Abstract
Background and Aims:
Alterations in periprosthetic bone are common sequlae of prosthetic implants. This prospective 3-year study was performed to follow regional periprosthetic bone turnover after uncomplicated total hip arthroplasty (THA) using single-photon emission computed tomography (SPECT).
Material and Methods:
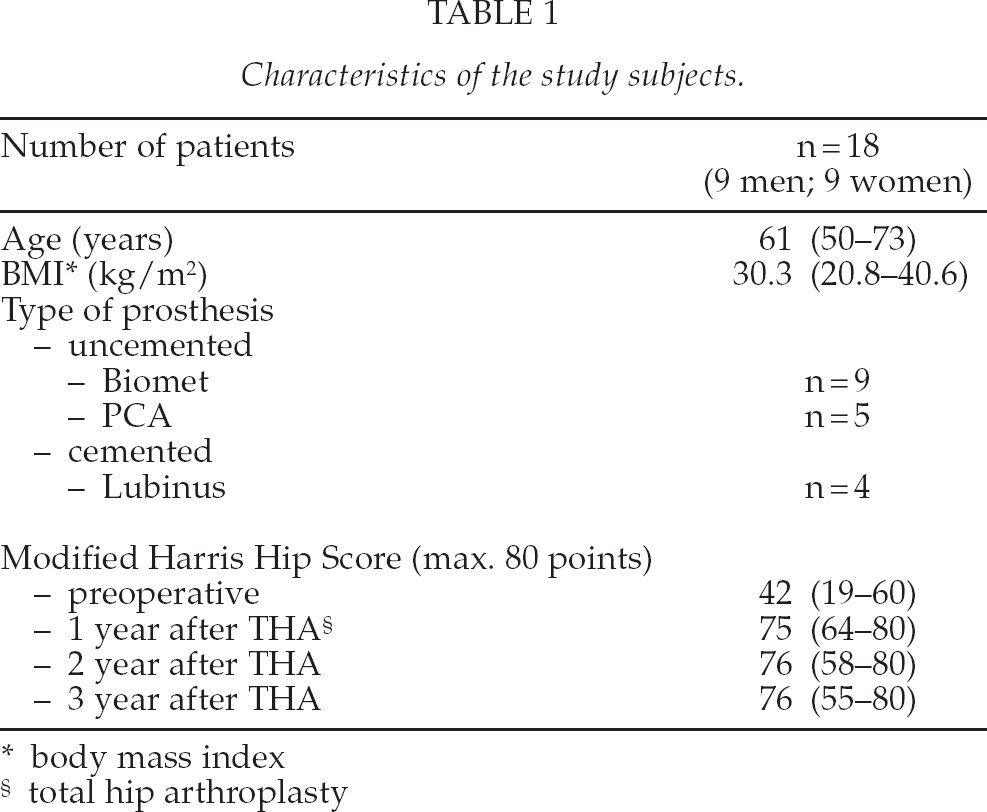
Eighteen patients (nine men, nine women: mean age 61 years, range from 50 to 73 years) with primary hip osteoarthritis underwent either uncemented or cemented THA. The SPECT measurements were taken 6, 12, and 36 months after THA. B one mineral density (BMD) measurements were performed on the patients during follow-up.
Results:
The mean SPECT uptake ratios decreased significantly in the regions of interest (ROIs) during follow-up compared to baseline value, in the trochanter major (p = 0.006), the trochanter minor (p = 0.009) and the total area (p = 0.018). Despite these decreases the uptake ratios in the medial cortex (p = 0.014), tip (p = 0.002) and total area (p = 0.016) remained significantly higher in the operated side than in the contralateral side (non-operated) 3 years after THA. Changes in bone turnover in the vicinity of the implant did not correlate with changes in periprosthetic BMD.
Conclusions:
Local periprosthetic bone turnover decreased slowly after THA and did not recover to the level typical of non-operated hips. This led us to suggest that bone turnover around the femoral implants remains increased more than 3 years after THA even if there are no signs of failure of the prosthesis.
Keywords
INTRODUCTION
Total hip arthroplasty (THA) is a common operation in patients with osteoarthritis of the hip (1). The insertion of metallic implant causes changes in bone mass and bone turnover in the vicinity of the implant. This is usually seen as a resorption of bone in the proximal part of femur because of altered loading patterns (stress shielding) (2, 3). Orthopedic surgeons are interested in following periprosthetic bone reactions in order to evaluate the results of THA and to discover whether some prostheses are more preferable than others. Several methods have been used to follow periprosthetic bone changes, such as serial standard radiographs, computed tomography, magnetic resonance imaging, and dual-energy X-ray absorptiometry (DXA) to follow periprosthetic bone changes (4–7).
Single photon emission computed tomography (SPECT) bone scanning using Tc-99m-diphosphonates is a useful method for detecting disorders of the skeleton and for measuring bone turnover (8–10). A few studies discussed the role of SPECT after arthroplasty. McMahon et al. studied femoral head vascularity after hip resurfacing and Soininvaara et al. studied bone turnover after uncomplicated total knee arthroplasty (TKA) (11, 12).
This prospective 3-year study aimed to follow regional periprosthetic bone turnover after uncomplicated THA with SPECT. The main purpose of the study was to examine whether or not bone turnover recovers to its ‘normal’ level after successful arthroplasty.
Furthermore, we studied correlations between periprosthetic SPECT uptake and changes in periprosthetic bone mineral density (BMD).
MATERIALS AND METHODS
PATIENTS
Eighteen patients with primary hip osteoarthritis were included in this prospective periprosthetic SPECT study. Patients (nine men and nine women) underwent either uncemented or cemented primary THA at Kuopio University Hospital from November 1993 to August 1994 using standard operative techniques. In order to be included in this study, the patients were required to be without any bone metabolic disease and to not have taken any bone-affecting drugs previously or during the follow-up. All patients provided written informed consent. The study protocol was approved by the local Ethics Committee (13).
The SPECT measurements were taken 6, 12, and 36 months after THA, and BMD measurements were taken within 2 weeks postoperatively, and at 3, 6, 12, 24, and 36 months after THA. Regular clinical and radiological follow-ups were organized. A modified Harris Hip Score (mHHS) was used (maximum 80 points) to evaluate clinical outcomes (14).
The femoral prostheses used in this study were uncemented Bimetric Titanium Alloy (Ti6A14V) implants (n = 9, Biomet Inc., Warsaw, IN, USA), uncemented PCA E Series Hip System (n = 5, Howmedica International Divisions of Pfizer), and cemented Lubinus SPII stems with collar (n = 4, Waldemar Link CMBH&CD, Germany). Uncemented femoral stems are proximal load carrying components, but they have some differiencies. PCA E Serie femoral stem is made from cobolt-chromium, the shape of the stem is anatomic and the surface is double layer porouscoated. The Bimetric stem is made from titanium alloy, it is straight and the surface of the stem is plasmasprayed. The bone cement used was Palacos® R-40 cum Gentamicin (contrast medium zirconium, Schering-Plough, Brussels, Belgium).
The mean age of the patients was 61 years (50–73 years). Their mean body mass index (BMI) was 30.3 kg/m2 (20.8–40.6 kg/m2) (Table 1). The normal postoperative mobilization schedule was followed, where patients with cemented arthroplasty were encouraged to perform full weight-bearing exercises immediately after the operation. Patients with uncemented prostheses were encouraged to perform partial-weight bearing exercises with crutches on the operated hip for 6 weeks after THA. In two patients, postoperative X-rays showed fissure fractures in the operated femur after uncemented arthroplasty, but these healed without any later complications.
Characteristics of the study subjects.
body mass index
total hip arthroplasty
SPECT
The SPECT scans were performed using a three-headed SPECT system (Siemens MultiSpect 3, Siemens Gammasonics Inc., Des Plaines, IL, USA) equipped with high-resolution collimators. The measurements were started 4 h after an intravenous injection of 740 MBq of Tc-99m-methylene diphosphonate (Tc-99m-MDP, Medronate II®, Amersham, Bucks., UK). A full 360° rotation was acquired (40 views/head, each for 35 s, using a 128 × 128 matrix).
After reconstruction using a filtered back-projection technique (Butterworth filter: order 6 and cut-off frequency 0.5 / cm), the data were carefully aligned in transverse, coronal, and sagittal projections in order to view both femurs and acetabular areas in the same plane. Thereafter, four original slices were summed to the final 14-mm-thick coronal slices that were used in quantification. Twelve symmetrical regions of interest (ROIs) (trochanter major, trochanter minor, lateral cortex, medial cortex, tip and reference area above the acetabulum) were manually drawn (lower threshold 7–9%) on the prosthesis and contralateral sides (Fig. 1). All regional uptake values (in counts/voxel) were divided by the averages obtained from the sum of reference areas to give regional uptake ratios. In addition, a total average uptake ratio (total area) was calculated over the femoral ROI in the ipsi- and contralateral sides (Table 2).
The mean SPECT uptake ratios (sd) in the regions of interest around the femoral stem during the 3-year follow-up.
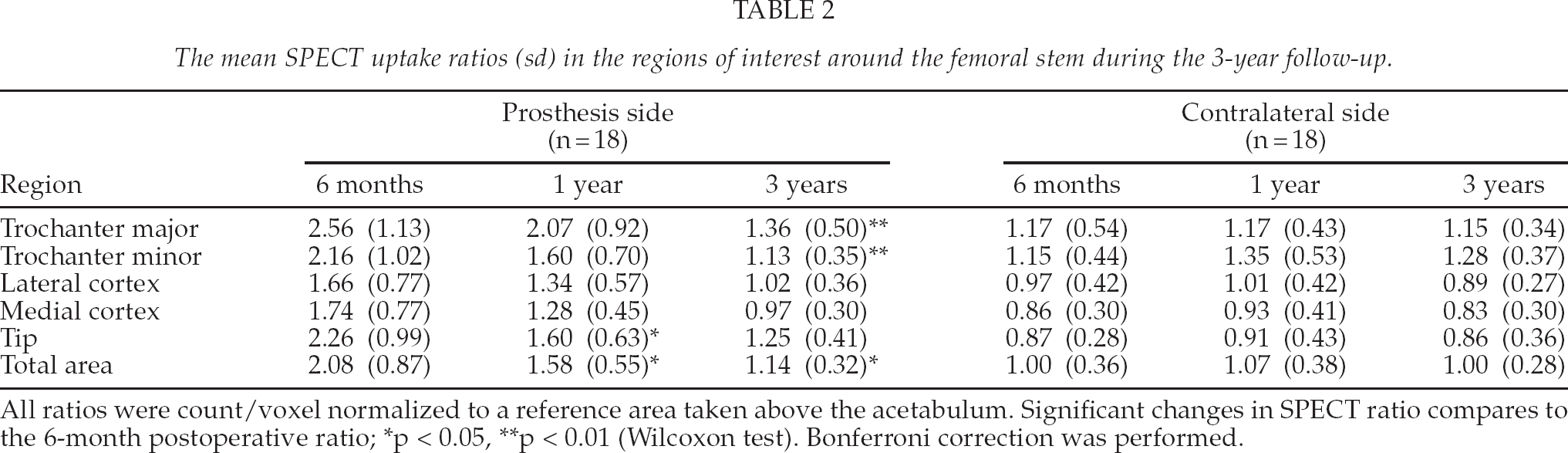
All ratios were count/voxel normalized to a reference area taken above the acetabulum. Significant changes in SPECT ratio compares to the 6-month postoperative ratio;
p < 0.05,
p < 0.01 (Wilcoxon test). Bonferroni correction was performed.
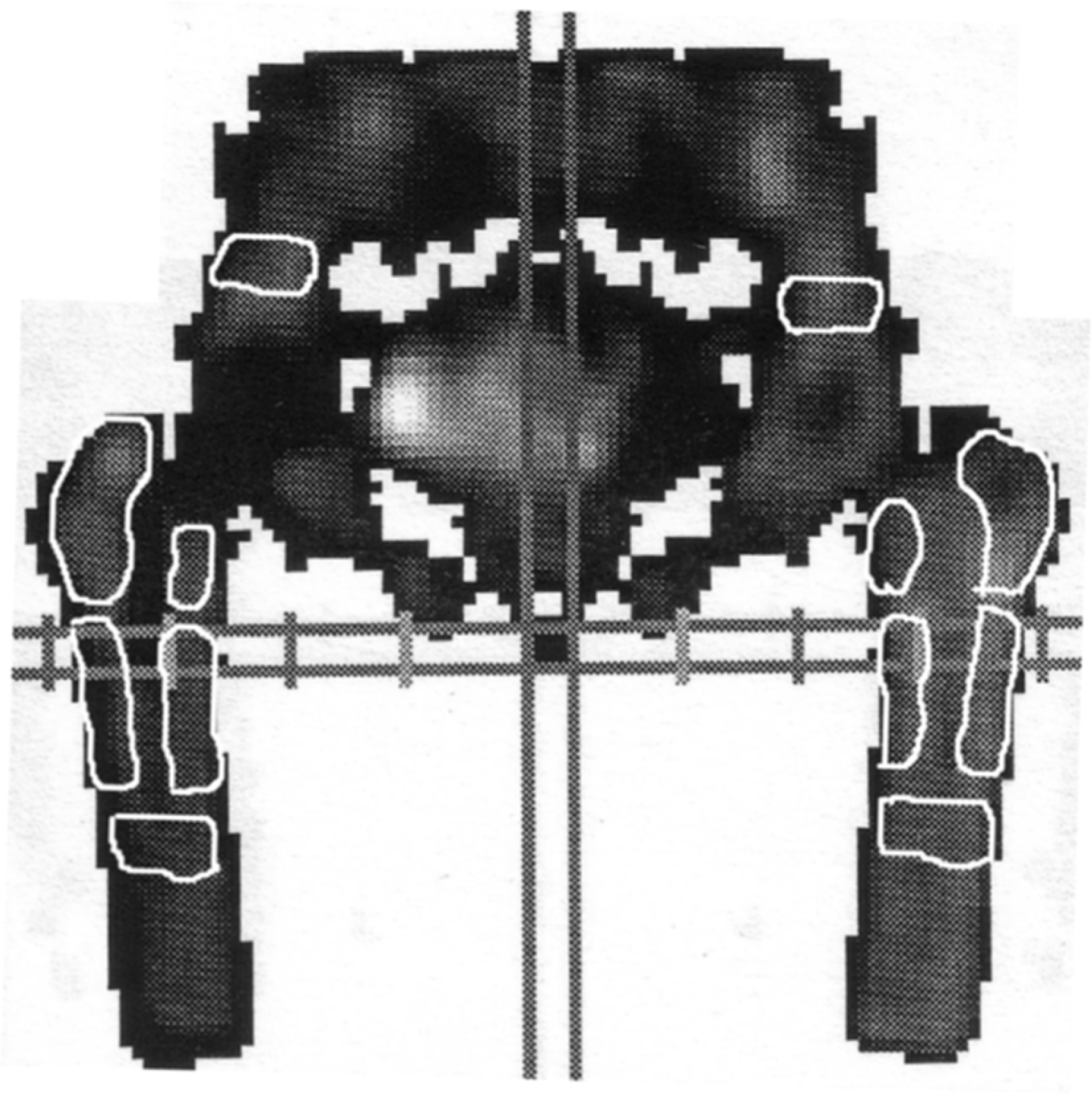
A SPECT image showing the twelve ROIs; a prosthesis side (right femur) and a control side (left femur).
DXA
The BMD measurements were taken using DXA (Lunar DPX; Lunar Co., Madison, WI, USA) with special orthopedic software (Lunar; version 1.2) according to the ROIs of Gruen and Lunar (15). The technique is reported in more detail in previously published articles both cemented and uncemented prostheses (7, 14).
Because the one purpose of the study was to compare results of SPECT and BMD measurements, five ROIs similar to those used in the SPECT measurements were created when the BMD values were calculated. Gruen ROIs 2 and 3 were combined in order to create the lateral cortex ROI and ROIs 5 and 6 were combined for the medial cortex ROI. The Gruen ROI 4 was same as the tip ROI. Lunar ROIs A and B were combined to form the trochanter major ROI and Lunar ROIs C and D formed the trochanter minor ROI. All five ROIs were combined to give the total area (Table 3).
The mean bone mineral density (g/cm2) (sd) in the different regions of interest during the 3-year follow-up.
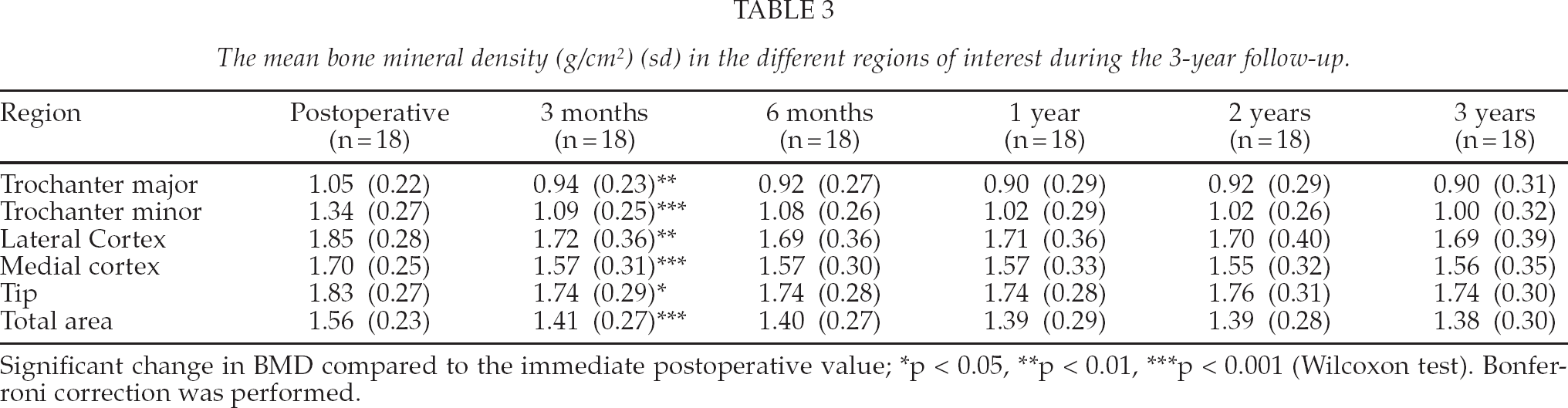
Significant change in BMD compared to the immediate postoperative value;
p < 0.05,
p < 0.01,
p < 0.001 (Wilcoxon test). Bonferroni correction was performed.
STATISTICAL ANALYSIS
SPSS software version 11.0 (SPSS Inc., Chicago, IL, USA) was used for the statistical analyses. The non-parametric statistical tests used were the Mann-Whitney test, the Wilcoxon test and Spearman's correlation. Statistical significance was defined as p < 0.05. The SPECT uptake ratios and BMD values were expressed as means and standard deviations (SD). Postoperative changes in the SPECT uptake ratios and BMD values were calculated using the first measured SPECT uptake ratio/BMD value as a reference: this SPECT uptake ratio/BMD value was compared to subsequent SPECT uptake ratios/BMD values in each ROI. The differences were expressed as percentages.
RESULTS
At the end of the follow-up there were no signs of loosening or other complications after THA. The mHHS changed from the average preoperative value of 42 to 75, 76, and 76 at 12, 24, and 36 months after the operation, respectively.
The mean SPECT uptake ratios (SD) and changes up to 3 years in the ROIs are shown in Table 2. The mean SPECT uptake ratios decreased in all ROIs from that of baseline values (six months after THA). The changes were significant at one year in the tip and total area (p < 0.05), and at three year in the trochanter minor (p < 0.01), trochanter major (p < 0.01) and total area (p < 0.05). Despite this decrease the uptake ratios in the medial cortex (p = 0.014), tip (p = 0.002) and total area (p = 0.016) remained significantly higher in the operated side than in the contralateral side (non-operated) at three years after THA. The uptake ratio in the trochanter minor uptake ratio was lower in the operated side (p = 0.034) (Fig. 3).
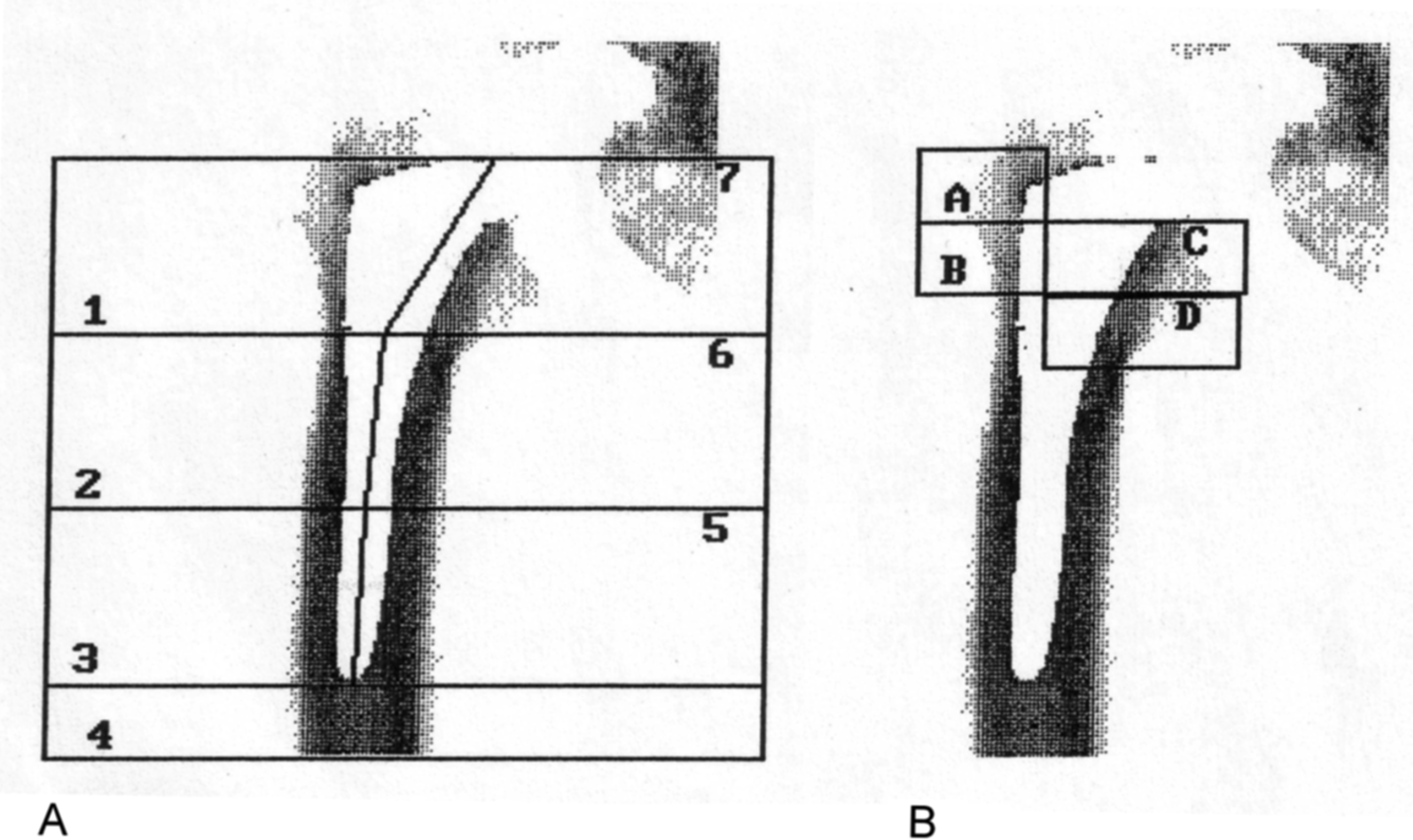
DXA images showing Gruen ROIs (A) and Lunar ROIs (B).
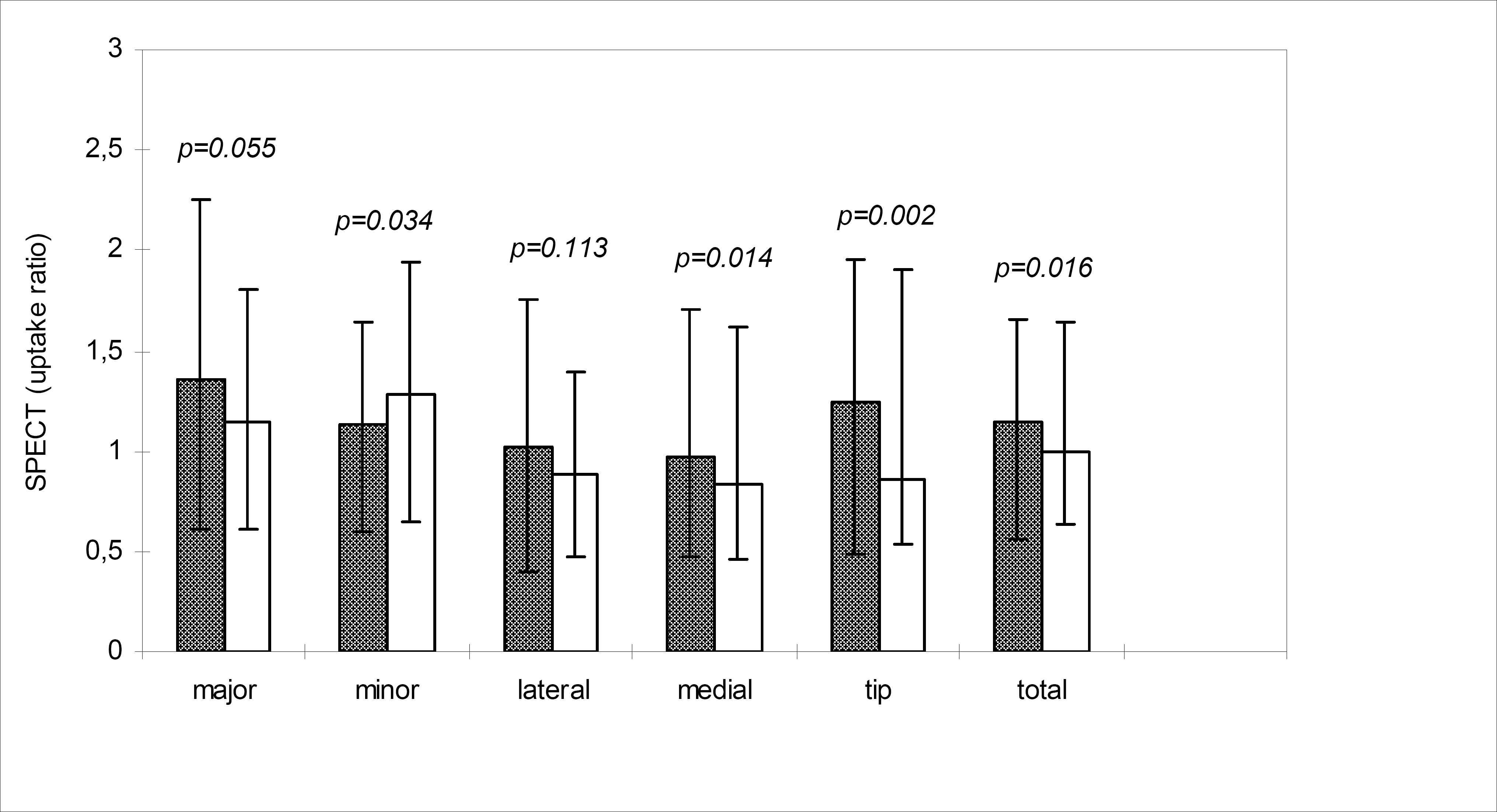
The mean SPECT uptake ratio (with the range of uptake ratios) in the prosthesis and control sides measured 3 year after THA. Grey bars represent uptake ratios in the prosthesis side and white bars represent uptake ratios in the control side. Significant differences between the sides were analysed using the Wilcoxon test (p < 0.05).
The mean BMD values (SD) and percentage changes are shown in Table 3 and Fig. 4. The BMD decreased by 5% to 19% in all ROIs during the first 3 months. All of these changes were statistically significant (p < 0.05 to p < 0.001). Following this, a slow decrease in BMD continued in the proximal part of femur, reaching −26% in the trochanter minor by the end of the follow-up. Only minimal BMD changes were found in the other ROIs after 3 months.
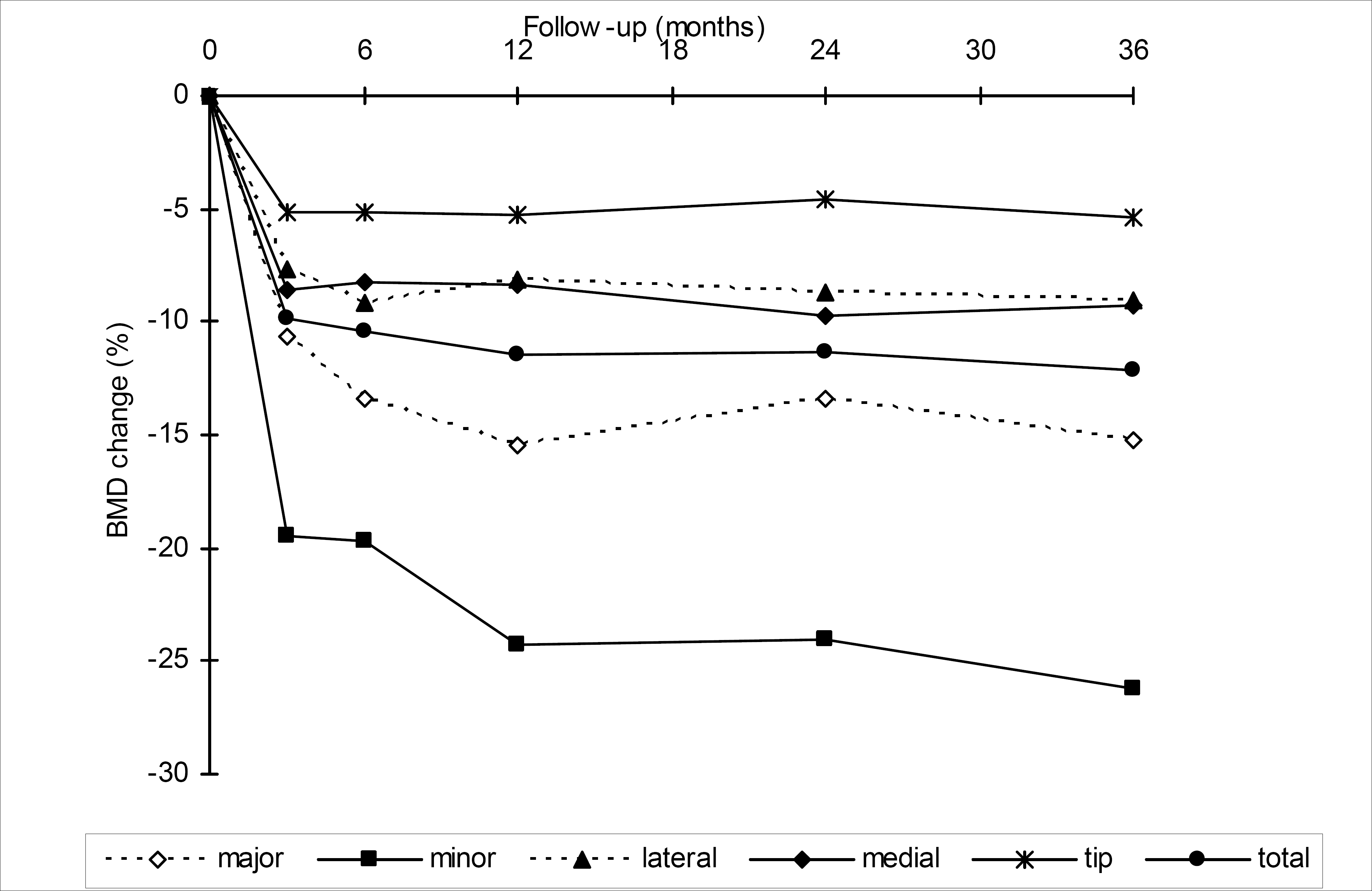
The mean change (%) in BMD in the five regions of interest and the total area.
No significant correlations were found between yhe SPECT uptake ratio and changes in BMD. Three different types of prostheses were implanted. The rate of bone turnover in the uncemented stems (n = 14) was lower than that in the cemented stems (n = 4) by the end of follow-up (Fig. 5).
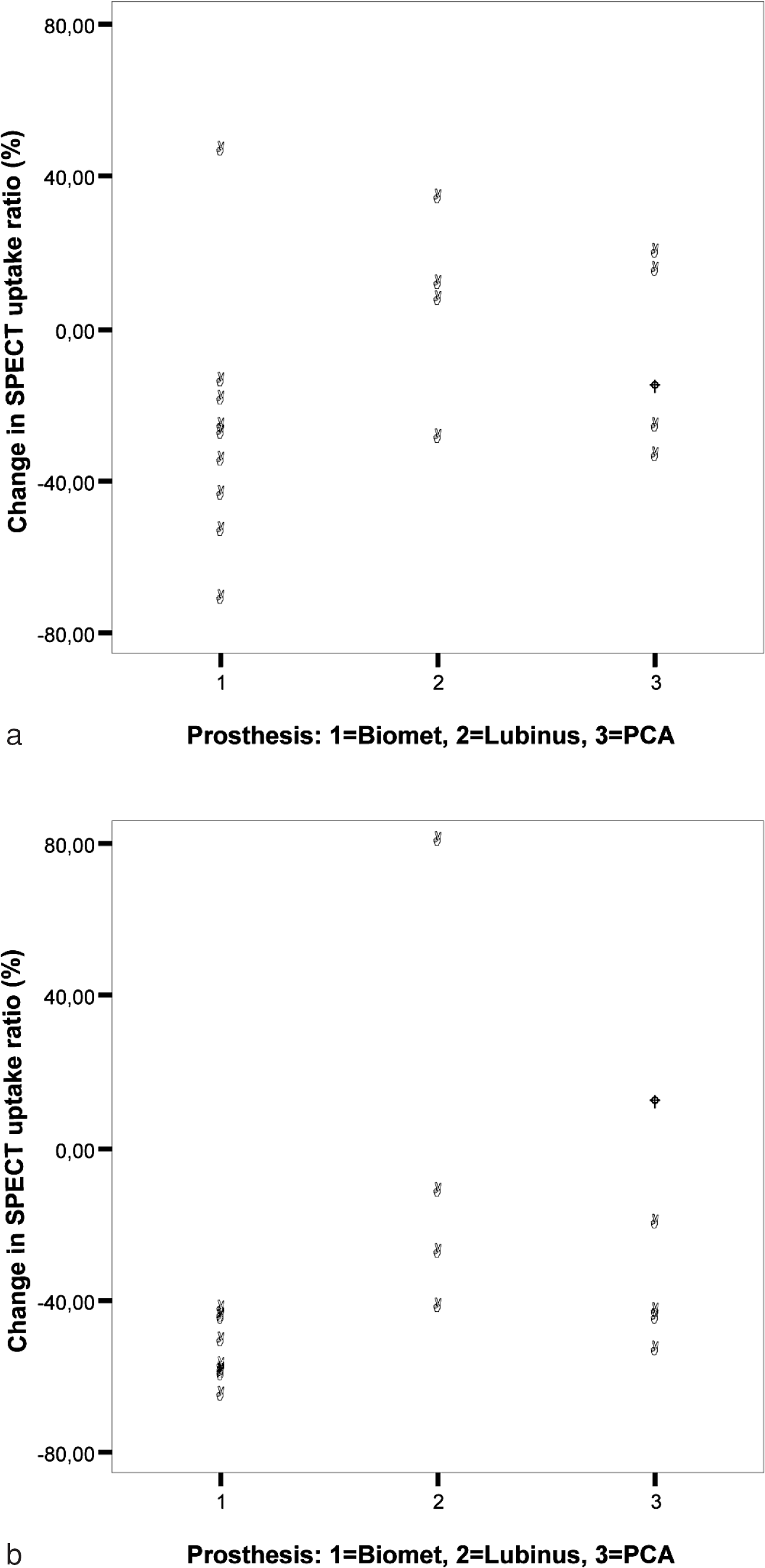
Changes in total average SPECT uptake ratios compared to the type of prosthesis over 6–12 months (A) and 6–36 months (B) of follow-up. The black circle represents the revision case.
DISCUSSION
In this prospective three-year study, 18 patients were followed with high-resolution SPECT in order to examine periprosthetic bone turnover around the femoral stem. We found that local bone turnover in the vicinity of the implant decreased slowly during the 3 years of follow-up. This decrease was statistically significant in the trochanter major, trochanter minor, and the total area by the end of follow-up. We also compared bone turnover rates in the operated hip and the contralateral hip (non-operated) at the end of follow-up. The SPECT uptake ratios remained higher in the operated hip except in one ROI (the trochanter minor). The bone turnover rate did not return to the level that seems to be typical of non-operated hips. During the 3-year follow-up, no signs of aseptic loosening or periprosthetic infection were observed in any of the patients. Thus, it seems that the normal phenomenon after THA is a decrease in bone turnover around the femoral stem, although still being higher than that found in normal bone, even if there are no signs of failure of the prosthesis.
Rahmy et al. studied 80 asymptomatic patients to establish the course and duration of bony ingrowths after uncemented hydroxyapatite-coated arthroplasty using a quantitative analysis of Tc-99m-MDP uptake. They found decreased bone turnover 1 year after THA, but this remained higher than the normal level. They concluded that this is normal shortly after THA. They also suggested that this ‘normal’ database could be used as a reference point to detect failures of THA (16). Utz et al. performed a 3-year prospective study with 97 patients to determine the natural postoperative appearance of bone scans using Tc-99m-MDP. They used a four-point grading system to classify patients according to the rate of bone turnover after THA and found that about 20% of patients have increased bone activity for more than 1 year, and 10% have a persistent increase in bone turnover after THA (17). Increased SPECT uptake indicating normal post-operative remodelling was also found 2 years after uncomplicated TKA in one study (12). The results of our study are in agreement with these findings.
Interestingly, we found that two patients had a high rate of bone turnover in the total area 3 years after THA (Fig. 5b). In the patient who underwent uncemented THA a markedly elevated SPECT uptake was found in the lateral cortex and the tip of the prosthesis. At that time, the patient was asymptomatic and X-rays showed no signs of loosening. However, 5 years after the operation, the patient suffered from thigh pain and new X-rays showed osteolytic lesions in the lateral side of the stem. The patient underwent a revision operation 6 years after primary THA because of aseptic loosening of the implant. In agreement with Utz et al. (17), we suggest, that a persistent increased SPECT uptake ratio may be a sign of aseptic loosening. It is thus prudent to follow patients with persistent increases in bone turnover rate around their prosthesis more carefully because this may be an early sign of arthroplasty failure.
There may be some other factors influencing the rates of bone turnover after THA. Rosenthall et al. studied the bone turnover rate for 12 months after THA where two different types of implants were used. In their study, the patients either received a titanium alloy S-ROM prosthesis or a cobalt-chrome alloy AML prosthesis (the elastic modulus of the S-ROM prosthesis is half that of the AMLprosthesis). They found significant differences in bone turnover in most of the ROIs between these two types of implants, with different levels of stiffness (18). Oswald et al. followed 21 patients with uncomplicated porous-coated THA for 2 years. In their prospective three-phase bone scan study, they concluded that the pattern of bone turnover differs in patients with uncemented THA from that seen in patients with cemented THA (19). In this study, three types of prostheses were implanted and two different fixation methods were used. Each type of prosthesis was associated with a different pattern of bone turnover, and bone turnover was lower around uncemented stems compared with cemented stems (Fig. 5b). Our study confirms that each implant seems to have own pattern of regional bone turnover after THA. This should be recognised when interpreting clinical patients. It is unlikely that different postoperative mobilisation schedule has influenced regional bone turnover three years after operation.
Several prospective BMD studies have shown that most bone loss occurs during the first 3–6 months after THA, and that by 1 year after the operation only minimal changes in BMD are found (7, 12, 20–22). A similar behavioral pattern of BMD was found in the study. Previously, we found that elevated SPECT uptake was related to early periprosthetic bone loss. In this study, SPECT uptake was compared with BMD changes during the period when maximal bone resorption usually occurs (the first 6 months after THA) (13). In the present study, SPECT uptake was compared with later BMD changes when the changes in bone density were only minimal, and no correlations were found. This is in agreement with results of Rosenthall, who suggested that the degree of radio-phosphate deposition is a reflection of bone turnover, but this may not correlate with BMD changes after THA (23).
Interestingly, the trochanter minor ROI uptake ratio was lower in the operated side than in the control side at the end of 3-year follow-up. The trochanter minor ROI is the smallest and more susceptible to measurement error which may explain the result.
This study had some limitations. For example, we did not measure the immediate postoperative bone turnover rate but only the rate 6 months after THA: also, the sample size was quite small, with different implants. Manual localization and drawing of the ROIs in SPECT analyses is not as accurate as localization of the ROIs in DXA analyses. However, we believe that reactions in proximal femoral bone are highly visible 6 months after THA.
The present study showed that SPECT is a feasible method for following periprosthetic bone turnover after THA. The local periprosthetic bone turnover rate decreased slowly after THA and did not recover to the level typical of non-operated hips. This led us to suggest that bone turnover around femoral implants remains increased for more than 3 years after THA, even if there are no signs of failure of the prosthesis. This should be noted in clinical practise when analyzing SPECT examinations. Although SPECT method is not suitable for routine THA follow-up, patients with persistent symptoms after uncomplicated arthroplasty may benefit from it. Larger prospective studies with preoperative baseline measurements, different stem designs and longer follow-up are warranted.
Footnotes
ACKNOWLEDGEMENTS
The authors thank Riitta Toroi, R.N. and Eila Koski, R.N., for their expert technical assistance.
