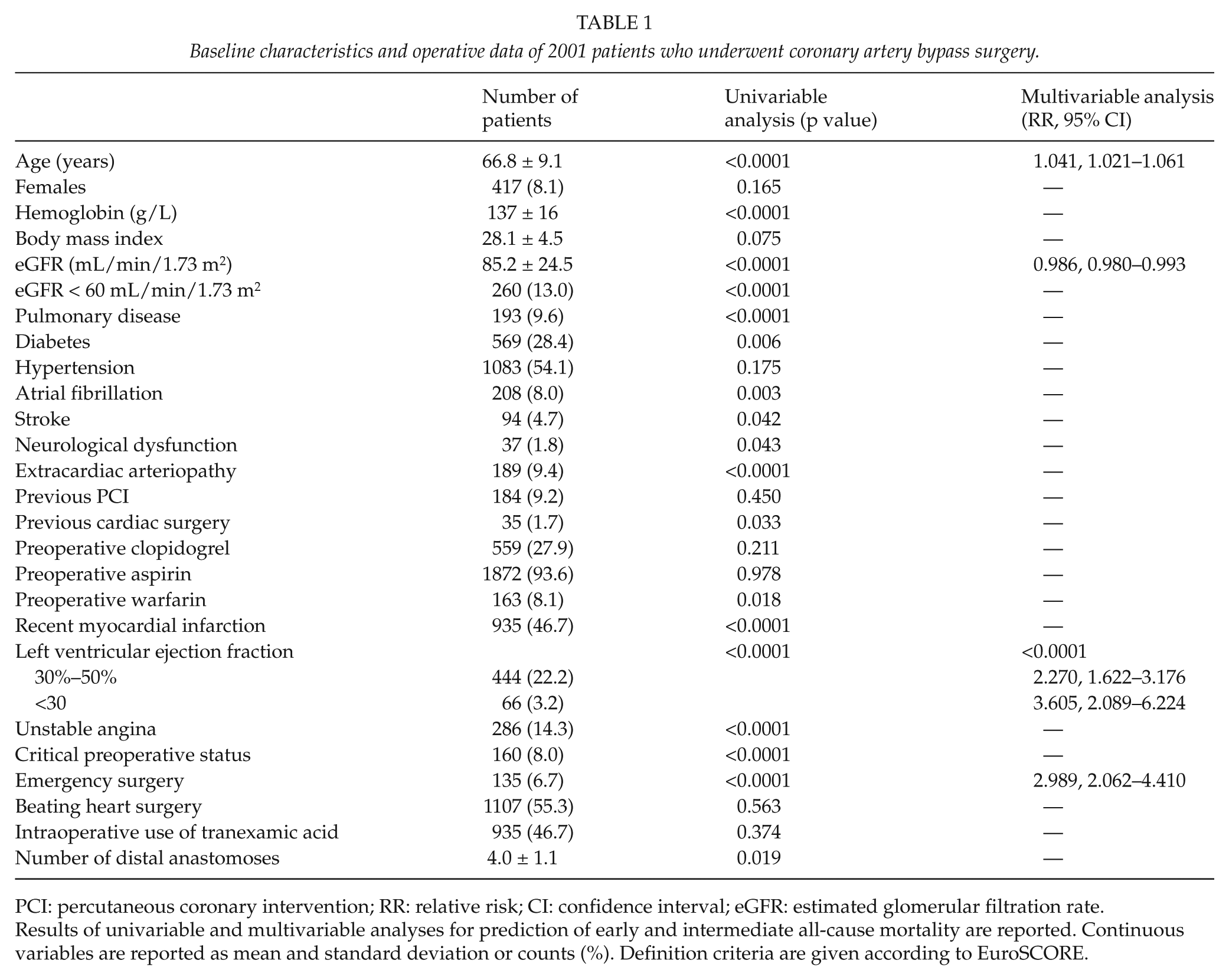
PCI: percutaneous coronary intervention; RR: relative risk; CI: confidence interval; eGFR: estimated glomerular filtration rate.
Results of univariable and multivariable analyses for prediction of early and intermediate all-cause mortality are reported. Continuous variables are reported as mean and standard deviation or counts (%). Definition criteria are given according to EuroSCORE.
Baseline and operative data were provided by a local institutional computerized database. The full medical records of the eligible patients were reviewed in order to determine the perioperative antithrombotic strategies and the incidence of major operative complications. Operative risk was assessed by the EuroSCORE risk scoring method (9). Follow-up was complete for all patients, and data on causes and date of death were acquired from the Finnish National Registry Statistics Finland.
Data on the use of blood products were retrieved from a prospective computerized database (VerSo), which collects information on all patients having or not received any transfusion of allogeneic blood products. For the purpose of this study, we collected data on the use of blood products (RBC, platelets, FFP, and Octaplas) during the operation day up to 30 days after surgery.
Perioperative Antithrombotic Treatment and Blood Transfusion Policy
Heparin (2.5–4.0 mg/kg) was administered intravenously after sternotomy to maintain an activated clotting time of more than 400 s, and it was neutralized at the end of the procedure by protamine sulfate (1.5–3.0 mg/kg). Furthermore, protamine was given in case of bleeding during closure of the chest or within the first hour after surgery according to activated coagulation time. Aprotinin was not used in any of these patients. Tranexamic acid was administered intraoperatively at the discretion of the anesthesiologist. Packed leukoreduced RBCs were transfused on the operation day if hemoglobin is less than 90 g/L. Later, RBCs were transfused if hemoglobin is less than 80 g/L. Octaplas as well as platelets were transfused according to the amount of intra- and postoperative bleeding, international normalized ratio (INR) levels, and platelet count.
All blood lost during the operation was collected in the cell saver reservoir, and washed, salvaged RBCs were autotransfused during or at the completion of the operation. Mediastinal blood/fluid was collected after surgery in a sterile collection chamber connected to 15-cm H2O wall suction via an underwater seal and then discarded.
Enoxaparin (40–80 mg once a day) was started from the evening of the operation day in those patients without excessive bleeding (<1000 mL). Aspirin (100 mg) was restarted on the first postoperative day. Warfarin was started on the first postoperative day in patients on chronic oral anticoagulation or started de novo in case of persistent atrial fibrillation. Clopidogrel was used postoperatively in these patients only in case of allergy to aspirin or recent percutaneous coronary intervention. Antiplatelet treatment strategy was the same in patients operated by off-pump or on-pump technique.
Operative Techniques
Intermittent antegrade and retrograde cold blood cardioplegia was used during conventional CABG. Proximal anastomoses were sutured to the ascending aorta during cross clamping, when the latter was considered safe. Epiaortic ultrasound was performed according to the surgeon’s preference. Octopus® stabilizer, intracoronary shunts and, in some instances, Starfish® stabilizer (Medtronic, Minneapolis, MN, USA) were used in patients who underwent off-pump coronary surgery.
Outcome End Point
The primary outcome end points of this study were all-cause and cardiac mortality. Secondary outcome end points were 30-day mortality, low cardiac output syndrome (cardiac index < 2.0 L/min/m2 at least twice), postoperative use of inotropics for more than 12 h, stroke, and de novo dialysis.
Ethical Considerations
The study protocol was approved by the Institutional Review Board of the Oulu University Hospital. This study was not financially supported.
Statistical Analysis
Statistical analysis was performed using a PASW v. 18 statistical software (IBM SPSS Inc., Chicago, IL, USA). Continuous variables are reported as the mean ± standard deviation. Correlation between continuous variables was evaluated by Spearman’s test. Logistic regression was used to adjust transfusion of any blood product for additive EuroSCORE. Survival analyses were performed by Kaplan–Meier’s method and Cox’s regression method with backward selection. Only variables having a p < 0.05 at univariate analysis have been included in the regression model. A p < 0.05 was considered statistically significant.
Results
In this series, 2.8% of patients died during the 30-day postoperative period, 12.2% had low cardiac output syndrome, 27.0% required use of inotropics for more than 12 h, 2.1% suffered stroke, and 1.4% required de novo dialysis. Re-exploration for bleeding was needed in 5.6% of patients. When adjusted for additive EuroSCORE, transfusion of any blood product was associated with significantly higher risk of 30-day mortality (p = 0.048, odds ratio (OR) = 4.496, 95% confidence interval (CI) = 1.012–19.972), low cardiac output syndrome (p < 0.0001, OR = 2.691, 95% CI = 1.728–4.191), prolonged use of inotropics (p < 0.0001, OR = 3.273, 95% CI = 2.387–4.486), and stroke (p = 0.016, OR = 3.763, 95% CI = 1.286–11.011). It also tended to be associated with de novo dialysis (p = 0.057).
The mean follow-up was 2.5 ± 1.5 years. Three-year all-cause mortality was 9.5% and cardiac mortality was 5.8%.
All-Cause Mortality
Results of univariable and multivariable analyses are summarized in Tables 1 and 2. All blood products were associated with significantly higher risk of all-cause mortality (Fig. 1). The impact of FFP/Octaplas on all-cause mortality was particularly evident (p < 0.0001). Multivariable analysis showed that patient’s age, estimated glomerular filtration rate (eGFR), left ventricular ejection fraction, emergency operation, and any blood product transfusion (relative risk (RR) = 1.678, 95% CI = 1.087–2.590) were independent predictors of all-cause mortality (Tables 1 and 2). When RBC transfusion, platelets transfusion, and FFP/Octaplas transfusion were included in this regression model, FFP/Octaplas transfusion was the only blood product associated with increased risk of all-cause mortality (RR = 1.692, 95% CI = 1.222–2.344). Only 64 patients received FFP, and when these patients were compared to those who received Octaplas, no significant differences were observed in terms of all-cause mortality (p = 0.631) and cardiac mortality (p = 0.703). RBC transfusion was marginally associated with all-cause mortality (RR = 1.503, 95% CI = 0.986–2.291). When the units of blood products were entered in this regression model, FFP/Octaplas units (RR = 1.066, 95% CI = 1.014–1.121) and RBC units (RR = 1.045, 95% CI = 1.010–1.082) were independent predictors of all-cause mortality. The additive effect of blood products was particularly evident when the sum of each blood product was included in the regression analysis (p < 0.0001, RR = 1.401, 95% CI = 1.203–1.630).
Data on postoperative bleeding and use of blood products in 2001 patients who underwent coronary artery bypass surgery.
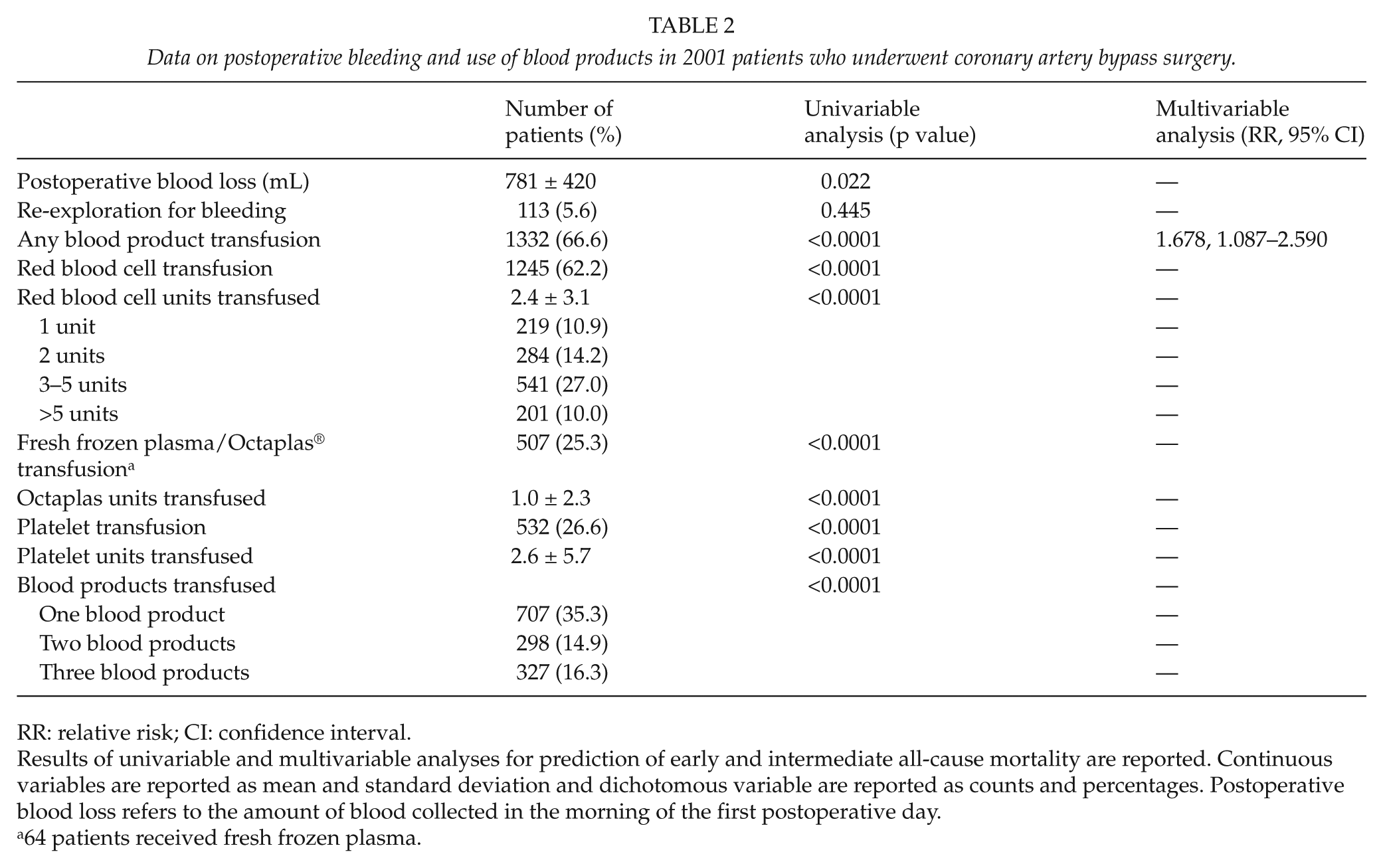
RR: relative risk; CI: confidence interval.
Results of univariable and multivariable analyses for prediction of early and intermediate all-cause mortality are reported. Continuous variables are reported as mean and standard deviation and dichotomous variable are reported as counts and percentages. Postoperative blood loss refers to the amount of blood collected in the morning of the first postoperative day.
64 patients received fresh frozen plasma.
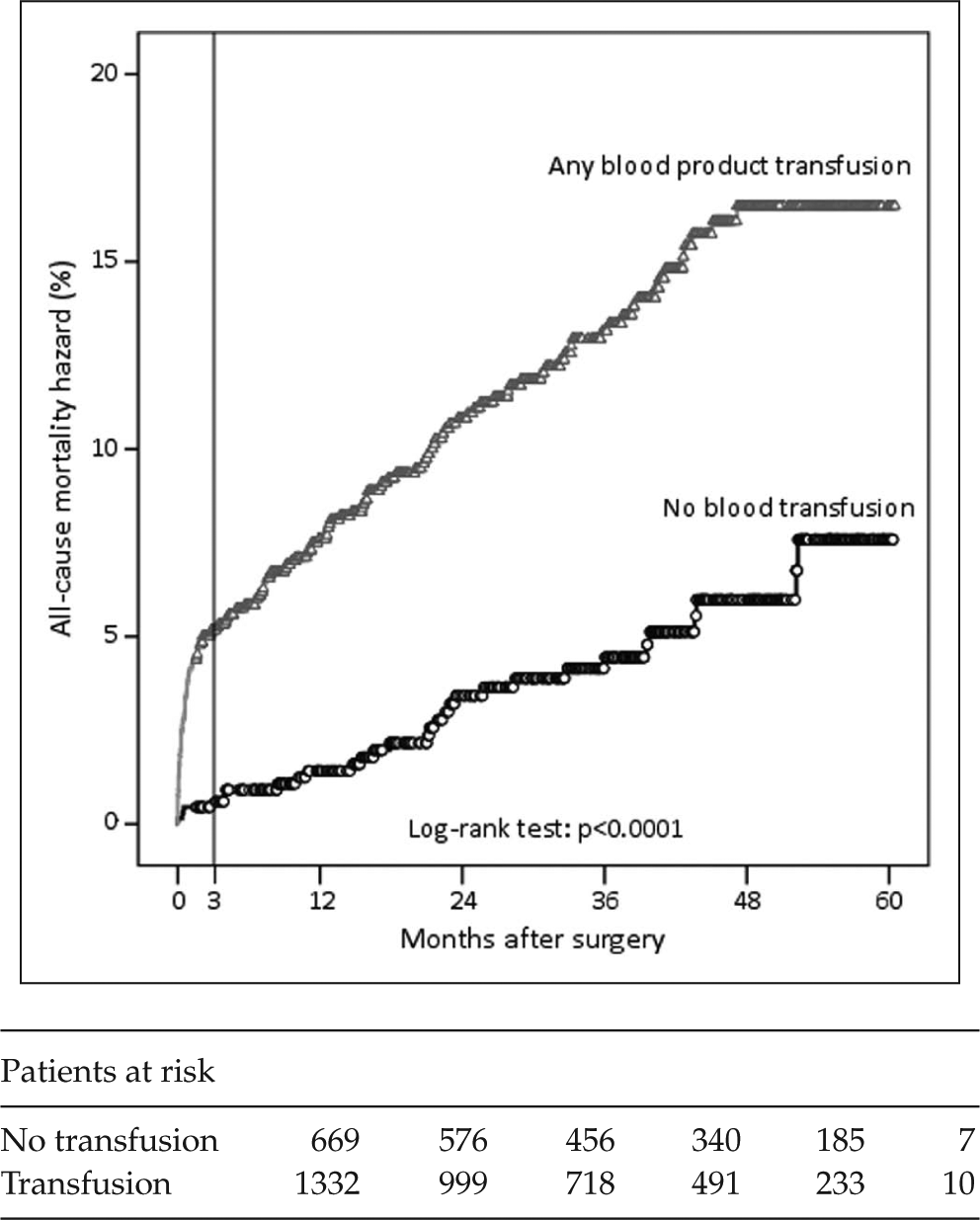
Kaplan–Meier’s estimate of all-cause mortality hazard in patients who received or did not receive any type of blood product transfusion.
Cardiac Mortality
Univariate analysis showed that age (p < 0.0001), eGFR (p < 0.0001), number of distal anastomoses (p = 0.005), diabetes (p = 0.018), extracardiac arteriopathy (p < 0.0001), prior cardiac surgery (p < 0.0001), recent myocardial infarction (p < 0.0001), left ventricular ejection fraction (p < 0.0001), emergency operation (p < 0.0001), critical preoperative status (p < 0.0001), atrial fibrillation (p = 0.005) as well as any kind of blood product transfusion (for all products: p < 0.0001) were associated with any cardiac mortality.
Cox regression analysis showed that any blood product transfusion was associated with significantly increased risk of cardiac mortality (p = 0.040, RR = 1.893, 95% CI = 1.030–3.478). When each blood product at once was entered into the regression model, RBC transfusion (p = 0.027, RR = 1.950, 95% CI = 1.078–3.526), FFP/Octaplas transfusion (p < 0.0001, RR = 2.292, 95% CI = 1.533–3.426), and platelet transfusion (p = 0.002, RR = 1.913, 95% CI = 1.265–2.893) were predictive of cardiac mortality. When all the three blood products were entered into the Cox regression model along with the other covariates, FFP/Octaplas was the only blood product associated with increased risk of cardiac mortality (p < 0.0001, RR = 2.125, 95% CI = 1.414–3.194). The synergic effect of blood products was evident when the sum of each blood product was included in the regression analysis (p < 0.0001, RR = 1.553, 95% CI = 1.273–1.895).
Impact of Blood Product Transfusion In Patients Surviving Longer Than 3 Months After Surgery
The effect of blood product transfusion was particularly marked during the first three postoperative months (Fig. 1). When the follow-up was truncated at 3 months, any blood product transfusion was a significant predictor of all-cause mortality (p = 0.040, RR = 2.998, 95% CI = 1.053–0.537; Fig. 2). In order to exclude a possible effect of transfusion only on the early outcome, we evaluated the outcome of 1880 patients who survived or had at least 3 months of potential follow-up. In this patient population, transfusion of any blood product was associated with all-cause and cardiac mortality only at univariable analysis, but not at multivariable analysis (all-cause mortality: p = 0.148, RR = 1.430, 95% CI = 0.880–2.323 (Fig. 3); cardiac mortality: p = 0.470, RR = 1.329, 95% CI = 0.614–2.877). The inclusion of each single blood product in the analysis did not significantly affect intermediate outcome of this subgroup of patients.
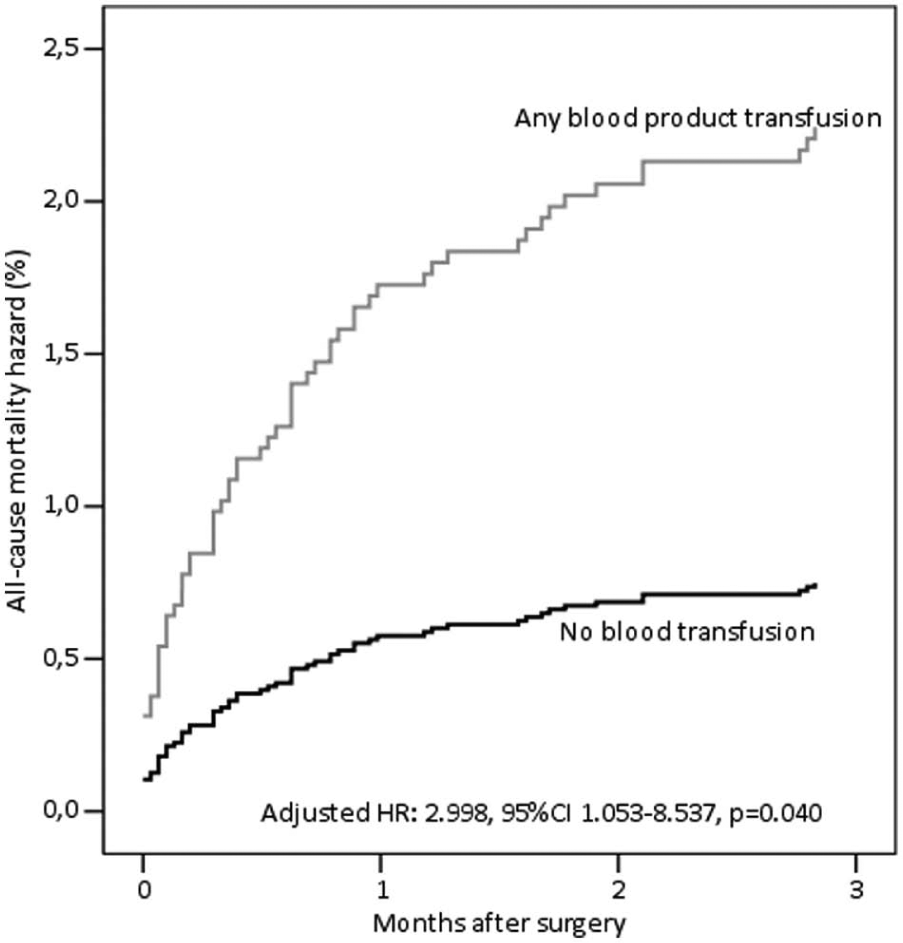
Cox proportional hazard estimate of all-cause mortality for any blood product transfusion at 3 months after coronary artery bypass surgery.
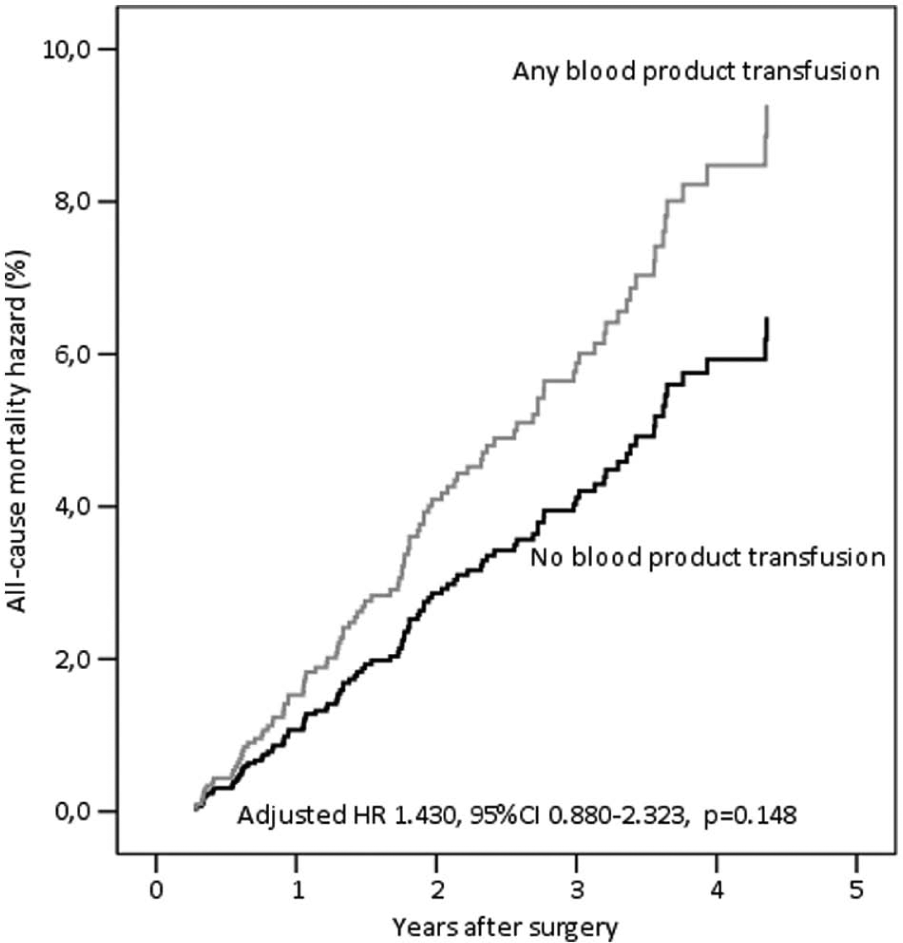
Cox proportional hazard estimate of all-cause mortality for any blood product transfusion in 1880 patients with a possible follow-up longer than 3 months after coronary artery bypass surgery.
Discussion
An increasing number of studies have shown that blood transfusion during cardiac surgery is associated with significant risk of postoperative mortality (1, 3–7, 10, 11), stroke (1), low cardiac output syndrome (1, 3), troponin release (12), infective complications (10, 11, 13–15), and renal failure (8, 11, 15). The negative impact of blood transfusion on the immediate postoperative mortality and morbidity has been confirmed in this study. Such an increased risk of adverse events seems to be independent of the severity of perioperative anemia (3, 12, 16). A recent study showed that nadir hematocrit has a major impact on immediate postoperative adverse events of patients who did not receive RBC transfusion after cardiac surgery (17). The authors did not provide data as to whether blood products other than RBCs were administered in these patients, and this may introduce a bias. However, this to some extent suggests that the individual impact of perioperative anemia and blood transfusion is not easily discernible, and both these risk factors may contribute independently to affect patients’ recovery.
Much research focused on the impact of RBC transfusion (3, 4–8). However, the use of FFP, Octaplas, and platelet transfusions may also affect the outcome after cardiac surgery as they are employed to promote coagulation and thus may potentially induce thrombotic complications. Koch et al. (10) reported on protective effect of FFP and platelet transfusions after CABG, but these findings were not coupled with others. In a recent multicenter study, Mikkola et al. (1) observed that the use of Octaplas was associated with an increased risk of postoperative stroke. Whitson et al. (15) reported on the prognostic impact of all the three blood products on the risk of postoperative complications after adult cardiac surgery. They identified a cutoff of 5.5 units of any blood product as a major determinant of poor outcome (15). Therefore, FFP and platelets also significantly contributed to adverse postoperative outcome. The present results suggest that using any allogeneic blood product may contribute negatively to the prognosis of patients undergoing CABG. At this stage, it is not clear whether patients receiving all the three blood products are at the highest risk of mortality because of the additive negative effects of each different type of allogeneic blood products or the severity of bleeding. However, we can assume that evaluation of the prognostic impact of blood transfusion should not be limited to transfusion of allogeneic RBCs.
A number of studies showed an association between blood transfusion and decreased long-term survival after CABG (6, 8, 18). Koch et al. (19) also showed that RBC and platelet transfusions have a negative impact on the health-related quality of life 6–12 months after cardiac surgery. However, this study and other studies failed to confirm these findings. Weightman et al. (20) evaluated a series of 1841 patients who survived longer than 60 days after CABG and did not find any association between transfusion of blood products and all-cause mortality up to 10 years after surgery. Using a similar approach, Surgenor et al. (4) showed that RBC transfusion had a significant impact on mortality up to 6 months, but during the late phase (6 months to 5 years), there was no significant association between allogeneic RBC exposure and mortality. Van Straten et al. (21) did not find any significant association between RBC transfusion and late mortality (>30 days) after CABG. Similarly, we observed that among patients with a follow-up of at least 3 months, perioperative use of any blood product was associated with a slight increased risk of late mortality, which was not statistically significant. These controversial findings can be explained by possible differences in the study populations and different transfusion policies. Furthermore, during the last few years, potent antiplatelet drugs were introduced in the perioperative treatment of patients undergoing CABG, and aprotinin use was discontinued. The latter may have significantly contributed to changes in the severity of bleeding, need of transfusion, and occurrence of cardiovascular events, and possibly also the prognosis of these patients.
The present results provide further evidence on the need of reengineering of preoperative management of anemia, prevention of intra- and postoperative bleeding, and a more parsimonious use of blood product management in patients undergoing CABG. This can be achieved by proactive screening for and treatment of anemia before surgery, preoperative discontinuation of potent antiplatelet drugs, use of acute normovolemic hemodilution, use of thromboelastometry, intraoperative use of tranexamic acid, and avoidance of overtransfusion (22, 23). Since surgeons may also play an important role in reducing the amount of perioperative bleeding (24), meticulous surgical technique remains a major goal to reduce the need for blood transfusion. Any of these measures may significantly contribute to the reduction of need for blood transfusion with possibly improved clinical outcome and decrease in costs.
Of concern is the negative prognostic impact of the use of FFP/Octaplas in these patients. Earlier, we observed an increased risk of stroke associated with its use during CABG (1). There is no specific data about the clinical impact of Octaplas transfusion in patients undergoing cardiac surgery, but De Maistre et al. (25) observed an increased risk of venous thromboembolism after FFP transfusion in patients who underwent abdominal aortic surgery. The reported lower level of protein S in Octaplas compared with FFP is of concern (26). Administration of solvent-/detergent-treated plasma has been shown to be associated with decreased protein S activity (27, 28), which may be detrimental in patients with congenital or acquired protein S deficiency. Therefore, we may suspect that a prothrombotic status may be further enhanced by solvent-/detergent-treated plasma in patients undergoing major surgery.
In conclusion, transfusion of any blood product is associated with a significantly increased risk of all-cause and cardiac mortality after CABG. Such a risk seems to be limited to the early postoperative period and diminishes later on. Among blood products, perioperative use of FFP or Octaplas seems to be a main determinant of mortality.
