Abstract
Complex abdominal wall defects refer to situations where simple ventral hernia repair is not feasible because the defect is very large, there is a concomitant infection or failed previous repair attempt, or if there is not enough original skin to cover the repair. Usually a complex abdominal wall repair is preceded by a period of temporary abdominal closure where the short-term aims include closure of the catabolic drain, protection of the viscera and preventing fistula formation, preventing bowel adherence to the abdominal wall, and enabling future fascial and skin closure. Currently the best way to achieve these goals is the vacuum- and mesh-mediated fascial traction method achieving close to 90% fascial closure rates. The long-term aims of an abdominal closure following a planned hernia strategy include intact skin cover, fascial closure at midline (if possible), good functional outcome with innervated abdominal musculature, no pain and good cosmetic result. The main methods of abdominal wall reconstruction include the use of prosthetic (mesh) or autologous material (tissue flaps). In patients with original skin cover over the fascial defect (simple ventral hernia), the most commonly used method is hernia repair with an artificial mesh. For more complex defects, our first choice of reconstruction is the component separation technique, sometimes combined with a mesh. In contaminated fields where component separation alone is not feasible, a combination with a biological mesh can be used. In large defects with grafted skin, a free TFL flap is the best option, sometimes reinforced with a mesh and enhanced with components separation.
Keywords
INTRODUCTION
An abdominal wall defect exists when one or more components of the abdominal wall are missing. The most important parts of the abdominal wall include the fascia and muscles providing support and function, and the skin which protects the inner layers. The abdominal wall defects can be divided in two groups depending on the presence or absence of normal skin coverage. In Type I defect there is intact or stable skin coverage, whereas Type II defects have absent or unstable skin coverage (1).
Open abdomen is a Type II defect with a skin defect and exposed viscera, and has been further categorized into different grades depending on the presence of contamination, lateralization and fixation of the abdominal wall, the presence of enteric fistula and finally a frozen abdomen, respectively (2). Recently, the World Society of Abdominal Compartment Syndrome Consensus Conference revised the classification by categorizing open abdomen into four categories with subgroups depending on the presence or absence of contamination and enteric fistula, respectively.
Planned ventral hernia refers to a management strategy where the abdominal fascial layer has been left unclosed and the viscera are covered with original or grafted skin (3).
The focus of this review is in the management of complex abdominal wall defects, mainly Type II defects in the above mentioned classification with special emphasis on the different surgical solutions that are available.
TEMPORARY ABDOMINAL CLOSURE
Although a full-thickness resection of a part of the abdominal wall for tumor or necrotizing infection obviously creates an abdominal wall defect, the most common cause of a complex abdominal wall defect today is one or other form of an open abdomen management strategy whether deliberate (damage control procedure with the abdomen left open) or unavoidable. Unless the patient undergoes an immediate abdominal wall reconstruction procedure (rare except in tumor cases), a form of temporary abdominal cover is used.
The short-term aims of temporary abdominal closure include closure of the catabolic drain (4), protection of the viscera and preventing fistula formation, preventing bowel adherence to the abdominal wall, and enabling future fascial and skin closure. The evolution of temporary abdominal closure techniques has reached its fourth generation (5). The first types of closure techniques aimed at protecting the viscera by closing the skin (often under tension) with towel clips or “skin only” running suture. If not possible, the viscera were covered with a plastic sheet, originally described by Oswaldo Borraez from Colombia (Bolsa de Borraez). The second generation involved the addition of a suction to remove excess fluid, the so-called vacuum pack dressing. With the invention of a negative pressure therapy utilizing a vacuum-assisted closure technique, the management of open abdomens reached a new stage. Finally, the combination of mechanical traction with a temporary mesh was combined with the negative pressure therapy, the vacuum-assisted wound closure and mesh-mediated fascial traction technique (6, 7). Recently, this temporary abdominal closure method has been shown to achieve fascial closure rates up to 90% (8).
PLANNED HERNIA
Leaving the fascia open can be deliberate or unavoidable and usually results from staged repair of the abdominal wall due to trauma, peritonitis, pancreatitis, abdominal vascular emergencies or abdominal compartment syndrome. Although the skin can sometimes be closed primarily (skin only closure), in most cases a split-thickness skin graft is used to cover the abdominal viscera (Fig. 1). In these situations the hernia is a favorable outcome with the aim of repairing the hernia at a later stage when it is safe, possible and tolerated by the patient.
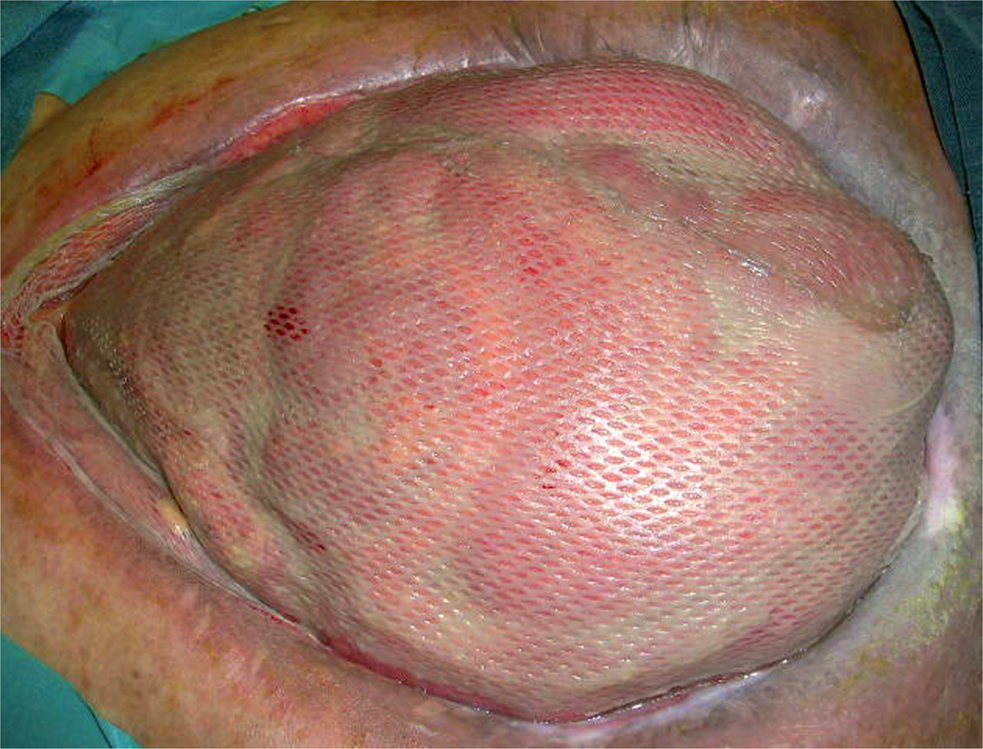
Split-thickness skin graft inserted over bowel loops
The main indications for a planned hernia approach are listed in Table 1. The long-term aims of abdominal closure following a planned hernia strategy include intact skin cover, fascial closure at midline (if possible), good functional outcome with innervated abdominal musculature, no pain and good cosmetic result.
Indications for planned hernia

RECONSTRUCTION METHODS
Overall, there are several methods available for abdominal wall reconstruction. The fascial defect can be repaired with prosthetic material or autologous tissue with multiple variations and combinations. Depending whether the fascial defect is covered with original or grafted skin, the repair methods can be broadly categorized in two groups. In Type I defects with stable skin coverage, bridging the fascial gap with prosthetic material or autologous tissue is sufficient, whereas in Type II defects with absent or unstable skin coverage, fascial repair alone is inadequate, and the repair must be covered with skin utilizing more complex reconstruction techniques. The criteria for special reconstruction techniques include a large size (40 cm2) defect, absence of stable skin coverage, recurrence of the defect after prior closure attempts, infected or exposed mesh, systemic compromise (intercurrent malignancy), local tissue compromise (irradiation, corticosteroid dependence) or concomitant visceral complications such as enterocutaneous fistula (1).
MESH REPAIR
In patients with original skin cover over the fascial defect (ventral hernia), the most commonly used method is hernia repair with an artificial mesh. There is a huge number of meshes available with a major commercial interest in the mesh market. The comparison of the different meshes is beyond the scope of this review. It is, however, important to be familiar with the different mesh placement options (onlay, sublay, inlay) and the interaction of the mesh with viscera if placed in direct contact with them. We prefer the use of an inlay mesh that can be placed in contact with the viscera and fix the mesh through all layers of the abdominal wall with interrupted, nonabsorbable sutures placed 1 cm apart about 5 cm or more overlap under the fascial layer. To reduce the size of the mesh needed, it is sometimes combined with a component separation (Fig. 2). If there is not enough fascial tissue at the edge of the defect to support the mesh, the mesh is fixed directly to the (usually iliac or pubic) bone with anchor sutures. Another technical trick is to create a big enough pouch with the mesh at the caudal part of the repair by extending the edges of the mesh deep into the pelvis. This puts the weight of the viscera, when the patient is standing, more on the mesh itself and less to the suture line.
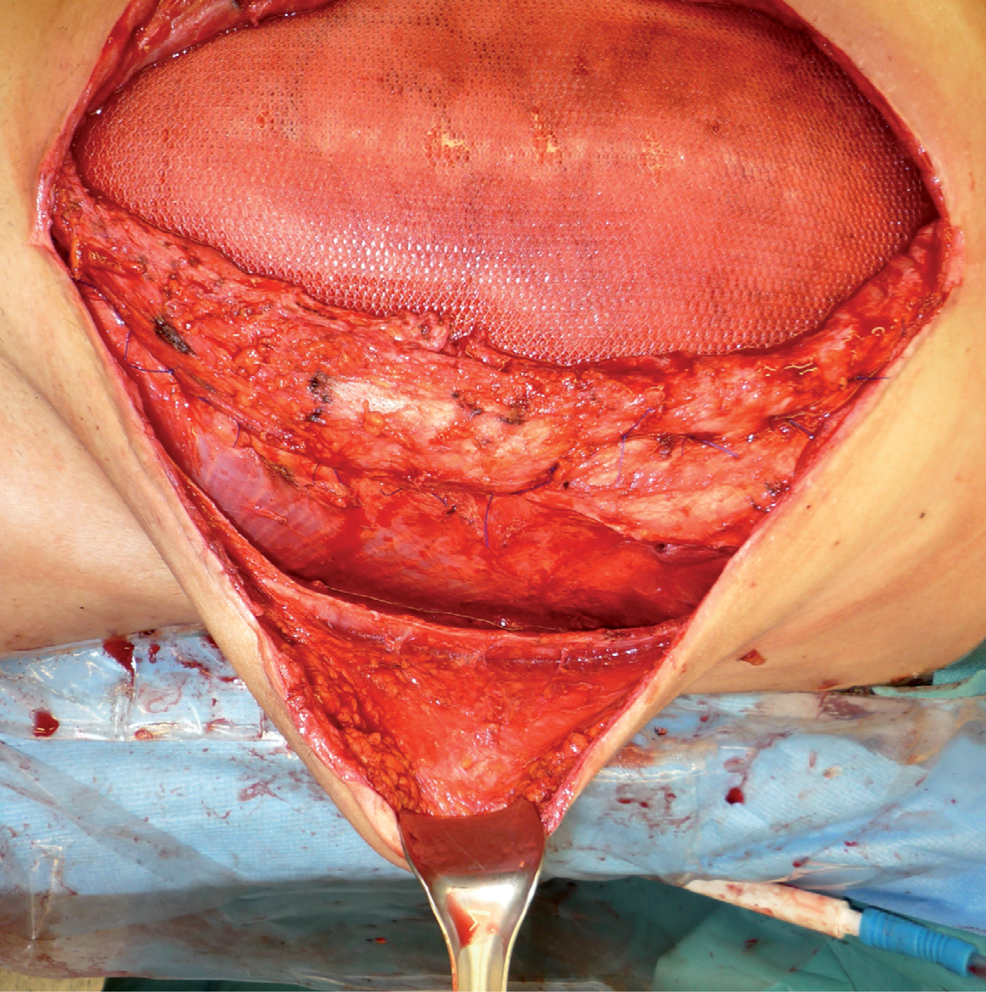
Inlay mesh repair combined with the component separation procedure
When a mesh is used for repair of extensive abdominal wall defects, following conditions need to be met: availability of normal skin to cover the mesh, applicability of the underside of the mesh directly over the bowel without causing bowel erosion with fistula formation or excessive adhesion formation (if intraperitoneal or inlay technique is used), and the risk of infection when used in a contaminated field (accidental bowel lesion during mobilization, presence of enteric fistula, simultaneous enterostomy closure).
More recently, biological meshes have been introduced to hernia repair surgery as a significant development in the armamentarium of surgeons, especially in patients with a contaminated field. The major advantage of these meshes is their ability to support revascularization and become part of human tissue. The application of combined advanced surgical techniques and biological materials may reduce the risk of recurrence and surgical site infection (9). However, more comparative studies are needed to evaluate the potential benefit of biological meshes. There is a concern that the new mesh types are put into use by surgeons world-wide without appropriate information of their long-term benefits. In addition, the current cost of the biological meshes is high.
COMPONENTS SEPARATION
The key surgical invention in the components separation procedure is the creation of a musculofascial rectus abdominis component that can be mobilized laterally and brought to the midline. Various methods to close the midline defects with sutures and fascial flaps were described by Guillouid in 1892, Chrobak in 1892, Gersuny in 1893 and Noble in 1895 (3). However, it was Alfonso Albanese from Argentina who in 1951 described the method of dividing the external oblique muscle vertically to enable the closure at midline by suturing together the rectus abdominis muscles (10). The method was rediscovered and refined by Ramirez and co-workers in their elegant study from 1990 (11). They used fresh cadavers to demonstrate that the external oblique muscle can be separated from the internal oblique muscle in a relatively avascular plane, and that the compound flap of the rectus muscle with its attached internal oblique – transversus abdominis muscle can be advanced 10 cm around the waistline. They subsequently used the technique in the reconstruction of abdominal wall defects in 11 patients with defect sizes ranging from 4 × 4 to 18 × 35 cm.
In the traditional method, the external oblique muscle is divided on both sides vertically about 2 cm laterally to the lateral edge of the rectus sheath. The external oblique muscle is then separated along the avascular plane from the internal oblique muscle all the way to the posterior groove. When done bilaterally, this procedure creates two rectus abdominis-transversus abdominis-internal oblique muscle flap complexes. These flaps complexes are then pulled medially and sutured together at midline with any technique used normally for midline abdominal closure. To get few more centimeters and decrease tension to the suture line, the posterior rectus muscle fascia can be divided at the middle part of the muscle. It is important to avoid injury to the epigastric blood vessels running below the muscle layer and not to create tissue pockets where bowel loops could be trapped.
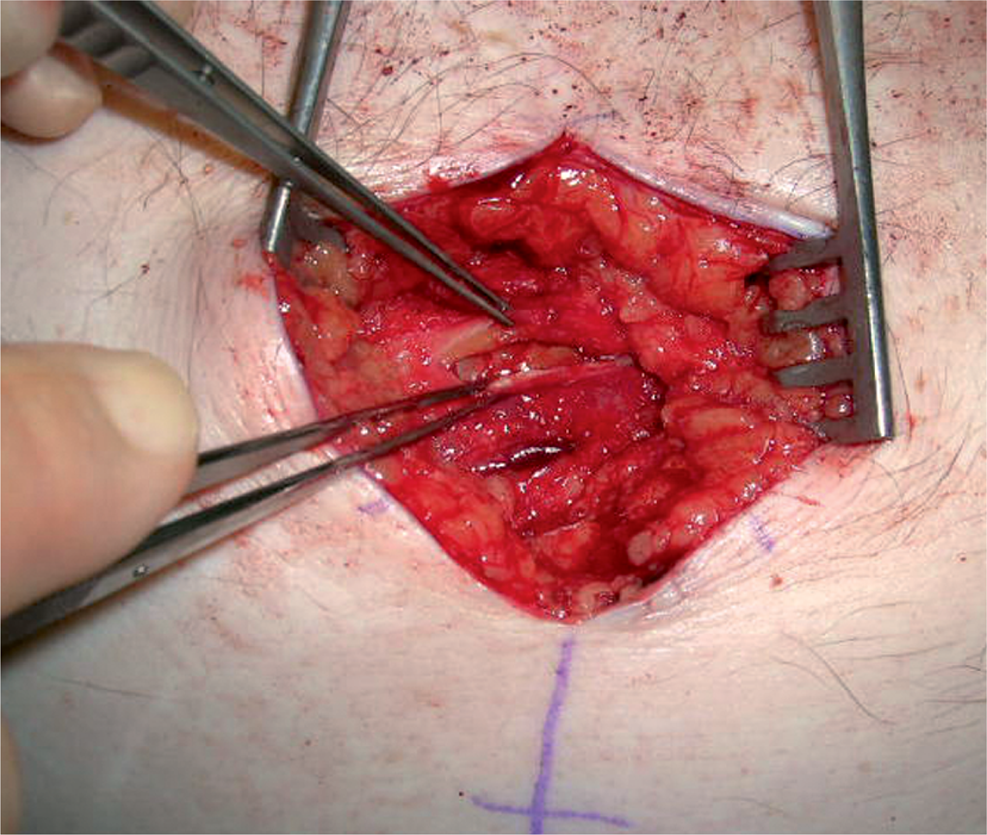
Minimally invasive components separation
A modification of the technique has been used where the muscle division and separation procedure is performed through small separate bilateral transverse incisions (Fig. 3). This technique allows the preservation of the perforant vessels to the skin flaps reducing the risk of subsequent skin necrosis.
The minimally invasive component separation procedure can also be performed endoscopically. In a retrospective study by Harth and Rosen comparing endoscopic and open techniques in 44 patients, both techniques had recurrence rates of about 30%, but the endoscopic group had shorter lengths of stay and less major wound complications (12).
MICROVASCULAR FLAPS
In comparison with prosthetic mesh repairs, autologous vascularized tissue flaps have the advantage of not implanting foreign material into the body thus reducing the risk of infection when applied to a contaminated field. They can also be used in large Type II defects where there is a large skin defect. Furthermore, they can be combined with other methods, such as components separation of mesh, to reduce the size of the flap needed. The disadvantages include the complex techniques required as well as the morbidity associated with the donor site. Although pedicled flaps can be used in small and mid-sized defects within the arch of the rotation of the flap, the size and location of the defect usually precludes the use of a rotational flap.
The main indication for using a microvascular flap is a large, full-thickness tissue defect (with grafted skin) extending into the upper abdomen. The tensor fascia latae (TFL) myocutaneous free flap was first described by Hill and co-workers in 1978 (13) and since then, its use has been reported in more than 100 patients. In a recent report from our institution, the free TFL flap was used in 20 patients with defects caused by tumor resection in 12, infected mesh in 2, and planned abdominal hernia in 6 patients where the visceral organs had been temporarily covered with split-thickness skin grafts. The skin graft was allowed to mature for 9–12 months until it could be freely rolled over the bowel. The repair was combined with a mesh in 9 patients, components separation in one patient, and the two additional techniques were used in one patient. The median follow-up was two (range 0.5–13) years. There was no perioperative mortality, or intra-abdominal or deep surgical site infections. There was one flap failure which required reconstruction with a second TFL flap from the contralateral side. In addition, there were two patients with minor distal tip necrosis, and one patient with recurrent hernia three months after TFL repair (14).
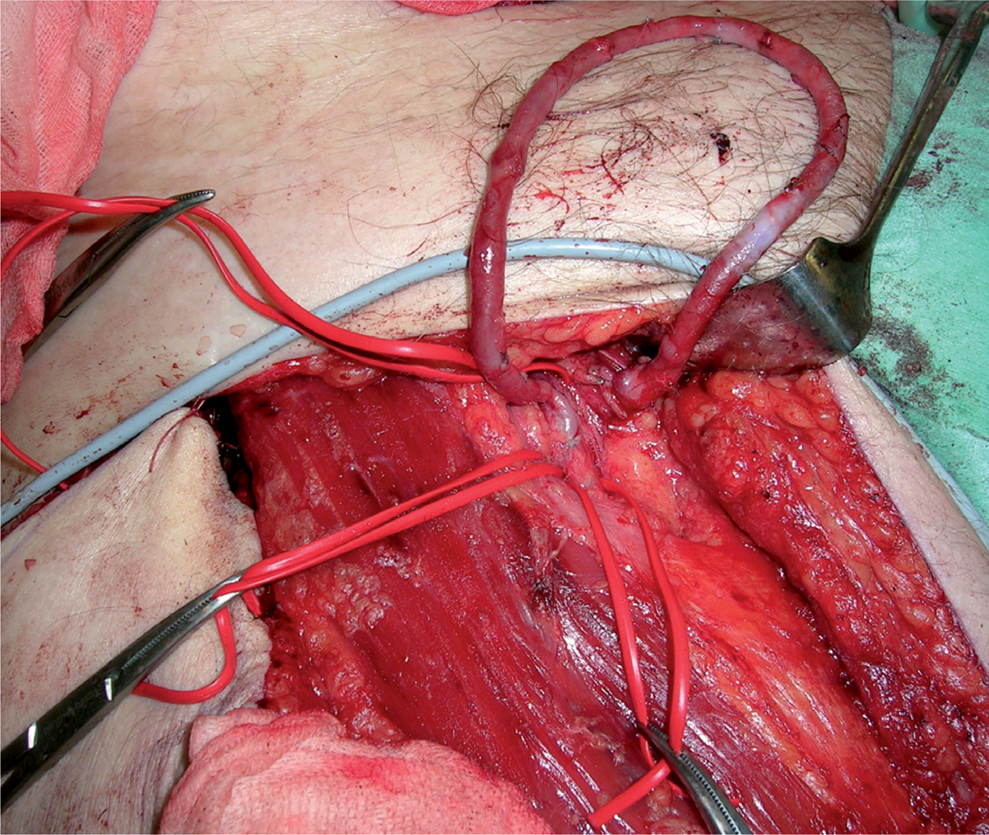
Microvascular TFL flap with the arteriovenous loop
The technique used at our institution is as follows: a musculofasciocutaneous TFL flap with a skin component measuring 30–35 cm × 15–20 cm and the underlying fascia with the tensor fascia latae muscle is harvested from the thigh and its pedicle dissected free towards the deep femoral artery and vein. In patients with large defects, the rectus femoris muscle can be included in the flap to ensure adequate perfusion of the distal tip. The ipsilateral great saphenous vein is divided distally above the knee, and its distal end is reflected proximally and anastomosed end-to-side to the common femoral artery creating an arteriovenous loop (Fig. 4). The loop is tunneled subcutaneously to the edge of the defect and divided at its apex. Arterial and venous anastomoses with the flap vessels are performed with continuous 7–0 or 8–0 vascular sutures. The flap fascial edges are sutured to the fascial edges of the original defect carefully avoiding any obstruction or kinking of the flap vessels. Drains are placed subcutaneously, and the subcutaneous space and skin are closed with interrupted sutures or staples. The donor site is closed directly as far as possible, and the remaining defect is covered with a split-thickness skin graft (14). Postoperatively, low molecular weight heparin is injected daily to the flap to reduce the risk of flap venous thrombosis, and the viability of the flap is monitored repeatedly. The extremity is supported in the bed to prevent extension of the vascular anastomosis area and any pressure or tight dressing on the vascular anastomosis site is avoided. Fig. 5 shows the end result after the flap has healed.
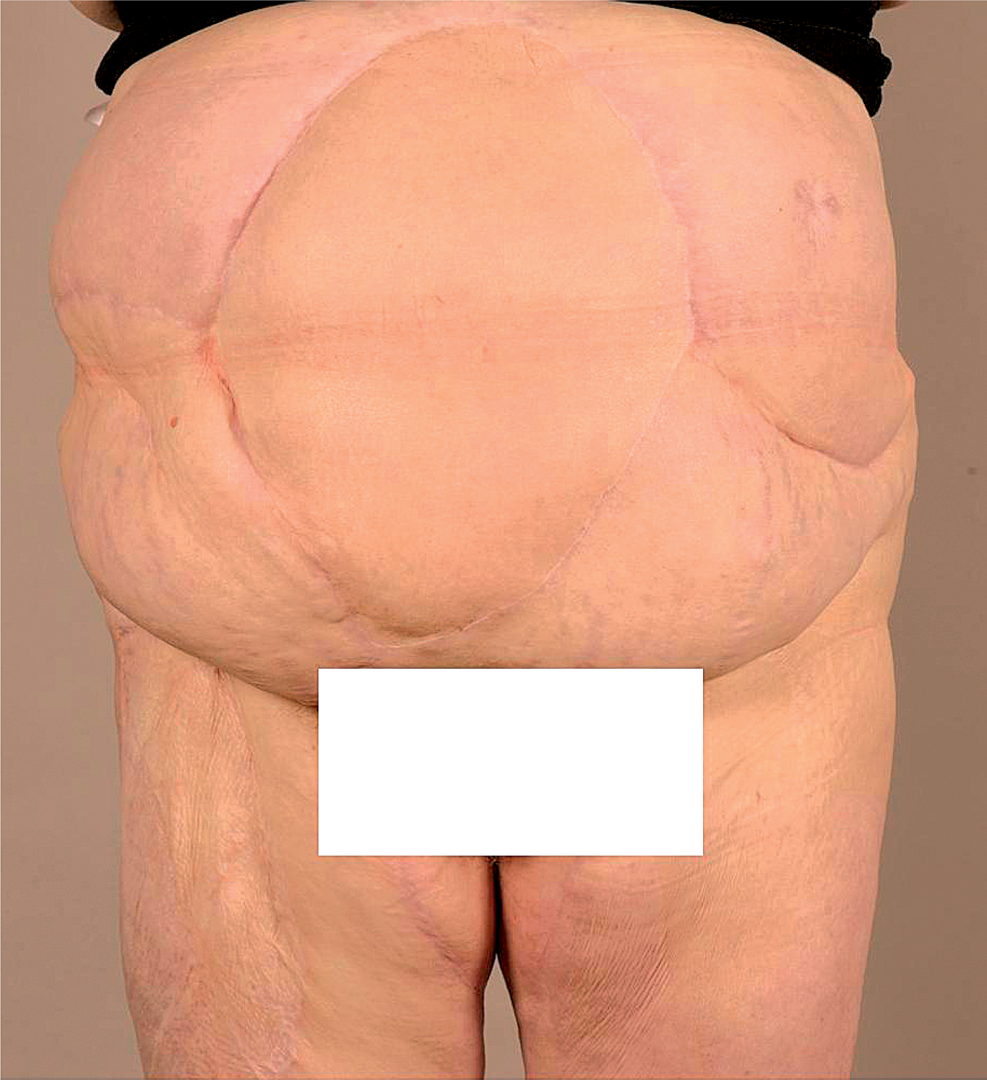
Healed TFL flap
The advantages of the TFL flap compared with an anterolateral thigh flap are the more constant anatomy of the TFL pedicle and larger vessel caliber matching the vessel size of the great saphenous vein loop. In addition, the size of the flap can be quite large although in very wide flaps the relative thinness of the anteromedial portion of the fascia, especially in women, sometimes requires mesh enforcement. Furthermore, the location of the donor site in the thigh has no effect on postoperative respiratory function, and usually heals well.
CHOICE OF RECONSTRUCTION METHOD
Most patients with Type I defects can be treated with mesh repair or components separation alone or in combination provided that there is enough original skin to cover the repair without tension. Because intuitively it seems beneficial to reconstruct the linea alba to provide a better functional repair, the components separation might be better in achieving this than a passive mesh, but there is little scientific evidence to support this. In contrast to inert material, the abdominal musculature provides dynamic support of innervated tissue to redistribute the stress applied from intra-abdominal forces. There is a randomized, controlled study where the components separation technique was compared with prosthetic repair with e-PTFE patch in 37 patients (15). The reherniation rates were higher after components separation (10/19 versus 4/18), but because of the high infection rate leading to removal of the prosthesis in 7/18 patients, however, the trial was discontinued at the interim analysis.
To reduce reherniation risk after components separation, a mesh can be inserted to enforce the repair. However, in a long-term follow-up study the use of prosthetic enforcement increased the recurrence rate 4-fold. Using a modified technique where an additional relaxing incision of the internal oblique muscle was followed by suturing the medial border of the posterior sheath to the lateral border of the anterior sheath, the authors were able to reduce the recurrence rate to 5% without using a prosthetic mesh (16).
There are reports indicating that biological meshes can be safely used in contaminated fields, and they might provide an option in patients with high risk of infection and unsuitable for components separation technique (17, 18).
More advanced reconstruction techniques are required mainly in extensive Type II defects or in patients with infected mesh. The most attractive method seems to be the microvascular TFL flap although it requires extensive experience and close collaboration with abdominal and plastic surgeons, and probably should only be used in specialized centers with sufficient institutional and surgeon-specific volume of patients with complex abdominal wall defects (19).
At our institution the first choice of reconstruction is the components separation technique, mostly performed in a minimally invasive way maintaining the integrity of the perforant vessels. If needed, the technique can be combined with a mesh place in inlay position provided that adequate skin cover is possible. In contaminated fields where component separation is not feasible alone, a combination with a biological mesh has been used in few patients. In large defects with grafted skin a free TFL flap is our first choice, sometimes reinforced with a mesh and enhanced with components separation.
ABDOMINAL WALL DEFECT WITH ENTERIC FISTULA
When an abdominal wall defect is complicated by the presence of an enterocutaneous or enteroatmospheric fistula, and especially when the abdominal cavity is open and scarred into a one solid mass with fragile small bowel loops adhered to each other (hostile abdomen), a multidisciplinary and individualized approach is needed (20).
The first step is to control a possible infection source and treat the systemic effects of sepsis. If the enteric fistula opens into the abdominal cavity and between the bowel loops, source control may be possible with intestinal repair or diversion, but often the only option available is to control the enteric leak with external drainage using multiple drains of sufficient diameter and decompressing the involved bowel segment with multiple intraluminal tubes. Once the fistula is controlled, the drains can be gradually removed by shortening the drains until a controlled enterocutaneous fistula tract is formed. An enteroatmospheric fistula requires complex stomal techniques to isolate the enteric content from the granulation tissue that has formed over the bowel loops. The raw surface should be covered with skin graft as soon as possible. The maturation after inserting the skin graft usually requires 6–12 months. By then the skin graft can be easily separated from the underlying bowel loops and the intra-abdominal adhesions can be divided without problems.
The second step concurrent with the maturation period consists of providing adequate nutritional support, preferably by enteral route, to create favourable local and systemic conditions for healing.
When the skin graft has matured and the patient is in good nutritional condition, definitive repair of the gastrointestinal tract and the abdominal wall defect can be planned. They can usually be performed in a single operation involving both gastroenterological and plastic surgeons. The restoration of the gastrointestinal continuity follows the established techniques of one or multiple intestinal anastomoses. The method of the reconstruction of the abdominal wall defect needs to be tailored individually following the principles outlined above. Smaller defects can be reconstructed with the component separation technique if enough original skin is available to reach the midline, whereas larger defects require a flap reconstruction.
